Abstract
Background:
Rare diseases (RDs) encompass over 6000–8000 conditions, with 94% lacking available therapies. These conditions affect 400 million people globally, including three million Canadians, who face numerous challenges throughout their healthcare journey. Patient engagement (PE) is increasingly recognized as essential for improving outcomes yet remains inadequate in RD and orphan drug research particularly in Canada, where a national strategy for integrating RD patients’ perspectives is lacking. To address this gap, this paper presents a Rare Disease Patient Engagement Framework (RDPEF), a structured model designed to support meaningful PE across all levels of healthcare, including research.
Objectives:
To develop a RDPEF that addresses barriers to engagement, reduces stigma, and incorporates patient experience as a core element in RD and orphan drug research and decision-making.
Design:
A conceptual framework development study informed by qualitative research and a targeted review of existing PE frameworks.
Methods:
The RDPEF was developed using a systematic approach that combined a review of existing literature on PE frameworks with new qualitative research on the experiences of RD patients in Canada. Semi-structured interviews examined patients’ healthcare journeys, focusing on disease management, access to orphan drugs, and opportunities for engagement. A thematic analysis of the existing literature and interview data identified common challenges, which guided the framework’s design. The RDPEF integrates elements from various other PE models, customizes them to the specific needs of RD patients, and emphasizes engagement across the entire orphan drug lifecycle.
Results:
Thematic findings from qualitative research highlighted limited to no patient involvement beyond clinical trials, significant stigma and discrimination, and the absence of structured engagement in drug review and reimbursement processes. These insights informed the development of the RDPEF, which outlines levels and forms of engagement, guiding principles (including stigma reduction), and mechanisms for integrating patient experience across healthcare, policy, and research domains.
Conclusion:
The RDPEF is a timely tool for enhancing PE in orphan drug research. By addressing engagement barriers, reducing stigma, and centering patient experience, the framework offers a roadmap for patients, researchers, healthcare providers, and policymakers to create a more inclusive and responsive system for RD patients in Canada.
Plain language summary
Rare diseases (RDs) include over 6,000 conditions that affect about 400 million people worldwide, with many lacking effective treatments. In Canada, rare disease patients face significant challenges, such as limited access to therapies, insufficient research participation, and a lack of integration in healthcare decisions. Patient engagement (PE) is increasingly recognized as essential for improving care and outcomes, but it is often inadequate in RD and orphan drug research. This paper introduces the Rare Disease Patient Engagement Framework (RDPEF), a new approach to enhance patient involvement across all aspects of healthcare, including research. The RDPEF was developed through a combination of reviewing existing patient engagement models and conducting new research on the experiences of RD patients in Canada. Semi-structured interviews with patients provided insights into their healthcare journeys, focusing on challenges like stigma, discrimination, and limited access to orphan drugs. These interviews revealed the need for a tailored engagement approach specific to RD patients. The framework incorporates findings from the interviews and elements from existing PE models, with a focus on the entire lifecycle of orphan drugs — from development to post-market use. The RDPEF aims to shift RD patients from being passive recipients of care to active collaborators in research and treatment development. By addressing barriers to engagement, reducing stigma, and emphasizing the importance of patient experiences, the framework fosters a more inclusive, patient-centered model of care. This tool is timely and necessary for enhancing PE in RD and orphan drug research in Canada. It provides a roadmap for patients, researchers, healthcare providers, and policymakers to create a more responsive healthcare system that values and integrates the perspectives of RD patients, ultimately improving research outcomes and the quality of care they receive.
Keywords
Introduction
Rare diseases (RDs) pose a significant global challenge, affecting over 400 million people worldwide, including 3 million Canadians.1,2 Of the 6000–8000 identified RDs to date, 94% lack available therapies. 3 Patients with RDs often experience a “diagnostic odyssey,” characterized by delayed diagnoses, inadequate access to social and healthcare services, and limited availability and accessibility of orphan drugs.4,5,6 In Canada, efforts to address these challenges have been fragmented due to the lack of a cohesive national strategy.7,8 After more than 25 years of discussions on establishing a pan-Canadian strategy for RDs and orphan drugs, the federal government announced in 2019 its plan to establish a national orphan drug strategy,9,10,11 which was formalized in March 2023 12 and launched in July 2024 through a bilateral agreement with British Columbia. 13 This pilot implementation involved two orphan drugs. 13 Despite this progress, patient engagement (PE) strategy remains underdeveloped, particularly in drug regulatory review and reimbursement decision-making processes (our use of patient engagement includes engagement with patients, caregivers, families, patient groups and other cognate stakeholders).
RDs have emerged as a significant public health concern, emphasizing the need for enhanced PE in RD and orphan drug research. RDs are typically characterized as heterogeneous, complex, chronically debilitating, and life-threatening conditions that impact a small percentage of the population.1,2 RD patients typically face a number of challenges throughout their journey, including lack of available therapies for the majority of RDs, high costs of orphan drugs, delayed diagnoses, limited access to specialized social and healthcare services, and significant barriers to obtaining available treatments. As a result, effective PE in the context of RD, generally, and orphan drugs in particular, is challenging. The physical, financial, and emotional burdens of managing a RD can limit patients’ capacity to engage actively. 4 The complexity and heterogeneity of rare conditions further hinder engagement, as even patients with the same rare disease may have varying needs and experiences.14,16,17,65 The healthcare landscape for RD patients is further complicated by factors such as limited engagement opportunities and fragmented care approaches, particularly in countries like Canada.4,6,8,18
Despite the implementation of a national orphan drug strategy in Canada in July 2024, significant gaps persist in incorporating patient perspectives into critical decisions related to their disease management. These gaps are particularly evident in the exclusion of patient voices from discussions on RD and orphan drug research, especially during the regulatory review and reimbursement decision-making stages that determine access to these therapies. 13
Recognizing the importance of PE, many jurisdictions emphasize its role in improving the quality of care, health outcomes, policies, and systems.19–23 However, the degree of patient involvement and the methods used to implement it vary significantly across regions.17,24 PE is defined by the World Health Organization (WHO) as “the process of building the capacity of patients, families, carers, as well as healthcare providers, to facilitate and support the active involvement of patients in their own care, in order to enhance safety, quality, and people-centeredness of health care service delivery.”25[p. 3]
This study addresses critical gaps in PE for RDs by first synthesizing insights from semi-structured interviews with patients and caregivers. We then conducted a review of established PE frameworks to identify essential components for the RD context. Building upon this analysis, we propose a patient-centered Rare Disease Patient Engagement Framework (RDPEF) designed to be both relevant and applicable to the unique challenges of the RD landscape. The RDPEF provides a structured, multi-level approach to integrating patient voices throughout the healthcare continuum, including direct care, research and program design, organizational governance, and policymaking. This article focuses specifically on PE in orphan drug research, spanning all phases of the drug lifecycle. It explores how the RDPEF can address engagement barriers, reduce RD-related stigma, leverage patient experience as a key metric, and elevate the role of RD patients as collaborators in orphan drug research.
