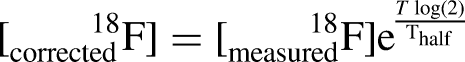Abstract
Background
Local and systemic side effects of glucose remain major limitations of peritoneal dialysis (PD). Glucose transport during PD is thought to occur via inter-endothelial pathways, but recent results show that phloretin, a general blocker of facilitative glucose channels (glucose transporters [GLUTs]), markedly reduced glucose diffusion capacity indicating that some glucose may be transferred via facilitative glucose channels (GLUTs). Whether such transport mainly occurs into (absorption), or across (trans-cellular) peritoneal cells is as yet unresolved.
Methods
Here we sought to elucidate whether diffusion of radiolabeled 18F-deoxyglucose ([18F]-DG) in the opposite direction (plasma → dialysate) is also affected by GLUT inhibition. During GLUT inhibition, such transport may either be increased or unaltered (favors absorption hypothesis) or decreased (favors transcellular hypothesis). Effects on the transport of solutes other than [18F]-DG (or glucose) during GLUT inhibition indicate effects on paracellular transport (between cells) rather than via GLUTs.
Results
GLUT inhibition using phloretin markedly reduced [18F]-DG diffusion capacity, improved ultrafiltration (UF) rates and enhanced the sodium dip. No other solutes were significantly affected with the exception of urea and bicarbonate.
Conclusion
The present results indicate that part of glucose is transported via the transcellular route across cells in the peritoneal membrane. Regardless of the channel(s) involved, inhibitors of facilitative GLUTs may be promising agents to improve UF efficacy in patients treated with PD.
Introduction
Glucose is added to peritoneal dialysis (PD) fluids to produce osmotic water removal across the peritoneal membrane. The presence of water channels in endothelial cells lining peritoneal capillary- and venular walls gives rise to free-water transport accounting for about 40–50% of total ultrafiltration (UF),1–3 essentially doubling UF efficiency in terms of mL UF per gram glucose absorbed. 4 Adverse effects from glucose exposure from PD fluids, both systemic and local, remain important clinical problems, resulting in the development of metabolic complications, 5 altering the structure and function of the peritoneal membrane,6,7 and reducing the efficiency of the treatment—ultimately shortening time on therapy.
Glucose is impermeable across cell membranes, and is transported into cells via sodium-independent, facilitative glucose transporters (GLUTs) and sodium–glucose co-transporters (SGLT). Most cells express more than one kind of GLUT. Fourteen GLUT proteins have been described in humans (GLUTs 1–14), and mediate the transmembrane movement of various monosaccharides and even myoinositol (GLUT13), urate (GLUT9), glucosamine (GLUTs 1/2/4), and vitamin C. 8 Among the 14 members in this family, GLUT1-4 is the most extensively studied and is believed to be most important for cellular glucose uptake. GLUTs have been detected in the peritoneal membrane.9,10 Schröppel et al. demonstrated GLUT1 and GLUT3 expression in cultured human mesothelial cells and induced GLUT1 and 3 mRNA expression and glucose uptake by cells exposure to high glucose and cytokines. 11 More recently, Schricker et al. found evidence of GLUT1 and GLUT3 expression in peritoneal membrane biopsies both by healthy controls, pre-dialytic patients, patients on PD, and in patients diagnosed with encapsulating peritoneal sclerosis, but analysis showed no significant differences in GLUT1 and 3 expressions between the different subgroups. 9 Balzer et al. showed in a mice model that GLUT 1 and 3 were upregulated, but GLUT4 was downregulated in response to chronic glucose-based PD fluid exposure. 12
