Abstract
Glucose transporter type 1 deficiency syndrome (GLUT1 Deficiency) is a rare epileptic encephalopathy first described in 1991. Impaired glucose transport leads to decreased glucose supply to the brain, resulting in an alteration of brain metabolism and function. Clinically, classical GLUT1 Deficiency is characterized by infantile-onset epilepsy, which is often drug resistant, movement disorders, intellectual disability, and speech and language disorders. There is a broad spectrum of symptoms and severity which may change over time. GLUT1 Deficiency is caused by pathogenic variants of the SLC2A1 gene that can be spontaneous or inherited in an autosomal dominant fashion, although a few cases of autosomal recessive transmission have also been reported. There is currently no cure for GLUT1 Deficiency; however, the recommended standard of care treatment is a medically supervised ketogenic dietary therapy, which helps improve some symptoms for some patients. The Glut1 Deficiency Foundation aimed to develop a strategic research plan centered on the needs and priorities of patients using a patient voice survey and gathering input from all stakeholders. The Collective Voices Survey content was developed through virtual focus group discussions and questionnaires with patients and families. We used learnings from our survey and our first scientific conference to assemble our strategic research plan, which we have called our Research Compass. The survey provided clarity and a more comprehensive representation of patient and family experiences. It also sheds light on their priorities for research, ideas around treatment development, and willingness to participate in clinical trials. Learnings from this survey, along with input from other stakeholders, helped us identify some of the research gaps in our disease field. These insights are incorporated into the Research Compass that is now guiding the scientific focus and priorities in our community and ensuring that patients are at the center of this effort.
Plain language summary
Glucose transporter type 1 deficiency syndrome (GLUT1 Deficiency) is a rare neurometabolic disease that was first described in 1991. The decreased glucose entering the brain causes a wide range of neurological symptoms. The classical symptoms include seizures that are often resistant to drugs, movement disorders, intellectual disability and speech and language disorders. Not all patients experience all symptoms and the spectrum of severity is broad. GLUT1 Deficiency is caused by mutations on the SLC2A1 that contains the information to produce the GLUT1 protein. There is currently no cure for GLUT1 Deficiency; however, the recommended standard of care treatment is a medically supervised ketogenic dietary therapy (KDT), which helps improve some symptoms for some patients. We aimed to develop a strategic research plan centered on the needs and priorities of patients using a patient voice survey and input from all stakeholders in the community. We identified a list of gaps in research on GLUT1 Deficiency and we developed a Research Compass that is now guiding research focus and priorities in our community with patients as the focal point of this guide.
Introduction
Glucose transporter type 1 deficiency syndrome (GLUT1 Deficiency) is a rare genetic disorder that impairs brain glucose metabolism 1 and disrupts brain function. The classical form of this disease is characterized by an infantile-onset encephalopathy with intellectual disability, movement disorders, speech and language impairments, and drug-resistant epilepsy.1–3 GLUT1 Deficiency patients exhibit several types of seizures, such as generalized tonic or clonic, focal, myoclonic, atypical absence, atonic, and unclassified. Additionally, infants can experience episodic eye-head movements that may precede onset of seizures. 4 Complex movement disorders characterized by ataxia, dystonia, and chorea may also occur. Additional atypical manifestations that some patients can experience include focal hand dystonia, intermittent ataxia, total body paralysis, Parkinsonism, alternating hemiplegia of childhood, hemiplegic migraine, cyclic vomiting, and stroke-like episodes.3,5,6
GLUT1 Deficiency is caused, in the majority of cases, by pathogenic variants of the SLC2A1 gene that encodes for the Glucose transporter type 1 protein (GLUT1). 7 As established by the GLUT1 Deficiency consensus guidelines, 1 a definite GLUT1 Deficiency diagnosis requires two of the following criteria: the presence of characteristic clinical features, hypoglycorrhachia or low cerebrospinal fluid glucose concentration <60 mg/dL, and a pathogenic variant in the SLC2A1 gene.
