Abstract
Sodium, which is present in NaCl (Sodium Chloride), is a vital nutrient required for numerous physiological processes. In animals, including Drosophila, low-salt concentrations induce attraction and high-salt concentrations trigger aversive behavior. Although the analysis of low and high salt concentrations in Drosophila has been described at the cellular level, the mechanisms governing high salt consumption and taste modulation remain unclear. Our study examined the neural basis of high NaCl consumption in adult Drosophila, focusing on how dietary adaptation influences salt acceptance. Our findings suggest that prolonged exposure to high salt alters the taste sensitivity of the pharyngeal labral sense organ (LSO) neurons, promoting increased salt intake, particularly under starvation conditions. This modulation requires active LSO neurons, as genetic suppression of their activity in high-salt-fed flies prevents excessive salt consumption under starvation or dopamine-supplemented feeding. Furthermore, multiple independent taste receptor neurons and pathways within the LSO contribute to this response. Silencing any one of these LSO neuron types can prevent excessive salt intake. In conclusion, our study suggests that Drosophila undergoes dietary salt adaptation over time, revealing a key mechanism for resetting salt appetite and related neural circuits.
Introduction
Similar to sugars, animals have a natural preference for lower concentrations of salt (sodium chloride – NaCl). Sodium, a crucial and frequently limited nutrient, is essential for maintaining ionic balance, nerve signaling, fluid regulation, and cardiovascular function. Animals require sodium from external sources to sustain these physiological processes. Low sodium levels in the body trigger specific appetite signals in the brain to drive sodium consumption. Recent research identifying a small cluster of neurons in the mouse hindbrain that regulates sodium appetite points to the involvement of chemosensory signals in this regulation. 1
Low salt concentrations in animals, including flies, elicit appetitive behaviors, while higher concentrations trigger aversion. 2 These opposing behavioral responses are mediated by distinct molecular and anatomical taste pathways. In mice, the epithelial sodium channel (ENaC) functions as a low-salt receptor.3,4 Interestingly, high salt does not activate ENaC in mice, but instead engages other taste pathways, 2 and recruits two primary aversive taste pathways by activating the sour-and bitter-taste-sensing cells. Similarly, in Drosophila, bitter-sensitive gustatory receptor neurons (GRNs) respond to high salt concentrations.5,6 In Drosophila larvae, the Pickpocket (ppk) DEG/ENaC (degenerin/epithelial sodium channel) channel family members ppk11 and ppk19 are involved in sensing both low- and high-NaCl concentrations, 7 though not in adult flies. 8 Moreover, the gene serrano (sano), along with gustatory receptor 66a (Gr66a), has been shown to be required for high salt avoidance in the terminal organs of third-instar larvae. 9
In Drosophila, low and high NaCl concentrations are detected by GRNs in I-type (intermediate) and S-type (small) sensilla located on the labellum.8,10-13 Aversion to high salt is mediated by pharyngeal Gr2a taste neurons. 14 Additionally, the Drosophila dpr locus (for defective proboscis extension response), a member of the immunoglobulin (Ig) superfamily, is also required for the aversive response to high salt. 15 The Drosophila ionotropic receptor- Ir76b detects both low and high-NaCl concentrations,8,13 contributing to a complex code for salt taste in flies. 16 Recent studies have shown that Ir56b mediates appetitive responses to salt, while Ir7c is responsible for avoiding high monovalent salt concentrations.17,18 Both Ir56b and Ir7c function with co-receptors Ir25a and Ir76b.17,18 Moreover, bitter taste neurons and a class of glutamatergic ‘high salt’ neurons expressing pickpocket23 (ppk23) have been implicated in driving high salt avoidance.6,16,19
While the cellular mechanisms of low and high salt taste detection are well-studied, those governing high salt consumption and taste modulation remain elusive. Recent functional and behavioral studies uncovered the role of distinct pharyngeal neuronal subsets in controlling avoidance responses to taste stimuli, including high salt in Drosophila. 20 Other studies identified an inhibitory mechanism that suppresses high salt intake, 21 and demonstrated that Ir60b in pharyngeal GRNs, along with co-receptors Ir25a and Ir76b, limits salt consumption. 22 Furthermore, salt sensitivity is known to increase in female flies after mating, and salt deprivation can trigger salt intake even in flies lacking appetitive salt taste.23,24 To investigate how dietary adaptation affects salt acceptance and subsequent salt taste behavior, we examined the neural basis of high NaCl consumption in adult Drosophila specifically. Wild-type flies are attracted to low NaCl concentrations and exhibit aversion to higher concentrations. However, we observed that exposing wild-type flies to a high-NaCl diet for 3 days altered their preference, increasing their acceptance of high salt under starvation. Critically, genetic suppression of labral sense organ (LSO) pharyngeal neuronal activity in high NaCl-fed flies inhibits this excessive salt intake. This adjustment is modulated by starvation or dopamine levels, which can diminish salt aversion, suggesting a critical role for these neurons in regulating dietary behavior. By identifying the involvement of various taste receptor pathways within the LSO (silencing any one of several LSO neuronal types prevents excessive salt intake), our study offers significant insights into the mechanisms of dietary influence on taste perception and behavioral responses in flies, with potential implications for broader biological processes. In conclusion, our study suggests that flies can adapt to the amount of salt ingested over several days, indicating the presence of a critical mechanism to reset the salt appetite and related neural circuits.
Results
Wild-Type Flies Show Aversion to High NaCl Concentrations
To understand how flies, respond to varying concentrations of NaCl, we first tested wild-type (CsBz) flies for their feeding preferences in a feeding assay. To examine the salt feeding behavior, batches of 20 flies per plate (10 males + 10 females) were presented with a choice between water and varying NaCl concentrations. In our 2-hours feeding assay, flies were allowed to choose between these options in an unbiased manner in the dark. Blue spots in the feeding plate contained just water and agar, while red spots provided NaCl combined with agar and water (Supplemental Figure 1A). The percentage of flies preferring NaCl (eating red) was scored based on the color of their abdomens. Flies were starved for 24 hours before performing the feeding assay. We found that wild-type flies are most attracted to 10, 50, and 100 mM NaCl concentrations, demonstrating the highest feeding preference at these concentrations (Supplemental Figure 1B). Conversely, they showed a significantly reduced preference for higher NaCl concentrations (200 and 500 mM; Supplemental Figure 1B). Our dye-swap experiment (NaCl presented in blue spots) yielded identical results and showed the same feeding preferences at 10, 50, and 100 mM NaCl concentrations (Supplemental Figure 2A) confirming that low NaCl concentrations act as an attractive cue and high NaCl concentrations elicit repulsion in flies.
When wild-type mated males and females were tested separately in feeding assays, both sexes exhibited highest feeding preference at 50 mM NaCl concentration (males at 31.44% and females at 51.67%), compared to other NaCl concentrations (Supplemental Figure 1C and D). Notably, mated female flies demonstrated a stronger feeding preference for NaCl (Supplemental Figure 1C and D and Supplemental Data File 2) than males at 10, 50, and 100 mM NaCl concentrations. This sexual dimorphism in salt appetite suggests that female flies may have a specialized requirement for sodium ions, likely to support oogenesis and the nutritional needs of their progeny. 23
We then used a different behavioral assay to test whether the response threshold to salt is modified and to investigate the external sensory responses to different concentrations of NaCl. We stimulated the tarsal taste hairs of wild-type (CsBz) flies to perform the tarsal Proboscis Extension Reflex (PER) assay (Supplemental Figure 1E and F) with a series of NaCl concentrations ranging from 10-500 mM (same concentrations used in the feeding assays). Flies were starved for 24 hours in these experiments. Consistent with our feeding results, we observed the highest proboscis extension response (~46%) in wild-type flies (mix of males and females) at 50 mM NaCl when compared with the other concentrations (Supplemental Figure 1G and Supplemental Video 1). While performing the assay, flies were closely monitored to ensure that there was no accidental contact with the labellum when the stimulus was applied to the tarsi. We observed 15 to 20% positive PER responses at 10, 100, and 200 mM (Supplemental Figure 1G and Supplemental Video 2) concentrations. Less than 10% responses were recorded at the highest concentration tested (500 mM; Supplemental Figure 1G) suggesting reduced attraction at high concentrations. Both our PER and feeding assay results suggest that flies are attracted to low concentration (50 mM) of NaCl and avoid NaCl as the concentration increases (Supplemental Figure 1F). In another set of PER experiments, we also tested sugar (100 mM sucrose) as a control stimulus at the beginning and end of the experiments to make sure flies were not losing sensitivity after testing various salt concentrations. About 85% to 95% flies showed positive PER and responded to 100 mM sucrose (Supplemental Figure 2B) in these experiments confirming that general gustatory sensitivity remained intact across the range of salt concentrations tested.
