Abstract
Families are a driving force in accelerating the understanding and science of SCN8A. The urgency felt by families facing the absence of treatments for their children makes them uniquely positioned to advance therapies through advocacy, data sharing, and partnerships. The International SCN8A Alliance (Alliance) brings families together to collaborate on advancing the science of SCN8A. The Alliance hosts SCN8A scientific meetings – facilitating coordination and collaboration among clinicians, researchers, industry, and the SCN8A community; funds early investigators to support research – building a new generation of investigators; builds and maintains a robust and dedicated International SCN8A Registry (Registry) providing longitudinal data on the natural history of the disorder and leading to over two dozen publications; cultivates partnerships with key stakeholders to accelerate innovation and progress including a Research Consortium, Global Clinicians Network, and the first global Consensus on the Diagnosis and Treatment of SCN8A; coordinates global community engagement by hosting families in virtual meetings in multiple languages and uniting advocates from across all epilepsies to call for more strategic and expanded investment in the epilepsies; builds and hosts the Global SCN8A Leaders Alliance (Leaders Alliance) promoting coordination and collaboration among leaders of SCN8A organizations worldwide; and advances a Global SCN8A Research Roadmap (Research Roadmap) – convening leading stakeholders in the SCN8A community to identify research priorities and accelerate progress toward better care, treatments, and outcomes. The outsized impact of small family advocacy organizations demonstrates that patient advocates can be effective agents in accelerating new therapeutics through maximizing their power to convene diverse stakeholders around a shared vision grounded in patient/caregiver priorities, maintaining a core focus on improving outcomes that are most important to families, and recognizing the importance of being bold, thinking big, and collaborating across disease areas.
Plain language summary
Many rare disease caregivers experience the dreaded realization that even with access to experienced clinicians the answers on how to best treat their children remain elusive.
Like leaders of many rare epilepsy patient organizations, the Alliance founders refused to accept the status quo and organized efforts to accelerate scientific progress in SCN8A. The foundation, initially called Wishes for Elliott (WFE), began in a Pediatric Intensive Care Unit motivated by delayed diagnosis, time lost on ineffective or exacerbating treatments, and countless hospitalizations where specialists had no evidentiary basis for life and death decisions. Many SCN8A families, motivated by these persistent challenges, are stepping forward to advance the understanding of SCN8A by sharing their experiences and data.
Over the past decade, the Alliance expanded efforts to include a wide range of partners and initiatives focused on building partnerships that break down silos. This includes work to: host SCN8A scientific meetings to promote coordination and collaboration among scientists and the SCN8A community; fund early investigators to support innovative research; build a dedicated SCN8A Registry providing longitudinal data on how the disease and treatments affect individuals; cultivate partnerships with all stakeholders to accelerate innovation and more effective treatments; coordinate global community engagement; build and host the Leaders Alliance to unite SCN8A advocacy groups; and advance the Research Roadmap to address critical gaps and advance a shared vision of research priorities grounded in patient/caregiver priorities.
While disease-focused patient advocacy organizations face many challenges in advancing new treatments, their impact is extended by convening stakeholders, engaging in broad collaborations beyond their specific disease, and being bold/thinking big. Effective collaboration enables the efficient coordination of scarce time, energy, and resources to address the complexities of a rare disorder like SCN8A.

Keywords
Background
Patient advocacy leadership to accelerate scientific progress in SCN8A is central to the work of the Alliance. The Alliance evolved from the first family-based foundation dedicated to advancing SCN8A research into a global collaborative of nearly all key stakeholders. The genesis of the work and mission of the Alliance and its partners mirrors nearly every rare epilepsy patient advocacy organization – receiving a diagnosis of a genetic epilepsy disorder that came with little information and no answers. The journey to a genetic diagnosis is frequently prolonged, marred by irreparable damage resulting from either the absence of treatment or inappropriate interventions for catastrophic seizures and profoundly delayed development.
Receiving a rare genetic epilepsy diagnosis initiates a new phase in the journey for answers as even the most experienced clinicians are often uncertain of how to best treat affected individuals or understand their prognosis. For families, it is clear that until more is learned, care for affected children rarely changes with a diagnosis. For most, treatment is still largely guessing which antiseizure medications to use, in what amounts, and in what combinations.