The current standard of care treatment for GLUT1 Deficiency is a medically supervised ketogenic diet therapy (KDT).1,6 KDT is a high-fat, adequate protein and low-carbohydrate diet that provides an alternative source of energy for the brain as it is metabolized into ketone bodies that can cross the Blood–Brain Barrier (BBB). 8 The KDT is highly effective for seizure control in some GLUT1 Deficiency patients, and it has also been reported to improve movement disorders and cognitive issues in some patients. 1 It is recommended that the diet be initiated as early as possible to provide the developing brain with an alternative metabolic fuel source. However, some patients may continue experiencing seizures or problems with cognition and social adaptive behavior even with diet compliance. 9
The number of GLUT1 Deficiency patients has been increasing since it was first described in 1991. A prospective study on incidence and phenotypes of childhood-onset genetic epilepsies in the Scottish population reported a birth incidence of 1:24,000 presenting with seizures before 3 years of age. 10 A second study, estimated a prevalence of GLUT1 Deficiency in the Danish population of 1:83,000. 11 Despite the progress made in diagnosis, these studies highlight the need to improve the diagnostic process.
The Glut1 Deficiency Foundation is a nonprofit patient advocacy organization dedicated to improving lives in the GLUT1 Deficiency community through the mission of increased awareness, improved education, advocacy for patients and families, and support and funding for research. In pursuing these mission priorities, we realized that while we had an abundance of anecdotal information, we lacked primary data about patient and family experiences, gaps in care, and what is most important to them in research and for outcomes in new treatment development. In order to be able to confidently speak for and represent what the patients and families want and need, we created a large-scale patient voice survey to capture these needs, experiences, and priorities. 12 As a starting point, we held focus groups and discussion sessions in Zoom gatherings with patients and families to start collecting ideas and insights that could be incorporated into the survey. We compiled these findings and asked for additional feedback and suggestions from the participants. We then worked to organize the information and create categories and a comprehensive set of questions with the following goals in mind: better define the range of symptoms; identify gaps in knowledge and understanding of the disease; better understand the burdens of the disease on patients and families; identify the most important components of our Natural History Study (NHS); develop and prioritize future service programs; create and prioritize a strategic research plan; and develop better and more effective clinical trials for potential future treatments. Herein, we describe results of the survey, as well as the outcomes that came from it, such as our Research Compass and the research tools and resources available in our community.
Methods
Collective voices in GLUT1 deficiency survey
The content of the survey was developed through virtual focus group discussions and questionnaires in the patient community. The questionnaires are not validated. A copy of the questions in the questionnaires can be found as a Supplemental File. The anonymous survey was conducted on the Qualtrics platform under Castle IRB and statistical analysis was provided by Insights Advisors Group.
Research compass
We took the learnings from our Collective Voices project and the first GLUT1 Deficiency scientific conference to assemble a list of the many gaps and needs regarding GLUT1 Deficiency. We collected information in small group focus groups from all conference attendees through surveys that asked specifically about research tools needed, research priorities, existing gaps, critical questions, and crucial projects necessary to advance research in the community. We sought to establish a systematic framework for classification and clustering of these elements, driven by the objective of developing an information repository conducive to streamlined access for researchers. Our overarching aim was to facilitate efficient information retrieval, enabling researchers to locate pertinent data, discover associated publications pertaining to existing endeavors in these domains, identify individuals engaged in this research, and engage in the collaborative exchange of ideas as the research progresses.
Results
Collective voices in GLUT1 deficiency survey
Demographics
There were 260 responses from 31 countries to the survey. While most of the respondents were located in the US (41%), there were also participants from Europe (Italy and the United Kingdom had the biggest representation in Europe at 8% each), South America (Argentina had the biggest representation at a 4%), Asia (Hong Kong had the biggest representation at about 1%) and Australia (2%). 95% of respondents were parents or caregivers representing their affected children, and 5% were patients. Fifty-seven percent of participants were females and the highest number of participants (37%) were in the 7–12-year-old age group. In addition, 79% of the participants reported to be white and 40% live in urban areas [Figure 1(a)].
