Abstract
Peripheral nerve injuries (PNIs) often cause persistent sensory and motor deficits that impair quality of life. To improve outcomes, cell-based therapies have been explored, and Schwann cells (SCs) are considered a promising option because of their essential roles in myelination and neurotrophic support. However, the clinical use of autologous SCs is limited by donor site morbidity and challenges in large-scale expansion, leading to growing interest in human induced pluripotent stem cell (iPSC)–derived SCs. This study aimed to verify the therapeutic effects of iPSC-derived SCs in vivo and to determine whether the differentiation stage of the SCs influences their efficacy. We generated dibutyryl-cAMP–treated mature SCs and untreated immature SCs from iPSCs and transplanted them into a rat sciatic nerve crush injury model. Morphometric analysis showed that myelinated fiber density (fibers/mm2) was significantly higher in the mature SC group than in the control group (12 602 ± 1419 vs 10 105 ± 1673). Myelinated axon diameter (µm) was also significantly greater in the mature SC group (4.73 ± 0.25) compared with the immature SC (4.06 ± 0.28) and control (3.86 ± 0.15) groups. Electrophysiological testing demonstrated that compound muscle action potentials in the pedal adductor muscle were detected only in the mature SC group within 28 days after surgery. Western blot analysis of the tibialis anterior muscle showed significantly higher myosin heavy chain 1 (MYH1) expression in the mature SC group than in the control group. Quantitative reverse transcription-polymerase chain reaction revealed higher expression of bNGF, CCL2, and LAMA2 in mature SCs than in immature SCs. Pathological analysis suggested accelerated Wallerian degeneration in the mature SC group. These results demonstrate that mature iPSC-derived Schwann cells, rather than immature ones, most effectively promote nerve regeneration both histologically and functionally in vivo, highlighting the critical importance of the SC differentiation stage for therapeutic efficacy.
Keywords
Introduction
The peripheral nervous system has an inherent, albeit limited, capacity to regenerate. Traumatic peripheral nerve injuries (PNIs) often result in long-term sensory and motor impairments, as well as chronic pain, which significantly reduce the patient’s quality of life despite advances in microsurgical repair techniques.1 -3 Autologous nerve grafting provides reliable treatment outcomes for severe PNIs. Such grafts contain a rich variety of cellular elements, including Schwann cells (SCs) and extracellular matrix (ECM) components of native tissues that are essential for nerve regeneration. 4 However, this procedure is limited by donor site morbidity 5 and by the restricted length of nerve defects that can be bridged using autologous nerve grafts. Therefore, cell-based therapies for peripheral nerve regeneration have been explored to replace or complement conventional treatments. 6 The aim is to improve prognosis after nerve repair surgery by transplanting supportive cells with defined regenerative functions. Research on stably generated cells with potent therapeutic effects holds promise for breakthroughs in treating PNIs.
Among the cells involved in peripheral nerve regeneration, SCs play a critical role. SCs are glial cells of the peripheral nervous system that wrap around axons to form myelin sheaths; they are normally responsible for maintaining homeostasis in the peripheral nervous system. During development, SCs pass through the stages of Schwann cell precursors (SCPs), immature non-myelinating SCs, and mature myelinating SCs. 7 After nerve injury, endogenous SCs release cytokines, neurotrophic factors, and ECM components. Together, these create a microenvironment suitable for nerve regeneration by facilitating the clearance of damaged axons through activation of macrophages (a process known as Wallerian degeneration), guiding regenerating axons, and promoting remyelination.8,9 Previous animal studies using rat models of nerve injury have shown that the addition of autologous SCs to acellular nerve grafts improves nerve regeneration. 10 Nevertheless, in clinical practice, treatment of PNIs with autologous SCs carries the risk of neurological deficits at the donor site during cell harvesting. Moreover, it is challenging to isolate or expand sufficient numbers of SCs from autologous sources without the influence of cellular aging, which limits their therapeutic potential. 4
Human induced pluripotent stem cells (iPSCs) are a promising cell source for creating SCs. iPSCs are stem cells that can be created from somatic cells through reprograming; they have unlimited self-renewal capacity and the ability to differentiate into multiple cell types.11,12 They offer potential for regenerative medicine in various fields13,14 and have been shown to differentiate into both SCPs and SCs. 15 Stable methods have been established to generate SCPs from iPSCs and to further differentiate them effectively into mature SCs. 16 In that study, a cell population in which more than 90% of cells were SOX10- and S100β-positive was obtained and was considered to represent a highly enriched SCP population. These cells expanded at a defined seeding density with a doubling time of approximately 26 hours and could be cryopreserved. Subsequently, these SCPs were differentiated into early SCs, which express immature markers with weak expression of neurotrophic factors. In contrast, mature SCs, generated by culturing immature SCs in medium containing dibutyryl (db)-cAMP, exhibit strong expression of neurotrophic factors and myelin basic protein (MBP), the main structural protein of myelin. 16 Compared with protocols requiring prolonged coculture with rat dorsal root ganglia (DRG) neurons or extended differentiation periods,15,17 this approach is conceptually advantageous in that it allows relatively rapid maturation of SCs for in vivo applications.
However, the in vivo therapeutic effects of iPSC-derived SCs on peripheral nerve regeneration remain to be fully elucidated. In particular, it is unclear whether their efficacy differs depending on the stage of differentiation. Therefore, in this study, we investigated the therapeutic effects of iPSC-derived immature and mature SCs transplanted into the epineurium of the sciatic nerve in a rat unilateral crush injury model. In addition to pathological and morphometric analyses of regenerated nerves, we evaluated motor and sensory function and examined muscles innervated by the sciatic nerve. To explore the mechanisms underlying regeneration, we also performed quantitative reverse transcription–polymerase chain reaction (RT-qPCR) of SCs for genes related to nerve repair.