Method
The RDPEF was developed through a systematic approach that combined a review of existing literature on PE frameworks and new qualitative research using semi-structured interviews with 30 Canadians affected by RDs. For the semi-structured interviews, participants were recruited from across Canada using first purposive sampling and then snowball sampling to ensure diverse representation of RDs. Eligible participants included adult patients or caregivers with a diagnosis of a RD. Recruitment was facilitated via websites, newsletters, and social media platforms of national rare disease organizations and foundations. Of the 30 interview participants, 16 were patients and 14 caregivers. All participants had navigated diagnosis, treatment access, and interactions with healthcare systems within the previous 3 years and were well-positioned to reflect on the rare disease care experience, healthcare systems, and engagement landscape (See Supplemental Appendix A for study details). Interview data were analyzed thematically, focusing on participants’ experiences with navigating healthcare systems, managing their rare conditions, accessing orphan drugs, and engagement opportunities in various levels of healthcare. 4 A review of existing literature on PE and other relevant frameworks further informed the analysis and recommendations drawn from the interview data. The review of literature included scholarly articles and books published during a period of two decades (between 2000 and 2019), as well as relevant reports issued by the Government of Canada and other OECD countries. The literature review process included searching databases such as Google Scholar, PubMed, Orphanet, and the websites of rare disease organizations. Search terms included a combination of: “rare disorder,” “rare disease,” “ultra-rare,” “orphan drugs,” “drugs for rare diseases,” “reimbursement,” “patient engagement,” “patient experience,” “health stigma,” “discrimination,” “decision-making,” “policy,” “research,” and “framework.” Sources were screened for relevance to the study’s aims and excluded if they lacked publication dates, were unpublished reports, or were limited to abstracts only.
Based on the literature review, key ingredients for a RD-specific PE framework were identified from existing prominent PE frameworks such as Carman et al.’s 26 Patient Engagement Framework, Stangl et al.’s 27 Health Stigma and Discrimination Framework, and the NHS Leeds 28 Patient Experience Framework. The selected frameworks from the literature were chosen for their conceptual rigor, applicability to rare disease policy, and alignment with the RDPEF’s objectives. Each provides a distinct but complementary perspective—on engagement levels (Carman et al. 26 ), stigma mechanisms (Stangl et al. 27 ), and patient experience within service delivery (NHS Leeds)—forming an integrated foundation for this tailored engagement model.
Insights from the interview results and key elements from three established frameworks were then integrated to formulate a RD-specific patient engagement framework into the RDPEF. The resulting RDPEF includes guiding principles, different levels and forms of engagement, and metrics for measuring patient experience and evaluating engagement activities.
Results
Qualitative study results
The analysis of patient interviews revealed several challenges faced by RD patients throughout their healthcare journey, including delayed diagnoses, limited access to specialized social and healthcare services, and difficulties in obtaining lifesaving therapies. 4 A common, recurring theme was the lack of engagement in healthcare decision-making and the exclusion of patients from decisions regarding their disease management and access to orphan drugs. Additional key issues highlighted by patients included experiences of health stigma and discrimination, as well as the omission of their input and experience in advancing scientific knowledge about their disease and in the research, development, and accessibility of orphan drugs. 4 The key findings are summarized as follows:
Patient engagement in orphan drug research
A recurring theme in the study was the lack of patient involvement throughout the orphan drug lifecycle, beyond participation in clinical trials, especially during the regulatory review and reimbursement decision-making stages. Only one participant (3%), affiliated with a research organization, reported serving on government-hosted advisory committees, and even then, their engagement was informal and ad hoc. As one Pompe patient explained, “It’s not a formal process. They were ad-hoc committees based on patient input to criteria for approval of the drug.” Notably, no participants reported involvement in reimbursement decision-making. Instead, engagement activities were largely limited to lobbying, contacting government officials in person or via mail, and using media to pressure governments to fund life-saving drugs.
The keen desire for comprehensive engagement with the orphan drug lifecycle was a dominant theme among participants in this study. Patients and caregivers alike asserted that such involvement should be both meaningful and structured, but that, essentially, they “would like to be more involved” (patient, Pompe). However, no patient-directed policy framework currently exists to steer patient involvement in decisions concerning orphan drugs in Canada.6,29,30 Patients specifically expressed their willingness and ability to take more active roles throughout all stages of the orphan drug lifecycle, extending beyond clinical trials to drug review and reimbursement decision-making. As one patient with Pompe disease articulated this need, stating, “I think insights provided by patients are very valuable. The regulators and decision-makers don’t know the specific things that really concern the patient; sometimes it’s not what they think.”
This sentiment was echoed by other participants who believe their experiences and insights could significantly enhance the research, development, and access to orphan drugs, contributing to the generation of RWE. Another participant with Morquio disease emphasized the importance of PE in the drug review process and generating RWE: “The real-world evidence is going to be helpful. You need to consider the patient’s expertise in that [drug review process] . . . Involve patients in the process to understand what they’re going through!” A parent of a child with Morquio disease emphasized the importance of their expertise in drug decision-making, saying, “The parents and the caregiver should be completely involved in the decision-making process. The reason number one is: we have been dealing with them every day and we are monitoring them closely. We know what changes they have been dealing with and what other challenges they have been facing. So, I think the parents should be involved in the decision-making process. I strongly recommend that.”
Impact of health stigma and discrimination
The findings from the qualitative study highlighted how health stigma and discrimination affected patients with RDs at all three levels: micro, meso, and macro. Participants described the profound effects of rare disease stigma on their social lives and relationships throughout their journeys.
In terms of intrapersonal stigma, many patients expressed the need to conceal their conditions, especially when symptoms were not outwardly visible. One patient with Fabry disease shared his struggle to keep his condition, including severe daily pain, a secret for decades: “I kept my disease secret . . . I never talked about it because I didn’t want people to know . . . I started to tell people this . . . when I was about just above 40 years old. That’s how long it took me before I mentioned it to anybody . . . none of my friends knew that I had anything. To them I look normal, I acted normal, but I kept it a secret . . . It was really hard to tell anybody to understand.” His experience underscores how internalized stigma can lead to isolation and a reluctance to share one’s reality with others.