High NaCl Fed Flies Show a Decline in High Salt Aversion Under Starvation
To determine if prior long-term exposure to high NaCl alters subsequent salt feeding behavior, wild-type flies were fed a high salt diet (200 mM NaCl mixed with standard fly food and fed continuously for 3 days; Figure 1A). This duration was selected based on preliminary experiments showing that 1- and 2-days of exposure were insufficient to shift preferences (Supplemental Figure 3A and B). Following a 24-hour starvation period, flies were subjected to a two-choice feeding assay to assess their preference for varying concentrations of NaCl (Figure 1A and B). In these experiments, flies maintained on standard fly media for 3 days served as controls (Figure 1A and B). We observed a significant shift in feeding preferences only after 3 days of exposure period (Figure 1A and B) but not with 1- and 2-days of feeding (Supplemental Figure 3A and B). Flies kept on high salt media maintained a preference for higher NaCl concentrations, with a notable increase in feeding preference for 100, 200, and 500 mM NaCl (black bars in Figure 1B), while no differences in feeding were observed at lower concentrations (10 and 50 mM, Figure 1B). As we found reduced aversion toward high NaCl concentrations when wild-type (CsBz) flies were pre-exposed to a high NaCl diet in our feeding assays, we validated these behavioral results by performing spectrophotometry on flies to quantify food intake (Figure 1C and D). Interestingly, this independent measure confirmed increased consumption of high NaCl in 2 distinct wild-type fly strains (CsBz and w1118) (Figure 1C and D). Together, these results suggest that prolonged exposure (3 days) to high NaCl likely triggers an adaptive mechanism allowing flies to tolerate, prefer and consume higher NaCl concentrations.
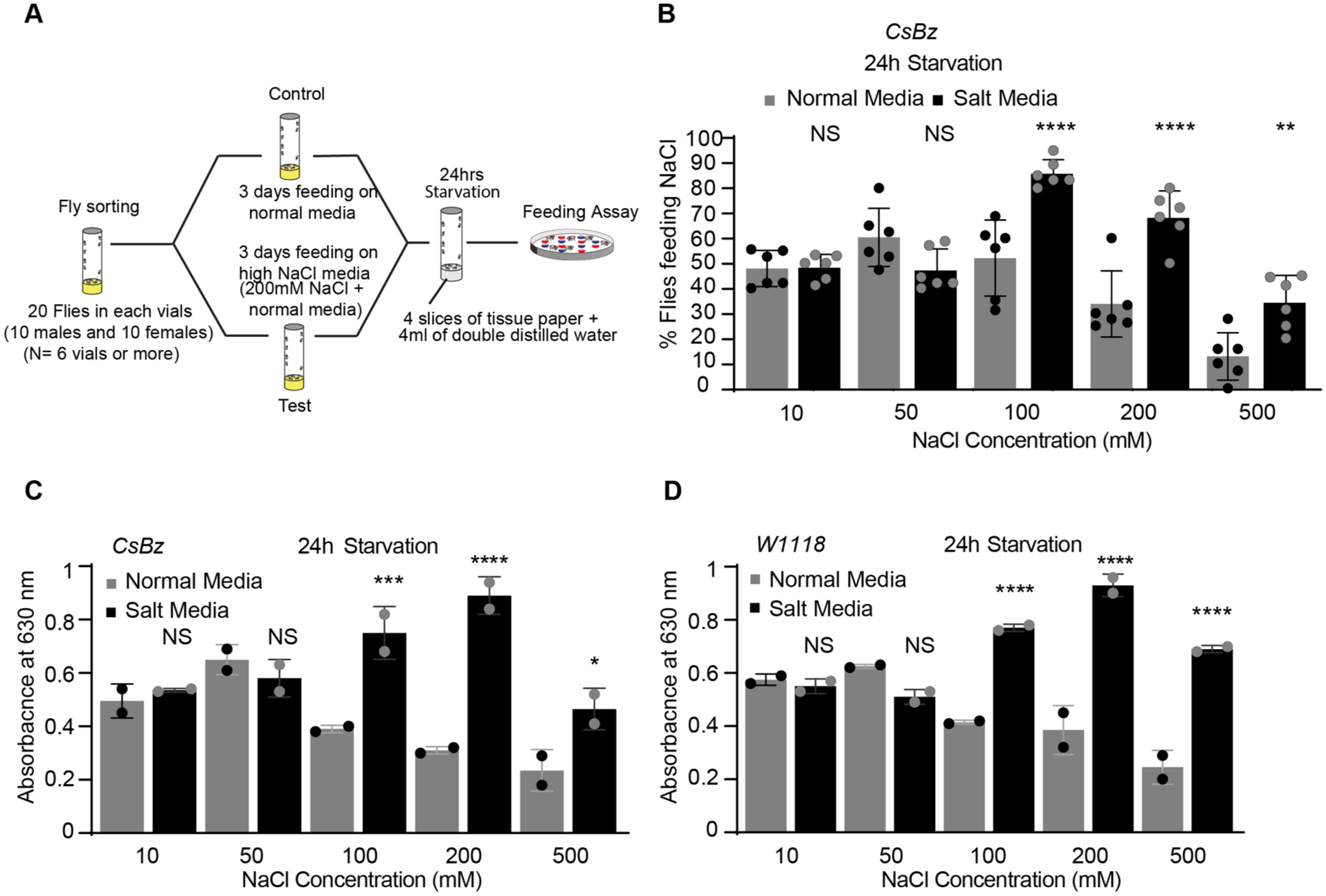
High NaCl-fed flies show reduced high salt aversion under starvation. (A) Schematic of feeding paradigm used to pre-expose wild-type flies to high-NaCl condition. (B) Dose response profile of CsBz wild-type flies showing feeding preferences for indicated concentrations of NaCl. Normal media- (3 days; gray bars) and high NaCl-fed flies (3 days; black bars; 200 mM NaCl mixed with normal fly food) used after 24 hours of starvation. N = 6 trials of 20 flies (10 males and 10 females) each concentration/condition. (C and D) Mean absorbance values for CsBz and w1118 flies kept on normal media and high-NaCl diet for 3 days and further tested for indicated concentrations of NaCl after 24 hours of starvation. Asterisks showing significant differences between flies fed on normal (gray bars) and high-NaCl media (black bars). N = 2 sets each concentration, 60 flies each set. For all graphs, error bars = SEM. Statistical analysis was performed using ANOVA Sidak’s multiple comparison test for obtaining P-values: *P < .05, **P < .005, and ***P < .0005. For all graphs, error bars = SEM and NS is not significant.
We next investigated whether the observed dietary adaptation is driven by changes in peripheral gustatory sensitivity or if it is restricted to the mechanisms governing ingestion. To address this, we performed tarsal PER assays using wild-type flies (CsBz) and tested whether chemosensory responses to NaCl at the external taste organs change following the 3-day high-salt diet exposure, and whether any such change tracks the direction of the salt-feeding phenotype we observed (Figure 1B-D). After 3 days on high-salt food, flies (normal media and high-NaCl fed) showed no significant difference in PER responses to NaCl tested at 50, 100, 200, and 500 mM concentrations under fed condition (no starvation). We observed a modest increase in PER only at 10 mM NaCl (black bars in Supplemental Figure 3C) in high-salt fed flies relative to normal media fed flies. Under 24-hour starvation period, flies maintained on normal media exhibited higher PER responses at 10, 50, and 100 mM NaCl compared to flies pre-exposed to high-salt food. No significant changes in PER were observed at 200 and 500 mM NaCl concentration (gray bars in Supplemental Figure 3D). Our NaCl PER and NaCl feeding results did not shift in the same direction after the 3-day high-salt diet eliminating the involvement of peripheral GRNs in driving the feeding phenotype. These results supported the role of internal chemosensory cells in modulating salt appetite, leading us to focus only on ingestion-based feeding behavior in subsequent experiments (Figure 1B-D).