Our group recently demonstrated that phloretin, a non-selective GLUT blocker, reduced glucose absorption and improved ultrafiltration in a rat PD model. 13 Due to the non-selective nature of phloretin, we could not answer the question via which GLUTs glucose absorption occurred, and whether GLUT-mediated glucose transport mainly occurs across or into cells. It is also possible that phloretin reduced para-cellular transfer, although no differences were observed for other small solutes. In order to cast light on these important questions, we investigated the effects of GLUT inhibition—using either a selective GLUT1 inhibitor BAY-876, non-selective GLUT blocker phloretin and GLUT1/4 blocker ritonavir 14 —on the transport of 18F-deoxyglucose ([18F]-DG) in the opposite direction, from plasma to dialysate, during dialysis with 2.3% glucose fluids (Figure 1). In a separate line of experiments, we studied the effects of GLUT4 inhibition during PD using intra-peritoneal indinavir. 14
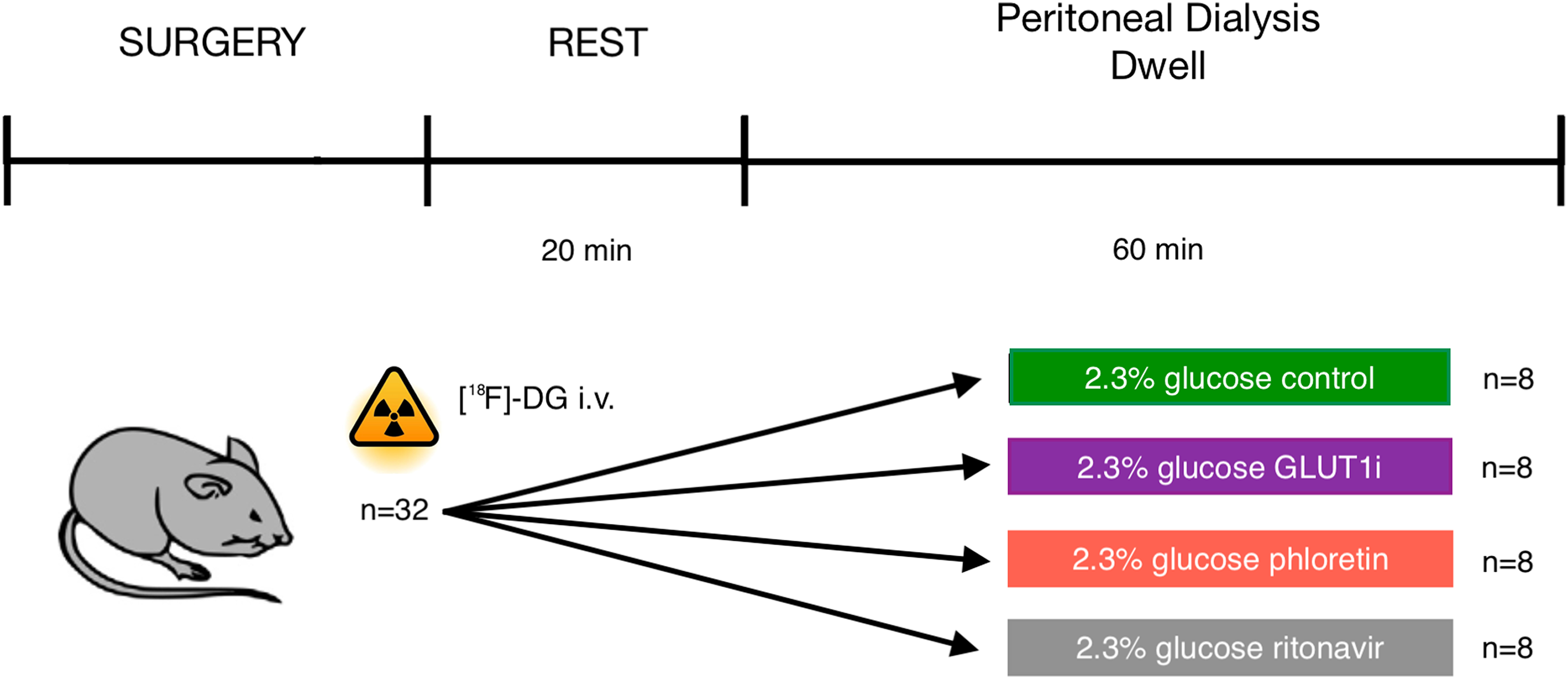
Schematic diagram of the experimental setup. Peritoneal dialysis was performed in anesthetized Sprague–Dawley rats using a fill volume of 20 mL with 2.3% glucose fluid with or without selective GLUT1 blocker BAY-876, non-selective GLUT blocker phloretin, and GLUT1/GLUT4 blocker ritonavir. Dialysate samples were obtained at baseline and after 30 min and 60 min dwell time. Routine blood samples were obtained before and after dialysis.
Methods
Animals
Experiments were performed in male Sprague–Dawley rats having an average body weight of 304 g (215−348) with unrestricted access to water and food (Special Diets Services RM1(P) IRR.25 no. 801157). All animals were treated according to the guidelines of the National Institutes of Health for the Care and Use of Laboratory Animals. The Ethics Committee for Animal Research at Lund University approved the experiments (Dnr 5.8.18-08386/2022). Exclusion criteria were signs of bleeding into the dialysate, premature death, signs of circulatory or respiratory failure, leakage of dialysis fluid, or technical malfunction of the equipment. Animal care staff and the technician performing the experiments were unaware of the purpose of the experiments to ensure that all animals in the experiment were handled/treated in the same way. Reporting of results is in compliance with the ARRIVE guidelines. Each animal was gently placed in a covered glass container to which a continuous supply of 5% isoflurane in air (Isoban, Abbott Stockholm, Sweden) was connected. The animal was taken out of the container after being properly anesthetized. After that, 1.6–1.8% isoflurane in air was administered using a small mask to maintain the anesthesia. After tracheostomy, the animals were connected to a ventilator (Ugo Basile; Biological Research Apparatus, Comerio, Italy) and ventilated in a volume-controlled mode using a positive end-expiratory pressure of 4 cm H2O. Body temperature was kept between 37.1°C and 37.3°C via a feedback-controlled heating pad. End-tidal pCO2 was monitored continuously and kept between 4.8 and 5.5 kPa (Capstar-100, CWE, Ardmore, PA, USA). The left femoral artery was cannulated for monitoring of mean arterial pressure and heart rate; and to obtain blood samples (95 µL) for measurement of glucose, creatinine, urea, electrolytes, hemoglobin, and hematocrit (i-STAT, Abbott Laboratories, Abbott Park, IL, USA) before and after dialysis. The right femoral vein was cannulated and used for continuous saline infusion of 50 µL/min. The right internal jugular vein was cannulated for drug infusion. Access to the peritoneal cavity was established percutaneously via a multiholed silastic catheter (Venflon, BOC Ohmeda AB, Helsingborg, Sweden; outer diameter 1.7 mm) secured to the skin using cyanoacrylate (Histoacryl, B. Braun Surgical, Rubi, Spain). After 60 min the dialysate was recovered from the peritoneal cavity, first by using a syringe, and thereafter carefully retrieving the rest of the fluid using pre-weighed gauze tissues. A small amount of creatinine (∼0.3 mmol/L) was added to the dialysis fluid prior to infusion to allow dialysate concentrations to exceed the limit of detection of the i-STAT device. Hematocrit was determined by centrifugating thin capillary glass tubes. PD solutions were pre-warmed to 37°C before instillation. Animals were euthanized with an intravenous bolus injection of potassium chloride after the experiment. Prior to dialysate sampling, 1 mL was flushed back and forth several times to ensure a valid sample from the dialysate. Solute mass balance (e.g., absorption) was determined as the mass (volume) out minus mass (volume) in. Ultrafiltration rates were calculated from the 30- and 60-min sodium dip according to the method by Helman et al. 15 Plasma-to-dialysate clearances were calculated as the average solute removal rate (based on mass balance) divided by the mean plasma concentration (mmol/L). Dialysate clearances were calculated as the mean solute removal rate (based on mass balance) divided by the average dialysate concentration (mmol/L). Isovolumetric and isocratic diffusion capacities are calculated as described previously. 16