At the meso level, personal experiences reveal the social isolation many face. A mother of a child with Hunter Disease shared her observations following her son’s diagnosis: “Socially, like some of the friends that we have, we don’t see anymore. In his first year of life, we were quite social with other friends that had children as well. And then he got diagnosed and I felt personally that it was almost as if those people didn’t want to expose their children to a child that was going to deteriorate and die. So, we didn’t see them anymore. It was almost like an immediate drop-off on being invited to any kind of outing with the same people as their children.” This illustrates the impact of stigma on personal relationships, leading to a withdrawal from social circles.
At the macro level, stigma can manifest in practical barriers to participation in community activities. The mother of a child with Hunter Disease recounted her difficulties enrolling her son in recreational programs: “People discriminate based on his disability of being non-verbal and not being as cognitive as the typical five-and-a-half-year-old and they don’t want them to go to their camp and demand that I send a one-on-one worker with him and I have to pay for that person out of my pocket . . . they will only say yes, he’s allowed to come there for the first week, and they will decide whether he’s welcome to come back after that. And that’s even with a one-on-one worker!” She further noted the daily challenges she faces, stating, “People are making judgments about my child based on a disease that they’ve never heard of.” This highlights how stigma not only affects access to services but also perpetuates discrimination within community settings.
Further complicating matters, RD patients expressed feelings of inequality in treatment access compared to those with more common conditions. Some even wished for a cancer diagnosis instead of a RD, as it would grant them better access to treatment. They emphasized their right to be treated equally. A participant with Gaucher disease articulated this sentiment, stating, “I’m a person just like anybody else . . . I was a tax-paying citizen just like anybody else. I’m a Canadian just like the next Canadian receiving healthcare . . . I go back to cancer, even though it’s an unfair comparison . . . but they don’t turn people away for chemo!” The reference to cancer points to the notable discrepancies in PE in Canada, particularly for access to therapies. In cancer treatments, initiatives such as the pan-Canadian Oncology Drug Review (pCODR) actively incorporate patient input throughout drug evaluations, emphasizing engagement as a core value. 31 The pCODR develops specific guidelines to inform patients about their participation in the drug review process, outlining eligibility requirements, feedback submission, and the utilization of patient input. This ensures that the voices of Canadian patients with cancer are not only heard but also considered in funding decisions. 31 In stark contrast, RD patients lack structured engagement strategies or support in the drug review process.17,30 Existing PE initiatives from federal and provincial governments remain informal and scarce, leaving these patients feeling marginalized and voiceless.4,17,30 This absence of engagement contributes to a profound sense of discrimination, as their input and experiences are frequently overlooked compared to those with other conditions, such as cancer patients.4,30
These insights underscore the urgent need for stigma reduction interventions that prioritize patient involvement and foster a more inclusive PE to care for and support, those living with RD.
Importance of patient experience measurement and analysis
Patient experience refers to the cumulative interactions that patients have with healthcare systems, professionals, and facilities. It encompasses the evaluation of specific events in healthcare settings, including how and how often they occur, as assessed by patients.32–35 Additionally, patient experience involves collecting and utilizing patient feedback to inform and improve policies, practices, and service delivery. 36 It serves as a vital tool for program planning, service redesign, and quality assurance in healthcare.28,32,34,35
Incorporating “patient experience” has become a cornerstone of PE as part of the growing efforts to expand the role of patients and their caregivers from being mere information-providers to becoming co-collaborators in the design and delivery of services. Our study highlighted that RD patients recognize the unique value of their experiences and insights in disease management, especially during drug evaluations in both pre- and post-market periods. They emphasized that their contributions play a crucial role in enhancing RWE. Participants noted the importance of structured ways to measure their experiences. A patient with Pompe disease shared, “I wish there was a way to show what we really go through, it would help decision-makers understand our everyday needs and struggles . . . If they can see and listen, things might change!” This desire to bring patient experiences to light underscores the importance of having their voices not just heard, but also counted, influencing meaningful changes in healthcare. By capturing patient experiences through metrics, health officials can better understand the real impact of therapies, promoting engagement and ensuring healthcare systems meet patient needs. Another patient diagnosed with Morquio emphasized the importance of measuring and considering patient experiences in decision-making, noting that this could lead to improved health outcomes. They stated, “if they say we will give you the medication, and we will monitor the situation. If everything is fine, we will continue. If not, we will stop . . . They were saying that the Vimizim doesn’t work for Morquio patients over the age of 12, . . . [but] I have seen many older patients getting Vimizim and it is doing tremendous change in their life . . . So, the experts’ decisions to deny older patients is totally wrong.”
Discussion
Addressing gaps in patient engagement across the orphan drug lifecycle
To fully understand patient engagement in the RD context, it is important to recognize the stages of the “orphan drug lifecycle,” which encompass the entire process of orphan drug research (Figure 1). This lifecycle begins with pre-clinical and clinical trials, followed by regulatory review and reimbursement decision-making, then moves into clinical practice, and concludes with post-marketing studies. 17
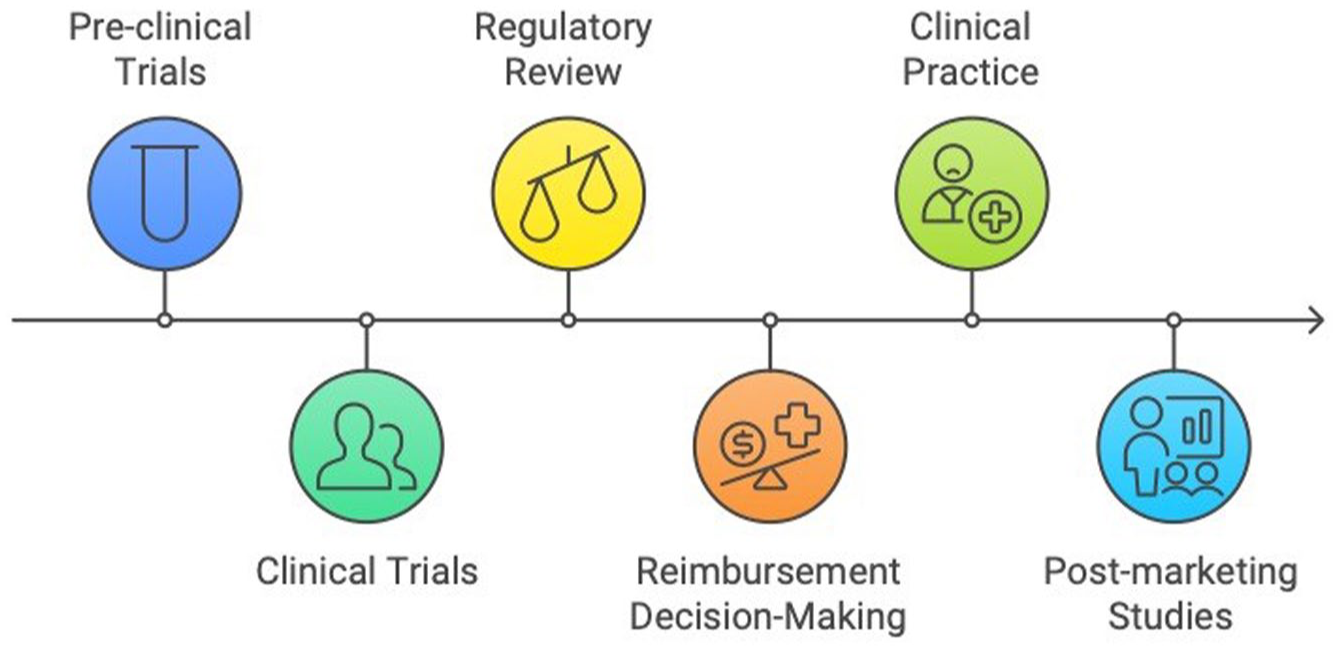
Orphan drug lifecycle. This figure illustrates the stages of the orphan drug lifecycle, highlighting the key phases from pre-clinical trials through to post-marketing studies. The lifecycle includes pre-clinical trials, clinical trials, regulatory review, reimbursement decision-making, clinical practice, and post-marketing studies.