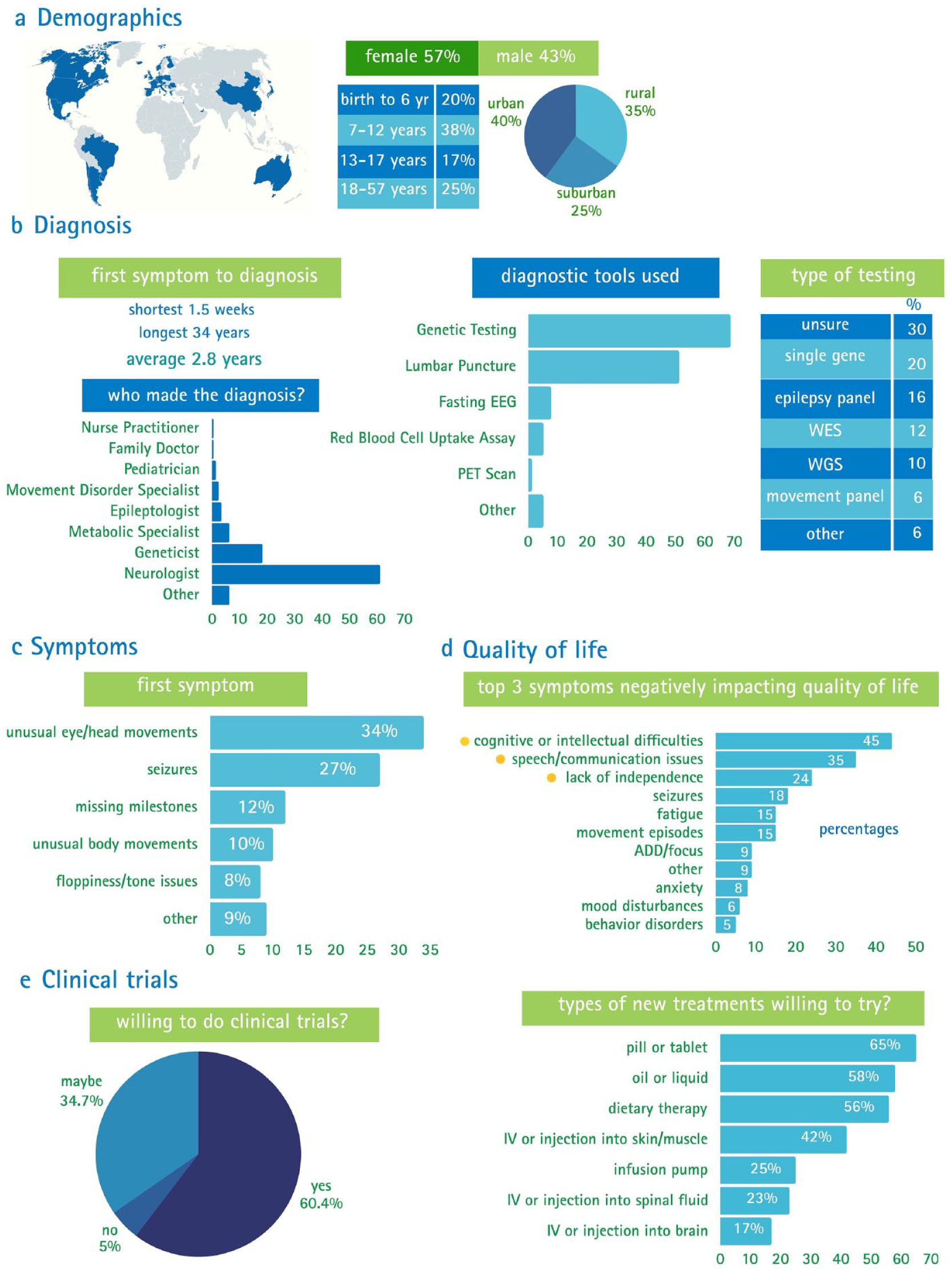
Collective Voices in Glut1 Deficiency Survey. (a) Demographics. Survey participants were located all over the world. The majority of surveys were completed by a parent or guardian. Most participants were females, most participants were in the 7–12-year-old age range and most participants lived in urban areas. (b) Diagnosis. Time elapsed from first symptom to diagnosis was approximately 3 years. Most diagnoses were made by neurologists, through genetic testing. The most common type of genetic testing test reported was single gene. (c) Symptoms. The most common first symptom experienced by most participants was the unusual eye-head movements. (d) Quality of life. The top three symptoms that affect patients’ quality of life are cognitive or intellectual difficulties, speech/communication issues, and lack of independence. (e) Clinical trials. Most of the participants are willing to participate in clinical trials and the most common types of new treatments they are willing to try include pill or tablet, oil or liquid, and dietary therapy.
