Abstract
Drug development for rare diseases can be long, complex, and costly. Desmoid tumors (DT), a rare type of soft-tissue tumor, are associated with substantial and debilitating burden, including disease-specific symptoms (e.g., pain, impaired mobility), reduce functioning for daily activities, and worsen quality of life for patients with this condition. These tumors can be potentially life-threatening when they invade surrounding tissues, affect vital structures, or interfere with the body’s functions. Until recently, there were no approved treatments specific to DT and little alignment on disease management. However, on November 27, 2023, the US Food and Drug Administration approved nirogacestat, an oral, targeted, and selective gamma secretase inhibitor, indicated for adult patients with progressing DT who require systemic treatment. This development milestone ascribes to nirogacestat the first approval of a gamma secretase inhibitor for human clinical use and the first therapy specifically indicated for treating patients with DT, thus addressing a long-term unmet need in this patient population. In the DeFi phase III trial of nirogacestat in adults with DT (NCT03785964), nirogacestat demonstrated statistically significant and clinically meaningful improvement in progression-free survival, objective response rate, DT-specific symptom burden (including pain), physical functioning, role functioning, and overall quality of life. This review chronicles the clinical development journey of nirogacestat from 2009 to the present day. Motivated to improve patient outcomes—and navigating considerable skepticism and uncertainty—the dedicated efforts of individuals within academic and medical institutions, industry, and patient advocacy groups shepherded nirogacestat through the development process, including those times when development stalled and might otherwise have been abandoned. Nirogacestat’s pathway to becoming a treatment for DT demonstrates how critically important collaboration and coordination are for identifying unique, creative solutions to overcome challenges in rare disease drug development.
Plain language summary
Rare diseases are diseases or conditions that affect a small number of people compared with the general population. Rare diseases often have no approved treatments. Desmoid tumors (DT) are a rare disease that can cause extreme pain and discomfort. DT can be life-threatening, depending on the tumor’s growth and location in the body. These tumors can make it difficult for people living with DT to perform daily activities. Symptoms caused by the condition can also reduce the quality of life (ability to do things that make life enjoyable) for people with DT. Doctors often have a hard time treating people with DT because tumor locations and behaviors differ from person to person. Developing and researching new drugs to treat rare diseases like DT is a long, complex, and expensive process. There had not been an approved medication for DT until November 27, 2023. This was when the US Food and Drug Administration approved a new drug called nirogacestat (Ogsiveo®) to treat patients with progressing DT who require systemic treatment. Nirogacestat works by shrinking the size of tumors or stopping the tumors from growing larger. In the phase 3 DeFi trial, nirogacestat treatment improved progression-free survival (how long the patient lived without disease progressing, DT-symptoms or health status getting worse, or dying from any cause) and objective response rate (when tumors shrink by at least 30% or disappear) when compared with a sugar pill placebo. Nirogacestat also decreased DT-specific symptom burden like pain and improved quality of life for people with DT in the study. The path to making nirogacestat available to people with DT had many challenges. Strong collaboration among doctors, scientists, pharmaceutical companies, and patient advocacy groups kept the development of nirogacestat moving forward. Because of the dedicated, tireless work of many people and organizations, nirogacestat became the first approved drug to treat DT.