Materials and Methods
Experimental Design
A preliminary experiment was conducted in one Lewis male rat (12 weeks old, weighing approximately 300 g; CLEA, Tokyo, Japan) to investigate the localization of human cells (SCPs, 1.0 × 106 cells) within the sciatic nerve immediately after transplantation by injection. In the main experiment, 18 F344 rnu/rnu male rats with immune deficiency (6 weeks old, weighing approximately 100-150 g; CLEA) were used. The rats were randomly divided into 3 groups: group A (administered immature SCs, 2.5 × 105 cells, n = 6), group B (administered mature SCs, 2.5 × 105 cells, n = 6), and the control group (administered Dulbecco’s phosphate-buffered saline: D-PBS, n = 6). Random numbers for group allocation were generated using the standard = RAND() function in Microsoft Excel. The number of transplanted cells (2.5 × 105) was based on previous studies demonstrating effective nerve regeneration in defect models supplemented with autologous SCs.18,19
Functional recovery was assessed using pinprick and toe-spread tests, as well as electrophysiological studies on postoperative days 7, 14, 21, and 28. On day 28, kinematic analysis was additionally performed. After completion of all functional assessments, regenerated sciatic nerves and tibialis anterior muscles were harvested for histological, morphometric, and Western blot analyses. The overall experimental workflow is illustrated in Figure 1.

Experimental design. Schematic overview of the preliminary and main experiments. In the preliminary experiment (n = 1), human cell localization was verified immediately after transplantation. In the main experiment (n = 6 per group), rats underwent functional evaluations, including pinprick, toe-spread, electrophysiological, and kinematic analyses, following sciatic nerve crush injury and transplantation of SCs or PBS injection.
A small sample size was selected because the therapeutic effects of iPSC-derived mature and immature SCs could not be estimated in advance, and the primary goal was to gather basic evidence for further research. Accordingly, no a priori power analysis was performed. Rats in all groups were housed in separate cages in pathogen-free rooms with ad libitum access to food and water and were sacrificed under general anesthesia with 5% isoflurane inhalation on postoperative day 28.
Differentiation of SCs From Human iPSCs
The human iPSC-derived immature and mature SCs used in this experiment were generated using the method of Tooi et al from the same healthy human iPSC line as the human peripheral blood-derived iPSCs previously reported. 16 Informed consent was obtained from all blood donors and the donations were made on a voluntary basis. Differentiated cells were confirmed to express SC marker proteins (S100β, MBP), as described by Tooi et al. 16 A suspension of SCPs was prepared at 2.0 × 106 cells per 0.1 mL of D-PBS (−). A suspension of the db-cAMP–treated mature SCs or untreated immature SCs was prepared at 5 × 105 cells per 0.1 mL of D-PBS (−).
Surgical Procedure
All animal procedures were performed in compliance with the guidelines of Kyoto University Animal Experimentation Committee, the ethics committee of Kyoto University Graduate School of Medicine. Surgical procedures were conducted under general anesthesia with isoflurane inhalation. With the rat in the prone position, a longitudinal skin incision was made on the right thigh, posterior to the greater trochanter. The gluteal muscle was split to expose the right sciatic nerve. A 2 mm section of the nerve posterior to the gluteal tuberosity was crushed for 10 seconds using a needle holder (No. 12501–13, Fine Science Tools, Inc., Vancouver, Canada). Then, 50 µL of a suspension of the cells or D-PBS was injected into the epineurium from the proximal side of the crush site using a 27-gauge needle (Terumo Needle, Terumo, Tokyo, Japan) and 1-mL syringe (Terumo) under a microscope. The proximal end of the crush site was marked at the epineurium with a 9-0 nylon suture and the wound was closed with 4-0 nylon sutures.
Pinprick and Toe-Spread Tests
The pinprick test was used to evaluate sensory recovery. The right hind limb of the rats was pinched from the toe to the heel with standardized forceps and the rat’s withdrawal responses to the stimulus was assessed. The pinprick test was graded from 0 to 3 as follows: grade 0, no response to the stimulus; grade 1, withdrawal response to the stimulus of the heel; grade 2, withdrawal response to the stimulus of the dorsal side of the foot; grade 3, withdrawal response to stimulus of the toe. Grade 0 indicates the poorest sensory recovery and grade 3 indicates the best sensory recovery.
The toe-spread test was used to evaluate motor recovery. Rats have a habit of extending and abducting their toes when lifted by grasping their tails; toe movements of the rats in this study were visually evaluated. Toe-spread was graded from 0 to 3 as follows: grade 0, no toe movement; grade 1, any sign of toe movement; grade 2, toe abduction; grade 3, toe abduction with extension. Grade 0 indicates the poorest motor recovery and grade 3 indicates the best motor recovery.
Electrophysiological Study
Electrophysiological studies were performed on both the injured and contralateral uninjured sciatic nerves to assess functional recovery. The compound motor action potential (CMAP) of the pedal adductor muscles of the rats was measured and the motor nerve conduction velocity (MNCV) of the sciatic nerves was calculated. The rats were placed in a prone position under general anesthesia and a needle electromyography (EMG) device (Neuropack S1, MEB-9404, Nihon Kohden, Tokyo, Japan) was used. Stimulation electrodes were inserted percutaneously behind the greater trochanter (S1) and into the popliteal fossa (S2) to stimulate the sciatic nerves, which are the proximal and distal sites of the nerve crush injury in the right hind limb, respectively. A pair of electrodes was inserted into the pedal adductor muscle to record EMGs. The CMAP (mV) was obtained by measuring the height of the EMG waveform evoked by stimulation of the S1 point. The MNCV (m/s) was calculated from the distance (mm) between the S1 and S2 points and the difference in latency (ms) between 2 EMG waveforms evoked by stimulation of the S1 and S2 points.
Kinematic Analysis
Kinematic analysis of the right hind limb of the rats walking on a treadmill using a 3D motion capture system (Kinema Tracer System, Kissei Comtec, Nagano, Japan) was performed as in our previous reports.20 -22 The rats were marked with colored hemispherical plastic markers on the posterior superior iliac spine, greater trochanter, knee joint, ankle joint, and fifth metatarsophalangeal (MTP) joint, and with acrylic resin ink on the toe. Then, each rat’s treadmill gait was filmed and the positions of these 6 landmarks were tracked. The angle of attack (AoA), which is the angle formed by the 3 landmarks of the ankle joint, the fifth MTP joint, and the toe just before the foot touches the ground at the end of the swing phase, was evaluated. The average angle was calculated for 8 to 10 consecutive steps on the treadmill. A smaller AoA suggests greater functional recovery of toe extension.