Studies of plasma-to-dialysate clearance of [18F]-DG
The study consisted of four groups of animals treated without intervention (SHAM; n = 8) or with BAY-876 (25 mg/L; n = 9), phloretin (50 mg/L; n = 8) or ritonavir (60 mg/L; n = 8; all from Merck, Darmstadt, Germany) in 20 mL of 2.3% glucose PD fluid (Balance, Fresenius, Bad Homburg, Germany). The BAY-876 dose was well above the IC50 concentration for GLUT1.
17
Similarly, the ritonavir dose used was nearly one order of magnitude larger than the IC50 concentrations, being ∼7 µM for both GLUT1 and GLUT4.
14
One animal in the BAY-876 was excluded from analysis due to a malfunctioning infusion pump. On the day of the experiment, [18F]-DG was retrieved in the morning from the radiopharmaceutical preparation unit (part of the Department of Radiation Physics) at Skåne University Hospital in Lund in quantities of 200–500 MBq. A smaller amount of [18F]-DG was then added to saline to produce fluid for intravenous administration at a rate of 50 µL/min. Experiments were performed in collaboration with a radiation expert, and all staff handling [18F]-DG were equipped with dosimeters. Samples for radioactivity measurements (20 µL) were obtained from the dialysate at 1, 10, 20, 30, 40, 50, and 60 min, and blood plasma (obtained by centrifugation) at 5, 15, 25, 35, 45, and 60 min and analyzed on a gamma counter (Wizard 1480; Wallac Oy, Turku, Finland) to determine [18F]-DG activities. Since the half-life (Thalf) of [18F]-DG is only 109.734 min, measured activities were corrected for the decay at time T in minutes from the start of dialysis as follows
Studies of intra-peritoneal indinavir 50 mg/l
This study consisted of two groups of animals treated without intervention (SHAM; n = 8) or with indinavir (50 mg/L; n = 9; Merck, Darmstadt, Germany) in 20 mL of 1.5% glucose PD fluid (Balance, Fresenius, Bad Homburg, Germany). The dose used (50 mg/L) is circa 4 times the IC50 concentration for GLUT4 and about half of the IC50 concentration for GLUT1. 14 In these additional experiments, no animal was excluded from analysis. 125I-albumin and 51Cr-EDTA were added to the dialysate to measure intra-peritoneal volume and to assess small solute diffusion capacity, identical to experiments in. 13 Urinalysis showed multiple flat colorless needle-shaped crystals in indinavir-treated animals (Supplementary Figure 1).
GLUT1 immunostaining
The surgically removed peritoneum tissue from rats was immediately transferred into buffered 4% formaldehyde (#02176; Histolab, Askim, Sweden) for 24 h, and subsequently tissue blocks were dehydrated in an automated dehydration machine (SAKURA-Tissue-Tek VIP® 6 AI; Sakura Finetek USA, Inc., Torrance, CA, USA). As part of the antigen retrieval process, slides (4 µm thickness) were baked at 60°C for 45 min and treated with a low pH target retrieval solution (#DM829; Dako, Glostrup, Denmark) in a DAKO PT Link HIER machine (PTlink 200; Dako, Glostrup, Denmark). The endogenous peroxidase enzyme was blocked with a peroxidase-blocking reagent (# DM821; DAKO, Glostrup, Denmark) for 10 min. Peroxidase-blocked slides were incubated with the primary antibody against GLUT1 (#SA0377; Thermo Fischer Scientific, Waltham, MA, USA) for 1 h, followed by the secondary antibody (#DM822, Envision Flex, Dako, Glostrup, Denmark) for 30 min and finally substrate the HRP-conjugated magenta dye (#DM857; DAKO, Glostrup, Denmark) for 10 min. For all the washing steps, readymade wash buffer (#DM831; Dako, Glostrup, Denmark) was used. After the immunostaining, the slides were counterstained with Mayer's hematoxylin (#01820; Histolab, Askim, Sweden), and the coverslip (ECN 631-1574; VWR, Radnor, PA, USA) was mounted with Pertex (#00840; Histolab).