Evolution of understanding the SCN8A gene
The voltage-gated sodium channel alpha subunit gene SCN8A encodes the channel Nav1.6, which regulates sodium levels in the brain. Voltage-gated sodium channel abnormalities can be broadly categorized into two groups: gain-of-function (GoF) or loss-of-function (LoF) mutations, with GoF mutations typically heightening neuronal excitability and LoF mutations often diminishing it. Pathogenic variants of SCN8A present with a wide spectrum of clinical phenotypes ranging from severe developmental and epileptic encephalopathy (DEE) with refractory seizures and profound intellectual disability to neurodevelopmental disorders (NDD) with or without seizures. 1 In 2005, Miriam Meisler (Meisler), Ph.D., identified a protein truncation mutation involving SCN8A in a 9-year-old boy 2 with neurological and developmental features, but no seizures. In 2012, Dr. Michael Hammer (Hammer), motivated to find the cause of his daughter’s severe childhood epilepsy, performed whole genome sequencing on his own family and discovered the first pathogenic SCN8A variant that causes DEE. 3 Early on, the vast majority of patients with SCN8A mutations were diagnosed with DEE; however, as more patients were discovered to carry pathogenic variants in this gene, the spectrum widened to include patients presenting with neurodevelopmental delays with or without epilepsy and milder forms of epilepsy. The incidence of SCN8A-related disorders is reported to be just over 1 in 56,000 births accounting for ~1% of all cases of epilepsy with encephalopathy. 4
There are currently an estimated 15 labs working on SCN8A, in Europe and the USA, with three published SCN8A mouse models,5–7 with a partial match to human phenotype, and three published patient iPSC cell lines. 8 There is a need for models that can represent the full spectrum of the disorder and associated comorbidities to yield more precision in developing comprehensive treatments beyond just seizures (including movement disorders, hypotonia, sleep issues, gastrointestinal (GI) complications, and Sudden Unexpected Death in Epilepsy (SUDEP)).
Evolution of the Alliance’s work
Gabrielle Conecker and JayEtta Hecker established a nonprofit in 2014, Wishes for Elliott: Advancing the Science of SCN8A (WFE) – pursuing their wish, and inferring Elliott’s – that no child, no family, should confront this diagnosis with such limited information and inadequate basis for treatments. Their advocacy comes from an unwillingness to accept the limited understanding of the disorder and a desire to see more effective treatments to mitigate the devastating effects of SCN8A on the lives of those impacted. They were fortunate to have a doctor who understood the intolerable circumstance, who shared the conviction that families could – and do – play an active role in significantly advancing scientific progress on rare disorders. Leveraging their physician’s guidance and expertise, they mobilized to convene the first gathering of SCN8A scientists, families, and pharmaceutical companies in 2015 – within 5 months of receiving Elliott’s SCN8A diagnosis. This pivotal gathering laid the groundwork for global collaborations to accelerate the understanding of SCN8A and its impacts.
Hammer continued his research and collaboration with Meisler. In search of other children with SCN8A mutations, Hammer launched an International SCN8A Registry (Registry) in 2015. The goal of the Registry is to improve understanding of the natural progression and varied presentation of the disease, identify predictors of the clinical outcome, describe the effect of varying treatments, and to inform the development of more targeted therapies. He organized a nonprofit to house the Registry, the Shay Emma Hammer Research Foundation (SEHRF), named for his daughter. WFE actively supported the Registry with financial aid and communicating its importance to families, aiding in recruitment, and sharing early results with the community, industry, and regulators.
In 2020, WFE and SEHRF united to form the International SCN8A Alliance, not only to coordinate efforts, but to provide a forum to bring together a growing number of dispersed organizations run by SCN8A families across the globe. The Alliance recognized the vital importance of collaboration in accelerating progress for rare diseases, understanding that uncoordinated and duplicated efforts could delay and undermine the progress toward improved outcomes each organization was seeking to make.
Just as WFE was expanding and deepening its work on SCN8A, it developed a broad partnership with patient organizations representing other DEEs to collaborate on providing education, support, and research focusing on those most profoundly affected by DEEs – known as the Developmental and Epileptic Encephalopathy Project or DEE-P Connections (DEE-P). It also launched a major global collaborative research effort in 2022, known as The Inchstone Project, 9 to accelerate adaptation of clinical outcome measures validated to capture small but important improvements (inchstones) in the severely affected DEE population. The larger mission is to improve the full assessment of efficacy of emerging new disease-modifying treatments in this largely unmeasured population.