Western Blotting of Tibialis Anterior Muscles
To evaluate the quality of the muscles innervated by the sciatic nerve, the right tibialis anterior muscles of the rats were harvested on postoperative day 28, immediately after sacrifice, and subjected to western blotting. Tissue fragments of the tibialis anterior muscle were lysed in RIPA buffer (16488-34; Nacalai Tesque Inc., Kyoto, Japan) containing 1× Halt Protease Inhibitor Cocktail (1862209, Thermo Fisher Scientific, Waltham, MA, USA). The lysates were centrifuged at 15 000× g for 10 minutes at 4°C, and 10 μg of protein per sample was separated on sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE), with samples run on 2 separate gels, and then transferred to PVDF membranes (1704274; Bio-Rad Laboratories, Hercules, CA, USA) using Trans-Blot Turbo Transfer System (1704150J1; Bio-Rad Laboratories). The membranes were incubated in EveryBlot Blocking Buffer (12010020; Bio-Rad Laboratories) for 5 minutes and blotted with primary antibodies overnight at 4°C. The primary antibodies used were anti-fast myosin skeletal heavy chain (MYH1) antibody (ab91506, 1:500; Abcam, Cambridge, UK), anti-nicotinic acetylcholine receptor epsilon (ACHRE) antibody (sc-376747, 1:500; Santa Cruz Biotechnology, Dallas, TX, USA) and anti-GAPDH antibody (c-32233, 1:20 000; Santa Cruz Biotechnology). Following 5 ×5 minutes washes, the membranes were incubated for 1 hour with StarBright Blue 700 goat anti-mouse IgG (12004158, 1:5000; Bio-Rad Laboratories) and CF770 goat anti-rabbit IgG (20078, 1:5000; Biotium, Fremont, CA, USA). The membranes were washed as described above, and the fluorescent signals were visualized using the ChemiDoc Touch MP Imaging System (17001402JA; Bio-Rad Laboratories), and fluorescence intensities were analyzed by using Image Lab software (Bio-Rad Laboratories). Protein expression levels of MYH1 and ACHRE were normalized to GAPDH as an internal control.
Morphometric Analysis
To evaluate both the quality and quantity of the regenerated axons, morphometric analysis was performed. A 15 mm section of the right sciatic nerve containing the crush site was harvested on postoperative day 28, immediately after sacrifice. The sample was cut 5 mm distal to the marked 9-0 suture, and the distal sample was used for myelinated fiber density measurement and electron microscopic evaluation. The sample was fixed with 1% glutaraldehyde and 1.44% paraformaldehyde and postfixed with 1% osmic acid and embedded in epoxy resin. The semi-thin transverse sections were stained with toluidine blue solution, and one full nerve cross-section per rat was examined using a light microscope at a magnification of 400× (BZ-X700; Keyence, Osaka, Japan). A rectangular area centered on the neural area was excised such that its area was maximal in the neural area and all of the myelinated fibers within it were counted using ImageJ (National Institutes of Health, Bethesda, MD). The number was divided by the analyzed area to calculate myelinated fiber density (fibers/mm2). Each animal was treated as the experimental unit for statistical analysis. An example of the analyzed area is shown in Supplemental Figure S1.
Additionally, using the same samples, 3 parameters of the regenerated axons (myelinated axon diameter, myelin sheath thickness, and G-ratio) were examined, as reported in our previous studies.20 -23 Ultra-thin transverse sections (60-80 nm), with one full nerve cross-section per rat, were stained with lead citrate and uranyl acetate and examined using a transmission electron microscope (JEM-1400Flash; JEOL, Tokyo, Japan). Seven randomly selected visual fields at 2000× magnification for each cross-section were analyzed. All myelinated axons within each field were included in the analysis. The shortest myelinated axon diameter (a) and bare axon diameter (b) were measured using ImageJ (National Institutes of Health). From these 2 measured values, myelin sheath thickness ([a ‒ b]/2) and G-ratio (b/a) were calculated in each axon. The mean values of the 3 parameters were calculated in each rat.
Evaluation of Viability and Localization of Human-Derived Cells by Immunostaining
Immunostaining with the human nucleus-specific Ku80 antibody was performed on specimens of the right sciatic nerve from one Lewis rat in the preliminary experiment and from 12 F344 rats from groups A and B. In the preliminary experiment, immediately after injection of SCPs, a 15 mm section of the right sciatic nerve including the crush site was harvested. In groups A and B, the proximal side of the 2-part sciatic nerve samples was used for immunostaining.
The samples were immersion fixed in 4% paraformaldehyde solution, paraffin-embedded, and then sliced using a microtome at 4 μm thickness. The tissue sections at the distal side and center of the crush site were deparaffinized and endogenous peroxidase activity was inhibited with 0.3% hydrogen peroxide. Subsequently, sections were incubated overnight at 4°C with Ku80 antibody (Cell Signaling Technology, Danvers, MA, USA) diluted at 1:200. After washing with PBS, the tissue sections were incubated for 1 h at room temperature with anti-HRP diluted at 1:500. After washing with PBS, the tissue sections were treated for 5 minutes at room temperature with DAB solution using Peroxidase Stain DAB Kit (Nacalai Tesque, Inc.). After washing with distilled water, the tissue sections were counterstained with hematoxylin and mounted with PathoMount (Fujifilm Wako Pure Chemical Corp., Osaka, Japan).
The localization of the SCPs inside of the nerve immediately after administration was examined in Lewis rat samples, and the viability of the SCs at 28 days after administration was examined in F344 rat samples by observation under a light microscope.
RT-qPCR
Total RNA was extracted from the db-cAMP–treated SCs and the untreated SCs using Sepasol-RNA Ⅰ Super G (09379-97; Nacalai Tesque) and reverse-transcribed using ReverTra Ace qPCR RT Master Mix with gDNA Remover (FSQ-301; Toyobo, Osaka, Japan) according to the manufacturer’s protocol. Real-time PCR was carried out using TB Green Premix Ex Taq (RR420A; TaKaRa Bio, Kusatsu, Japan) and StepOnePlus Real-Time PCR System (Applied Biosystems, Walthum, MA, USA). The primer list is shown in Supplemental Table 1. The expression levels of each gene were normalized to that of GAPDH.
Pathological Analysis
In groups A and B, the proximal side of the 2-part sciatic nerve samples was used for pathological evaluation. Transverse sections prepared 5 mm distal to the marked 9-0 suture placed at the proximal end of the 2 mm crush site during the initial surgery were stained with hematoxylin and eosin (H&E), and one full nerve cross-section per rat was examined under a light microscope at a magnification of 400× (BZ-X700; Keyence). All enlarged cells with foamy and vacuolated cytoplasm (foamy cells) infiltrating between SCs within the nerve cross-section were counted for each section using ImageJ (National Institutes of Health). These foamy cells are considered macrophages associated with clearance of myelin and axonal debris after PNIs.