Many recent studies have demonstrated that fly taste pathways are modulated by internal physiological states.25-30 We therefore investigated whether changes in the internal state, specifically a 24 hours starvation period, was responsible for modulating the salt feeding preferences of salt-acclimated flies. To this end, we compared the feeding preferences of flies pre-exposed to a high-NaCl diet that were either kept unstarved (Supplemental Figure 4A) or starved (Figure 1B) prior to testing. In the fed state, no significant differences were observed in feeding preference between normal media and high salt-fed flies (Supplemental Figure 4A). Furthermore, no significant differences were found in the body weights of male and female flies across dietary conditions (Supplemental Figure 4B). Even under fed conditions, flies showed the highest attraction to salt at 50 mM concentration (Supplemental Figure 4A, gray bars). The overall feeding preferences were reduced compared to starved flies (Supplemental Figure 4A, gray bars and Figure 1B) under fed conditions. Thus, our data suggest that the internal metabolic state is a critical regulator of salt-feeding decisions, with starvation acting as a necessary gate to reveal the dietary-induced shift in salt preference.
High NaCl Fed Flies Show No Alteration in Consumption of Potassium Under Starvation
Next, we evaluated whether flies on a high-NaCl diet discriminate between potassium and sodium salts and would show any alterations in feeding preferences for potassium salts as those obtained for sodium salts. To determine this, we tested their feeding preferences for various potassium chloride (KCl) concentrations following a 24-hour starvation period. Unlike the shift observed with sodium, flies fed with a high-NaCl diet showed no increased feeding preferences for KCl at any tested concentration. No significant differences were detected between the normal and high-NaCl fed groups (Supplemental Figure 4C). These data suggest that the adaptive mechanism triggered by high-NaCl exposure is cation-specific and does not alter the behavioral processing of potassium salts. This distinction highlights that the observed plasticity is a specialized response to sodium intake rather than a generalized change in mineral appetite or osmotic tolerance.
High NaCl-Fed Flies Show Alteration in Consumption of Metabolizable Sugars
Flies are capable of evaluating both the nutritional value and palatability of sugars from low to higher concentrations showing peak attraction and feeding at intermediate concentrations of sucrose. To determine whether pre-exposure to a high-NaCl diet cross-modulates sweet taste, we tested a range of sucrose concentrations (1, 5, 10, 15, 20, 25, 50, 100, and 200 mM) using both spectrophotometry and two-choice feeding assays. Following a 24-hour starvation period, we observed no significant differences in consumption or feeding preferences between flies fed on a high-NaCl or normal-media at 50, 100, and 200 mM concentrations (Supplemental Figure 5A and B) . However, when lower concentrations of sucrose (5, 10, 15, and 20 mM) were presented, high NaCl-fed flies showed increased consumption, as indicated by spectrophotometry analysis (Figure 2A), despite showing no change in feeding preferences (Figure 2B). No differences were detected at 1 mM or 25 mM sucrose concentrations between test and control flies (Figure 2A and B). These findings suggest that while a high-NaCl diet does not shift the valency of sucrose, it significantly alters the quantitative consumption of low-concentration of sucrose.

High NaCl-fed flies show consumption for selective sugars. (A) Absorbance values for different concentrations of sucrose (1, 5, 10, 15, 20, and 25 mM) between high NaCl- and normal media-fed wild-type flies (CsBz). (B) Feeding assay showing feeding preferences for different concentrations of sucrose as used in A between high NaCl- and normal media-fed wild type flies. (C) Absorbance values measured for different sugars (fructose, trehalose, galactose, D-glucose, sucralose, and L-glucose) at 100 mM concentration between high NaCl- and normal media-fed flies. (D) Feeding assay showing feeding preferences for different sugars used in C at 100 mM concentration between NaCl- and normal media-fed wild type flies. For A and C, n = 2 sets each concentration, 60 flies each set. For B and D, n = 6 to 12 trials × 20 flies each concentration. In all graphs, black bars represent high NaCl-fed flies compared with flies fed on normal media (gray bars). Flies were starved for 24 hours in all the experiments. Asterisks showing significant differences between flies fed on normal (gray bars) and high-NaCl media (black bars). Statistical analysis was performed using ANOVA Sidak’s multiple comparison test for obtaining P values: *P < .05, **P < .005, and ***P < .0005. For all graphs, error bars = SEM and NS is not significant.
We further examined a variety of sugars to determine whether the dietary adaptation to NaCl influences the intake of other sugar varieties . We observed significant increase and measurable changes in the ingestion of the nutritive, metabolizable sugars including D-fructose and D-glucose at 100mM concentration (Figure 2C). In contrast, no significant differences were detected for D-trehalose, galactose, or non-nutritive sugars such as sucralose, or L-(-) glucose at the same concentration (Figure 2C). Consistent with our sucrose data, these shifts in ingestion volume were not accompanied by changes in feeding preference, as all sugars tested at 100 mM yielded similar preference indices between groups (Figure 2D). Furthermore, no significant differences were observed in the consumption of any tested sugar at 10 mM concentration (Supplemental Figure 5C), nor was there any change in feeding responses to the bitter compound caffeine (10 mM) between flies fed normal-media or high-NaCl diet (Supplemental Figure 5D). Taken together, our data indicate that pre-exposure to high-NaCl diet specifically promotes the consumption of certain metabolizable sugars, particularly D-fructose and D-glucose at 100 mM concentrations, suggesting that high salt intake may alter metabolic homeostasis and the subsequent drive for caloric energy.
Recent studies have shown that flies can detect different categories of salts.18,21 To investigate if pre-exposure to high NaCl diet modulates flies feeding preferences for other categories of salts, we tested high NaCl-fed flies with different salts (sodium hydrogen carbonate, di-sodium hydrogen O-phosphate, magnesium chloride and potassium di-hydrogen O-phosphate) following starvation period of 24 hours. We tested 25 and 100 mM concentrations of sodium hydrogen carbonate, di-sodium hydrogen O-phosphate, as well as 10 and 100 mM concentrations of magnesium chloride and potassium di-hydrogen O-phosphate. No significant differences in feeding behavior were observed between high NaCl- and normal media-fed flies across different salt categories (Supplemental Figure 5E). These results suggest that the dietary-induced shift in preference is specific to NaCl and does not generalize to other sodium-containing compounds or divalent salts.
Role of Neuronal Activity Within Sweet Peripheral and Pharyngeal LSO Neurons in Modulating High Salt Consumption Under Starvation
Next, we sought to identify the neural substrates underlying the increased salt intake in salt-acclimated flies. We first evaluated the role of neuronal activity in peripheral sweet taste neurons, as adaptive changes in neuronal activity can influence food intake in response to shifting internal and external conditions. 31 We began by testing the Gr5a-positive sugar receptor neurons (Figure 3A). The Gr5a receptor is expressed specifically in the peripheral sweet neurons of the labellum, and not in the internal taste organs of the pharynx). 32 To probe the role of Gr5a gustatory receptor neurons in regulating high salt intake, we silenced their activity by expressing an active form of tetanus toxin (Gr5a-GAL4 > UAS-TNT) and pre-exposed the flies to a high-NaCl diet. Expression of tetanus toxin (TNT) disrupts neuronal activity and blocks neurotransmitter release. 33 In our spectrophotometry and feeding assays, high NaCl-fed flies (Gr5a-GAL4 > UAS-TNT) showed increased NaCl consumption (high absorbance values) and a heightened feeding preference for both 200 mM (high) and 50 mM (low) NaCl concentrations (Figure 3B and Supplemental Figure 6A, black bars) compared to flies fed on normal media (Figure 3B and Supplemental Figure 6A, gray bars). Flies were starved for 24 hours in all these experiments. Notably, at 50 mM concentration, high salt-fed flies exhibited more than double the absorbance values and feeding preference (Figure 3B and Supplemental Figure 6A). Since high salt (200 mM) is typically an aversive stimulus, to ensure this was not a generalized loss of aversion, we also tested feeding preference to the bitter compound caffeine (10 mM). No significant differences were observed between normal media-fed and high salt-fed Gr5aGAL4 > UAS-TNT flies for caffeine (Supplemental Figure 6D). Furthermore, both parental controls (Gr5aGAL4/+ and UAS-TNT/+; Figure 3C and D; Supplemental Figure 6B and C) behaved as wild-type flies (Figure 1B-D), showing increased feeding preferences and absorbance values at 200 mM (high salt-fed conditions), but not at 50 mM.