Diagnosis
The average age of diagnosis was 6 years old, and time elapsed from first symptom to diagnosis was approximately 3 years. The average number of physicians consulted to obtain a diagnosis was eight and those diagnoses were performed by neurologists in 61% of the cases. In addition, the diagnostic tool used in 69% of the cases was genetic testing, and the most common type of test was single gene test [Figure 1(b)].
Symptoms
The most common first symptom was unusual eye/head movements with 34% of patients reporting this symptom [Figure 1(c)]. Approximately 58% of the participants experienced classical symptoms of GLUT1 Deficiency such as seizures, cognitive or intellectual difficulties, not meeting developmental milestones, and speech and/or language issues. The most common seizures reported were absence seizures. In addition, patients reported experiencing other symptoms including memory problems (31%), sleep disturbances (32%), retina and vision issues (21%), and chronic pain (8%), among others.
Quality of life
We were particularly interested in identifying the symptoms that most significantly impact patients’ quality of life. The findings revealed that cognitive or intellectual difficulties (44%), speech/communication issues (35%), and lack of independence (24%) were the symptoms exerting the most pronounced impact [Figure 1(d)]. We also learned which outcome improvements are priorities for the community. The majority of the respondents (35%) indicated that being able to eat a normal diet is their top priority, followed by improved cognition (27%) and better speech/communication (25%).
Clinical trials
Lastly, the survey sought to define their willingness to participate in clinical trials and the types of new treatments they are willing to try. Approximately 60% of participants are willing to participate in clinical trials and about 35% indicated they may participate. Most of the participants (65%) reported being willing to try a pill or tablet, 58% an oil or liquid, and 56% a dietary therapy [Figure 1(e)].
Learnings from this survey 12 in addition to input from other stakeholders helped us identify some of the research gaps in our community and to develop our patient-reported NHS. We have used the data collected from the Collective Voices project to create multiple in-depth reports available on our website and have presented findings in a number of settings, including scientific posters and both professional and family meetings.
Research compass
We leveraged insights from our Collective Voices project and our inaugural virtual scientific conference, conducted during the pandemic, as the foundation for compiling a comprehensive list of the many gaps and needs in GLUT1 Deficiency. We identified classification methods that provided optimal coherence. Our overarching objective was to establish an information repository aimed at facilitating seamless accessibility for researchers to simplify data retrieval, identify associated publications, locate individuals engaged in this research and allow diverse participants to actively contribute ideas as research advances.
We decided to call our research roadmap a ‘Research Compass’ because we believe progress will come quickest and easiest not in a straight path or roadmap, but by creating relationships and collaborations across the many scientific corners that impact GLUT1 Deficiency (Figure 2). This is an open-source tool available on our website to anyone interested.

Schematic of the GLUT1 Deficiency Research Compass. Each area of focus is listed with some of the specific topics of interest. GLUT1 Deficiency patients and families are at the center of this compass guiding research in the community. Areas of focus for the Research Compass include GLUT1 – The transporter. Some of the topics of focus in this area include understanding its functions, activators, and what happens at the Blood–Brain Barrier (BBB) when GLUT1 does not function. Open-source research tools. For this area, our goal is to define the most relevant and reliable research tools and make them easily accessible to any researcher. Some of the resources currently available include a biorepository, disease animal models, and assays. Metabolism. Some of the topics of interest in this area comprise the downstream effects in the brain of GLUT1 malfunction and the role of glycogen and adenosine. Cells. Some of the topics of interest in this area include the types of cells involved in the disease, their location within tissues, optimal cell models, and cell lines. Pathogenesis. Some of the topics of interest in this area involve understanding the long-term effects of the disease, other organs or body systems affected, and the role of GLUT 1 in other diseases. Genetics. Some of the topics of interest in this area comprise the disease-causing variants, their functional effects, and the possibility of using gene therapy to cure this disease. Therapy development. Some of the topics of interest in this area include alternative treatments, and which treatments can be based on symptoms. Clinical tools. Some of the topics of interest in this area involve understanding the phenotypic spectrum of the disease, endpoints and outcome measures, and biomarkers that could help diagnose and determine treatment efficacy in clinical trials.