Pre-clinical trials involve laboratory and animal studies to assess a drug’s safety, biological activity, and potential toxicity before human testing. If successful, the drug moves into clinical trials, which are conducted in phases (I, II, and III) to determine safety, dosage, and efficacy in humans. Regulatory review follows, where bodies such as Health Canada, FDA, or EMA assess clinical trial data to determine a drug’s safety and efficacy and decide on its approval for marketing. After approval, reimbursement decision-making involves public or private payers conducting a Health Technology Assessment to evaluate the drug’s cost-effectiveness and coverage. 17 The final stages of the orphan drug lifecycle include its use in clinical practice and post-marketing studies, which gather long-term data on the drug’s safety and effectiveness in real-world settings.
Existing patient engagement initiatives in RD focus predominantly on clinical trials (33% (n = 10) of respondents in this study reported their participation as study subjects in clinical trials. Participation in other research activities was further limited: 10% (n = 3) were involved in data collection for pre-clinical trials, 7% (n = 2) participated in post-marketing studies, and 50% (n = 15) joined registries). While participation in clinical trials is a recognized form of PE, it represents only one aspect of the broader involvement necessary throughout the orphan drug lifecycle. Effective PE requires patient input at every stage of the drug lifecycle, from early research design to access decisions and post-marketing evaluations.
The exclusion of patient voices from discussions on RD and orphan drug research, especially during the regulatory review and reimbursement decision-making stages, creates a significant gap in Canada’s evolving strategy. 13 Our findings demonstrate that patients possess a keen desire for comprehensive and meaningful engagement throughout this lifecycle, yet their reported activities were largely limited to lobbying and informal, ad-hoc roles.
Patient input in the regulatory review and reimbursement processes is crucial, as it provides insights into real-world experiences with diseases, including symptoms, side effects, and the overall impact of treatments on daily life. This input is particularly valuable in RD research, where achieving statistical significance in clinical trials is challenging due to small patient populations.24,30 Such limitations often delay access to innovative therapies.24,30 Real-world data (RWD), centered on patient-reported outcomes and experiences, can help bridge the gap created by limited clinical evidence from small trials, addressing uncertainties about the effectiveness of new treatments.15,17,30,37 Consequently, RWD plays a critical role in enabling health officials to make informed decisions about drug approval and coverage, ultimately facilitating faster and safer access to life-saving therapies.24,15–17,37–39
Furthermore, incorporating PE in the regulatory review and reimbursement processes can enhance overall drug evaluation, increase patient trust in these processes, potentially reduce the number of appeals against negative reimbursement decisions, and facilitate the implementation of drug review recommendations.4,30 Increased transparency in decision-making about how and when to incorporate patient perspectives can also be achieved through PE at this stage.17,18,30
Furthermore, expanding engagement into the often-overlooked clinical practice and post-marketing stages is equally vital, as it aids in generating real-world evidence (RWE) that integrates insights from clinical practice, patient experiences, and observational data. 40
Overall, PE throughout the orphan drug lifecycle offers essential information regarding the performance of current policies and programs that influence access to orphan drugs. This comprehensive involvement provides a fuller picture of treatment effects and long-term outcomes, and it can be leveraged to improve healthcare, enhance the management and monitoring of orphan drug programs, inform national strategies for orphan drugs, and promote strategic regulatory decision-making and patient safety.17,22,23,39,41
The gap between patient willingness and actual involvement underscores a systemic failure to leverage a critical evidence base.
The findings from our semi-structured interviews emphatically underscore the critical need for a structured patient engagement framework tailored specifically to the RD population. To achieve this, we identify the key building blocks for a RDPEF by reviewing and adapting existing engagement frameworks to the RD context. The resulting RDPEF is designed to help healthcare systems respond effectively to patient needs, ultimately leading to improved health outcomes. It highlights the importance of patients’ active involvement in healthcare decisions while addressing significant barriers to engagement, such as health stigma and discrimination, which remain underrecognized in the RD domain despite their prevalence.42–46 Furthermore, the framework emphasizes the value of patient experience as a key measure for identifying patient needs and assessing the effectiveness of engagement interventions.
The integral role of patient engagement frameworks
Patient Engagement Frameworks from various jurisdictions—such as Canada, the United States (US), the European Union (EU), and the United Kingdom (UK)—have recognized the unique value patients bring through their lived experiences of managing specific illnesses and their insights into the potential health outcomes of various treatments.19–23,47,48 Several strategies and frameworks have emerged to facilitate and measure PE. Notable examples include Angela Coulter’s 21 roadmap for engaging patients in the UK, the Institute for Healthcare Improvement’s (IHI) Framework for Safe, Reliable, and Effective Care in the US, Carman et al.’s 26 Patient and Family Engagement Framework, and Ontario’s Patient Engagement Framework. 33 These frameworks offer valuable guidance on involving patients and families in healthcare decision-making and designing patient-centered health systems.
Coulter’s roadmap emphasizes building effective partnerships between healthcare providers and patients, proposing strategies such as consultation, professional training, and institutional restructuring to enhance engagement.21,49 Similarly, the IHI framework highlights patient and family engagement as a key component of operational excellence, recognizing that patient perspectives can drive improvements in healthcare systems. 45 However, both Coulter’s and the IHI’s frameworks focus primarily on the micro-level of patient engagement, without addressing the integration of patient voices at the policymaking level.