Statistical Analyses
All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). Specifically, it is a modified version of R commander designed to add statistical functions frequently used in biostatistics. 24 Data are presented as mean and standard deviations (SD), except in figures, where data are shown as mean and standard error of the mean (SEM). One-way analysis of variance (ANOVA) was used to compare data between the 3 groups. This approach was applied given the balanced group sizes and the robustness of ANOVA to moderate deviations from normality. When a significant difference was found, a post hoc test was performed using the Tukey-Kramer test. The t-test was used to compare data between groups A and B. For ordinal data, such as pinprick test and toe-spread test grades, the Kruskal-Wallis test was used. No post hoc tests were performed because no significant differences were observed. Values were considered statistically significant at P < .05 for all data.
Results
Transplantation of SCs
Representative phase-contrast images of the SCs prepared for transplantation are shown in Figure 2A and B. The db-cAMP–untreated immature SCs exhibited an elongated spindle-like morphology (Figure 2A), whereas the db-cAMP–treated mature SCs showed a tadpole-like morphology (Figure 2B). Representative intraoperative photographs from the main experiment are shown in Figure 2C and D. Figure 2C shows the right sciatic nerve immediately after induction of a crush injury. Immediately after intraneural injection of the cell suspension, localized bulging of the epineurium was observed at the crush site (Figure 2D).

Induction of a crush injury and cell transplantation. (A) A representative phase-contrast image of db-cAMP–untreated immature SCs. (B) A representative phase-contrast image of db-cAMP–treated mature SCs. (C) Right sciatic nerve immediately after induction of a crush injury in the main experiment. (D) Right sciatic nerve immediately after injection in the main experiment, showing localized bulging of the epineurium at the crush site. (E-H) Transverse sections of the right sciatic nerve harvested immediately after transplantation of SCPs in the preliminary experiment. Transplanted SCPs were identified by immunostaining with a human nucleus-specific Ku80 antibody (deep brown). (E and F) SCPs diffusely distributed between nerve bundles within the epineurium on the distal side of the crush site. (G and H) In the central region of the crush site, partial disruption of the perineurium (red arrows) and endoneurium (yellow arrows) was observed, with some SCPs located between axons. Scale bar: 200 µm (A, B and E); 50 µm (F); 100 µm (G and H)
In the preliminary experiment using a Lewis rat, transverse sections of the sciatic nerve harvested immediately after transplantation of SCPs demonstrated that the transplanted cells were successfully administered within the epineurium and had diffused between nerve bundles from the proximal to the distal side of the crush site (Figure 2E and F). In the central region of the crush site, partial disruption of the perineurium and endoneurium was observed, and some transplanted SCPs were found located between axons (Figure 2G and H).
Functional Evaluation of Regenerated Nerves
Rats in all groups showed gradual improvement in sensory and motor test grades. By postoperative day 28, all rats had recovered to grade 3 in the pinprick test, and most rats in all groups reached grade 2 or higher in the toe-spread test. Statistical analysis using the Kruskal-Wallis test revealed no significant differences among the 3 groups at any time point. The transition of grades over 28 days is shown in Figure 3A.

Functional recovery of the sciatic nerve in postoperative 28 days. (A) Transition of grades of pinprick and toe-spread tests. Median grades are shown as line graphs. All groups showed improvement over time, with no significant differences among the 3 groups at any time point (Kruskal-Wallis test). (B) Electrophysiological study of the right sciatic nerve detected significant electromyogram (EMG) waveforms of the pedal adductor muscle only within group B. (−): No significant waveform; (+): significant waveforms detected. Representative EMG findings at day 28 in each group are shown. Scale bar: 200 µV. (C) Results of kinematic analysis, schematic diagram illustrating an angle of attack (AoA) and representative photographs of the AoA in each group are shown. The mean AoA was smallest in group B but there was no significant difference among the 3 groups (1-way ANOVA). Data are presented as mean ± SEM (n = 6).
Regarding the electrophysiological studies, stimulation of the contralateral (uninjured) sciatic nerve consistently elicited normal electromyogram (EMG) waveforms in the corresponding pedal adductor muscles throughout the study period. In contrast, the right sciatic nerve did not produce detectable EMG waveforms in the pedal adductor muscle in any rats in the control group or group A during the 28-day period. In group B, one rat on each of days 14, 21, and day 28 showed detectable EMG waveforms (Figure 3B). The CMAP and MNCV values obtained from rats with detectable waveforms are summarized in Supplemental Figure S2.
Kinematic analysis of the right hind limb of the rats in treadmill walking by using a 3D motion capture system was performed on postoperative day 28 (Figure 3C). The mean value of angle of attack (AoA) was 14.43 ± 9.06° in the control group, 12.73 ± 5.52° in group A, and 7.35 ± 5.04° in group B (n = 6 for each group). The mean value of AoA was the smallest in group B, indicating the greatest recovery of toe extension, but there was no significant difference between the 3 groups.
Western blotting of the right tibialis anterior muscle showed that the expression levels of ACHRE, a marker of mature neuromuscular junction (NMJ), and MYH1 were highest in rats transplanted with mature SCs (group B), followed by those in group A (Figure 4). The expression level of MYH1 was significantly higher in group B than in the control group (P < .05). The images of full-length gels and blots are shown in Supplemental Figure S3. The wet weight ratio of the tibialis anterior muscle was also measured and tended to be greater in the SC groups (Supplemental Figure S4), but the difference was not statistically significant.

Expression of the neuromuscular junction marker and the muscle marker. Western blot analysis of ACHRE and MYH1 in the right tibialis anterior muscles harvested on postoperative day 28. Protein expression levels were normalized to GAPDH and expressed relative to the control group. MYH1 expression was significantly higher in rats transplanted with mature SCs (group B) than in the control group, whereas no significant differences were observed for ACHRE among groups. Data are presented as mean ± SEM (n = 6). Statistical analyses were performed using 1-way ANOVA followed by the Tukey-Kramer post hoc test.