Statistical methods
Data are shown as median (intraquartile range) unless otherwise indicated. Sample sizes were chosen to be similar to those in our previous experiments using GLUT inhibitors. 13 Significant differences were assessed using a Kruskal–Wallis test followed by post-hoc pairwise comparisons using Dunn's test when significant. All P-values and confidence intervals are reported “as is” without corrections for multiple comparisons. 18 Calculations were performed using R for mac version 4.1.1.
Results
Non-selective GLUT inhibition using phloretin reduced diffusion of radio-labeled [18F]-DG whereas selective GLUT1 inhibition with BAY-876 had no effect
The radionuclide [18F]-DG is widely applied for positron emission tomography imaging. The released positron is immediately annihilated upon collision with an electron, producing gamma radiation. After entering the cell via the same pathways as glucose, [18F]-DG is converted by hexokinase to [18F]-deoxyglucose-6-phosphate which is essentially trapped in the cell (Figure 2(A)). Thus, one may expect that inhibiting cellular uptake in the peritoneal membrane would improve diffusion of [18F]-DG. In contrast, during a 60 min dwell utilizing 2.3% glucose, we noted marked decrements in [18F]-DG diffusion capacity during non-selective inhibition of GLUT channels using phloretin, and GLUT1:GLUT4 blocking by ritonavir, but apparently not during selective GLUT-1 inhibition by BAY-876 (Figure 2(B); Supplementary Table 1). The decrease in [18F]-DG diffusion capacity using phloretin was confirmed using the three-pore model (Supplementary Table 2). Glucose diffusion capacities (mass transfer area coefficients [MTACs]) were not significantly different between groups (Figure 2(C)), but [18F]-DG MTACs were closely correlated with glucose