In contrast, Carman et al.’s 26 multidimensional framework provides a more comprehensive approach by incorporating various forms and levels of PE. It identifies a continuum of engagement, ranging from consultation—where patients have limited decision-making power—to partnership and shared leadership. 26 This framework outlines how patient roles can evolve at different levels of engagement, supporting the development of strategies that promote meaningful engagement.
Carman et al.’s framework further distinguishes different levels of engagement: direct patient care, organizational design and governance, and policymaking. This multi-level perspective highlights the importance of integrating patient values and experiences into every aspect of healthcare, from individual care interactions to broader organizational and policy decisions. The framework also acknowledges various factors that can influence engagement, such as patient characteristics, organizational policies, and broader socio-political contexts. By considering these factors, healthcare organizations can create environments that promote higher levels of PE across the care continuum. 26
Additionally, the framework supports the assessment of existing patient-reported outcomes and experiences or the development of new measures to evaluate the effectiveness of engagement activities at all levels. It allows for tracking outcomes for each intervention, ensuring that engagement efforts are meaningful and impactful at every level and stage.
Carman et al.’s framework has been widely adopted by healthcare institutions to guide patient engagement in service delivery, program design, and policymaking. This framework is especially relevant to RD and orphan drug research due to the challenges arising from the heterogeneity of RDs, as it considers factors that influence patient’s ability to engage, including poor health and low health literacy—both common in RD patients. Importantly, Carman et al. 26 highlight that the highest forms of engagement, such as partnership and shared leadership, may not always be feasible or lead to better outcomes, especially at the policy-making level. They advocate for moving patients along the continuum of engagement, gradually increasing decision-making power as evidence of improved outcomes accumulates. This is particularly relevant in RDs due to the lack of evidence of the benefits of power-sharing in engagement interventions. Carman et al.’s 26 framework is also relevant to RDs and orphan drugs, as it supports the use of RWE through the creation and analysis of patient-reported outcomes and experiences for assessing outcomes.17,30 Therefore, by building upon the comprehensive, multi-level foundation of Carman et al., the RDPEF is designed to tailor these principles specifically to the orphan drug lifecycle and the unique vulnerabilities of the RD population, while integrating crucial additional dimensions of stigma and patient experience.
Confronting health stigma and discrimination as a barrier to engagement
A second set of findings from our interviews underscores the impact of stigma and discrimination on RD patients. Any PE framework for the RD sector ought to recognize and integrate the pervasive impact of health stigma and discrimination identified in our study. Stigma is widely recognized as barriers to engagement in healthcare and in seeking care and wellness.27,51 They create serious health disadvantages for stigmatized individuals, including patients with complex conditions.27,44,52–57 Due to its pervasiveness and its disruption of various life domains—such as access to resources and physical and psychological well-being—stigma is considered a crucial social determinant of health.27,53–57
Stigma exists at intrapersonal (micro), interpersonal (meso), and structural (macro) levels 58 ; however, most studies on stigma have primarily focused on intrapersonal and interpersonal aspects, analyzing how stigmatized individuals perceive and respond to stigma across health domains such as HIV, obesity, and mental illness.52,58 This leaves a gap in understanding and addressing structural stigma across diverse health conditions, including RDs. 27 In the context of RDs, health stigma at the structural level has often been an overlooked barrier to PE in decision-making and in seeking treatment and support, contributing to health disadvantages and disparities.42–46 Our interview participants expressed a sense of discrimination, particularly when comparing their access to treatment with that of cancer patients. This perspective highlights how macro-level policies significantly limit PE access for patients with RDs, a clear manifestation of structural stigma.52,58,59
These examples illustrate how the pervasive nature of health stigma across different levels disrupts the lives of individuals with RDs. Micro and meso-level stigma can discourage patients from participating in clinical trials and openly communicating with healthcare providers, limiting their access to resources and support networks. Meanwhile, structural stigma marginalizes patients, making them feel unwelcome and unsupported in healthcare settings, thereby actively discouraging their engagement in treatment and research.
Therefore, a structured, stigma-sensitive PE framework is essential to address this gap and tackle stigma at all levels. This is crucial for promoting PE and improving health outcomes for individuals with RDs. In 2019, Stangl et al. introduced a comprehensive health stigma and discrimination framework designed to connect stigma with its health and social impacts. 27 This framework addresses social and structural stigma, aiming to reduce its detrimental effects on health outcomes. It has been applied to various health conditions, including cancer, mental illness, leprosy, epilepsy, HIV, and obesity. The framework emphasizes the importance of eliminating stigma facilitators and drivers to combat health stigma effectively. Once stigma is established, it focuses on mitigating its harmful consequences. Stangl et al.’s framework is particularly relevant for RD patients as it provides a nuanced understanding of the complex experiences of vulnerable and marginalized populations, such as those with RDs. It offers practical strategies to recognize and address stigma’s facilitators, drivers, and manifestations while proposing interventions for monitoring stigma at both program and national levels, filling a significant gap in previous stigma frameworks.
Stigma reduction interventions
Stangl et al. 27 emphasize that a single-level approach is inadequate for addressing health stigma and discrimination, given the complex factors that sustain stigma, requiring a system-wide response.60–63 Various stigma-reduction strategies have been developed including:
Information-based interventions aim to raise awareness among both stigmatized and non-stigmatized individuals about patients’ conditions and available resources. These interventions enable patients to make informed health decisions, develop coping strategies, and maintain stability throughout their disease journey.27,61 Additionally, increasing awareness and promoting knowledge within society and organizations support patient-centered programs and improve health professionals’ attitudes and behaviors toward stigmatized individuals, enhancing their care. 63
Contact interventions facilitate interactions between those affected by specific diseases and key societal figures (e.g., legislators, policymakers, employers, etc.), effectively reducing stigma, especially when combined with education.27,60,61
Advocacy interventions play a critical role in reducing stigma at all levels.27,61 They focus on influencing government policies to ensure equitable access to care for stigmatized populations, while also seeking recognition and commitment from policymakers to develop patient-centered policies and programs.27,62,64,65
Overall, addressing stigma in health systems requires a comprehensive approach that integrates education, contact, advocacy, and policy changes.27,60
Stigma measurement and monitoring
Stigma measurement is essential for evaluating the effectiveness of stigma prevention and reduction interventions, as well as for refining strategies.27,63 While stigma measurement tools exist for conditions like mental illness, HIV, leprosy, and obesity, no specific tools have been developed for RDs. The most effective way to capture health-related stigma is through patient-reported experiences, as patients and their families offer valuable insights into healthcare barriers and quality.62,63
Carman et al. 26 emphasize the importance of a measurement system within a PE framework to monitor progress and evaluate the effectiveness of PE approaches. For RD patients, it is essential that patient experience measures not only assess and track the impact of PE interventions but also gauge stigma experiences. Employing a combination of quantitative (e.g., surveys and questionnaires) and qualitative (e.g., interviews and focus groups) methods can effectively capture the diverse challenges faced by RD patients, including stigma, while mitigating the limitations posed by small sample sizes.32,62,66,67
Existing frameworks for PE, including Carman et al., 26 often overlook the nuanced realities of health stigma and discrimination. The RDPEF that we develop below incorporates Stangl et al.’s health stigma and discrimination framework into its guiding principles to analyze the impact of stigma on health outcomes and to mitigate its harmful consequences. By incorporating core principles focused on stigma reduction, including sensitivity to stigma, advocacy, education, and care and support, the RDPEF is uniquely positioned to support the complex experiences of patients with RDs.