Overall, no significant differences were observed among the 3 groups in the behavioral assessments within the 28-day observation period. In contrast, electrophysiological activity was detected at earlier postoperative time points in a limited number of rats in group B. In addition, MYH1 expression in the tibialis anterior muscle was significantly higher in group B than in the control group, whereas no significant differences were observed in ACHRE expression among the groups.
Morphometric Analyses of Regenerated Nerves
Toluidine blue–stained transverse sections on the distal side of the crush site were evaluated using light microscopy (Figure 5A and B). To calculate myelinated fiber density (fibers/mm2), an average neural area of 161 808 ± 40 122 µm2 of the semi-thin transverse nerve section was analyzed for each rat. The mean value of myelinated fiber density was 10 105 ± 1673 in the control group, 10 210 ± 1366 in group A, and 12 602 ± 1419 in group B (n = 6). Myelinated fiber density was the highest in group B and significantly higher in group B than in the control group (P < .05).
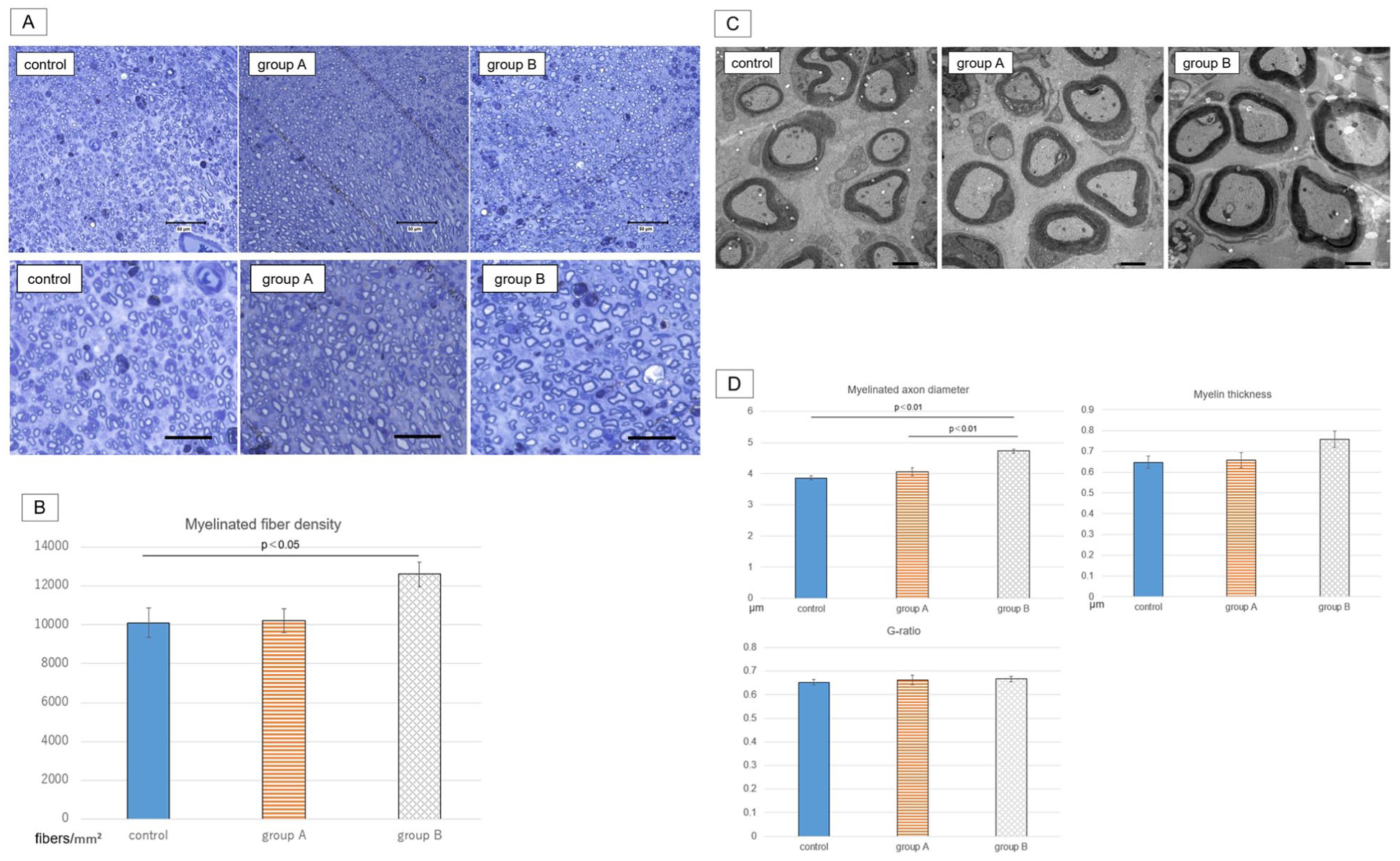
Morphometric analysis. (A) Representative images of sections stained with toluidine blue. The upper row shows low-magnification images with a scale bar of 50 μm, and the lower row shows high-magnification images with a scale bar of 25 μm. (B) Myelinated fiber density. The mean density was highest in group B and significantly higher than in the control group. (C) Representative images of ultra-thin sections observed with transmission electron microscopy. Scale bar: 2 μm. (D) Myelinated axon diameter, myelin thickness, and G-ratio. The mean values of myelinated axon diameter and myelin thickness were highest in group B, and the mean myelinated axon diameter was significantly higher in group B than in the other groups. The G-ratio did not differ significantly among the groups. Data are presented as mean ± SEM (n = 6). Statistical analyses were performed using 1-way ANOVA followed by the Tukey-Kramer post hoc test.
Ultra-thin transverse sections of the same samples were examined using a transmission electron microscope (Figure 5C and D). The mean diameter of myelinated axons was 3.86 ± 0.15 µm in the control group, 4.06 ± 0.28 µm in group A, and 4.73 ± 0.25 µm in group B (n = 6), and was significantly greater in group B than in the other groups (P < .01). The mean myelin thickness was 0.64 ± 0.06 µm in the control group, 0.66 ± 0.08 µm in group A, and 0.76 ± 0.09 µm in group B (n = 6). The G-ratio was 0.653 ± 0.032 in the control group, 0.663 ± 0.054 in group A, and 0.667 ± 0.033 in group B (n = 6).
Thus, morphometric analyses demonstrated increased myelinated fiber density and larger myelinated axon diameter in group B, with no clear differences in the G-ratio among the groups.