MTACs (R2 = 0.49; Figure 2(D)) and Bland–Altman analysis indicated no difference indicating that the capacity for glucose transport across the peritoneal membrane is the same—regardless of the direction of transport. The amount of UF in mL per gram glucose was markedly increased by both phloretin and ritonavir (Figure 2(F)). In contrast to results using [18F]-DG, neither intervention affected MTACs for creatinine (Figure 3(A)) or any other measured solute (Figure 3(C)–(F)), with the expected exception of urea (Figure 3(B); phloretin inhibits urea transport) but also bicarbonate (Figure 3(G)). No effects were observed regarding glucose plasma concentration or any other systemic parameter before or after dialysis (Supplementary Table 3).
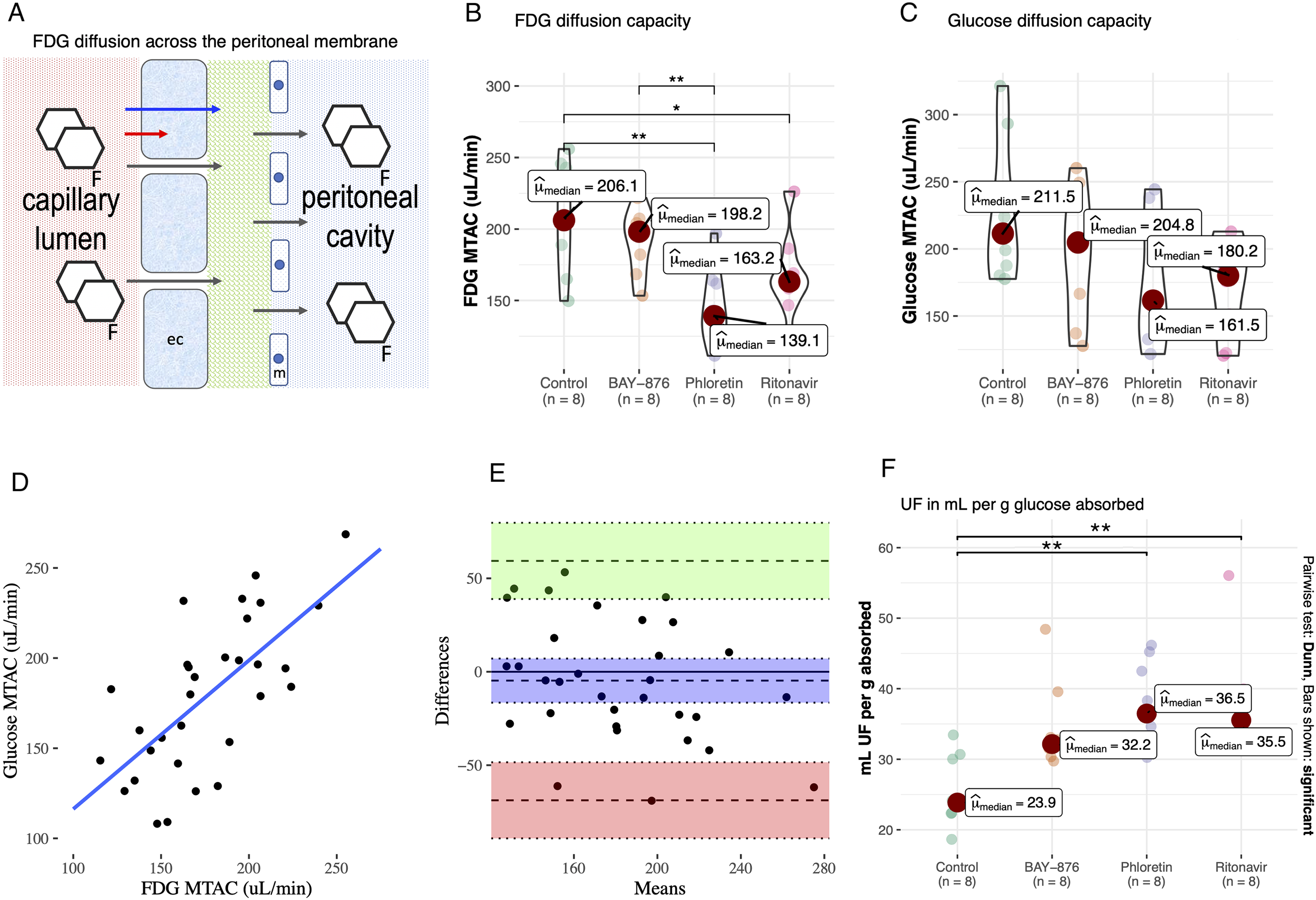
Pan-GLUT inhibition using phloretin and GLUT1:GLUT4 inhibition using ritonavir resulted in decreased diffusion capacity for [18F]-deoxyglucose. (A) Schematic illustration of [18F]-DG diffusion across the peritoneal membrane. (B) Impact on [18F]-DG diffusion capacity using selective GLUT-1 inhibitor BAY-876, non-selective GLUT inhibitor phloretin and GLUT1/4 inhibitor ritonavir. (C) Glucose diffusion capacity using BAY-876, phloretin and ritonavir. (D) Linear regression between [18F]-DG and glucose MTACs. (E) Bland–Altman plot of the difference in MTAC between [18F]-DG and glucose. (F) Ultrafiltration efficiency for the different treatment groups.
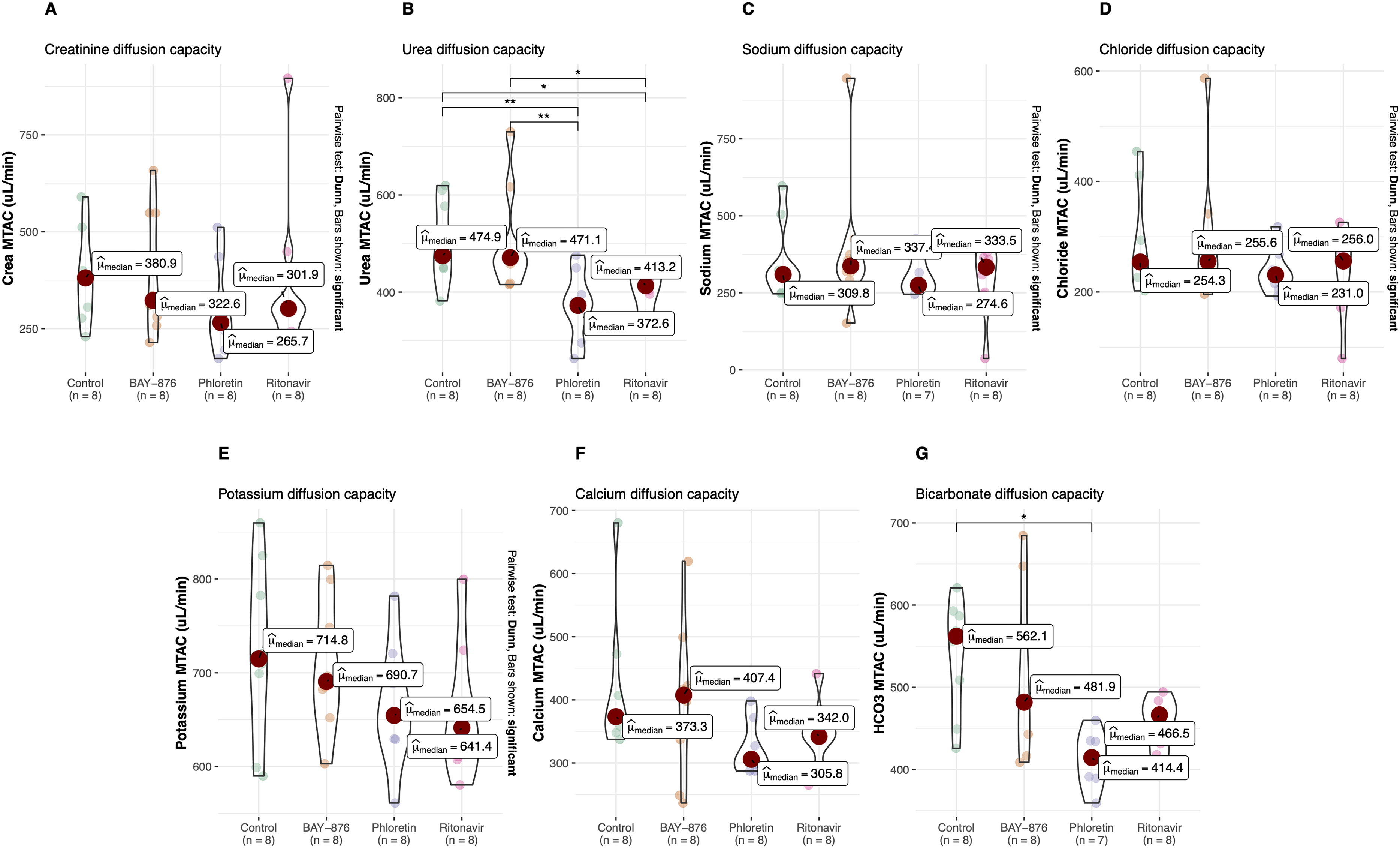
No other solutes were significantly affected with the exception of urea and bicarbonate. Impact on (A) creatinine, (B) urea, (C) sodium, (D) chloride, (E) potassium, (F) calcium, (G) bicarbonate diffusion capacity using selective GLUT-1 inhibitor BAY-876, non-selective GLUT inhibitor phloretin and GLUT1/4 inhibitor ritonavir.