Measuring and analyzing patient experience to generate evidence
A third set of thematic findings from our interviews revolve around measuring and analyzing patient experience. Assessing patient experience is a widely used method in patient engagement, offering rich data on how patients and their caregivers perceive the quality of care they receive.14,32,68–70 These experiential data have been instrumental in informing health research, guiding program design, and setting priorities,19,20,22,23 as well as driving policy and practice changes. 36 It also supports ongoing quality assurance in healthcare services.28,32,34,35
To leverage patients’ experiential data effectively, health organizations need robust systems for data collection, analysis, and management.32,71 A mixed-methods approach, combining quantitative methods like surveys with qualitative ones such as interviews and focus groups, is often recommended to ensure comprehensive insights and yield reliable and valid data, particularly for patients with complex needs, such as those with RDs.32,66,67
Several frameworks have been developed globally to measure and analyze patient experience, including Staniszewska et al.’s 35 Warwick Patient Experiences Framework (WaPEF), HQO’s 44 Patient Experience Measurement Strategy, the NHS Leeds 28 Patient Experience Framework in the UK, and Oben’s 34 Conceptual Framework for Understanding the Patient Experience. These frameworks integrate patient experiences into health programs and guideline development. For instance, WaPEF emphasizes the importance of patient participation in care, using their experiences as evidence alongside clinical and economic data to develop clinical guidelines. 35 This approach informed the National Institute for Health and Clinical Excellence’s (NICE) guidance and quality standards.
HQO’s 32 Patient Experience Measurement Strategy considers patient experience as a critical source of information for evaluating care, though it stops short of treating it as equivalent to clinical and economic evidence. The strategy aims to standardize patient experience data collection, analysis, and reporting across Ontario, using both quantitative (e.g., surveys) and qualitative (e.g., interviews) methods, with a focus on marginalized patient groups. This framework is designed to enhance patient experience and serve as a foundation for performance monitoring and quality improvement.
Oben’s 34 framework treats patient experience as an essential dimension of healthcare quality, equal to clinical effectiveness and safety. It provides a deep understanding of the patient journey as a “human” experience throughout the continuum of care but does not detail strategies for measuring or analyzing this experience.
The NHS Leeds Patient Experience Framework 28 aims to integrate patient and caregiver experiences into all commissioning activities and decision-making processes to enhance not only the safety and effectiveness of care but also the overall patient experience. 28 Building on the WaPEF, this framework positions patient experience as a core quality indicator alongside clinical effectiveness and safety.
The relevance of the NHS Leeds framework extends to orphan drug programs, particularly in regulatory reviews and reimbursement processes, where uncertainties about drug effectiveness and safety can delay patient access to vital therapies. Patient experience data can help reduce these uncertainties, providing insights and data into potential clinical outcomes and supporting real-world evidence that facilitates quicker and safer decision-making and access to orphan drugs.17,30,38,72
Additionally, the NHS Leeds framework highlights the connection between patient experience and institutional reputation. It recognizes that negative patient experiences can harm an institution’s standing. In Canada, institutions like Health Canada and CADTH have faced criticism for excluding RD patients from decision-making processes in drug reviews and reimbursement decision-making, an approach that is not applied to patients with other conditions, such as cancer. Unlike the engagement strategies in place for cancer drug evaluations (e.g., pCODR), RD patients in Canada often feel marginalized and uninformed about the review processes for orphan drugs. They report a lack of communication channels, transparency, and appeal processes, contributing to feelings of exclusion and discrimination. 73 This has led to tensions between rare disease communities and governmental institutions, who are often perceived as viewing rare diseases as a financial burden.45,46
Globally, drug review programs often rely on clinical and economic evidence to inform funding decisions, yet patient experience data remains underutilized, particularly in the realm of orphan drugs.16,17,24,30,74 Despite progress in integrating patient experiences into RD strategies—such as the UK’s Rare Disease Strategy (RDUK) established in 201375,76—challenges persist in achieving a balanced incorporation of this evidence. There are limited progress and examples of using patient experience as a quality indicator in orphan drug programs and review processes. For instance, the National Institute for Health and Clinical Excellence (NICE) Highly Specialised Technologies (HST) evaluation committee in the UK, have implemented processes to gather patient input, including opportunities for stakeholder consultation during orphan drug reviews.47,77–79 These examples of how patient experience is utilized or weighted in decision-making processes remain limited and unclear, and the impact of such integration on outcomes is not well documented.17,30,80
The reluctance to engage RD patients in decision-making drug reviews and reimbursement decision-making processes is mainly rooted in concerns about the potential financial impact of high-cost therapies with uncertain benefits on government budgets and the overall sustainability of the healthcare system. The continuum of engagement, as envisioned in Carman et al.’s 26 PE framework, allows for the measurement of engagement outcomes, providing a way to balance patient input with system sustainability in the context of orphan drugs. By initially educating patients about their conditions and the drug review process, they can progressively move toward more significant roles in consultation and deliberation, ultimately reaching levels of partnership and shared leadership in the decision-making process.
To ensure effective and meaningful engagement while maintaining the sustainability of the healthcare system, the RDPEF must be measurable. Therefore, it is essential to develop and validate RD-specific measurable patient-reported indicators, such as Patient-Reported Outcome Measures (PROMs) and Patient-Reported Experience Measures (PREMs), which, among other things, include psychometric assessments of stigma. 81 PREMs assess overall patient experiences, evaluate healthcare performance, and drive improvements toward patient-centered care.82–84 Recognized globally as quality indicators for patient care and safety,85,86 PREMs are widely used by OECD countries for national surveys across various healthcare settings.84,87 However, their application at higher levels of healthcare, such as organizational governance and policymaking, remains limited.
