Abstract
Interest in spinal interneurons (SpINs), their heterogeneity in the naive spinal cord and their varying responses to central nervous system injury or disease has been steadily increasing. Our recent review on this topic highlights the vast phenotypic heterogeneity of SpINs and the efforts being made to better identify and classify these neurons. As our understanding of SpIN phenotype, connectivity, and neuroplastic capacity continues to expand, new therapeutic targets are being revealed and novel treatment approaches developed to harness their potential. Here, we expand on that initial discussion and highlight how SpINs can be used to develop advanced, targeted cellular therapies and personalized medicines.
Characterizing SpINs and their neuroplastic potential
Spinal neurons comprise a vastly heterogeneous population of neurons that have both their cell body and their projections within the spinal cord. Developmental neurobiology has provided a wealth of information on transcription factor expression that enables the classification of SpINs into “cardinal” classes (dorsal interneurons 1-6 (dI1-6) and ventral interneurons 0-3 (V0-3) 1 ). The expression of the transcription factors also changes as the cells mature, 2 and can change based on neuroanatomical location (eg, Chx10 expression of V2a interneurons differs between cervical and lumbar spinal levels). 3 Indeed, it has become clear that even within each cardinal class there are many subtypes of cells, for example, the V1 population can be divided into approximately 50 subtypes that settle at discrete locations in the spinal cord.4,5 Genomic screening has revealed that in the V2a class, which is identified by the developmental expression of Chx10 (VSX2 in humans), expression of other genes varies depending on location (eg, cervical vs lumbar), but may also vary based on the network in which they reside.
Behavioral studies in knockout transgenic mice have elucidated potential functions of the cardinal classes of SpINs, and electrophysiology experiments in cell culture and spinal tissue slice preparations indicate functional diversity within those classes. Notably, however, characterization based on function may not always align with cardinal classification. In addition, although most research has focused on SpINs within locomotor networks (and for the most part at the lumbar levels), the cells also act within several other motor, sensory, and autonomic networks. Insights from technologies such as single-cell and nuclei RNA sequencing have challenged the cardinal classification, and have raised an important question: How granular should we get in trying to understand these cells, especially if the interest is in how they function in a circuit rather than in isolation?
Perhaps one of the most fascinating questions for which there is still no clear answer is how do these cells change after a traumatic event like spinal cord injury (SCI). SCI may cause SpINs to lose their original connections, resulting in the formation of new synapses and a novel anatomical network, and their neuronal activity and gene expression may also change. With such fluctuations, should the cellular phenotypes be “recharacterized”? The concept of phenotypic switching has been proposed previously, especially during development, 6 but how this occurs and what that might mean for the new anatomy of the injured spinal cord remains unknown. Does traumatic injury re-open a window for plasticity among SpINs and allow for a type of phenotypic switching, as has been seen in non-neuronal cells (eg, glia, immune cells, etc.)? If one thing is for certain, it is that SpINs are highly adaptable, many of their “defining” characteristics change post-SCI, and we should not underestimate their neuroplastic potential. In the words of Heraclitus, “All is flux, nothing is stationary.”
Developing treatments to harness SpIN therapeutic potential
With the recognition that neural networks within the central nervous system are not hardwired has come an appreciation for the remarkable neuroplastic potential of the neural substrate that is spared in the context of injury. Even after losing all input from the brain and brainstem, spinal networks retain the ability to function. While denervated spinal networks typically fall silent post-SCI, activity can be elicited by stimulation (eg, primary afferent activation via peripheral movement or stimulation, or direct exogenous stimulation via neural-device interfacing such as intraspinal, epidural, transcutaneous, or transmagnetic devices). Activity-based therapies (eg, locomotor rehabilitation, respiratory training, and other physical therapy interventions) and neural interfacing enhance activity in spinal neurons and pathways spared by injury. 7 Continued stimulation promotes plasticity, contributing to additional alterations in anatomical connectivity, which can support continued functional change. Several pre-clinical studies have investigated how this plasticity may recruit SpINs into novel neural networks that lead to lasting improvements in recovery and functional outcomes post-SCI.