Role of neuronal activity within sweet peripheral and pharyngeal LSO neurons in modulating high salt consumption under starvation. (A) Cartoon showing expression of Gr5a. (B-D) Comparison of mean absorbance values of Gr5a-GAL4 > UAS-TNT and parental control flies (Gr5a-GAL4/+ and UAS-TNT/+). N = 2 to 4 sets each concentration, 60 flies each set. The graphs displaying absorbance values for low (50 mM) and high (200 mM) concentrations of NaCl after 24 hours of starvation. In all the graphs, black bars represent food consumption of flies pre-exposed to high-NaCl diet for 3 days and compared with flies fed on normal media for 3 days (gray bars). Asterisks show significant differences between black versus gray for genotypes Gr5a-GAL4>UAS-TNT, Gr5a-GAL4/+, and UAS-TNT/+. (E, H, K, and N) Cartoon showing expression of Gr43a, Gr64a, Gr64e, and Gr64f. (F-M) Mean absorbance value of flies after genetically silencing the neuronal activity of sweet taste receptor neurons by expressing tetanus toxin (UAS-TNT) using Gr43a-GAL4 (F), Gr64a-GAL4 (I)), Gr64e-GAL4 (L), and Gr64f-GAL4 (O) drivers, and parental control flies (G) Gr43a-GAL4/+, (J) Gr64a-GAL4/+, (M) Gr64e-GAL4/+, and (P) Gr64f-GAL4/+. N = 2-4 sets each concentration, 60 flies each set. Asterisks showing significant differences between flies fed on normal (gray bars) and high-NaCl media (black bars). Statistical analysis was performed using ANOVA Sidak’s multiple comparison test for obtaining P values: *P < .05, **P < .005, and ***P < .0005. For all graphs, error bars = SEM and NS is not significant.
Next, we evaluated whether sweet taste receptors expressed within the labral sense organ are involved in this adaptation. For this, we examined the consumption and feeding preference of flies expressing other sweet taste receptors, including Gr43a (Figure 3E and F and Supplemental Figure 6E), Gr64a, Gr64e, and Gr64f (Figure 3H, I, K, L, N, and O and Supplemental Figure 6G, I, and K) after silencing their neuronal activity with tetanus toxin. Unlike Gr5a, which is restricted to labellum neurons, these receptors are expressed in LSO pharyngeal neurons (Figure 3E, H, K, and N) and have an additional expression in the taste neurons of the labellum or in other regions of pharynx.34-37 Notably, Gr43a and Gr64e have been reported to co-express with other sweet receptor members specifically within the LSO circuit. 38 In striking contrast to our Gr5a results, silencing these LSO-expressed neurons (Gr43a, Gr64a, Gr64e, and Gr64f) using tetanus toxin (GAL4>UAS-TNT) completely abolished the dietary-induced shift. We observed no significant differences in NaCl consumption or feeding preferences at either 50 mM or 200 mM concentrations between normal media- or high NaCl-fed flies for any of these lines (Figure 3F, I, L, and O and Supplemental Figure 6E, G, I, and K). These findings suggest that silencing sweet LSO neurons may reduce feeding preferences back to levels similar to those observed in normal media-fed flies. All parental controls (UAS-TNT/+, Gr43aGAL4/+, Gr64aGAL4/+, Gr64eGAL4/+, and Gr64fGAL4/+; Figure 3D, G, J, M, and P and Supplemental Figure 6C, F, H, J, and L) behaved like wild-type flies (Figure 1B-D), exhibiting the typical increase in consumption and feeding preference at 200 mM concentration under high NaCl-fed conditions but not at 50 mM. Overall, our results suggest that functional and active LSO pharyngeal neurons, particularly under starvation (as seen in Gr5a > TNT or wild-type flies; Figures 1B-D and 3B and Supplemental Figure 6A), play a role for mediating the adaptive increase in high NaCl intake. Interestingly, while Gr43aGAL4 > UAS-TNT and Gr64eGAL4 > UAS-TNT flies showed basal increase in consumption at 200 mM NaCl concentration under normal media condition (Figure 3D, F, G, L, M, and P and Supplemental Data File 2), Gr64eGAL4 > UAS-TNT flies exhibited a significantly reduced feeding preference compared to parental control flies at 50 mM NaCl concentration under control conditions (Supplemental Figure 6C, I, and J and Supplemental Data File 2). Taken together, these data suggest that while labellar input is dispensable, active LSO neurons are a critical requirement for mediating the homeostatic resetting of salt appetite.
Role of Neuronal Activity Within Bitter Peripheral and Pharyngeal LSO Neurons in Modulating High Salt Consumption Under Starvation
To further investigate the role of pharyngeal neurons further in mediating adaptive increase in high NaCl intake, we next targeted bitter-sensing neurons. We hypothesized that if LSO activity is required for this behavioral shift, silencing LSO-specific bitter populations would abrogate the adaptation, while silencing non-LSO populations would not. We first examined the role of Gr33a-positive bitter receptor neurons. Gr33a is a known receptor for the bitter compound caffeine. 39 Since Gr33a is not expressed in LSO pharyngeal neurons,35,39 we investigated whether silencing bitter neurons in the periphery and the ventral cibarial sense organ (VSCO; Figure 4A) would lead to increased high-NaCl intake, similar to what we observed in Gr5a > TNT flies. Upon silencing the activity of bitter neurons (Gr33a-GAL4 > UAS-TNT) using tetanus toxin (Figure 4B and Supplemental Figure 7A), high NaCl-fed flies still exhibited significantly increased consumption and feeding preference at 200 mM NaCl compared to normal media-fed flies (Figure 4B and Supplemental Figure 7A). Flies were starved for 24 hours in all these experiments. Parental control flies (Gr33a-GAL4/+ and UAS-TNT/+; Figure 4C and D and Supplemental Figure 7B and C) behaved as wild-type flies (Figure 1B-D). In general, Gr33a-GAL4 > UAS-TNT flies showed lowered feeding preference at 200 mM NaCl concentration under normal media condition when compared with parental controls (Figure 4B-D; Supplemental Figure 7A-C and Supplemental Data File 2) but no significant difference in consumption was observed. Next, we extended our analysis to other classes of bitter receptor neurons, including Gr93a, which does not exist in the LSO17,35,40 (Figure 4E and F and Supplemental Figure 7D), and Gr66a, which is expressed in LSO35,41 (Figure 4H and I and Supplemental Figure 7F). Silencing Gr66a-expressing LSO neurons (Gr66a-GAL4 > UAS-TNT) did not affect feeding preference between normal and high NaCl-fed flies (Figure 4I and Supplemental Figure 7F). In contrast, silencing Gr93a-expressing neurons (Gr93a-GAL4 > UAS-TNT) resulted in increased consumption and feeding preference at 200 mM NaCl in high salt-fed flies (Figure 4F and Supplemental Figure 7D), similar to Gr33a-GAL4 > UAS-TNT high NaCl-fed flies. Parental controls (Gr93aGAL4/+, Gr66a-GAL4/+, and UAS-TNT/+; Figure 4D, G, and J and Supplemental Figure 7C, E, and G) again behaved as wild-type flies (Figure 1B-D). We also noticed increased consumption by Gr93a-GAL4 > UAS-TNT flies at 200 mM NaCl concentration under NaCl media condition (Figure 4D, F, and G and Supplemental Data File 2) when compared with parental control flies. These results demonstrate that the dietary-induced shift in salt appetite is not a generalized loss of peripheral bitter sensitivity. Instead, our results suggest that neuronal activity plays a key role in driving specific adaptations and it specifically requires functional LSO pharyngeal neurons, which likely undergo a sensitivity shift following prolonged NaCl exposure to promote increased intake.