Development of the RDPEF
The foregoing discussion of findings from the qualitative study on Canadian patients with RDs and a review of the extant literature demonstrate that a structured rare disease-specific framework is essential to address the PE gap and tackle stigma at all levels. Such a framework is crucial for promoting PE and improving health outcomes for individuals with RDs. In the remaining part of the article, we offer a RDPEF that attends to the critical needs of rare disease patients and caregivers. This multi-level conceptual framework integrates elements from the three key PE models discussed above such as Carman et al.’s 26 Patient Engagement Framework, Stangl et al.’s 27 Health Stigma and Discrimination Framework, and the NHS Leeds 28 Patient Experience Framework. The goal of establishing a structured RDPEF is to ensure that the voices of rare disease patients are heard and considered in decisions affecting their care, ultimately enhancing health outcomes for this vulnerable population.
The RDPEF (Figure 2) is structured around three main components: (1) levels and forms of patient engagement, (2) guiding principles, and (3) patient experience.

Rare disease patient engagement framework (RDPEF). This figure illustrates the three main components of the RDPEF: (1) levels and forms of patient engagement, (2) guiding principles, and (3) patient experience. The framework highlights a continuum of patient engagement, ranging from information-sharing to collaboration, and emphasizes the evolving roles and responsibilities of patients across all levels of healthcare (direct care, research and program design, organizational governance, and policymaking). The RDPEF is guided by core principles, including sensitivity to stigma, education, transparency, responsiveness, advocacy, care and support, and empowerment. At the core of the framework is patient experience, positioned at its center, and consisting of robust metrics within the framework.
The patient engagement component of the RDPEF outlines a continuum of engagement approaches built upon Carman et al.’s 26 multi-level model, which describes various forms of PE, ranging from information-sharing to shared decision-making. The RDPEF introduces a new foundational form of engagement called “information-sharing,” which focuses on educating patients about their condition, treatment options, and relevant systems and resources—particularly important in complex systems like Canada’s. This type of engagement is crucial not only for educating patients about disease-related matters but also for equipping them with the knowledge needed to progress to higher levels and forms of engagement. At the lower end of the continuum, patients have limited power and receive information passively. As they progress toward collaboration, they gain more authority, actively participate in decision-making, and collaborate to define goals and solutions.
The second form of engagement in the RDPEF is Consultation, which involves gathering patient feedback on health issues, research, and policy. The two lower levels of the continuum are defined by patients’ limited decision-making power and unidirectional communication, where information flows in one direction—providers/organizations/systems to patient, or reverse.
The third form is Deliberation (expanding on the “involvement” level in the Carman et al. framework), where patients actively participate in discussions related to health decision-making, such as deliberative polling, town hall meetings, or community deliberations. At the highest level of the continuum, Collaboration (similar to partnership and shared leadership in the Carman et al. framework) involves patients sharing leadership, decision-making, and responsibilities.
It is important to note that the four forms of engagement outlined in the framework represent a continuum of possibilities. They are not the only forms of engagement but rather markers of the most commonly used approaches along the continuum. Other forms of engagement are also possible. The continuum ranges from lower levels of decision-making power and unidirectional information flow on the left to increasing patient roles, power-sharing, and bidirectional communication on the right.
The second component of the RDPEF is levels of patient engagement. The RDPEF includes four levels of activity where patient engagement is possible: direct patient care, research and program design, organizational governance, and policymaking. Compared to other models, the RDPEF introduces a distinct, standalone level for “Research and Program Design,” positioned before the organizational governance and policy-making levels. This level ensures that patients have the opportunity to shape research priorities and study designs early in the healthcare process. Levels of engagement are centered on the idea that engagement should not be limited to individual direct care interactions, but that it should be more wholistic, spanning the entire spectrum of RD-related organizational and systems-level activity.
As mentioned before, a key feature of the RDPEF is that patient roles and degrees of power-sharing shift with different forms of engagement, evolving along the continuum from passive recipients of information to active partners in decision-making. This notion of progressively greater rights and responsibilities applies across all levels of the RDPEF. For instance, at the direct care level, patients begin by passively receiving information about their conditions. As they progress along this continuum, they start to actively collaborate with healthcare providers in developing care plans and setting personalized goals. This shift not only empowers patients but also fosters a partnership that enhances the overall quality of care.
At the level of organizational governance, patients initially may only receive information about the organization’s structures and changes. However, as they engage more actively, they provide valuable input on the design and structure of healthcare organizations and facilities. Patients can participate in advisory committees and councils, contributing to improvements in healthcare quality, patient safety, and staff training and development. By partnering at this level, patients help determine priorities and set agendas, sharing in decision-making authority.
Moreover, at the policymaking level, patients similarly start by passively receiving information about relevant health policies. However, as they advance along the continuum, they gain the opportunity to influence the development and implementation of these policies. This involvement can lead to a form of collaborative governance41,88–90—a model that, while still uncommon in the health domain, can be realized through a structured, measule patient engagement framework. By empowering patients to participate in policymaking, healthcare systems can better reflect the needs and values of those they serve, ultimately resulting in more effective and relevant health policies.
In the context of PE in RD research, it is worth exploring how the forms and levels of engagement apply to the stages of the orphan drug lifecycle. The forms of engagement help us understand the extent to which patients are involved in decision-making in a particular stage of the drug lifecycle. For instance, during the regulatory approval process, patients can transition from being passive recipients of information about the drug review process to actively advocating for their perspectives to be considered by decision-makers, sharing their experiences with the disease and treatment. The extent of power-sharing can increase as feasible, allowing patients to participate in setting treatment objectives and outcome measures, and even sharing responsibility for decisions at certain points (e.g., funding may cease if there is poor compliance or inadequate clinical outcomes). While advanced forms of engagement, such as partnership, may seem daunting and potentially jeopardize the healthcare budget, they could become feasible in the future as PROMs and PREMs are developed along with advances in technology and artificial intelligence, and demonstrate positive results for both health and healthcare systems.
It is not our position that the highest forms of PE are feasible in every case at every stage of the orphan drug cycle. Rather, the appropriate approach to engagement should be determined case by case, based on patient abilities and willingness, system capacity, and the policy environment. However, it is likely that patients will want steadily rising involvement and more decision-making power at every stage of the drug cycle, given recent research findings connecting PE in research with better health outcomes.38,91
Guiding principles, the second component of the RDPEF, form the basis for planning, implementing, and evaluating PE interventions across various health domains. These principles draw from study findings emphasizing transparency and responsiveness, while also incorporating concepts and aspects focused on reducing stigma, such as sensitivity to stigma, education, care and support, empowerment, and advocacy. This component is essential for guiding data analysis and addressing the literature gap on health stigma and discrimination experienced by RD patients.