Introduction
Desmoid tumors (DT) are rare, soft-tissue tumors with a variable natural history. Although DT are neoplasms that do not metastasize, tumors can be locally aggressive and are associated with high morbidity and reduced quality of life.1–3 These tumors arise in connective tissue and can occur throughout the body. Depending on growth and location, DT can cause severe pain, impair physical functioning, and, in rare cases, be lethal.2–4 The disease course is unpredictable, with cycles of tumor progression, stabilization, and sometimes spontaneous regression making disease management difficult.1,5–7 Given the low mortality but high morbidity associated with DT, treatment goals should go beyond clinical markers like progression-free survival (PFS) to include patient-reported outcomes that evaluate the effect of symptom burden on daily life, functioning during daily activities, and overall quality of life. 7
As a rare disease with approximately 1000–1650 new cases diagnosed each year in the United States,8–11 there is little awareness around this debilitating condition. Current guidelines recommend an initial evaluation and personalized management by a multidisciplinary team with expertise in DT.1,4,7,12 However, few clinicians have experience with DT, making inaccurate and delayed diagnoses commonplace.1,7
Before the approval of nirogacestat (PF-03084014; Ogsiveo®, SpringWorks Therapeutics) on November 27, 2023, by the US Food and Drug Administration (FDA),13,14 there were no approved systemic therapies specifically indicated for patients with DT. As such, effective treatment options for DT were limited. While active surveillance is initially recommended for patients with DT, active treatment is indicated for patients who are symptomatic or progressing.4,12 Treatment options or decisions are generally influenced by the location of the tumor. 7 Because of the high morbidity associated with surgery and high rate of tumor recurrence,8,15–18 surgical resection is only recommended in certain clinical situations. For most tumor locations, systemic therapy that may shrink DT and alleviate symptoms has been recommended as first-line treatment. Prior to the approval of nirogacestat, systemic therapy options included nonsteroidal anti-inflammatory drugs, which have few side effects, but limited effectiveness.19–21 Tyrosine kinase inhibitors have demonstrated varying degrees of efficacy and toxicity,22–25 and chemotherapy is occasionally used for the treatment of DT, especially when tumors require rapid shrinking.25–29 Locoregional therapy modalities, such as cryoablation and high-intensity focused ultrasound, are also used to treat small- and moderate-sized extra-abdominal DT.1,12 Because of the limited safety and efficacy profile of available therapeutic options, an effective, well-tolerated therapy with an acceptable safety profile remained a significant unmet need.
The drug development process is long, complex, and costly, with nearly 90% of drug candidates failing clinical investigation. 30 These challenges are augmented in the rare diseases setting, where a lack of disease awareness means limited research funding, interest among researchers, and data about the disease’s natural history.31,32 Even when drugs demonstrate preclinical promise in rare diseases, the high costs of research and development limit the incentive to continue drug advancement. Clinical trial design can be challenging in the rare disease space, as relevant endpoints and outcomes may not be well defined and biomarkers may be unknown. A small, often geographically dispersed patient population also means fewer available patients to fully enroll trials with appropriate control arms.31,32
These challenges have been particularly hinder some to DT drug development. Several agents have been assessed in clinical studies of DT over the years but never advanced to phase III trials (e.g., tamoxifen, imatinib, pazopanib, vinblastine, methotrexate, and sulindac). For new drugs being evaluated for DT, the variable clinical course (including spontaneous regression and stabilization) and lack of standard of care requires randomized, placebo-controlled, double-blind studies to robustly assess treatment efficacy and safety, with enough patients for at least two treatment arms. In addition, the unique nature of DT—a chronic, potentially debilitating disease with variable pain and functional compromise depending on tumor growth and location—makes traditional clinical endpoints like overall survival and PFS less able to capture the nuances of DT symptom burden. As such, patient-reported outcome (PRO) tools are essential to fully evaluate new therapeutics in this small population. Tolerability of chronic administration and ability to improve DT-associated symptoms need to be assessed using validated questionnaires that cover the spectrum of symptoms experienced by patients with DT. 3
To navigate the numerous challenges of rare disease drug development, coordinated efforts of investigators from academic organizations, medical study partners and clinicians, pharmaceutical industry partners, and patient advocacy groups are essential for success. This coordination can provide long-term and comprehensive support to enable scientific breakthroughs for rare diseases (Figure 1). In this review, we detail the real-world clinical development journey of nirogacestat, the first therapy specifically indicated to treat adults with progressing DT.14,33 We highlight how collaborations among academia, industry, and patient advocacy groups overcame critical challenges, uncertainties, and surprises as nirogacestat advanced from development to regulatory approval for the treatment of DT (Figure 2).