In 2023, Decoding Developmental Epilepsies (DDE) became the umbrella nonprofit, housing the Alliance, DEE-P, and The Inchstone Project and other initiatives advancing collaboration across the rare epilepsies. DDE is a multipronged strategic effort designed to work on diverse fronts to improve treatments and outcomes for all those affected by SCN8A and other developmental and epileptic encephalopathies.
Building a strategic approach to accelerating the science of SCN8A
From the earliest initiatives of the Alliance, the focus was on building a patient advocacy community that would collaborate to fill gaps in ongoing coordination, communication, and collaboration among researching scientists, clinicians, and families. The central focus was on building partnerships and keeping families at the center of all efforts. Figure 1 represents how families are at the center of the diverse partnerships built with government, researchers, industry, clinicians, other patient advocates, and global leaders to advance the science of SCN8A.

Families are at the center of Alliance Partnerships with all stakeholders.
Ten years of strategic investments
The focus and nature of the various initiatives pursued in search of better treatments and a cure were incremental, but with a clear focus on being strategic. All efforts sought to address the highest priority gaps in the SCN8A research landscape. Seven strategic steps taken to advance SCN8A therapeutics over the past 10 years are illustrated in Figure 2.

Ten years of strategic investments collaborating for a cure.
Each of the seven areas of effort has continued and evolved into ever more global collaboration across the widest range of stakeholders. While interrelated, the journey to accelerate new therapeutics is codified into these strategic steps, outlined below, that continue to advance the science and improve care and treatment for those with SCN8A.
Host SCN8A scientific meetings
The earliest focus was on hosting scientific meetings and facilitating coordination among clinicians, researchers, government, and industry stakeholders as a means to amplify the urgency and priorities of SCN8A families. The seminal gathering of SCN8A researchers, clinicians, industry, and caregiver advocates in 2015 culminated in the publication of the first consensus document on the state of SCN8A research, the first GeneReviews entry on SCN8A, and new collaborative ventures.10,11 We included other patient leaders from The Cute Syndrome (TCSF) and a growing Facebook community and agreed WFE would focus on accelerating the science of SCN8A while TCSF would focus on family support and host an annual family meeting.
A key takeaway from the second SCN8A scientific meeting convened in 2016 was the importance of and need for multidisciplinary centers to improve the understanding of and care for those with this complex disorder. In response, WFE supported the first open multidisciplinary clinic for SCN8A at Children’s National Medical Center, contributing to expanded knowledge about the range and diversity of SCN8A phenotypes. 10 In 2018, WFE partnered in organizing the Sodium Channelopathy Coalition, uniting researchers across three major sodium channel epilepsies – Dravet Syndrome/SCN1A, SCN2A, and SCN8A – to expedite learning and sharing. Recognizing the critical need to continue convening stakeholders, adaptations were made to more readily and effectively gather stakeholders and are outlined in other steps below.
Fund early investigators
In 2014, WFE’s inaugural grant funded a research coordinator to assist in the organization of the first SCN8A scientific meeting and coordinate preparation of the consensus manuscript. To institute a funding mechanism for the most promising early investigators, assessed through a rigorous peer-review process, WFE partnered with the American Epilepsy Society (AES) and funded nine early investigators. 11 Not only did all grants result in publications, but several recipients have now emerged as leaders in SCN8A research with their own labs.
In addition, to ensure the inclusivity and representation of those most severely affected in models, WFE provided bridge funding to ensure the continuity of a complex and challenging effort to develop a mouse model of a variant consistently associated with a more severe phenotype (the conditional R1872W model). 7 Nearly all basic research to date had been built on a variant with a more moderate phenotype (N1768D), and efforts to create a mouse with a severe phenotype had failed with the mice dying before any research could be conducted. Soon after the completion of the work on R1872W, another team, active in the Research Consortium, published on their success in producing a new mouse model of one of the most severe SCN8A variants (R850Q).12,13 As the team prepared its research plans for studying distinct impacts on the new severe model, WFE facilitated a discussion between the research team and a group of families with children with that variant, leveraging its unique position as a family advocacy group to directly inform the direction and priorities for research.
Build dedicated International SCN8A Registry
Another early priority was to gather comprehensive and reliable data on the diverse presentations of SCN8A, recognized from the outset as a disorder with highly heterogeneous manifestations. Creation and maintenance of the Registry was led by Hammer. The Registry questionnaire was designed to provide: (1) critical aspects of the history of onset and progression of disease; (2) pertinent information on patient experiences that may not be reported to clinicians and often do not get documented in medical records or case reports; (3) a unified platform integrating different clinics, countries, and groups; and (4) useful data for clinicians, families, and industry regarding existing treatments, genotype-phenotype correlations, outcome measures, and improved prognosis.