We are focusing on eight major areas: Open-source research tools, cell-centric questions, GLUT1-the transporter, metabolism, genetics, pathogenesis, therapy development, and clinical tools. Within each major focus area, we developed a list of questions and topics of interest, accompanied by a catalog of related publications. We also provide a roster of researchers actively engaged in these subject topics and who are both amenable to communication and open to collaborative endeavors.
Regarding open-source research tools, our goal is to define the most relevant and reliable tools to use for research and make them easily accessible to any researcher. The topics for focus in this area include: To determine and develop the most optimal cell and animal models for research; to standardize the research assays and procedures; and to determine and/or develop the optimal research and clinical tools. In particular, we want to focus on cell lines and biosamples, animal models, assays (including assays to quantify functional GLUT1 transporter), and clinical tools such as reliable biomarkers that will aid with future clinical trials. In this regard, we have established a biorepository available (Table 1), containing a diverse array of biological specimens, including plasma, serum, PBMCs, patient-derived iPSCs, fibroblasts, and other tissues. Researchers also have reliable mouse models available to them that they use to conduct their studies on GLUT1 Deficiency (Table 1).
Models available to study GLUT1 Deficiency.

We currently have two biorepositories, one at Coriell where we have one patient-derived iPSC cell line, 1 fibroblast cell line, and the LCL (lymphoblastoid cell lines). All other samples are stored through our membership with COMBINEDBrain at Van Andel Institute. Both GLUT1 Deficiency iPSC clones generated by CRISPR/Cas9 are currently stored at the researcher’s academic institution.
Another major area of focus is to more clearly define the cell types involved in GLUT1 Deficiency, their locations within tissues, and which ones can and should be targeted for treatment. Some of the topics for focus are to clearly determine which cell types are involved in the disease, to identify where the impacted cell types are located, and to develop comprehensive, diverse, and representative cell lines. In this area of focus, we are learning that the impacts of GLUT1 Deficiency go beyond the endothelial cells that were once thought to be the only cell type involved, with astrocytes now becoming a major focus as well as the downstream impacts on neurons. One group has developed a cell growth assay to study the functional effect of the SLC2A1 variants. 18 An additional group has genetically generated GLUT1 deficient iPSC cell lines 17 (Table 1) and BBB models to study the impact of GLUT1 Deficiency in brain energy metabolism and these have been added to our open-source research tools.
With regards to the focus area of GLUT1 – the transporter protein, our goal is to better understand the function of this protein, what influences its function, and how malfunctions can be corrected. Topics for focus in this area include to better understand the function of the GLUT1 transporter in different tissues throughout the body, determine how GLUT1 function is influenced and regulated, and to identify methods to correct protein malfunctions. Among some of the things we specifically want to learn are: what other factors at the BBB impact GLUT1 activities, what are other functions of GLUT1, and how is GLUT1 regulated at the transcriptional and post-transcriptional level. There has been some progress in the area with a recent publication on additional functions of GLUT1. 19 This publication reports that GLUT1 significantly contributes to transport of L-fucose and that this transport does not compete with D-glucose transport by GLUT1.
Metabolism is another major area of focus in our Research Compass. The goal for this area is to better understand the metabolic functions that are impaired by GLUT1 Deficiency and identify ways to compensate. Some of the topics include to better understand the role of glucose in metabolism, to identify other metabolites, enzymes substrates, and pathways involved, and to find methods to compensate for metabolic defects. Significant advancements have also been observed within this specific area of focus as well. Earlier this year, a proof-of-concept study was published, in which the authors postulated that the presence of glucose in red blood cells, combined with the deficiency of GLUT1 transporters in red blood cells of GLUT1 Deficiency patients, might offer an opportunity for improvement. They suggested that replacing these deficient cells with healthy red blood cells through exchange transfusion could potentially enhance glucose transport from red blood cells to neural cells through mass action. This effect is more likely to be observed when there is a healthy red blood cell count or an abundance of plasma glucose. 20
Another major area of focus is genetics. The goal for this area is to identify all of the genes that may play a role in GLUT1 Deficiency, how defects in the genes contribute to disease, and how their function can be repaired. Some topics for focus in this area include to better understand the functional implications of variants, clarify genotype/phenotype relationships, and identify regulators of SLC2A1 gene expression. A recent publication 13 shows progress in this area of focus, where the researchers developed a cell growth assay to quantify the functional impact of single nucleotide variants in SLC2A1.