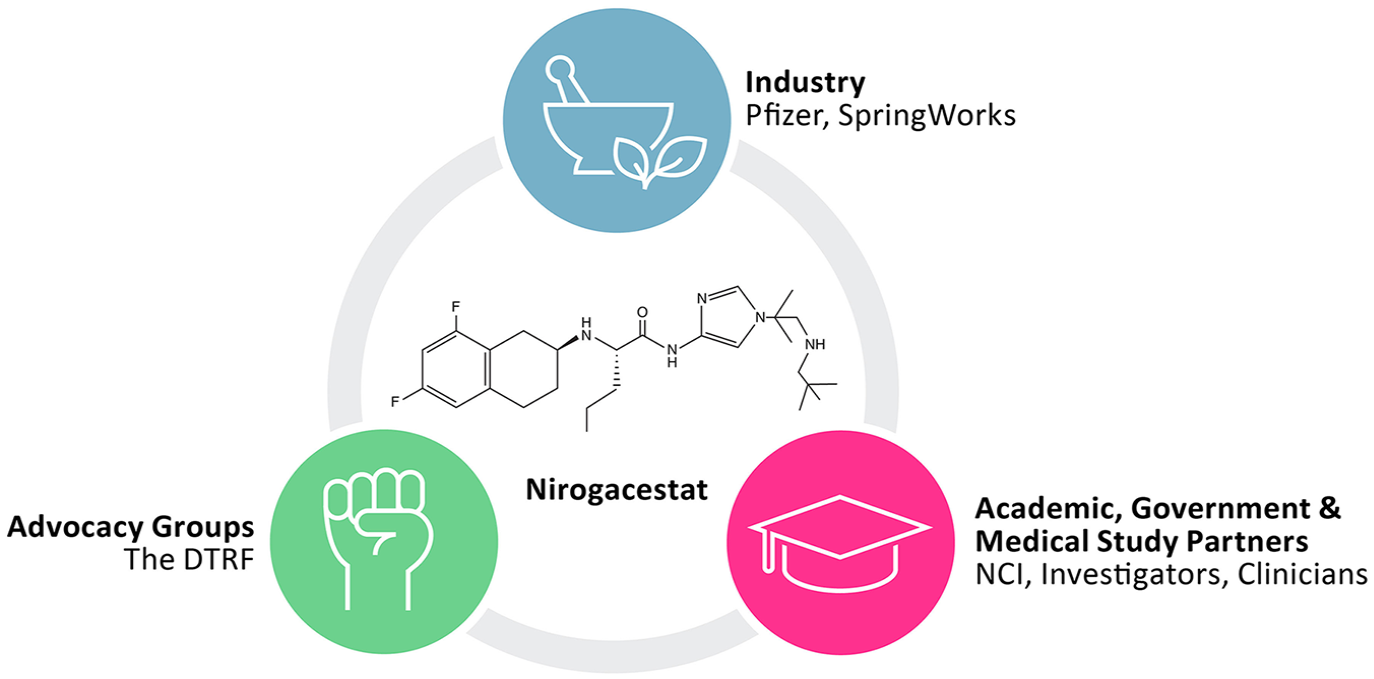
Cross-functional collaborations during the clinical development of nirogacestat.

Timeline of challenges and key collaborations during nirogacestat clinical development.
Initial development of Nirogacestat
Nirogacestat is an oral, targeted, and selective small molecule gamma secretase inhibitor (GSI).33–35 It was originally developed by Pfizer, Inc. (New York, NY, USA) 36 as an Alzheimer disease drug because gamma secretase cleaves amyloid-β peptides, which form the amyloid-β plaques that contribute to pathogenesis of Alzheimer disease. 37 However, many GSIs failed clinical trials for Alzheimer disease often due to negative side effects of decreased Notch signaling, 38 and nirogacestat did not advance to clinical trials for Alzheimer disease.