Role of neuronal activity within bitter peripheral and pharyngeal LSO neurons in modulating high salt consumption under starvation. (A) Cartoon showing expression of Gr33a (B-D) Mean absorbance values of Gr33a-GAL4 > UAS-TNT, Gr33a-GAL4/+ and UAS-TNT/+ flies for different NaCl concentrations after feeding normal or high-NaCl diet following 24 hours of starvation. Asterisks showing significant differences between flies fed with normal (gray bars) and high-NaCl media (black bars). (E) Cartoon showing expression of Gr93a. (F and G) Spectrophotometry analysis of Gr93a-GAL4 > UAS-TNT and parental control flies (Gr93a-GAL4/+) showing comparison between normal- and high-NaCl media conditions (gray vs black bars) following 24 hours of starvation. (H) Cartoon showing expression of Gr66a. (I and J) Spectrophotometry analysis of Gr66a-GAL4>UAS-TNT and parental control flies (Gr66a-GAL4/+) showing comparison between normal- and high-NaCl media conditions (gray vs black bars) following 24 hours of starvation. Asterisks showing significant differences between flies fed on normal (gray bars) and high-NaCl media (black bars). N = 2 sets each concentration, 60 flies each set. Statistical analysis was performed using ANOVA Sidak’s multiple comparison test for obtaining P values: *P < .05, **P < .005, and ***P < .0005. For all graphs, error bars = SEM and NS is not significant.
In our assays, we observed increased feeding preference (Supplemental Figure 7C, F, and G) at 200 mM and consumption at 50 and 200 mM NaCl concentration (Figure 4D, I, and J) in Gr66a > TNT flies maintained on normal media (Supplemental Data File 2) when compared with parental flies. Gr66a is known to play a critical role in bitter taste perception and avoidance, particularly in detecting and rejecting toxic compounds like caffeine and other aversive chemicals. The weakened aversion to 200 mM NaCl observed under normal media conditions (compare normal media-fed flies gray bars only, Figure 4I and Supplemental Figure 7F), suggests that Gr66a-positive LSO bitter neurons are intrinsically involved in detecting high- salt concentrations. These neurons likely serve as a primary aversive pathway that regulates salt intake; thus, their silencing reduces the fly's innate rejection of high salt even in the absence of dietary acclimation.
Genetic Suppression of LSO Pharyngeal Neurons Inhibits Excessive Salt Intake in High NaCl-Fed Flies
To further characterize the role of active LSO pharyngeal neurons in regulating high salt intake, we selectively silenced pharyngeal LSO neurons using two distinct drivers: Gr2a-GAL4 14 and Poxn-GAL4 35 (Pox neuro). Both drivers exhibit restricted expression in these pharyngeal neurons (Supplemental Figure 8A-C and F white arrow in right-hand side images). Silencing these populations (Gr2a-GAL4 > UAS-TNT and Poxn-GAL4 > UAS-TNT) completely abolished the dietary-induced shift; we observed no significant differences in feeding behavior (Supplemental Figure 8D and G, left hand-side graphs) or NaCl consumption between high NaCl- and normal media-fed flies at both concentrations (Supplemental Figure 8E and H, left hand-side graphs). The parental control flies (Gr2a-GAL4/+, poxn-GAL4/+ and UAS-TNT/+; Supplemental Figure 8D-H, right-hand side graphs; Figure 3D and Supplemental Figure 6C) showed absorbance values and feeding preferences similar to wild-type flies (Figure 1B-D). Flies were starved for 24 hours in these experiments. We also observed increased consumption in case of Gr2a-GAL4>UAS-TNT flies when compared with parental controls (Supplemental Figure 3D and 8E and Supplemental Data File 2) for normal media condition at 200 mM concentration. On the other hand, lowered feeding preference and consumption was noticed in case of poxn-GAL4>UAS-TNT flies when compared with parental controls (Supplemental Figure 3D and 8G and H and Supplemental Data File 2) for salt media condition at 200 mM concentration. Collectively, these results confirm that the activity of LSO pharyngeal neurons is the primary requirement for inducing the behavioral plasticity that allows flies to tolerate and consume high concentrations of NaCl under starvation.
Next, we tested Drosophila Ir76b neurons to investigate if the activity in Ir76b + LSO neurons is involved in causing the feeding behavioral shift under starvation in high NaCl-fed flies. Ir76b is a critical receptor required for mediating both attractive and repulsive taste responses to salt,13,16,42 and is broadly expressed in pharyngeal neurons. We independently confirmed its expression in many LSO neurons (Supplemental Figure 9A-C, white arrow). After silencing activity of Ir76b positive neurons (Ir76b-GAL4 > UAS-TNT), we found no difference in feeding preference between high NaCl-fed and normal media-fed flies (Ir76b-GAL4 > UAS-TNT; Supplemental Figure 9D) at 50 and 200 mM NaCl concentrations . Silencing Ir76b neurons reduced feeding preferences at both concentrations relative to parental controls (gray bars in Supplemental Figures 6C and 9D-E), consistent with the known role of the Ir76b receptor in low and high salt detection. Ir76b GAL4/+ flies behaved like wild-type flies, showing the expected increase in feeding preference at 200 mM NaCl when high NaCl-fed flies were compared to normal media-fed flies. We observed overall lowered feeding preferences under normal media and high-NaCl conditions for both 50 and 200 mM concentrations (Supplemental Data File 2). Collectively, these data indicate that Ir76b-mediated activity within the LSO is essential for both the detection of salt and the subsequent behavioral adaptation triggered by chronic high-salt ingestion.
Role of Dopamine and Activity in Regulating High Salt Consumption
Recent studies have shown that both starvation and L-dopa supplementation increase behavioral sensitivity to sucrose in flies.13,16,29,42,43 Given our observation that salt-feeding adaptation only emerges during food deprivation, we investigated if, this starvation-mediated behavioral change can also be replicated by L-dopa supplementation. We administered 3-(3,4-dihydroxyphenyl-2,5,6-d3)-L-alanine (L-Dopa) through normal food and measured feeding preferences, without starving flies in any of these experiments. For L-Dopa experiment, we tested only those flies that previously exhibited a robust preference shift (Figures 3B, 4B and F and Supplemental Figure 6A, 7A and D) under starvation. Consistent with our findings from starved flies, we found that high NaCl-fed Gr5a-GAL4 > UAS-TNT flies showed increased feeding preferences at both 50 and 200 mM NaCl concentrations (Figure 5A, black bars) after L-Dopa feeding (L-Dopa + food). Enhanced feeding was notably observed at 50 mM NaCl concentration (Figure 5A, black bars) when compared to the respective parental control flies (Gr5a-GAL4/+ and UAS-TNT/+; Figure 5B and C) fed with a high-NaCl diet (black bars). Crucially, in the absence of L-Dopa (normal media-fed conditions), no significant differences were found between normal media- and high NaCl-fed flies across any of the genotypes (Gr5a-GAL4 > UAS-TNT, Gr5a-GAL4/+, and UAS-TNT/+) tested at either 50 mM or 200 mM NaCl concentrations (Figure 5D-F). These results indicate that the behavioral adaptation triggered by chronic salt intake is gated by dopaminergic signaling, which is endogenously activated by starvation but can be exogenously induced by L-Dopa.

Role of Dopamine in modulating salt taste behavior. (A-C) Comparison of feeding preferences of Gr5a-GAL4>UAS-TNT and parental control flies (Gr5a-GAL4/+ and UAS-TNT/+) after L-Dopa feeding. In all graphs, black bars represent preferences of high NaCl-fed flies compared with flies fed on normal media (gray bars). Asterisks showing significant differences between black versus gray bars for genotypes Gr5a-GAL4 > UAS-TNT, Gr5a-GAL4/+ and UAS-TNT/+. (D-F) Comparison of feeding preference of Gr5a-GAL4 > UAS-TNT flies with parental control flies (Gr5a-GAL4/+ and UAS-TNT/+) after feeding on normal fly food (no L-dopa treatment). (G-N) Mean feeding preference of Gr33a-GAL4 > UAS-TNT and Gr93a-GAL4 > UAS-TNT flies and their parental controls (Gr33a-GAL4/+ and Gr93a-GAL4/+) with (G-J) and without L-Dopa treatment (K-N). No starvation was done in any of these experiments. Asterisks showing significant differences between flies fed on normal (gray bars) and high-NaCl media (black bars). For each bar, n = 6 to 12 trails of 20 flies each (10 males and 10 females). Statistical analysis was performed using ANOVA Sidak’s multiple comparison test for obtaining P values: *P < .05, **P < .005, and ***P < .0005. For all graphs, error bars = SEM and NS is not significant.