Pathogenesis is another area of focus in the Research Compass. The goal in this area is to better define the clinical course of GLUT1 Deficiency, what causes the symptoms, and why they change over time. Some topics of focus include better understanding the patient journey, clarify and explain the long-term clinical course, and identify synergies with other GLUT1-involved diseases. One of the ways the GLUT1 Deficiency Foundation has contributed to this area is the Collective Voices in GLUT1 Deficiency Survey 12 (Figure 1). The enthusiastic engagement of the GLUT1 community has transformed this resource into a valuable source of information for patients, clinicians, and researchers. In addition, there have been publications on other conditions such as Alzheimer’s disease (AD) where it was described that the expression of GLUT1 transporter is reduced in certain areas of the brain in Alzheimer’s patients, 21 highlighting possible synergies between GLUT1 Deficiency and AD.
Another important area of focus is therapy development, where the goal is to better understand the benefits and limitations of current therapies as well as develop better ones. Some of the topics of focus include to develop better, easier treatments for all symptoms, optimize ketogenic dietary therapies, and target treatment outcomes most meaningful for patients and families. We have also gotten valuable insights for this area of focus from the Collective Voices in GLUT1 Deficiency Survey 12 [Figure 1(d)], as well as from other studies done on the ketogenic diet regarding its effectiveness,1,22 and exploring other possible therapies to treat GLUT1 Deficiency such as triheptanoin, 23 diazoxide 24 or lactate. 25
The last area of focus is clinical tools. The goal for this area is to develop tools to improve the diagnostic process, support better clinical care, and advance meaningful treatment development. Some of the topics for focus include to develop better and easier biomarkers, to identify the most appropriate outcome measures, and to develop a newborn screening tool. There has also been progress in this area of focus with publications describing different tools that can be used for monitoring the effect of the ketogenic diet in GLUT1 Deficiency patients 26 or tools for diagnosis 27 among others.
Natural History Study
This is an ongoing project for which we have established two opportunities to bring the power of this tool to the GLUT1 Deficiency patient community. The first opportunity is the GLUT1 Deficiency NHS on the Matrix platform, established in collaboration with other rare neurodevelopmental disorder organizations through COMBINEDBrain. This platform provides a secure place to share information about symptoms, development, and medical history based on information and data reported by patients and families, also known as patient-reported outcomes. The information is collected through a series of ClinGen-validated surveys, and the first questionnaire is a health and development survey. If patients report issues related to any of the body systems in the general health and development survey, other more in-depth surveys will be assigned with questions specific to that system. Patients enrolled in this study generate a unique identifier (CRID Number) 28 to facilitate research and link all data sources and biosamples donated by them in a de-identified manner.
One of the goals of the study is to include members of our global GLUT1 Deficiency community and make the data as diverse and representative as possible. To help meet this goal, surveys are available in multiple languages, including English, Spanish, German, Italian, French, Portuguese, Korean, and Hebrew. There are also capabilities to store and organize Electronic Health Records (EHR) and other important documents through the Matrix portal, along with the ability to track medications, symptoms, and events that are most important to the patient and family and analyze this data for trends and triggers.
The importance of collecting patients’ health information in order to understand how the disease develops and changes throughout time has also been highlighted by the Italian GLUT1 association, through the development of a patient registry in their community, 29 but these are clinician-reported outcomes and do not capture the patient perspectives through patient-reported-outcomes. In the future, as the Matrix platform evolves, we hope to add clinician-reported data and repositories for EEGs and other imaging files and be able to link all of these sources of data through a unique identifier system. Therapy developers and researchers interested in getting access to the current de-identified data for the 37 patients we have enrolled can contact the Glut1 Deficiency Foundation, COMBINEBrain, or Across Health.
The second opportunity is our digital NHS, which also was established in collaboration with other rare disease organizations and Ciitizen. This opportunity allows the collection of de-identified EHR that can be analyzed in a retrospective manner to help inform a better understanding of GLUT1 Deficiency and further research toward potential treatments, as well as to give patients and families the opportunity to collect all their EHR and store them digitally in one place. Therapy developers interested in getting access to the data can contact Ciitizen directly.