Based on the mechanistic rationale that GSIs can inhibit the Notch pathway, investigations into other potential indications began and Pfizer sponsored an exploratory phase I solid tumor trial in 2009. 39 This phase I dose-finding trial enrolled patients with advanced cancer or acute T-cell leukemia/lymphoblastic lymphoma that was resistant to standard therapy or for which no standard therapy was available (NCT00878189). Sixty-four patients with solid tumors, including nine patients with DT, were enrolled. Nirogacestat, administered twice daily for cycles of 21 consecutive days, was generally well tolerated with mild-to-moderate adverse events. Importantly, patients with DT had an unexpectedly high response, with 5 of the 7 response-evaluable patients having a partial response (71.4% objective response rate (ORR)). Further, none of the DT cases had progressed at the time of data cutoff (maximum 24-month follow-up). 39
Although interesting, these preliminary results could not be seen as predictive of a true treatment effect in DT, given the unpredictable nature of DT growth and historical challenges with translating phase I clinical trial data into later-stage studies. Additionally, all the DT patients evaluable for a response were enrolled at a single site, with no eligibility criteria around baseline tumor progression or other tumor characteristics, further highlighting the uncertain nature of the observed clinical signal. The exact mechanism of action in DT also remained unconfirmed. In DT and other tumors, GSIs are hypothesized to act through inhibition of the Notch pathway. Preclinical data suggest that DT can be driven by overexpression of Notch1 pathway components and alterations in the Wnt/APC/β-catenin pathway,1,40–42 and crosstalk between these two pathways may contribute to DT pathogenesis.34,43 Thus, a hypothesis emerged that by targeting a pathway that is critical for DT development and growth, GSIs like nirogacestat could have a potentially unique therapeutic role relative to other systemic agents used to treat patients with DT.39,41,44
Given the promising, but not definitive, clinical responses to nirogacestat treatment in the phase I study among patients with DT, the University of Colorado Cancer Center subsequently conducted a retrospective analysis of these patients with DT with long-term follow-up. 45 All five patients who achieved a partial response, including four patients who had stopped treatment, continued to maintain duration of responses between 47.9 and 73.6+ months. Beyond reductions in tumor size, this trial noted that every patient had “dramatic, sustained symptomatic relief (decreased tumor pain and/or increased mobility), usually within weeks of starting therapy, often preceding radiographic changes by months.” 45 Though these outcomes were not objectively characterized with a PRO tool to evaluate pain, DT symptom burden, functioning, and/or quality of life, and the trial did not include a control arm, the results were intriguing nonetheless.
Nirogacestat clinical development journey for treatment of desmoid tumors: Challenges, collaborations, and solutions
CHALLENGE: Gaining traction and attention to develop nirogacestat for DT treatment
Gaining traction for therapeutics directed toward rare diseases like DT is difficult, and Pfizer did not conduct additional trials. At the time, there was skepticism about the prospects of the GSI drug class in general. However, in 2012, data from the phase I trial were presented at the joint meeting of the European Organisation for Research and Treatment of Cancer, National Cancer Institute (NCI), and American Association for Cancer Research. 46 Here, the results caught the attention of an investigator from the NCI, Dr. Shivaani Kummar. Although the rarity of DT presented the challenge of recruiting enough patients to conduct a follow-up phase II trial at a single institute, the unique resources of the NCI, with its ability to provide travel support and bring in patients from across the United States to Bethesda, Maryland, made it the ideal location to conduct such a trial.
Collaboration between the investigators at the NCI and the clinical development team at Pfizer finally led to the design of an open-label, single-arm phase II trial to further explore the efficacy and safety of nirogacestat in patients with unresectable DT, with clinical response as the primary endpoint. 47 Ultimately, this phase II trial, along with others, would lead to the creation of NCI’s Rare Tumor Initiative, which was formally launched in 2013 to foster collaborations between basic and clinical scientists, patient advocacy groups, and industry partners to facilitate development of therapeutic approaches to rare tumors. 48 This type of collaborative research network continues to be supported through programs such as MyPART, a Cancer Moonshot NCI-led effort for rare cancers. 49
CHALLENGE: Recruiting enough patients for a rare disease trial being conducted at a single institution
Despite the resources of the NCI, reaching enough patients with DT to complete enrollment of the trial proved problematic. As the trial leads at the NCI prepared to implement the phase II trial, they realized they would need the help of a patient organization. In May 2013, the NCI became aware of the Desmoid Tumor Research Foundation (DTRF), 50 a nonprofit patient advocacy organization that supports DT research funding, as well as education and collaboration among patients, physicians, and researchers. The DTRF had been cofounded in 2005 through the determined efforts and partnership between two women living the DT experience: Jeanne Whiting (a patient) and Marlene Portnoy (a caregiver). After nearly a decade of working to fund and facilitate DT research, they were elated to receive news of a clinical trial and request for collaboration.
A close and enthusiastic partnership between the NCI and the DTRF brought forward the phase II trial of nirogacestat in adult patients with progressing DT, which opened in November 2014 (NCT01981551). 47 Although it was initially anticipated that it could take up to 2 years to complete patient enrollment due to the rare nature of the disease, with the DTRF’s outreach, the trial fully enrolled all 17 patients in about half a year.