The Registry includes multiple modules designed to inform a comprehensive understanding of the disorder illustrated in Figure 3.

Twelve International SCN8A Registry modules.
There are currently over 400 unique individuals included in the Registry representing ~300 different genetic variants. See Figure 4.
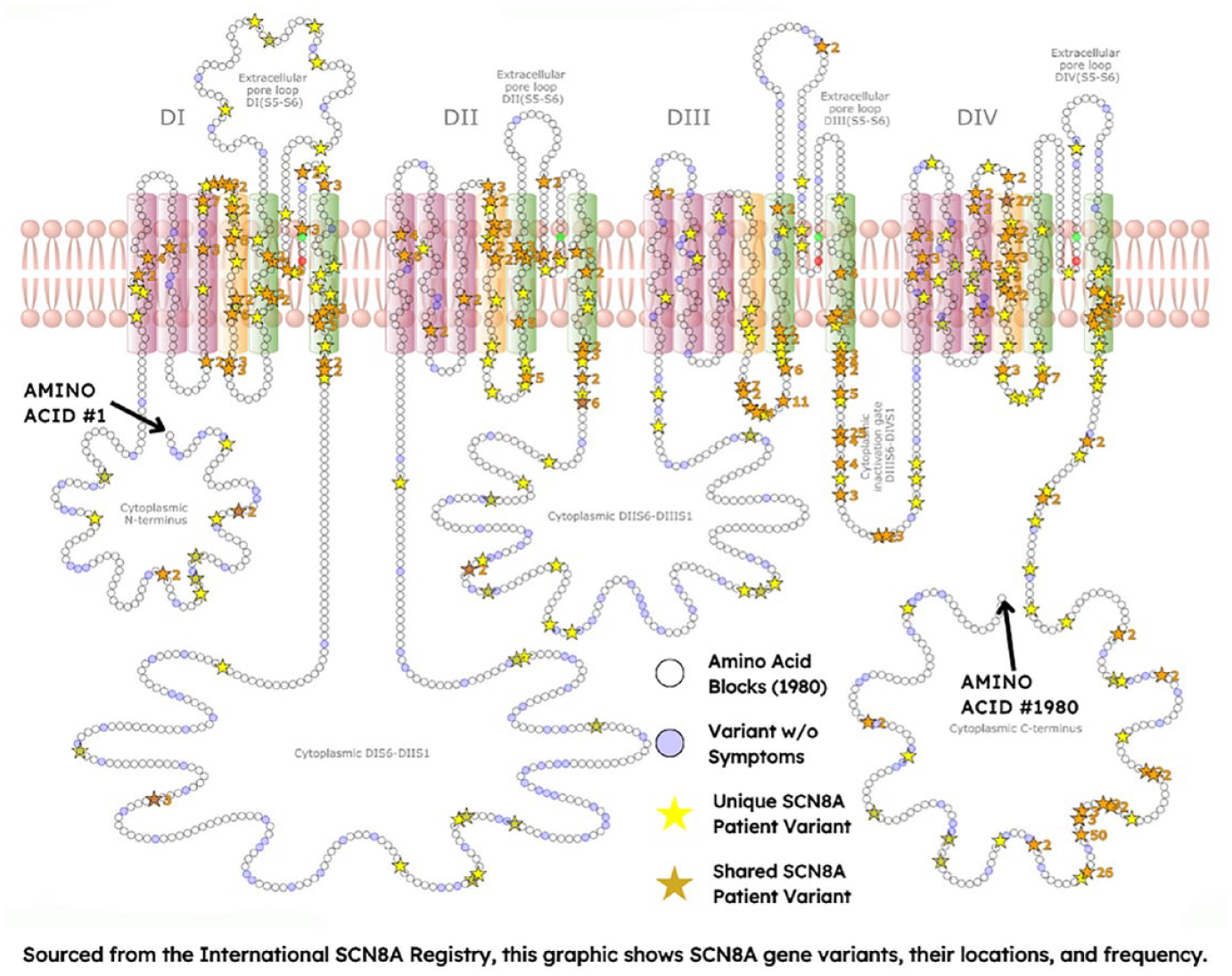
SCN8A gene variant map from the International SCN8A Registry.