Inhibition of GLUT4 using indinavir did not affect glucose- or osmotic water transport during experimental PD
In the next step, we wanted to elucidate whether the observed effects of GLUT1:GLUT4-blocker ritonavir were due to blocking of GLUT4. We performed separate experiments in 16 animals with 20 mL dialysis fluid with either GLUT4 inhibitor indinavir or sham. Baseline parameters are shown in Supplementary Table 5 and main outcomes in Supplementary Table 6. Taken together the results show that indinavir did not significantly affect glucose transport or any parameter related to osmotic water transport. In particular, there was no difference in UF per g glucose absorbed. A summary of key outcomes for the different interventions in the present study is displayed as forest plots in Figure 4.
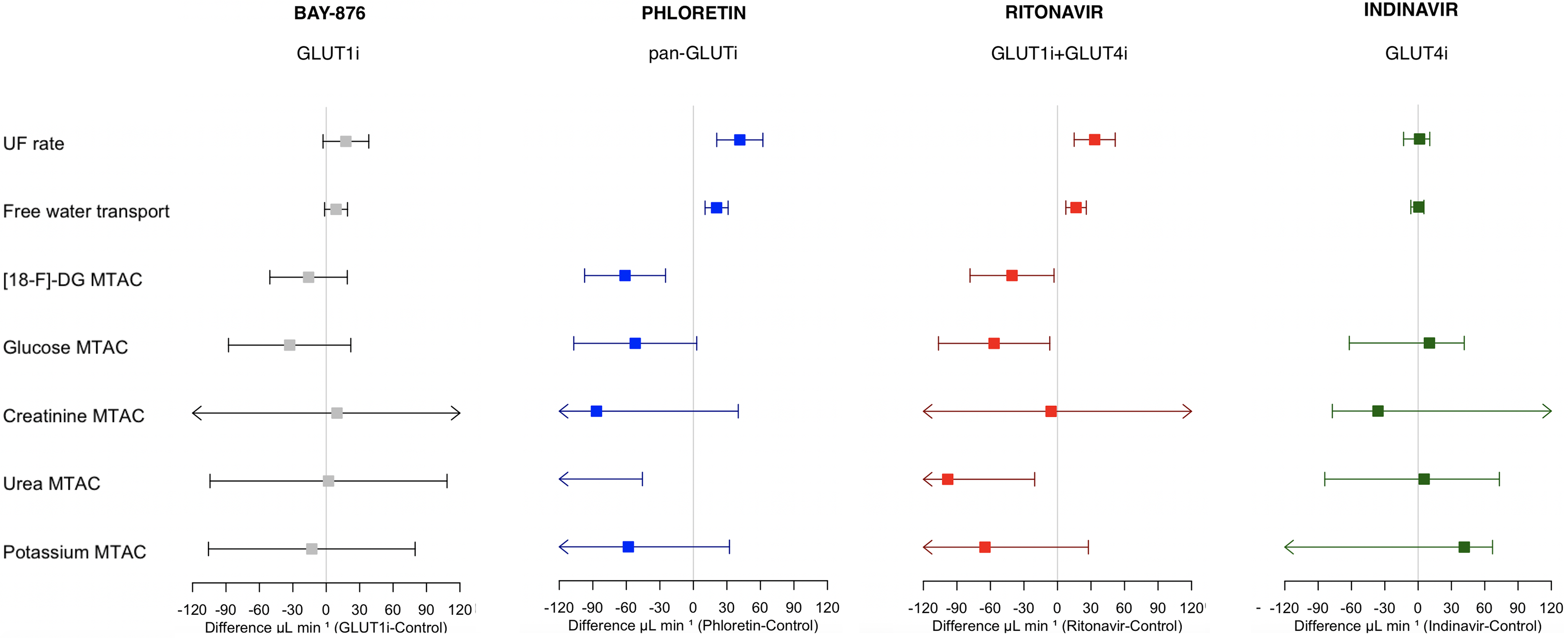
Treatment effects for selective GLUT1 inhibitor BAY-876, non-selective GLUT-inhibitor phloretin, non-specific GLUT1/4 blocker ritonavir, and non-specific GLUT4 blocker indinavir. 95% CIs of the drug effects on UF rate (UFR), free-water transport, and isocratic diffusion capacities (MTACs) for [18F]-deoxyglucose (not present in indinavir experiments), glucose, creatinine, urea, and potassium. Square boxes represent medians of the difference (drug control). Free water transport was approximated as UFR - sodium clearance.