Evaluation of Viability of Transplanted SCs
In all of the sciatic nerve sections harvested from F344 rnu/rnu rats in groups A and B on postoperative day 28, no cells stained with the human nucleus-specific Ku80 antibody were identified. These findings indicate that transplanted human SCs were no longer present in the sciatic nerve at this time point.
Expression Analysis of Genes Related to Nerve Regeneration by RT-qPCR
To elucidate the molecular cytological activity of the SCs, we analyzed expression levels of beta nerve growth factor (bNGF), C-C motif chemokine ligand 2 (CCL2), and laminin alpha2 subunit (LAMA2) in immature and mature SCs (Figure 6). bNGF is a typical neurotrophic factor and CCL2 is a chemokine released by SCs to recruit macrophages to the site of nerve regeneration. 25 LAMA2 is a component of the basal lamina and is associated with myelination by SCs. 26 Mature SCs tended to show higher expression of bNGF, CCL2, and LAMA2 than immature SCs, although no statistically significant differences were detected. In addition, a single enzyme-linked immunosorbent assay (ELISA) indicated increased CCL2 protein levels in the culture supernatant of mature SCs (Supplemental Figure S5), consistent with the transcriptional tendency of CCL2.

Expression levels of genes related to nerve regeneration. Quantitative PCR analysis of the nerve regeneration–related genes bNGF, CCL2, and LAMA2. The ratio of each mRNA level is normalized to that of GAPDH, and data are presented as mean ± SEM (n = 3 independent experiments). Levels of bNGF (encoding a neurotrophic factor), CCL2 (encoding a chemokine for macrophage), and LAMA2 (encoding an extracellular matrix protein) were numerically higher in mature SCs (cells of group B) than in immature SCs (cells of group A); however, no statistically significant differences were detected between groups (1-way ANOVA).
Pathological Analysis of Regenerated Sciatic Nerves
To investigate the mechanism by which the transplanted SCs influence nerve regeneration, we conducted a pathological evaluation of sciatic nerves harvested on postoperative day 28 from groups A and B (Figure 7). H&E-stained transverse sections from the distal side of the crush site showed significantly greater infiltration of foamy cells in group A than in group B (P < .01). The mean number of foamy cells was 72.2 ± 36.1 in group A and 19.5 ± 3.9 in group B (n = 6). Consistent with previous reports,27,28 these foamy cells are interpreted as macrophages associated with clearance of myelin and axonal debris following PNIs.

Foamy cells in the sciatic nerves. (A) Representative images of hematoxylin and eosin (HE)-stained transverse sections of the right sciatic nerve from groups A and B. Yellow arrows indicate the typical appearance of foamy cells. Infiltration of enlarged cells with foamy and vacuolated cytoplasm was observed among SCs. Scale bar: 100 µm. (B) HE-stained sciatic nerve sections showed significantly more foamy cell infiltration in group A than in group B (t-test). Data are presented as mean ± SEM (n = 6).
Discussion
In recent years, efficient protocols to generate iPSC-derived SCs via an SCP stage have been established.15,16,29,30 However, only a few studies have reported the therapeutic effects of iPSC-derived SCs in vivo, and no studies have evaluated effects on nerve regeneration from a multifaceted perspective, both histologically and functionally. In addition, it remains unclear whether the differentiation stage of iPSC-derived SCs influences their therapeutic efficacy. Therefore, when developing SC-based therapies, it is essential to determine which stage of differentiation provides the greatest regenerative benefit.
The 2 main objectives of this study were to verify the regenerative effect of iPSC-derived SCs in vivo and to compare the therapeutic efficacy of immature and mature SCs. Our results demonstrated that iPSC-derived mature SCs improved nerve regeneration in a rat sciatic nerve crush model and produced stronger therapeutic effects than immature SCs.
We used the rat sciatic nerve crush injury model, which is the most common in vivo model for PNIs because of its technical simplicity. 31 A 27-gauge needle was selected for cell administration, based on a previous study showing higher cell viability compared with finer needles. 10 This needle size is also practical and commonly used in clinical settings. Furthermore, we used a standard clinical syringe, and the injection volume of 50 μL was chosen as the minimum volume that could be reproducibly delivered using this setup. This experimental model was selected to evaluate the in vivo effects of iPSC-derived SCs generated using the present differentiation protocol under clinically relevant transplantation conditions, while maintaining technical simplicity. In the preliminary experiment, partial disruption of the perineurium and endoneurium was observed at the crush site, whereas the epineurium remained intact. Some of the injected cells were found to have infiltrated around axons at the crush site. However, the volume of the injected cell suspension exceeded the tissue volume of the crush site itself, and thus the majority of the cells were likely to have diffused distally within the epineurium. Because this animal model does not retain all transplanted cells at the injury site, it inherently limits the extent to which the direct effects of SCs on regenerating axons can be evaluated. Rather, the observed therapeutic effects are likely mediated predominantly through indirect paracrine mechanisms exerted by the transplanted cells within the epineurium. This interpretation is consistent with previous studies, including those by Lopez-Verrilli et al and Zhang et al, which demonstrated paracrine mechanisms in peripheral nerve regeneration. Lopez-Verrilli et al reported that SC-derived exosomes, when injected into the sciatic nerve distal to the crush site in a rat sciatic nerve crush injury model, enhanced axonal regeneration. 32 Zhang et al used a scaffold seeded with neural progenitor-like cells derived from human gingiva-derived mesenchymal stem cells, which was wrapped around the injury site in a rat sciatic nerve crush injury model, and reported improved remyelination. 33 While the experimental approaches differ, these studies support the idea that transplanted cells or their secreted factors can facilitate nerve regeneration through indirect paracrine effects.
To quantitatively assess limb function, kinematic analysis was performed to measure toe extension lag during treadmill walking. Although the mean toe extension lag was the lowest in group B, no significant differences were detected among the groups. This suggests that evaluation at different time points may be necessary to detect potential functional differences. Furthermore, the absence of footprint analysis, 34 which might have provided a more sensitive assessment of functional recovery, should be noted as another limitation of this study.