Rehabilitation and neural interfacing have great promise in treating people living with SCI, and no matter what treatments are developed in the future, they will likely be most effective if used in combination with one or more of these strategies. However, used alone these treatments have a limit to their potential because they are reliant on the activation of neural substrates spared by injury, and do nothing to account for the very significant loss of tissue, often accompanied by large cystic cavitation at the injury site. Accordingly, treatments capable of either limiting tissue loss (neuroprotection) or restoring tissue integrity and connectivity (neural repair/regeneration) are necessary to increase the potential for neuroplastic change. One approach that has received a great deal of attention both pre-clinically and clinically is the transplantation of cells to promote tissue repair. Transplantation of neural (neuronal and glial) progenitor cells (NPCs) is particularly interesting as the approach not only provides injured tissue with the building blocks to construct new anatomy, but the donor cells are typically rich with interneurons. Pioneering work in this field obtained NPCs for transplantation from developing spinal cord tissue, in which the spinal motor neurons usually did not survive, leaving behind a vast range of developing SpINs. Regardless of whether these tissues were: (i) mechanically dissociated and transplanted into the injured spinal cord8,9; (ii) mechanically and chemically dissociated and transplanted10-12; or (iii) dissociated and cultured to yield neuronal and glial lineage-restricted precursors,13,14 the donor cells used were consistently rich with SpIN progenitors. Transplanting tissue that has been only mechanically dissociated is likely the least disruptive to the full complement of SpIN progenitors, whereas culturing these tissues is a selective process that alters the resulting donor cell phenotypes. 15 Compared to gene expression in the normal developing spinal cord, cells cultured for 2 days exhibit a significant downregulation of transcription factors associated with V2a and V0 SpINs, with some retention of Engrailed 1 expression, which is associated with V1 SpINs. This apparent selection for inhibitory V1 interneurons, and potential loss of V2a and V0 SpINs, may have a great impact on the potential of these donor cells to repair the injured spinal cord. 15 This finding further supports the need for enhanced control over the preparation and engineering of donor cells for transplantation.
Regardless of the method used, however, obtaining donor NPCs from developing spinal tissues is suboptimal from a translational perspective. Therefore, the pioneering work of Shinya Yamanaka in developing human induced pluripotent stem cells (iPSCs) from readily available cell sources (eg, skin fibroblasts) 16 represents a significant advance in cellular engineering that may change the way injured neural tissues can be repaired. By culturing iPSCs or embryonic stem cells with a series of morphogens, researchers can differentiate them into distinct populations for transplantation to restore function following SCI.17,18 Notably, engineering neuronal precursors from stem cells likely also results in a heterogeneous mixture of SpIN progenitors for transplantation, which may be quite different from donor cells obtained from developing spinal tissue.
In the pre-clinical animal studies that have been conducted so far, transplanted human PSC-derived neurons have been shown to extend axons and promote recovery following SCI through functional integration into the host spinal cord circuitry.19,20 These promising results have even led to the advancement of early PSC-derived products to clinical trials for SCI in Japan. 21 However, the specific identity and function of the PSC-derived neurons responsible for reconnecting disrupted spinal cord circuits remains unknown. This may limit the therapeutic potential of the approach, and the development of a more specific cell therapy for SCI is necessary to improve safety and efficacy. Key questions remain: What donor cell populations are needed to repair specific networks that were compromised by SCI, and how pure do they need to be? Might a cocktail of cells be more optimal to promote repair and recovery?