The phase II trial completed its primary analysis in 2017. The primary results were promising—5 of 16 evaluable patients had a partial response that persisted through the trial, and 11 patients had stable disease.47,51 Long-term (7-year) follow-up found that none of the 16 patients had disease progression while on the trial and that nirogacestat continued to be well tolerated. 52 However, the single-site, single-arm, open-label design coupled with uncertainty around the natural history of the disease prevented definitive conclusions from being drawn. The primary PRO assessment, using the MD Anderson Symptom Inventory (MDASI) questionnaire, showed improvements in symptom severity, though patients were aware that they were receiving nirogacestat. 47 The DTRF also noted that patients participating in the trial were experiencing positive changes in their quality of life. However, the MDASI is a general PRO tool, emphasizing the need for a disease-specific PRO for DT.
CHALLENGE: Creating a DT-specific patient-reported outcomes tool
As DT are soft-tissue tumors that do not metastasize and have a variable clinical disease course, traditional oncology clinical trial endpoints, such as PFS and response rates determined by the Response Evaluation Criteria in Solid Tumors (RECIST; version 1.1), may not fully capture the patient experience with DT. 3 Factoring in the effect of treatment on disease-specific PROs—such as reduced pain, alleviated DT-specific symptom burden and the consequent impact on patients’ lives, and improved functioning with daily activities and overall quality of life—creates a new dimension for evaluating success. A DT-specific assessment tool would also allow for a potential new endpoint in DT clinical trials. 3 In 2015, the DTRF funded and participated in a collaboration with Memorial Sloan Kettering Cancer Center (MSKCC) and IQVIA, a leader in the development of PRO instruments, to produce an assessment tool specific to DT. Creating this tool allowed for the success of future DT trials with new endpoints that capture the patient voice, as PFS and ORR are not sufficient to tell the complete story of the effect of potential DT treatments. However, development of a PRO instrument is labor-intensive, time-consuming, and costly. For rare disease PRO tools, the additional challenges of identifying investigators to lead development, engaging regulatory agencies, acquiring funding support, recruiting patients, and validating the PRO tool required the collaboration of academia, patient advocacy groups, and industry. 3
To develop the DT-specific assessment tool, researchers first prospectively evaluated patients with DT through concept elicitation interviews to understand their symptoms, experiences with treatment, and impact of the disease on their functioning and daily living. 3 Saturation of concepts was met during these interviews (i.e., additional interviews did not contribute unique concepts or information). Following development of a draft PRO tool, cognitive interviews were conducted in a second cohort of patients with DT to assess their understanding of the tool’s instructions, items, and response scales. Multiple rounds of refinement were used to establish its content validity as a clinical trial endpoint. The final 28-item questionnaire was termed the Gounder/DTRF Desmoid Symptom/Impact Scale (GODDESS®) and captures symptoms and impacts related to DT. 3 Blinded, pooled data from the prospective phase III DeFi trial of nirogacestat (NCT03785964) were used to validate GODDESS as a reliable and responsive clinical trial endpoint to assess improvements in DT symptom severity and quality of life in patients with DT. 53
With the phase I trial results, commencement of the NCI phase II trial, and development of the DT-specific assessment tool, continued progress of nirogacestat development seemed promising—if not for a major setback.
CHALLENGE: Finding a path forward for nirogacestat after halted clinical development during the phase II trial
In 2015, during the phase II trial, Pfizer made the decision to deprioritize nirogacestat development. Although patients in the phase II trial were still served by Pfizer’s compassionate use program, this was a disheartening decision to patients, physicians, and researchers in the DT community. To continue the drug development of nirogacestat, the DTRF leadership and NCI investigators began to meet with Pfizer to review the promising results of the ongoing phase II trial and find a path forward. 47 While uncertainty existed around the true effects of nirogacestat, there was enthusiasm and momentum to keep the clinical development going. Ultimately, as awareness of these meetings spread, DT clinicians and researchers in academia voiced their support.
CHALLENGE: Continuing nirogacestat clinical development
As a result of the continued advocacy effort and collaboration with academia and industry, Pfizer decided it would out-license the rights to nirogacestat. 36 The next challenge was engaging investors with the only available clinical data in DT coming from uncontrolled phase I and II trials. Investors would have to be convinced that a successful phase III trial in DT could be executed given the challenges associated with trials in rare diseases.31,32 Ultimately, SpringWorks Therapeutics was founded in 2017 to continue the clinical development of nirogacestat, with Pfizer, two institutional investment firms, and a medical research charity providing the required Series A funding to commence the company’s operations. 36 With the rights to nirogacestat, SpringWorks could begin design of a phase III trial.