Discussion
The Glut1 Deficiency Foundation, a parent-led patient advocacy organization, is dedicated to increasing awareness, advancing education, advocating for patients and families, and supporting and funding research. Our overall objective is to position patients and their families as central figures in our advocacy, education, and research initiatives. With this in mind, the GLUT1 Deficiency Foundation launched a comprehensive survey aimed at gaining deeper insights into the experiences of individuals and families impacted by this disorder.
We received an impressive response from the GLUT1 community, with 260 individuals representing diverse demographics and geographic regions worldwide out of an estimated 2000–3000 who have received a diagnosis. However, it is noteworthy that certain countries in Latin America and Europe exhibited lower participation rates, despite our awareness of a larger number of patients in these regions based on correspondence and mailing lists. This observation underscores the importance of devising more effective strategies to ensure active participation of patients within these communities in such research studies, thereby achieving a more comprehensive representation of our diverse patient population.
The survey results highlighted several important points with a particular focus on the diagnosis process. The results reveal that, on average, patients require nearly 3 years from the onset of first symptom to receive a diagnosis. In addition, in most cases, diagnosis is provided by a neurologist, and genetic testing is the most common diagnostic tool. Interestingly, a significant number of the patients diagnosed by genetic testing lack clarity regarding the specific type of test employed. These findings underscore the need for more education concerning genetic testing within the GLUT1 Deficiency community. Moreover, there is potential for improvement regarding physician-patient communication during the diagnosis process, particularly in elucidating the type of genetic testing and the nature of genetic variants identified.
We were also able to determine that most of the patients participating in this survey exhibit the classical symptoms associated with GLUT1 Deficiency, including seizures and movement disorders. The results also indicated that most of the patients experience unusual eye/head movement as the first symptom. It is noteworthy that patients reported additional symptoms not typically documented in the existing GLUT1 Deficiency literature, including sleep disturbances (32%), attention issues (34%), anxiety (23%), obsessive-compulsive disorder (19%), and excessive drooling (19%), among others (data not shown). Furthermore, the survey outcomes indicate that the top three symptoms impacting patients and families in the GLUT1 Deficiency community are cognitive or intellectual difficulties, speech and communication issues, and lack of independence. These findings are particularly striking, as existing literature predominantly highlights seizures and movement disorders as the primary symptoms experienced by patients, with movement disorders often posing significant treatment challenges. However, it is evident from the GLUT1 Deficiency community perspective that these symptoms do not carry the same degree of impact on overall quality of life.
The outcomes pertaining to inquiries about clinical trials and patient priorities for treatment outcomes reveal that a substantial majority of patients and their families express a keen interest in participating in clinical trials. There is unified interest in new therapies aimed at restoring normal dietary habits, enhancing cognitive abilities, and improving speech and communication skills. Data from the survey underscore that a significant proportion of participants are willing to consider new treatment modalities in pill form, while a noteworthy percentage also voiced openness to oil or liquid forms or dietary therapy.
The willingness of the GLUT1 Deficiency community to participate in clinical trials inspired us to develop a Research-Ready Series. These meetings are a series of in-person and virtual meetings and represent a special ongoing project to build a strong, patient-centered research foundation and increase research literacy in our community. The goals of this project are to educate the community on the importance of patient-centered outcomes research, increase research literacy and clinical trial readiness, help ensure research is focused on the needs and priorities of patients, to strengthen relationships, and form new partnerships and collaborations across all stakeholders.
An important aspect of clinical trial preparedness involves cultivating a robust and dependable data repository that can serve as a guiding resource for researchers, clinicians, and industry stakeholders in the development of improved and innovative treatment modalities for clinical evaluation. The implementation of the NHS is poised to significantly contribute to this goal. It will facilitate research aligned with patients’ preferences and requirements, enhance symptom characterization, and delineate variations within the patient population. Thus, these efforts will provide the necessary insights to strategically design more effective treatments and clinical trials tailored to the specific needs of the GLUT1 Deficiency patient cohort.