The third component, patient experience, is central to the RDPEF. Integrating patient experience into the RDPEF is essential for enhancing the relevance and effectiveness of PE across all levels. Quantitative patient experience measures, such as PROMs and PREMs, should be developed and collected at various points along the engagement continuum. These measures inform decision-making processes, guiding both the delivery of care and the shaping of health policies. Additionally, developing tailored interview and focus group questions for each case is crucial for capturing an in-depth understanding of the complex experiences of RD patients. A mixed-methods approach—combining qualitative and quantitative methods—offers a more nuanced understanding of the unique challenges faced by patients with rare conditions, including stigma. For example, surveys and focus-group interviews can address issues related to small sample sizes in RD research, ensuring reliable and valid data collection.
By capturing patient experiences, this component provides critical insights into patient needs, identifies areas of healthcare requiring improvement, and guide research efforts. It also aids in monitoring the progress and effectiveness of engagement and stigma-reduction interventions over time. Additionally, patient experience can serve as a quality measure of treatment, alongside effectiveness and safety, which helps address uncertainties surrounding the clinical benefits of new drugs, thereby boosting RWE.
In synthesizing these elements—engagement forms, stigma mitigation, and experiential evidence—the RDPEF offers a pioneering RD-specific framework that addresses the unique challenges faced by RD patients, particularly involving health stigma and discrimination. The framework provides a structured pathway for patients, researchers, healthcare providers, advocacy groups, and policymakers to develop patient-centered programs, research, and policies. It emphasizes the importance of integrating guiding principles into PE practices, ensuring patient voices are included throughout all stages of healthcare, including research. Additionally, it highlights the need to systematically collect and analyze data to ensure that engagement efforts translate into improved health outcomes and sustained PE, ultimately driving positive change in healthcare.
Implications
The RDPEF represents a significant advancement in efforts to include RD patients as active partners in RD and orphan drug research. By providing a clear pathway for PE across the entire drug lifecycle, the framework addresses long-standing gaps in patient involvement, particularly at the stages of regulatory review and reimbursement decision-making. The integration of patient experience as a core metric ensures that healthcare systems remain responsive to the needs of RD patients, while the focus on reducing stigma fosters a more inclusive environment for patient engagement. As the framework is implemented, it holds the potential to inform national strategies in Canada and beyond, offering a roadmap to guide healthcare providers, policymakers, and researchers in incorporating patient perspectives throughout the drug lifecycle, healthcare activities, and policymaking. This will foster a more patient-centered approach in the rare disease space. Collaborative efforts among stakeholders, including policymakers, patient advocacy groups, and clinicians, will ensure the framework are applied effectively to improve outcomes for rare disease patients.
Strengths and limitations
The RDPEF is underpinned by a robust qualitative study that provides valuable insights into the lived experiences of RD patients. By integrating qualitative data and elements from multiple established frameworks, the RDPEF offers a systematic approach to PE specifically tailored for RDs. Moreover, its emphasis on addressing structural stigma marks a significant contribution to the existing literature on PE in the RD domain.
However, the effectiveness of the RDPEF across all levels of healthcare remains unclear and uncertain until it is measured through various approaches and levels of power sharing. A key shortcoming is the lack of clear specification of patient-reported indicators, which limits the ability to evaluate the framework’s impact effectively. Furthermore, the RDPEF has been developed primarily within the Canadian context, and this may restrict its applicability to other healthcare systems.
Recommendations for further research
Future research should focus on the practical implementation of RDPEF within healthcare systems, assessing its impact on patient outcomes and engagement levels. As PE frameworks continue to evolve, it is essential to gather data on which aspects of engagement lead to the most significant improvements in healthcare outcomes and treatment access for RD patients. Stakeholders—including healthcare providers, policymakers, and patient advocacy groups—are encouraged to collaborate in promoting the RDPEF to ensure that the voices of patients with RDs are heard and valued.
Additionally, further research should evaluate the long-term effects of the RDPEF on health outcomes, patient experience, and the overall healthcare system.
It is also important to validate the RDPEF across different jurisdictions and healthcare settings to assess its generalizability and impact on PE in orphan drug research. Further development and testing of patient-reported indicators to evaluate the framework is needed. Furthermore, studies should explore the integration of patient experience data with clinical and economic evidence in regulatory review and reimbursement processes for orphan drugs. Research on the effectiveness of stigma reduction interventions within the RDPEF could also yield valuable insights into enhancing patient involvement in decision-making.
Conclusion
The RDPEF is a timely and essential tool for improving PE in RD healthcare, marking a significant advancement by addressing a longstanding gap in the field. It empowers patients to actively participate in health-related matters, including research planning and design. By emphasizing stigma reduction and integrating patient experience as a central measure, the framework enhances engagement throughout the orphan drug lifecycle, ultimately contributing to better health outcomes and more responsive healthcare systems.
As the RDPEF gains traction, it has the potential to reshape the landscape of orphan drug research in Canada, offering a roadmap for healthcare providers, researchers, and policymakers to create a more inclusive and equitable system for RD patients. This framework fosters an environment where patients’ voices are heard at every stage of the drug lifecycle, including regulatory review and reimbursement decisions.
Moving forward, ongoing evaluation and refinement of the RDPEF will be crucial to adapt to the evolving needs of the vulnerable RD population and effectively addressing the challenges they face within the healthcare system.
Supplemental Material
sj-pdf-1-trd-10.1177_26330040251404519 – Supplemental material for From care to cure: a patient engagement framework for rare disease and orphan drug research
Supplemental material, sj-pdf-1-trd-10.1177_26330040251404519 for From care to cure: a patient engagement framework for rare disease and orphan drug research by Nahya Awada and Anil Varughese in Therapeutic Advances in Rare Disease
Footnotes
Acknowledgements
The authors would like to acknowledge Carleton University for providing the academic environment and resources that supported this research. We are especially grateful for the guidance and support from the professors in the Health Sciences and Public Policy and Administration departments.
Declarations
Ethics approval and consent to participate
Since human subjects were involved in the qualitative study, ethics approval was required to collect data related to patient experience with rare disease management. Thus, an application form was submitted to the Carleton University Research Ethics Board (CU-REB) for review and approval. CU-REB’s approval was granted in February 2019 before any data being collected for this study. Ethics Protocol Clearance ID: Project # 110105. Informed consent (written or verbal) was obtained from all participants prior to each interview. Verbal consent was utilized and digitally recorded in instances where participants lacked the means to provide written consent (e.g., no access to a printer or scanner); this method was approved by the CU-REB due to the low-risk nature of the study.
Consent for publication
Not applicable.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Competing interests
The authors declare that there is no conflict of interest.
Availability of data and materials
Please contact the corresponding author for data requests.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