GLUT1 is mainly localized to the endothelium of peritoneal blood vessel walls
Given the negative results of GLUT4 inhibition using indinavir, but positive results using GLUT1:GLUT4 inhibitor ritonavir, there is reason to believe that the present effects are, at least in part, mediated via the GLUT1 transporter. Immunohistochemical staining of GLUT1 in biopsy specimens from the peritoneum (Figure 5(A)) using specific antibodies revealed diffuse positivity for GLUT1 in the endothelium of vessel walls, while the mostly denudated surface didńt show any reaction in mesothelial cells, nor any reaction in myocytes or noticeable reaction in fibroblasts (Figure 5(B)).
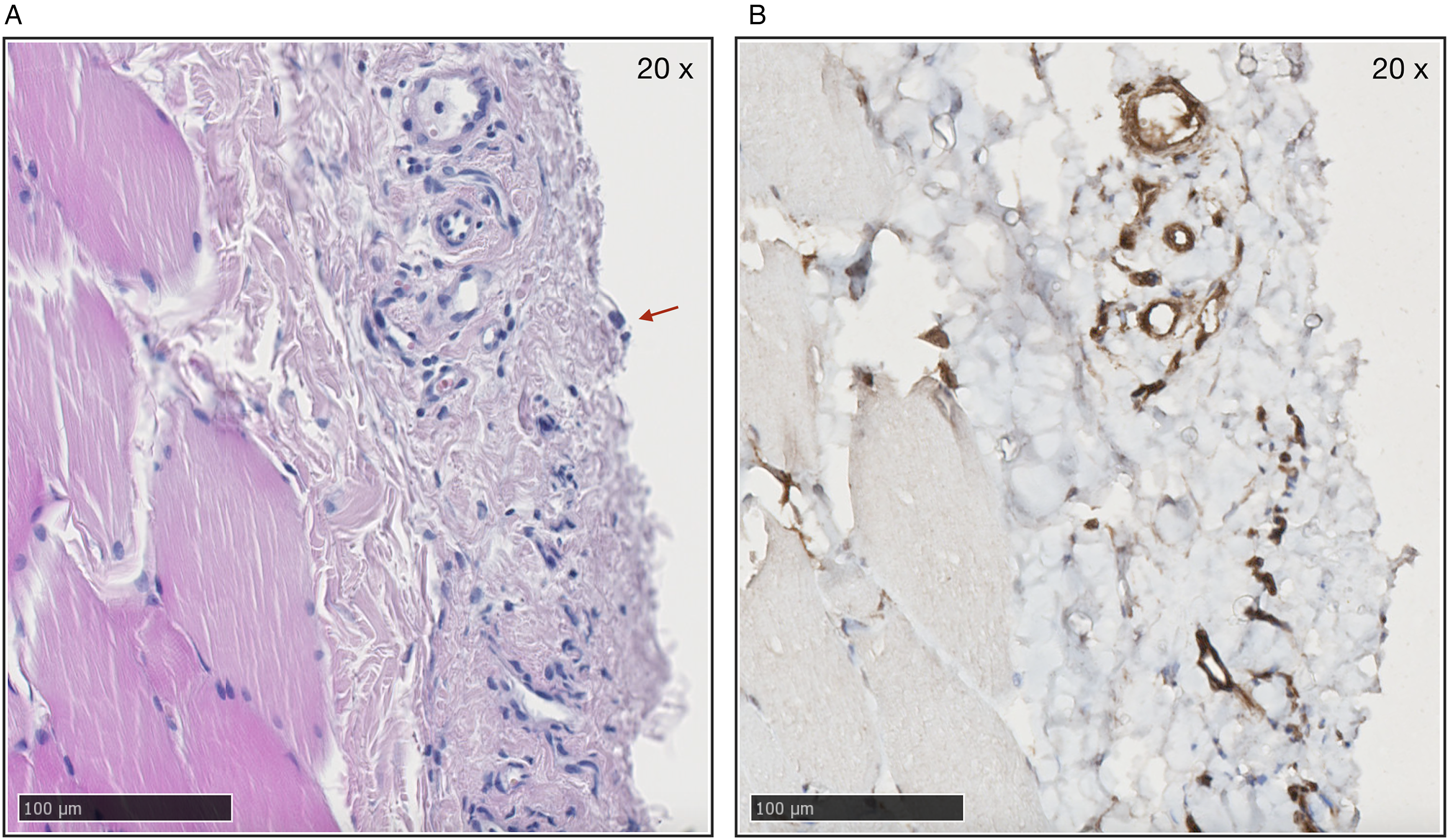
GLUT1 is localized to the endothelium of peritoneal blood vessel walls. (A) Parietal peritoneum (hematoxylin and eosin stain 20×) with several small vessels. Note single mesothelial cell (arrow). (B) Immune-staining of parietal peritoneum for GLUT1 (brown). Positive reaction in endothelium of small vessels shown in picture. Note the absence of reaction elsewhere.