Another contributing factor to the development of a research-ready GLUT1 Deficiency community is the Research Compass, which has already played a pivotal role in multiple areas and is a popular resource on our website. First, it has allowed us to identify key focal areas based on the priorities of our patients and families, as well as input from additional stakeholders. It has also enhanced our comprehension of the research landscape regarding GLUT1 Deficiency. Second, it has revealed gaps in research, treatments, and tools necessary for research and patient diagnosis and care. Third, we have utilized the Compass to identify key researchers who were previously outside our research network, thereby expanding our collaborative research efforts and amplifying and accelerating progress. Lastly, the Research Compass has enabled us to compile scientific publications relevant to each area of focus, consolidating this information in one place.
Some of the limitations of the Collective Voices Survey were that we did not get a full representation of the GLUT1 Deficiency patient population. We know there are more GLUT1 Deficiency patients in Latin America and Spain because we have been in contact with them through virtual gatherings and social media groups. However, their participation in the survey was low compared to the participation of the US patient population. Similarly, we had a lower participation of racial minorities. Our goal at the foundation is to include the voices of our global community, independent of location, ethnicity, or race. On this front, we continue to offer virtual gatherings for all the members of our community around the world, and we are planning on identifying better strategies to reach out to racial minorities in our community to have a better understanding of their experience and to better voice their needs and wants. An additional limitation of this survey is that the questionnaire used was not validated. While this is not optimal, the fact that we have only used the data as a guide to have a better understanding of the patients’ and families’ experience, and as a starting point to develop other programs and tools such as the Research Compass, gives us confidence that the data gathered is a good starting point for our community. Finally, another limitation of the survey, was that a power analysis was not performed. The purpose of this survey was to capture as many voices in our patient community as possible. For this reason, we launched the survey in multiple languages, so that our global community could participate.
Moving forward, we will continue to expand our research network to fill the research gaps in GLUT1 Deficiency. We are also committed to increasing patient engagement in the NHS, providing strong, relevant, meaningful, and significant data to researchers, clinicians, and industry stakeholders dedicated to the advancement of improved treatments. Furthermore, we aspire to foster the development of treatments that demonstrate efficacy across the spectrum of symptoms, benefiting all members of our patient community. Lastly, we aim to establish a Center of Excellence to improve patient care, focus collaborative research, and speed translation to the clinic. This comprehensive care center will offer streamlined and interconnected multidisciplinary care, will allow consolidated data collection to learn more about the patient experience, and improve service for patients and families in the community. Coordinating care in one center with the appropriate expertise, care, and services will improve patient care, advance research, and facilitate the rapid translation of scientific progress from bench to the clinic.
Conclusion
The mission of the Glut1 Deficiency Foundation is to improve lives in the GLUT1 Deficiency community through increased awareness, improved education, advocacy for patients and families, and support and funding for research. In order to better serve our community we launched the Collective Voices Survey to hear the voices of patients and families in our community to have a better understanding of their needs and wants. Overall, the results from the survey showed that there is a diverse community of patients with GLUT1 Deficiency presenting a wide range of symptoms which can vary over time. The results highlight that patients and families prioritize the need for developing better treatments that improve their quality of life, emphasizing the importance they place on the social aspects, such as being able to eat a normal diet and improving their cognitive and communication skills. In addition, learnings from the survey enabled us to identify a list of research gaps regarding GLUT1 Deficiency and to develop a Research Compass that is now guiding research focus and priorities in our community with patients and their needs at the center.
Supplemental Material
sj-pdf-1-trd-10.1177_26330040241265421 – Supplemental material for The road toward patient-led research in the GLUT1 Deficiency community: a patient organization perspective
Supplemental material, sj-pdf-1-trd-10.1177_26330040241265421 for The road toward patient-led research in the GLUT1 Deficiency community: a patient organization perspective by Sandra Stella Ojeda Torres, Chantal Marie Sanchez and Glenna Spencer Steele in Therapeutic Advances in Rare Disease
Footnotes
Acknowledgements
The authors thank the GLUT1 Deficiency community for their support through participation in the Collective Voices in GLUT1 Deficiency Survey and the Natural History Study, for donating samples to the biorepository and for participating in the Foundation’s conferences. Additionally, we thank our scientific advisor Dr. Matthew Gentry for reviewing this manuscript. Finally, we thank all the researchers and clinicians working to find better treatments and eventually a cure for GLUT1 deficiency.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