CHALLENGE: Building the infrastructure to run a multisite, international trial
At this point in the clinical development journey, a randomized, placebo-controlled trial was necessary to demonstrate the safety and efficacy of nirogacestat in DT, given the uncertainties around the phase I and II trial data. As a newly founded organization, SpringWorks focused on building the infrastructure to initiate a global, randomized, placebo-controlled phase III clinical trial for nirogacestat in DT. SpringWorks engaged with the FDA to negotiate the phase III development program for nirogacestat, including trial design and endpoints. In June 2018, the FDA granted Orphan Drug Designation for nirogacestat for the treatment of DT, and in November 2018, they granted Fast Track Designation for the treatment of “adult patients with progressive, unresectable, recurrent or refractory desmoid tumors or deep fibromatosis” 54 (Figure 3). This designation was based on phase I and phase II data evaluating nirogacestat as a monotherapy in patients with DT.

Major regulatory designations for nirogacestat development and DeFi trial milestones.
In late December 2018, the trial record was posted to ClinicalTrials.gov under its official title: “A Randomized, Double-Blind, Placebo-Controlled, Phase III Trial of Nirogacestat in Adult Patients with Progressing Desmoid Tumors/Aggressive Fibromatosis (DT/AF) (DeFi)” (NCT03785964; Figure 3). However, the high costs of implementing an international phase III trial in 7 countries with 52 investigational sites 55 meant that the funds initially raised by SpringWorks would be insufficient to run DeFi. SpringWorks commenced another financing round, securing several investors to support the company and its main program, nirogacestat for DT. 56 This Series B financing was completed in April 2019.
Finally, DeFi could commence. As it had with the NCI in the phase II trial, the DTRF collaborated with SpringWorks to facilitate patient recruitment, which was challenging given the stringent enrollment criteria of the phase III trial as well as the fifty-fifty chance that patients might receive a placebo. 35 Reaching thousands of patients through its patient and caregiver database, the DTRF raised awareness of the DeFi trial. In May 2019, the first patient was randomized and dosed in the DeFi trial, and in only 15 months, enrollment was completed (N = 142 across 37 sites that randomized patients in the United States, Canada, and Europe). 35 In August 2019, the FDA granted Breakthrough Therapy Designation to nirogacestat for the treatment of “adult patients with progressive, unresectable, recurrent, or refractory DT or deep fibromatosis.” 57
An unforeseen challenge for this trial was the onset of the COVID-19 pandemic beginning in early 2020. Implementation of creative strategies to enable continued patient engagement in the clinical trial included direct-to-patient shipping of study drug and supplies and increased flexibility on where trial assessments were conducted (i.e., local laboratory and imaging assessments were allowed with prior approval).
CHALLENGE: Facilitating new drug approval for a novel therapeutic agent in a rare disease
In May 2022, SpringWorks announced positive topline results for the phase III DeFi trial, representing the first time that nirogacestat’s safety and efficacy in DT had been established with a randomized and placebo-controlled trial. 58 Nirogacestat treatment reduced the risk of disease progression or death by 71% compared to placebo (p < 0.001). The likelihood of no disease progression in 2 years was 76% for patients in the nirogacestat group and 44% in the placebo group. 35 Patients treated with nirogacestat had an ORR of 41% (8% for placebo; p < 0.001) and a complete response rate of 7% (0% for placebo). 35 This trial was also the first to prospectively study the DT-specific GODDESS tool, which was included as one of the key secondary efficacy endpoints.35,53 The DeFi trial also included various other PROs, including the Brief Pain Inventory—Short Form and the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire—Core 30. 35 As a result, nirogacestat demonstrated statistically significant and clinically meaningful improvements in pain, disease-specific symptom burden, physical and role functioning, and overall quality of life compared to placebo. Consistent with previous trials, adverse events were mostly grades 1 and 2, and nirogacestat seemed to be well tolerated over time. These phase III results generated excitement in the field with the study outcomes presented at the 2022 European Society for Medical Oncology Presidential Symposia, 59 the 2022 DTRF International Desmoid Tumor Research Workshop, 60 the 2022 Connective Tissue Oncology Society Meeting, 61 and eventually published in the New England Journal of Medicine in 2023. 35 The results of the first completed randomized controlled trial of a GSI to demonstrate clinical benefit in any indication represented a breakthrough for a drug class that had long been thought to not have therapeutic utility.