Similarly, in Gr33a-GAL4 > UAS-TNT (Figure 5G) and Gr93a-GAL4 > UAS-TNT (Figure 5I) flies, L-Dopa supplementation phenocopied the starvation state, leading to significantly higher feeding preferences at 200 mM NaCl in high NaCl-fed flies compared to normal media-fed flies at 200 mM, but not at 50 mM. The parental control flies (Gr33a-GAL4/+, Gr93a-GAL4/+, and UAS-TNT/+; Figure 5C, H, and J) showed the typical wild-type pattern (Figure 1B), showing an adaptive shift only at 200 mM NaCl and no change at 50 mM between normal media- and high NaCl-fed flies (gray and black bars). Crucially,under normal media-fed conditions without L-Dopa, no significant differences were observed between normal media- and high NaCl-fed flies for any genotype at either concentration (Figure 5F and K-N). Our results suggest that internal states (such as starvation) or elevated dopamine levels can modulate salt taste behavior, reducing salt aversion after prolonged high-NaCl exposure, provided LSO neuron activity remains intact. Our results further solidifies dopamine as a hunger signal in the fly nervous system that can alter feeding behaviors to molecules other than sugars.
Discussion
Adult Drosophila possess distinct internal taste organs located in the pharynx, including the LSO (Labral Sense Organ), VCSO (Ventral Cibarial Sense Organ), and DCSO (Dorsal Cibarial Sense Organ), in addition to peripheral taste cells found on the labellum and tarsi. Once feeding initiates, the pharynx plays a key role in regulating food intake, ensuring that only appetitive food is consumed. Recent advances in molecular mapping have revealed a sophisticated receptor-to-neuron map of the pharyngeal taste organs, identifying distinct functional groupings among pharyngeal neurons. 35 However, while the mechanisms by which peripheral taste systems detect salt are well-characterized, the specific role of the pharyngeal neurons in modulating high salt intake, particularly under prolonged high-salt dietary conditions, remains largely unclear. Although recent work has identified an inhibitory mechanism that suppress high salt intake, 21 and specific pair of pharyngeal neurons expressing Ir60b, that facilitate rejection of high-salt foods, 22 the broader circuit changes underlying chronic dietary adaption remain to be elucidated.
Beyond gustatory detection, recent studies have identified a population of enteric neurons, designated as Internal Sodium-Sensing (INSO) neurons, that detect sodium levels within Drosophila gut demonstrating that sodium sensing extends beyond taste receptors to include internal mechanisms. 24 This work uncovers a distinct pathway for sodium detection and response, critical for maintaining homeostasis. Activation of INSO neurons directly influences sodium preference ; after a period of sodium deprivation, flies with functional INSO neurons exhibit an increased preference for sodium-rich food to restore physiological balance. The study highlights the importance of postprandial sensing, emphasizing the dynamic regulation of sodium levels following food intake. Integrating these findings with our own results reveals a sophisticated, multi-layered regulatory system: while INSO neurons monitor internal state, the pharyngeal LSO circuit serves as the adaptive gatekeeper, undergoing dietary-induced plasticity to calibrate salt intake before it reaches the digestive system
Previous calcium imaging studies suggested that Gr5a- positive sweet neurons mediate low salt attraction in insects, 6 although this driver (Gr5a-GAL4) also labels additional non-sweet GRNs outside the traditional sweet neuron class. 36 Other studies have implicated the ionotropic receptor Ir76b13,42 and Gr66a GRNs6,44 in salt sensing. Gr64f and Ir94e mediate attraction toward low salt and Gr66a and Ppk23 glut drive avoidance to high salt concentrations, 16 highlighting an added layer of complexity in salt coding in flies. Recently, 2 groups demonstrated the involvement of different ionotropic receptors in promoting appetitive responses to salt 17 and mediating avoidance of high concentrations of monovalent salt. 18 Of note, in all these studies, the role of internal organs, including pharyngeal areas, was not tested for salt taste behavior. Our study provides the neural basis for adaptation suggesting high dietary salt modulates and restructures salt and other taste curves, where flies adapt their acceptance of high salt depending on diet and exposure time through activity- and state-dependent mechanisms involving LSO pharyngeal neurons.
A key finding of our study is the functional disscociation between reflexive tarsal PER and cumulative feeding behavior. Crucially, the tarsal PER outcomes did not mirror the direction of the salt-feeding phenotype after a 3-day high-salt diet. This dissociation argued against a model in which the increased salt ingestion after diet exposure is driven primarily by a global increase in peripheral/external NaCl sensitivity. Instead, these findings are most consistent with the interpretation that diet exposure engages internal chemosensory circuitry, aligning with our LSO-focused model. While PER does not support a peripheral-sensitivity explanation for the feeding phenotype, PER is an assay of reflexive proboscis responses and does not exclude all conceivable peripheral contributions. Nonetheless, the lack of concordance between PER and feeding, together with the requirement for LSO-associated GRNs, supports our conclusion that internal chemosensory cells are a key driver of the diet-associated change in salt ingestion. Additionally, our findings implicate dopaminergic signaling as the metabolic gate that reveals this adaptation under starvation.
Our results demonstrate that multiple taste receptor neurons within the LSO region contribute independently to a common output. When one of them is inhibited, we observed a partial reduction of heightened salt responses, highlighting their critical role in this adaptive mechanism under starvation. Overall, our findings suggest that flies can reset their salt appetite and related neural pathways based on prior dietary salt exposure. Moreover, beyond salt, we observed that high salt pre-exposure cross-modulates the consumption of metabolizable sugars (Figure 2), but not non-metabolizable ones. Compared to fructose, which is detected by Gr43a, 45 sucrose is detected by multiple receptors.46-50 The abundance of sucrose receptors may allow flies to detect even low concentrations of sucrose (below 100 mM; Figure 2). Our findings suggest that pre-exposure to high-NaCl diet modulates flies’ consumption of sucrose and shift it to lower concentrations (Figure 2), indicating that sodium intake may modulate the neural processing of sweet taste information through complex interactions involving homeostasis, neural pathways, and metabolic processes. While comparing across control genotypes and diet groups, we also observed that some cell silencing experiments appeared to impact feeding in the salt assays even in control animals (eg, Gr66a > TNT, IR76b > TNT; Supplemental Data File 2). This was expected, as many cell types are known to contribute to salt feeding at a range of concentrations. Overall, our findings suggest that flies can reset their salt appetite based on prior dietary history, utilizing the pharynx as a critical gatekeeper for nutritional homeostasis.
The significant reduction in salt preference observed when silencing the LSO using Gr2a-GAL4 and poxn-GAL4 drivers, underscores the necessity of these pharyngeal neurons in regulating high-salt intake (Supplemental Figure 8). However, further experiments are needed to determine whether LSO neuron activation alone is sufficient to trigger a loss of salt aversion, as this remains untested. Consistent with a recent study suggesting that salt taste is encoded by the combined activity of multiple GRN classes at the periphery, 16 our findings also suggest that under starvation conditions, neurons in the LSO region regulate high salt ingestion through combinatorial pathways rather than a single dedicated channel. However, we do not know whether pre-exposure to high salt alters downstream processing in higher brain centers, as the precise circuit architecture for pharyngeal salt integration has yet to be fully mapped (Figure 6).

Working model of high salt intake. Three distinct internal taste organs are present in the adult fruit fly pharynx: the labral sense organ (LSO), ventral cibarial sense organ (VCSO), and dorsal cibarial sense organ (DCSO). Our results suggest that flies typically prefer low salt concentrations and avoid high ones. However, extended consumption of a high-NaCl diet diminishes this aversion. This shift is mediated by active pharyngeal LSO neurons and can be further influenced by the flies’ hunger or dopamine levels (thick red arrow). The feeding preference for high-NaCl get suppressed in case of reduced neuronal activity in LSO neurons and under fed state (thin red arrow) as observed with many classes of gustatory receptor neurons expressing Gr43a, Gr64a, Gr64e, Gr64f, Gr66a, Gr2a, Ir76b, and poxn (all express in LSO). The identity of salt metabolic sensor in the brain and the role of central salt neurons causing high salt taste modulation is an open area of exploration.