In the electrophysiological study, only rats in group B (3 out of 6) exhibited EMG waveforms in the pedal adductor muscles within 28 days after surgery. This finding suggests a tendency toward earlier reinnervation of distal muscles innervated by the sciatic nerve in some animals transplanted with mature SCs. Consistent with this observation, Western blot analysis of the tibialis anterior muscle showed higher MYH1 expression in group B compared with the control group, while no significant differences were observed in ACHRE expression among groups. MYH1 is known to decrease in association with muscle atrophy,35,36 and the increased MYH1 expression in group B may therefore reflect reduced denervation-associated muscle changes in this group. ACHRE was included as a marker related to neuromuscular junction status, as its expression has been reported to decrease following denervation. 37 However, the lack of significant differences in ACHRE expression among groups indicates that protein-level changes at the neuromuscular junction could not be conclusively demonstrated in the present study. Importantly, behavioral assessments did not reveal significant differences among the groups within the 28-day observation period. In addition, electrophysiological activity was detected only in a limited number of animals, and protein-level analyses were restricted to a small number of markers. Therefore, findings related to functional recovery should be interpreted with caution.
The apparent discrepancy between findings from the pinprick/toe-spread tests and the electrophysiological findings likely reflects differences in what each assessment captures. The pinprick test evaluates sensory recovery, and the toe-spread test reflects coordinated movement involving multiple foot muscles. CMAP recordings were obtained from the pedal adductor muscle, which is one of the most distally located muscles innervated by the sciatic nerve. As a result, recovery detected by the pinprick or toe-spread tests may occur before electrophysiological activity becomes detectable in the pedal adductor muscle.
Morphometric evaluation of the sciatic nerve showed that rats in group B had the highest myelinated fiber density and greatest myelinated axon diameter, the latter being significantly greater than that of group A and the control group. In terms of axonal regeneration, these results suggest that mature SCs exerted a significantly stronger therapeutic effect than immature SCs. In this study, no significant differences in the G-ratio were observed among the 3 groups. The G-ratio values seem to be higher than those estimated from a previous study on normal rat sciatic nerves, 38 suggesting that remyelination was still in progress in all groups at the time of evaluation.
RT-qPCR in vitro revealed that both mature and immature SCs expressed bNGF, CCL2, and LAMA2, with all 3 genes expressed at higher levels in mature SCs. LAMA2 encodes laminin, an essential ECM component that supports SC attachment and promotes neurite extension.26,39,40 CCL2 is a chemokine primarily released by SCs that plays an important role in the recruitment of macrophages during Wallerian degeneration.8,25
In the pathological analysis, a greater number of foamy cells remained within the nerve tissue at postoperative day 28 in group A than in group B, suggesting prolonged persistence of macrophage-associated changes in group A. This observation raises the possibility that Wallerian degeneration progressed earlier or resolved more rapidly in group B. Given that CCL2 expression was higher in mature SCs in vitro, differences in CCL2-related signaling may be involved in this process. However, because foamy cell infiltration was evaluated at a single postoperative time point and in vivo CCL2 levels were not measured, it is not possible to conclude that Wallerian degeneration was accelerated by transplantation of mature SCs, and further analyses will be required to clarify this relationship.
In our analysis of the viability of transplanted SCs, engraftment of the SCs in vivo could not be confirmed at 28 days after surgery, even though immunodeficient rats were used to minimize the host’s immune response. Kim et al transplanted human iPSC-derived SCs, matured by coculture with rat DRGs, into a mouse sciatic nerve defect model using Matrigel and reported that the GFP-tagged SCs had engrafted 8 weeks after transplantation. 15 In addition to engraftment, they also reported functional improvement assessed by the sciatic functional index (SFI); however, differences in injury models and cell delivery strategies limit direct comparison with the present study. In that model, the transplanted SCs were positioned within a nerve gap and maintained direct and sustained contact with regenerating axons. In contrast, in our crush injury model, only a fraction of the transplanted SCs were in direct contact with regenerating axons at the crush site, while most were likely distributed more distally within the epineurium. We therefore hypothesize that this limited direct association between transplanted SCs and regenerating axons contributed to the lack of detectable engraftment at 28 days.
Within 48 hours of PNIs, chemokines and neurotrophic factors rapidly increase around the lesion, accompanied by aggregation of macrophages, host SCs, and fibroblasts. 9 Infiltration of macrophages involved in Wallerian degeneration into the lesion peaks approximately 7 days after injury. 27 These temporal dynamics suggest that transplanted SCs may exert their effects primarily through paracrine mechanisms during the early phase of nerve regeneration, after which they likely decline and become undetectable.
Previous studies have examined how the differentiation stage of Schwann-lineage cells influences their regenerative effects in vivo. Endo et al transplanted syngeneic SCs derived from adult Lewis rats into a rat sciatic nerve crush model, comparing developing and mature phenotypes. They found that mature SCs significantly enhanced axonal regeneration, whereas developing SCs did not. ELISA of cultured cells revealed higher secretion of NGF and brain-derived neurotrophic factor (BDNF) by mature SCs, and transcriptomic profiling showed upregulation of inflammation-related genes, suggesting that enhanced regeneration was mediated by paracrine neurotrophic and immunomodulatory mechanisms. 41
In our study, human iPSC-derived mature SCs also promoted regeneration more effectively than immature SCs in an immunodeficient rat sciatic nerve crush model. This concordance suggests that the enhanced regenerative effects of mature SCs are associated with their greater paracrine functional maturity. Differences in pathological findings, including the persistence of foamy cells, between the mature and immature SC groups may likewise reflect variations in inflammatory modulation, as previously reported. 41
In contrast, Huang et al investigated a rat sciatic nerve defect model in which human iPSC-derived neural crest stem cells (NCSCs) or NCSC-derived SCs were seeded into a biodegradable conduit. They reported that NCSCs achieved greater functional recovery, higher cell survival at 2 weeks, and elevated NGF and BDNF protein levels within the conduit compared with NCSC-derived SCs. Most surviving NCSCs expressed S100β, indicating in vivo differentiation toward the Schwann lineage. 42 Given the differences in injury type (defect vs crush), delivery method (conduit vs intraneural injection), and cell phenotype at transplantation, the mechanisms underlying Huang’s results likely differ from those in our study.
In our model, transplanted SCs were present within the epineurium immediately after injection but were no longer detectable at 28 days postoperatively. Cell viability at intermediate time points was not evaluated, which remains a limitation of the study.