SpringWorks collaborated with the FDA to ensure timely FDA review through the Real-Time Oncology Review (RTOR) program in June 2022. 14 In December 2022, SpringWorks filed a New Drug Application (NDA) with the FDA for nirogacestat in DT, 62 which was accepted in February 2023. 63 Finally, on November 27, 2023, nirogacestat became the first FDA-approved systemic therapy for DT. 14
In the approximately 8 years since its founding, SpringWorks—with collaboration from the DTRF and academic study partners—reignited the clinical research of nirogacestat in patients with DT and conducted a rigorous phase III clinical trial that produced, for the first time, positive safety and efficacy data to support the approval of an NDA for DT. This approval represented a breakthrough along two dimensions: the first being confirmation of the finding that a GSI was efficacious in treating DT, and the second being a successful clinical development campaign in a rare disease like DT regardless of the drug’s mechanism of action. Nirogacestat is a novel treatment for DT that achieves patient-relevant treatment goals by improving PFS and PROs, including quality of life. 35 In addition to its effectiveness in DT, nirogacestat is also in clinical development for the treatment of ovarian granulosa cell tumors (NCT05348356) and for combination treatment of multiple myeloma with other therapies (NCT05556798).
This review has limitations to note. First, the scope of this review is narrow, as it focuses exclusively on the clinical development of nirogacestat. As such, an exhaustive review of the DT or GSI drug literature is not presented. Lastly, perspectives that could not be supported by a citation to published literature were not included.
Conclusion
Our goal was to place the clinical development journey of nirogacestat into a larger context to showcase the surprises, challenges, and learnings to be considered when developing new drugs for rare diseases. A key lesson is the importance of collaboration among multiple groups with an aligned purpose, throughout the long and arduous process. In the case of nirogacestat development, partnerships between clinicians and investigators at academic institutions and the NCI, patients and advocacy groups, and executives in the pharmaceutical industry were essential to moving the development of nirogacestat forward, particularly during the many times when skepticism arose, development stalled, and nirogacestat could have been abandoned. The effectiveness of these types of partnerships is an important takeaway from the nirogacestat story, and the cultivation of similar partnerships can be applied to drug development in other rare disease areas.
Collaboration also solved the difficulty of recruiting a limited, distributed patient population for clinical trials of a rare disease such as DT. Involving advocacy groups early in the initial concept stages of trial design improved outreach to meet the recruitment needs of the nirogacestat clinical trials. This type of collaborative recruitment strategy with patient advocacy groups can be pursued for other rare disease clinical trials.
Another key learning from the nirogacestat development program was the importance of having a disease-specific PRO tool. For many rare diseases like DT, traditional trial endpoints, such as OS and PFS, may not fully capture a drug’s effect on patient outcomes. Evaluating the changes in quality of life meaningful to patients and incorporating appropriate PRO assessments into other rare disease clinical trials may enrich results.
The final lesson from the nirogacestat journey that is applicable to new drugs in other rare diseases is the benefit provided by early engagement and collaboration with the FDA. The FDA’s recognition of the potential clinical benefit of nirogacestat (e.g., granting Orphan Drug, Fast Track, and Breakthrough Therapy designations and accepting into the RTOR program) was essential to accelerate the approval process for nirogacestat. From the first phase I trial of nirogacestat in 2009 to its approval on November 27, 2023, the nirogacestat clinical development program has continued to progress because of dedicated individuals and organizations who never stopped advocating for patients to provide access to the first FDA-approved treatment for DT.
Footnotes
Acknowledgements
The authors would like to acknowledge the countless integral individuals to this rare success story who were not named directly in this manuscript, including patients, families, caregivers, and advocacy groups as well as investigators and study staff involved in nirogacestat clinical development. Writing and editing support was provided by Alexandra L. Thomas, PhD and Jacqueline Benjamin, PhD from Prescott Medical Communications Group, a Citrus Health Group, Inc. company (Chicago, IL) with funding from SpringWorks Therapeutics, Inc.