Insects such as fruit flies, moths, and locusts exhibit a known capacity to reduce their avoidance of certain bitter compounds following prolonged exposure. 51 For instance, dietary exposure to unappealing yet safe additives like camphor leads to a decline in camphor rejection, driven by reversible down-regulation of TRPL (transient receptor potential-like) levels in the proboscis. 42 Similarly, state- dependent modulation of salt taste behavior, mediated by Ppk23 glut neurons, has been demonstrated under conditions of salt feeding (3 days on food containing 10 mM NaCl) and salt deprivation. 16 In humans, salt depletion likewise increases salt palatability, 52 and metabolic hunger signals are known to override aversive responses to otherwise unappetizing foods. 14 Our study highlights a previously underexplored, state-dependent role of internal pharyngeal taste neurons in modulating salt taste behavior, particularly in response to high, aversive salt concentrations. We used a paradigm in which flies were pre-exposed to 200 mM NaCl (an aversive concentration) mixed with normal food for three consecutive days. This differs from the approach by Jaeger et al, 16 who used salt concentrations typically found in yeast (5%-15%) added to normal fly media. Notably, the basal salt levels present in our standard rearing media did not impact the PER or other feeding behavior responses, confirming that the observed adaptation is a specific response to the high-salt dietary challenge.
In our study, prolonged exposure to high salt recalibrated the taste sensitivity of pharyngeal LSO neurons, ultimately leading to increased salt consumption. This contrast with short-term exposure studies 17 that highlight the LSO’s role in promoting the avoidance of concentrated salts. These seemingly contradictory findings can be reconciled by considering the timescale and physiological context of exposure: the LSO acts as a dynamic sensory organ, switching between adaptive responses (prolonged exposure) and protective aversion (short-term exposure) mechanisms depending on the duration and intensity of salt exposure. This underscores the complexity of taste perception and its critical role in maintaining homeostasis in fluctuating environments. Future studies aimed at identifying the molecular mechanisms underlying adaptive changes in LSO sensitivity and examining the interaction between the LSO and other taste organs in regulating salt preference and avoidance will deepen our understanding of the intricate mechanisms that govern taste perception and behavior in flies and potentially extend these insights to other organisms, including humans.
Mating significantly modifies feeding behavior in female Drosophila, 53 inducing a salt appetite by enhancing the gustatory response to sodium. 23 Consistent with this, we also found that mated females show a higher preference for salt-containing food (Supplemental Figure 1D), likely reflecting a compensatory reproductive investment required for oogenesis. Beyond reproductive state, neuropeptides such as dNPF (Drosophila neuropeptide F) and sNPF (short neuropeptide F) are known to modulate multiple feeding-related behaviors, including the regulation of food intake during starvation.54-58 Under conditions of energy deficit, animals generally become less selective, showing increased sensitivity to nutritious resources like sugars,29,59-63 highlighting the role of neuromodulatory cascades as key mediators of state-dependent behavioral control.64-67 While our study identifies a clear dopamine-dependent modulation of salt taste behavior (Figure 5), whether dNPF/sNPF signaling through LSO neurons contributes to this modulation remains an open question for future investigation (Figure 6). Collectively, our data suggest that previous exposure to high salt concentration activates distinct internal taste pathways that are further gated by metabolic state. This integration of sensory history and internal state, as proposed by the state-dependemt models of Devineni et al, 68 ultimately allows the fly to switch between innate aversion and adaptive consumption of high-salt sources.
The role of neuronal activity in modulating taste perception remains largely unexplored. In mice, acute manipulation of the ventromedial hypothalamus (VMH) steroidogenic factor 1 (SF1) neurons has been shown to alter food intake 69 by integrating energy status with appetite drive. Similarly, our findings identify a valence for NaCl preference linked to LSO neuronal activity, which is gated by starvation or dopamine signaling in high salt-fed flies. While we have identified the LSO as a critical regulator of this decision-making process, how these pharyngeal signals integrate into central circuits such as SEZ to govern feeding behavior across different internal states remains an open question. Notably, such a state-dependent analysis was missing in the study by Chen et al, 20 where functional and behavioral studies in poxn mutants suggested avoidance of many aversive compounds, including high salt, 20 mediated by pharyngeal neurons. Because our experiments utilized constitutive silencing with UAS-TNT, our neuronal silencing experiments do not distinguish whether LSO GRNs are required for: (1) induction of a diet-dependent adapted state during the 3-day exposure, or (2) for the acute expression of altered salt feeding during the assay. Our current genetic silencing experiments establish that LSO GRN output is necessary somewhere within this combined exposure-and-assay timeframe for diet associated increase in salt feeding, but do not yet isolate the temporal locus of necessity (induction vs expression). Future experiments using temporally restricted inhibition (eg, GAL80ts, Shibirets with appropriate temperature controls, or optogenetic inhibition such as GtACR) would resolve whether LSO GRNs act primarily during induction or expression.
Our results establish that pharyngeal taste organs, like LSO, serve as an essential regulatory hub for sodium homeostasis. This pharyngeal circuit appears to be part of an evolutionarily conserved framework designed to finely balance calorie intake with energy metabolism. Investigating the molecular and behavioral impacts of appetitive cues, such as sugar and varying salt concentrations, on the brain’s reward circuitry 70 will be crucial for understanding how these cues drive changes in neural activity that can lead to overeating and metabolic disorders. Ultimately, addressing these mechanisms may inform new therapeutic strategies and public health interventions aimed at reducing sodium intake and managing the global burden of salt-associated metabolic disease.
Materials and Methods
Fly Stocks
CsBz and w1118 (from NCBS, Bangalore), UAS-TNT (BL 28838), Gr5a-GAL4 (BL 57592), Gr33a-GAL4 (BL 57623), Ir76b-GAL4 (BL 51311), Gr43a-GAL4 (BL 57636), Gr64f-GAL4 (BL 57669), Gr64a-GAL4 (BL 57661), Gr64e-GAL4 (BL 57667), Gr66a-GAL4 (BL 57670), Gr93a-GAL4 (BL 57679), Gr2a-GAL4 (BL 57590), UAS-mcd8GFP (BL 5137), and poxn-GAL4 (Bl. 66685) were obtained from the Drosophila Bloomington Stock Center. Drosophila stocks were reared on standard cornmeal dextrose medium (normal media) at 25°C unless specified otherwise.
Drosophila media composition used (for 1 l of media) – corn flour (80 g), D-glucose (20 g, SRL, Cat. no. 50-99-7), sugar (40 g), agar (8 g, SRL, Cat. no. 9002-18-0), yeast powder (15 g, SRL-REF-34266), propionic acid (4 ml, SRL, Cat. no. 79-09-4), Tegosept (1.25 g in 3 ml of ethanol, Fisher Scientific, Cat. no. 99-76-3), and orthophosphoric acid (600 ul, SRL, Cat. no. 7664-38-2).
Feeding Behavior Assays
For feeding assays, flies of the required genotype were raised from eggs to adults at 25°C on normal fly food. About 1 to 3 days old flies were sorted in vials as 10 males and 10 females (20 flies/vial) on normal fly food upon eclosion and maintained at 25°C for 3 days on fresh media (high-NaCl or normal fly food). Following starvation of 24 hours at 25°C, these flies were tested for feeding behavior as described previously,43,71 and abdominal coloration was scored as positive if there was any pink or red eating (red or pink- feeding on salt or taste compound; white- no feeding; blue-feeding on agar and water; purple- feeding on both red and blue). Purple was also scored if flies ate both red and blue in any of the experiments. About 60 × 15 mm feeding plates (Tarsons India) were used for the assay. In these assays, flies were provided with red dots as taste stimuli and blue spots as control containing dye, water, and agar.
Percentage of flies feeding on taste compound was calculated as follows: First, % flies feeding on red, blue or purple (Supplemental Figure 1A) for each plate was calculated. The Mean of % flies feeding (6 or more plates) was taken as a final value.
Tarsal Proboscis Extension Reflex (PER) Assay
For all PER experiments, flies were tested as described previously. 43 Flies were collected after 2 to 3 days of eclosion and kept on normal media for 2 days. Both male and female flies were used for the PER assay. The flies were starved for 24 hours in vials with water-saturated (4 ml) tissue papers. No starvation was done when flies were tested under fed condition. Prior to the PER experiment, flies were immobilized by cooling on ice for at least 15 minutes and then mounted using nail polish, vertical aspect up, on glass slides (76 mm × 26 mm × 1 mm from Borosil). Flies were allowed to recover in a moist chamber (plastic box with wet tissues) for at least 2 hours prior to testing. Tastant solutions prepared in water were applied to tarsi via a drop extruded using 2 µl pipette. Before testing the taste solutions, flies were allowed to drink water ad libitum. Flies not responding to water were excluded before the assay. Flies satiated with water were then tested with NaCl or other tastant solutions. Following each tastant application, flies were retested with water as a negative control. Each fly was tested 5 times with each tastant solution stimulus. The interval between consecutive tastant solution applications was at least 2 to 3 minutes to minimize adaptation. Flies showing 3 or more proboscis extensions were considered responders. About 100 mM sucrose was used as a control stimuli at the beginning and end of the experiments to make sure flies were not losing sensitivity after testing various salt concentrations in another set of PER experiment. Responses of flies showing positive PER to 100 mM sucrose were plotted (Supplemental Figure 2B). The tastant was administered without sucrose in our PER experiments.