There are several other limitations in the present study. First, the number of rats in each group was small. Kinematic analysis showed no significant differences between the groups, although toe extension was numerically greatest in group B. Western blotting of tibialis anterior muscles also showed no significant difference between groups A and B. In both analyses, considerable variability in the data may have contributed to the lack of statistical significance. By increasing the sample size, more reliable results regarding the functional recovery of the sciatic nerve may be obtained. Second, the therapeutic effect was examined only in a nerve crush injury model, in which 50 µL of the cell suspension was injected. To further validate the therapeutic potential of iPSC-derived SCs, it will be necessary to evaluate their effects in more severe injury models, such as nerve defects. Moreover, direct mechanisms of action should be assessed using other experimental settings that allow closer association between transplanted cells and regenerating axons. Third, this study did not determine the optimal therapeutic cell dose and used one cell dose (2.5 × 105 cells) per transplant in the main experiment. In previous studies using nerve defect models, good nerve regeneration could be achieved by adding 106 autologous SCs to each acellular nerve graft.18,19 In the preliminary experiment, the same cell number (106 cells) was administered because the intraneural distribution of transplanted cells was unpredictable at that stage. The preliminary experiment confirmed that intraneural delivery within the epineurium could be achieved. Based on these observations, a lower cell dose was selected for the main experiments. This decision also considered that the present study employed a crush injury model, which is less severe than nerve defect models. However, graft-based nerve defect models differ substantially from the crush injury model used here, particularly with respect to injury severity and cell delivery strategy. As a result, the optimal number of transplanted cells in the current model may differ considerably from that used in this study. In addition, in the present experimental setting, some of the injected cells may have leaked outside the epineurium due to animal movement after the transplantation. Accordingly, future studies should consider alternative delivery strategies as well as multiple cell doses to more precisely evaluate dose-dependent therapeutic effects. Fourth, this study did not include a sham-operated group, which limits direct comparison with uninjured nerves and prevents evaluation of baseline nerve morphology and function. Including such a control group in future studies would help clarify the extent of true regenerative recovery. Fifth, histological assessment was based on toluidine blue and H&E staining, without immunohistochemical analysis using specific markers such as S100β or neurofilament (NF), which could have provided more detailed insights into SC distribution and axonal regeneration. In addition, this study did not include detailed histological evaluation of the exact crush site at 28 days after injury. Specifically, precise identification of the original lesion at day 28 is difficult, particularly because the crush length was very short (2 mm). Therefore, to allow consistent comparison across animals, pathological and morphometric analyses were performed at standardized distal nerve segments.
Finally, the evaluation of paracrine factor expression by mature and immature SCs prepared for the transplantation was limited to mRNA analysis. Although mature SCs tended to have higher expression levels of bNGF, CCL2, and LAMA2, no statistically significant differences were observed. Protein-level validation was not systematically performed, except for a limited ELISA assessment of CCL2, which suggested higher levels in mature SCs. CCL2 was selected for protein-level evaluation due to its known involvement in macrophage recruitment. A previous study using similarly derived SCs reported significantly higher NGF protein levels in mature SCs compared with immature SCs, as measured by ELISA. 16 In addition, only 3 genes were analyzed by RT-qPCR, and other important neurotrophic or growth factors, such as BDNF or fibroblast growth factor 2 (FGF-2), were not evaluated. Additional studies will be needed to comprehensively assess the paracrine profiles of these cells.
Conclusion
In this study, we evaluated the in vivo therapeutic efficacy of 2 iPSC-derived SC populations at different stages of differentiation using a clinically relevant transplantation method. Among the transplanted cells, dibutyryl-cAMP–treated mature SCs showed the greatest therapeutic efficacy, as evidenced by significantly improved sciatic nerve morphometric outcomes and the earlier detection of electrophysiological activity in a subset of animals, together with muscle-related molecular findings indicative of reduced denervation-associated changes. Further research is needed to understand the mechanism by which the SC transplantation affects nerve regeneration, including investigations in other experimental settings.
Supplemental Material
sj-docx-1-exn-10.1177_26331055261443172 – Supplemental material for Mature Schwann Cells Derived From Human Induced Pluripotent Stem Cells Promote Peripheral Nerve Regeneration In Vivo
Supplemental material, sj-docx-1-exn-10.1177_26331055261443172 for Mature Schwann Cells Derived From Human Induced Pluripotent Stem Cells Promote Peripheral Nerve Regeneration In Vivo by Kazuaki Fujita, Ryosuke Ikeguchi, Tomoki Aoyama, Takashi Noguchi, Koichi Yoshimoto, Daichi Sakamoto, Terunobu Iwai, Tetsuya Miyamoto, Yuta Takeuchi, Momoko Nagai-Tanima, Daiki Nohara, Norie Tooi, Koichi Igura, Takayuki Kiboku, Rio Okama, Hiroyuki Ogasawara and Shuichi Matsuda in Neuroscience Insights
Footnotes
Acknowledgements
We thank the Division of Electron Microscopic Study, Center for Anatomical Studies, Graduate School of Medicine, Kyoto University for technical assistance in electron microscopic analysis.
Ethical Considerations
All experimental protocols were approved by the Kyoto University Animal Experimentation Committee, and all animal procedures were performed in compliance with its guidelines. Studies involving human iPSCs, SCPs, and SCs were approved by the Life Ethics Review Committee of Stem Cells & Device Laboratory, Inc (EA-2023 001). All methods are reported in accordance with ARRIVE guidelines.
Consent to Participate
Informed consent was obtained in writing, and the donations were made voluntarily.
Consent for Publication
This study does not include any data from individual persons; therefore, consent for publication was not required.
Author Contributions
K.F., R.I., T.A., and S.M. designed the experiments. N.T. and K.I. generated SCPs and SCs from iPSCs and performed immunostaining on nerve sections. T.K., R.O., M.N., and D.N. performed western blotting. T.K. and R.O. performed RT-qPCR and prepared Figures 3 and ![]() . H.O. supported pathological evaluation. K.F. performed all other experiments. T.N., K.Y., D.S., T.I., T.M., and Y.T. supported interpretation of the data analysis. K.F., R.I., and T.A. wrote the manuscript. S.M. gave final approval of the manuscript.
. H.O. supported pathological evaluation. K.F. performed all other experiments. T.N., K.Y., D.S., T.I., T.M., and Y.T. supported interpretation of the data analysis. K.F., R.I., and T.A. wrote the manuscript. S.M. gave final approval of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by JSPS KAKENHI Grant Number 24K02796.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.*
Supplemental Material
Supplemental material for this article is available online.