Growing knowledge of the cues that drive cell development and phenotypic fate has also provided new opportunities to direct NPC fate toward specific subpopulations of SpINs. 17 The careful characterization of SpINs (1) that contribute to function within intact motor, sensory or autonomic networks, or perhaps (2) that can effectively contribute to plasticity and functional recovery after SCI, may enable engineering of these populations for transplantation. One such cell type is the excitatory pre-motor V2a SpIN, which has been shown to be anatomically connected with the locomotor 22 and respiratory networks.23,24 Following high cervical SCI, the number of V2a SpINs that are connected to the phrenic motor network significantly increases. 24 Preliminary assessments also suggest that the number of V2a SpINs connected to the injured phrenic network correlates with the extent of functional plasticity and degree of recovered diaphragm function. More recently, transcriptomic analysis of injured spinal cords from animals treated with rehabilitation and/or epidural stimulation found that V2a SpINs are key components of the therapeutically driven locomotor recovery in rodents. 25 Accordingly, these cells represent an excellent candidate for targeted cell transplantation strategies, from commonly used cell sources summarized in Figure 1. In fact, enriching donor NPCs with V2a neurons significantly improved phrenic motor recovery following cervical SCI. 15 Building off our growing knowledge of SpINs and plasticity post-SCI, we propose that donor NPC preparations can be tailored for transplantation of specific SpIN subtypes, or even cocktails of SpIN sub-populations that are known to contribute to restoration of function.
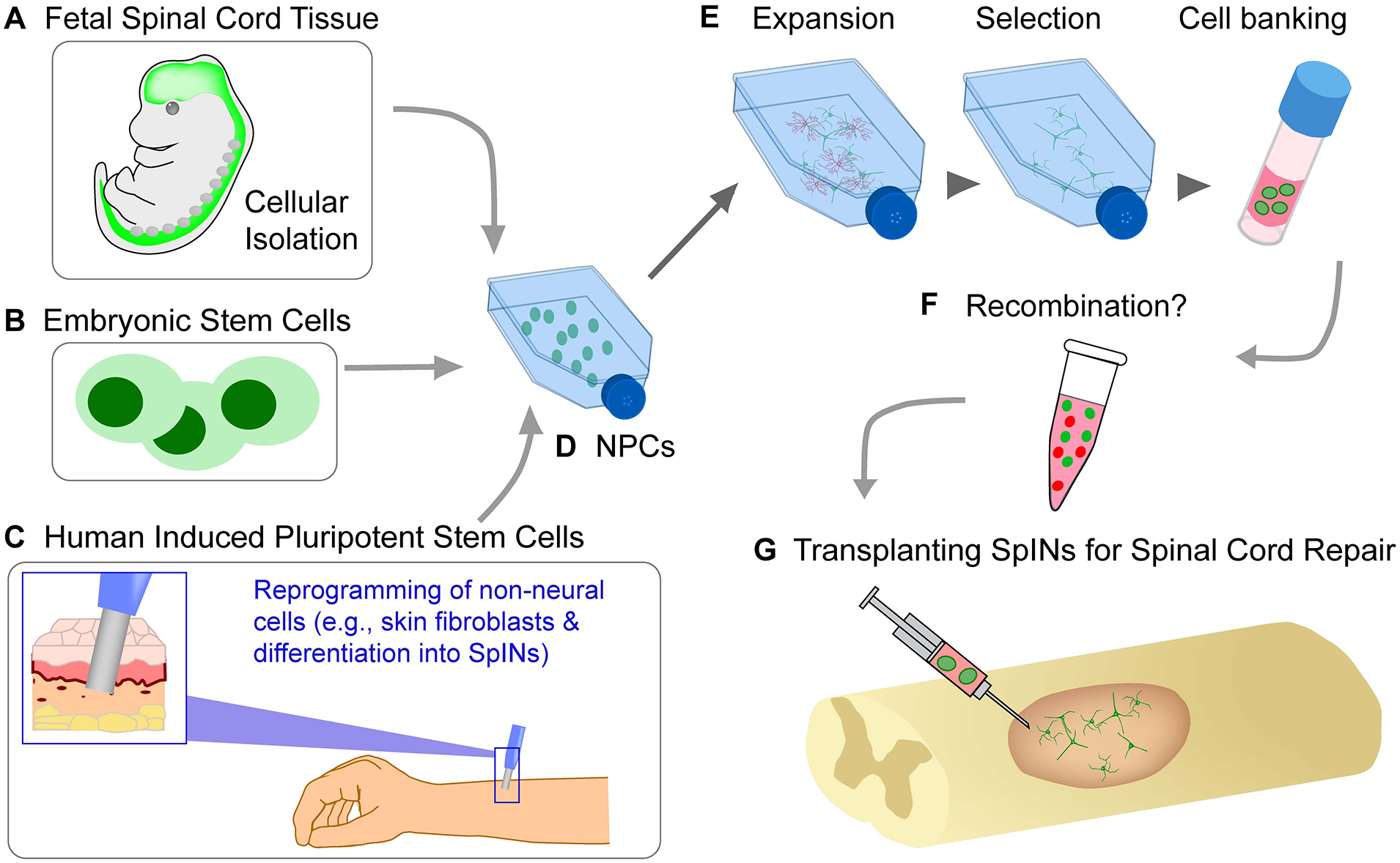
This schematic diagram highlights 3 commonly used cell sources (A-C) for obtaining neural progenitor cells (NPCs, (D)) inherently rich in interneuronal populations. The process of isolation and preparation of cells for transplantation (eg, expansion, selection and cell banking of specific phenotypes, (E)), allows for cellular engineering of specific subtypes of spinal interneurons. Multiple phenotypes of neuronal and non-neuronal precursor cells can be recombined (F) and transplanted (G) into the injured spinal cord.
Closing Remarks
There’s little doubt that SpINs, like most neural cells, are highly plastic. Major questions for the field moving forward are how we should characterize SpINs, and how crucial is it to understand their phenotypes—and in what detail—in order to develop effective, timely treatments. What defines a cell type? For instance, is it sufficient for a cell to express a specific combination of transcription factors (eg, ISL1, LHX3, HB9) for it to be classified as a spinal motor neuron? Or does it also need to project an axon from