More than 50 flies were tested in batches for all PER experiments, and the percentage of responders was calculated for each set. Graphs depict mean responses, and error bars indicate SEM (Standard error of the mean).
Chemicals
Sugars used in the experiments were all obtained from Sigma Aldrich- Sucrose (57-50-1), Fructose (57-48-7), Trehalose (6138-23-4), Sucralose (56038-13-2), Galactose (59-23-4), L-(-) Glucose (Sigma –921-60-8) and D-(+)-Glucose (50-99-7). The other compounds used in the study- NaCl salt (Fisher Scientific- 7647-14-5) of 99.9% purity; Caffeine (Sigma-Aldrich # 58-08-2); Blue dye- Indigo carmine (Sigma: 860-22-0); Red dye-Sulforhodamine B (Sigma- 3520-42-1); Potassium Di Hydrogen Orthophosphate (Fisher Scientific -7778-77-0); Magnesium Chloride (Fisher Scientific- 7791-18-6); Di Sodium Hydrogen O-phosphate (Fisher Scientific- 7558-79-4); Sodium Hydrogen Carbonate (Fisher Scientific- 144-55-8); Potassium Chloride (Fisher Scientific- 7447-40-7) and Calcium Chloride (Fisher Scientific- 10043-52-4).
Spectrophotometry Analysis
After eclosion, 3-days old flies were sorted (10 males and 10 females) into batches on normal media which served as a control (6× vials). Similarly, 3 days old flies were separated as 10 males and 10 females on different salt concentrations (10, 50, 100, 200, and 500 mM NaCl) mixed with normal fly food (6× vials). Flies kept on these media conditions for 3 days were treated the same way as done for feeding assays in Figure 1B (starvation was done for 24 hours at 25°C). Feeding plates in this experiment contained blue (test stimuli) and white (control) dots.
Spectrophotometry analysis: after feeding assays, total 120 flies were divided into 2 sets (2 sets × 60 flies each) for each concentration. Later, flies (all the flies were used in the assay in an unbiased manner) were placed in 2 ml Eppendorf tubes in 70% ethanol (60 flies in 1 Eppendorf). Flies were first crushed in 150 µl of 70% ethanol and then 150 µl 70% ethanol was added to crush them more. After crushing, 200 µl of double distilled water (to get the content sticking to pestle and the wall of the Eppendorf tube) was added in the same soup and centrifugation was done at 3000 rpm for 15 minutes. After centrifugation, pellet was discarded and supernatant was transferred in the fresh Eppendorf’s. For spectrophotometry analysis, supernatant was further diluted with double distilled water to make up the total final volume of 650 µl in the cuvette. Food consumption (mixture of NaCl and blue dye) was measured using blue dye that can be measured spectrophotometrically at 630 nm wavelength. One reading was taken for each set and only the mean values were plotted. Spectrophotometer used was Perkin Elmer, lambda 35 UV/VIS Spectrometer.
Weight Analysis
For weight analysis, 30 CsBz wild-type flies were used per media condition (n = 4 × 30 flies). Newly eclosed flies were collected within 48 to 72 hours of emergence and sorted by sex. After being maintained on their respective media for 3 days, the flies were weighed. Throughout the experiment, flies were maintained at 25°C.
Immunohistochemistry
Immunohistochemistry for labellum was performed as mentioned before. 43 After anesthetizing flies on ice, labellum was dissected in chilled 1X PBS and fixed for 30 minutes in 4% paraformaldehyde (0.3% Triton X-100) at room temperature. After washes with PBST (PBS with 0.3% TritonX-100) 4 × 20 minutes, samples were blocked with 5% normal goat serum in PBST for 1 hour. Samples were incubated in appropriate primary antibody solutions overnight at 4°C. Primary antibody used was rabbit anti-GFP (1:1000, Invitrogen, catalog no. A11122). Next day, samples were washed with PBST (4 × 20 minutes) after primary antibody step. Secondary antibody used was Alexa Fluor 488 goat anti-rabbit immunoglobulin G (IgG; 1:200, Invitrogen). Samples were washed with PBST (4 × 20 minutes) after secondary antibody step before mounting on the slides in 70% glycerol in PBST.
L-Dopa Feeding Experiments
For experiments in Figure 5, L-Dopa (Sigma-Aldrich, CAS no. -53587-29-4) was first dissolved in water (5 mg/ml). 43 Then, freshly prepared solution (750 µl of 5 mg/ml for 3 ml of fly food) was spread on fly food and small holes were made with the needle to make L- Dopa absorbed in the media. After this, flies were maintained on this L-Dopa food by wrapping the vials with aluminum foil at 25°C incubators. The medium was changed once after 24 hours and freshly made L-Dopa was added to the media. Control flies were fed on normal fly food without any L-Dopa. No starvation was involved in these experiments.
Microscopy Used for Image Analysis and Video Recording
GFP Imaging
Adult labellum was mounted in 70% glycerol in PBST after immunohistochemistry. Samples were analyzed and GFP fluorescence was visualized using a confocal microscope (Leica TCS SP5 II), and image stacks were generated acquired at 0.5 micron optical sections. Olympus SZX10 dual tube microscope was used for generating videos and Olympus SZ61 stereomicroscopes for doing the general fly pushing. Images were processed using ImageJ, Adobe Photoshop, and Illustrator software.
Statistical Analyses
Unless otherwise stated, all the results from behavioral and spectrophotometric experiments were analyzed for statistical significance using one-way analysis of variance (ANOVA), followed by Sidak’s multiple comparisons post hoc test to determine specific group differences. For datasets involving comparisons between only 2 groups (eg, Supplemental Figures 5D and 6D), statistical significance was assessed using an unpaired two-tailed Student’s t-test. A P-value of less than .05 was considered statistically significant. All analyses were performed in GraphPad Prism.
Supplemental Material
sj-docx-12-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-docx-12-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-10-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-10-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-11-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-11-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-3-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-3-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-4-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-4-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-5-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-5-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-6-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-6-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-7-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-7-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-8-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-8-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-jpg-9-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-jpg-9-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-xlsx-1-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-xlsx-1-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Supplemental Material
sj-xlsx-2-exn-10.1177_26331055261440786 – Supplemental material for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster
Supplemental material, sj-xlsx-2-exn-10.1177_26331055261440786 for Activity and State-Dependent Modulation of Salt Taste Behavior Via Pharyngeal Neurons in Drosophila melanogaster by Shivam Kaushik, Kartik Singh, Rahul Kumar, Sachin Kumar, Srishti Sanghi, Teiichi Tanimura, Diego E. Rincon-Limas and Pinky Kain in Neuroscience Insights
Footnotes
Acknowledgements
We are grateful to PK lab members for helpful comments on the manuscript. We thank National Centre for Biological Sciences, Bangalore and Bloomington Drosophila Stock Center, USA for the fly stocks.
ORCID iDs
Ethical Considerations
Drosophila strains used in the study were reviewed and approved by the institutional ethical committee.
Author Contributions
Conceptualization, P.K.; Methodology, T.T. and P.K.; Investigation, S.K, S.S, S.K, R.K; Validation, S.K., K.S., R.K and P.K.; Formal Analysis, S.K., K.S., D.E.R.L. and P.K.; Writing – Original Draft, P.K.; Writing – Review & Editing, T.T., D.E.R.L. and P.K.; Visualization, S.K. and P.K.; Figures Preparation, S.K., K.S. and P.K.; Supervision, P.K.; Funding Acquisition, P.K.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by Wellcome trust DBT India Alliance grant (IA/I/15/2/502074) to P.K.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and analyzed during the current study is present in the manuscript as the supplementary data file 1 and 2. Confocal files can be available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