Discussion
The main finding of this study is that [18F]-DG diffusion capacity was markedly reduced by the non-selective GLUT blocker phloretin while the transport capacity of most other solutes appeared unaffected. These results support the hypothesis that a significant part of glucose is transferred via GLUTs in a trans-cellular fashion during PD. The findings also confirm recent results by our group using phloretin 13 and our previous findings using phlorizin 19 (which is converted to phloretin). Actual transcellular transfer of glucose across endothelial cells in peripheral capillaries has as yet not been formally demonstrated. 20 The present data nevertheless suggest such direct transport of glucose across cells, as we observed a marked decrease in transport from plasma to dialysate during non-selective GLUT inhibition, with minimal effects on other solutes. However, in order for [18F]-DG (and glucose) to be transported across a cell, it must evade enzymatic phosphorylation by hexokinase, since it would otherwise be trapped inside the cell. It is possible that hexokinase becomes saturated during the rather extreme glucose concentrations during PD. However, an early study by Betz et al. has shown that not all [18F]-DG is converted by hexokinase, and 30–60% remains unphosphorylated 21 already at normal glucose concentrations.
The next question is via which transporters such transcellular passage of [18F]-DG occurs. Experiments using GLUT1:GLUT4 inhibitor ritonavir (positive) and GLUT4 inhibitor indinavir (negative), clearly point towards GLUT1 as being the main channel responsible for the present effects. The negative results using the selective GLUT1 inhibitor BAY-876 thus appear difficult to explain, with results being negative for glucose and [18F]-DG transport. However, it is well known that BAY-876 is poorly soluble in water and, thus, we suspect that actual dialysate concentrations may have been far lower than the IC50 of ∼2.5 nM, rendering BAY-876 inefficient when administered in the dialysate. It is well established that pan-GLUT inhibitor phloretin blocks urea transporters 22 but the present results show that using ritonavir was also effective in decreasing urea transport to the dialysate, indicating a potential inhibitory action by ritonavir on urea transporters.
The limitations in this study are mainly related to the low number of animals in each group and the acute nature of the experiments, meaning that long-term effects cannot be evaluated from these data. Next, glucose measurements had an error (VC) of about 6% which likely contributes to variance between groups, which might explain the finding of no difference between groups when it comes to glucose. Also, control animals did not receive solvent (100 µL dimethyl sulfoxide) used to dissolve the interventions (BAY-876, phloretin, and ritonavir), and interventions were also given in a single dose. Lastly, the present experiments compared three interventions (BAY-876, ritonavir, and phloretin) with a single control (SHAM) group which may have decreased statistical power to detect significant differences.
We conclude that data so far support a role for GLUT1 in trans-cellular peritoneal glucose transport. Further experimental studies using more selective blockers are important as a next step, since ritonavir may have other effects unrelated to GLUTs. Given the availability of several medications with inhibitory action on GLUTs, and the fact that the expression in peritoneal tissues of these transporters appears to be increased during PD, clinical trials to assess the therapeutic effects of glucose transport inhibitors in PD patients are warranted as the next step. Our recent pre-clinical study 13 indicates that approximately 20–30% of glucose may be transported via GLUTs. This appears to be in contrast with clinical results in a previous study from our group showing that creatinine and glucose diffusion capacities in PD patients were highly correlated. 23 However, such a correlation should be expected since most glucose is transferred via the same pathway as creatinine, via inter-endothelial channels (small pores).
Supplemental Material
sj-docx-1-ptd-10.1177_08968608241299928 - Supplemental material for Transcellular transport of 18F-deoxyglucose via facilitative glucose channels in experimental peritoneal dialysis
Supplemental material, sj-docx-1-ptd-10.1177_08968608241299928 for Transcellular transport of 18F-deoxyglucose via facilitative glucose channels in experimental peritoneal dialysis by Giedre Martus, Premkumar Siddhuraj, Jonas S Erjefält, András Kádár, Martin Lindström, Karin Bergling and Carl M Öberg in Peritoneal Dialysis International
Footnotes
Acknowledgments
The authors are very grateful for the excellent experimental work by Helén Axelberg. We are also grateful for all helpful advice from radiation expert Hanna Holstein, and the excellent people at the Department of Medical Radiation Physics at Lund University.
Data availability
All original data reported in this article have been deposited in Dryad (doi:10.5061/dryad.f1vhhmh52) and can be accessed online via http://doi.org/10.5061/dryad.f1vhhmh52.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Giedre Martus has a collaboration with Triomed AB (Lund, Sweden) unrelated to the present work. Karin Bergling has pursued two master thesis projects with Gambro Lundia AB (unrelated to this work). Karin Bergling reports research funding from Baxter Healthcare and patents or royalties with Baxter Healthcare. At the time of publication, Karin Bergling was no longer employed at Lund University. Karin Bergling and Carl M. Öberg are inventors of a pending patent filed by Gambro Lundia AB (Baxter; unrelated to this work). Carl M. Öberg reports research grants (unrelated to this work) from Baxter Healthcare and Fresenius Medical Care and speaker's honoraria from Baxter Healthcare and Boehringer Ingelheim. Carl M. Öberg reports a consultancy agreement with Baxter Healthcare and an advisory or leadership role with the Peritoneal Dialysis International editorial board and the editorial board of Nature Scientific Reports. All other authors have nothing to disclose.
Ethical approval
The Ethics Committee for Animal Research at Lund University approved the experiments (Dnr 5.8.18-08386/2022).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Crafoord Foundation (ref. no. 20230690), Lund University Medical Faculty Foundation grant (YF 2020-YF0056), the Swedish Kidney Foundation grant (F2023-0051), and The Inga-Britt and Arne Lundberg’s Research Foundation (all to Carl M. Öberg).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.