the spinal cord into the periphery to innervate its target organ? If it loses any of these characteristics, it is no longer a motor neuron? With that in mind, what features must SpINs possess to be harnessed therapeutically for spinal cord repair? If we transplant a single type or cocktail of SpINs into the injured spinal cord, is it problematic that they do not maintain their “defining” characteristics? Perhaps the “identity” of the cell is defined by the experimental approach at the time of the measurement, and cells, can in fact be multiple “identities” at the same time, and even equally efficacious. A useful metaphor is the dual nature of light, which has the “identity” of a wave as well as the “identity” of a discrete quantity of energy (eg, photon), both of which are differentially described depending on the tools used to study light at the time of the measurement.
The future of stem cell engineering and neural cell therapy is filled with promise for all those living with neural injury and disease. With the first stem cell clinical trial being used to treat individuals living with spinal cord injury it will be especially exciting to see advances for those individuals with now more refined and tailored treatment approaches. Ongoing preclinical studies in parallel will be crucial to better understand the optimal application of engineered cell therapies (timing, dose, and phenotypic cocktail to be used). Consideration for the phenotype of non-neuronal cells in transplant preparations, such as glia, and endothelial cells, will also become important and these elements may need to be engineered to optimize repair interventions. Similarly, we will need to better understand how the injured cellular and molecular environment changes following transplantation, either working in concert with donor cells to facilitate repair, or inadvertently resulting in changes to the donor phenotypes. Finally, whether the phenotype of transplanted neurons will be retained long term after being transplanted into the injured spinal cord needs to be further explored. Given the demonstrated plasticity in SpIN populations, can the phenotype of donor cells change? Ongoing research is beginning to address these important questions to comprehensively understand the therapeutic potential of these cells and the promise of cellular engineering and transplantation for neural repair.
Footnotes
Acknowledgements
Our sincerest gratitude to the Gladstone Editing Team for their helpful edits on the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are supported by National Institutes of Health, NINDS, R01 NS104291 (MAL), and F32 NS119348 (LVZ), and the Lisa Dean Moseley Foundation (MAL).
Declaration of Conflicting Interests:
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Both authors contributed equally to this article.