The Hammer Lab at the University of Arizona combines ongoing rigorous analysis of SCN8A patient data with a mouse lab for rapid testing of hypotheses emerging from the robust caregiver-reported data.5,14–20 Important findings from the Registry are continually shared with families 21 via regular Zoom meetings, an online community and disseminated through peer-reviewed articles.20,22–26
The unique Registry resource has benefited pharmaceutical companies in designing clinical trials that are now testing novel SCN8A-specific drugs. The robust caregiver-reported data not only provide the most complete characterization of the heterogeneous disorder, but have informed inclusion and exclusion criteria, site selections, and realistic outcome measures. Participants of the Registry update their profiles with any new information annually, and results from ongoing data analyses are presented at frequent online Citizen Scientist sessions (families are referred to as Citizen Scientists given the bidirectional flow of information between scientists and families). In advance of any SCN8A-specific new drug applications to the Food and Drug Administration (FDA), WFE and SEHRF collaborated to submit a detailed filing with FDA on the diverse characteristics of the SCN8A population, the significant unmet need, and urgency of families in search of effective treatments for their children.
Form partnerships with diverse stakeholders
SCN8A partnerships
The work expanded in multiple ways in 2019 with a major focus on cultivating partnerships across a wide range of stakeholders to improve focus, impact, and results. This evolution led to the formation of the International SCN8A Alliance, a union of the endeavors of WFE and SEHRF. Noteworthy initiatives include the establishment and hosting of a global SCN8A Clinicians Network, which convenes to coordinate and advise the Alliance on priorities, as well as an ongoing SCN8A Research Consortium, which provides a regular forum for leaders and early investigators in major SCN8A-focused basic research labs to collaborate.
These deepening relationships facilitated the Alliance team working with Meisler to help extend her most recent work identifying a promising antisense oligonucleotide to modify expression of the SCN8A gene. 27 The Alliance team collaborated in efforts to identify further support needed to translate these important findings into therapeutic development and brought the patient voice to the outreach.
The Alliance works to deepen collaboration with leading researchers across the globe including the Danish Epilepsy Center 4,18,28, participating in their biannual research conference and collaborating with leading clinicians in China29,30 treating and reporting on a large community of SCN8A patients.
Critical to meeting the ultimate goal of realizing improved treatments, and ultimately cures for all forms of SCN8A, the Alliance forges relationships with a wide range of industry partners. The Alliance works closely with both industry partners who are actively pursuing therapeutics in SCN8A as well as working to expand the number of companies investing in new therapeutics for SCN8A. The focus is on communicating the needs of the SCN8A community and priorities for new treatments. The longitudinal Registry provides critical information on the diverse population and allows the Alliance to be data driven and precise in discussions. In addition, the regular family meetings and intermittent community surveys give insights into the community to ensure that the Alliance is speaking about the community’s breadth of experiences, needs, and priorities.
Global consensus on the diagnosis, care and treatment of SCN8A
The consolidation of stakeholders through various initiatives of the Alliance increased both the efficiency and effectiveness of efforts aimed at “collaborating for a cure.” Recognizing the shared priority of both families and clinicians, the Alliance initiated and managed a modified Delphi process to develop the first Consensus on SCN8A, published in two companion articles in Epilepsia.9,31 The depth and breadth of the first global consensus include features to aid in earlier diagnosis of 5 distinct phenotypes, variations in the best and contraindicated treatments by phenotype and variant function, the presence, severity and prognosis of 17 comorbidities in the 5 phenotypes, and overall prognosis by phenotype for epilepsy, cognition, and development. See Figure 5 illustrating Highlights of Consensus Areas on the Diagnosis and Treatment of SCN8A.

Highlights of global consensus on the diagnosis, care and treatment of SCN8A.
The review panel for the process included 30 clinicians, 1 researcher (Hammer) and 13 families from 16 countries across 5 continents. Consensus was achieved on all topics included in the process including diagnosis, phenotypes, treatment, comorbidities, prognosis, and counseling and resources for families. Next steps include both a global education and a dissemination plan to maximize the reach of the important areas of consensus as well as expanded collaboration to address the many gaps remaining in understanding and providing more effective treatment for all those affected by SCN8A (see plan to develop a Research Roadmap below).
Collaboration across DEEs
Since its inception, WFE has been active in championing various collaborative fora, which recognize the many areas of shared interests that are better achieved working together. 32 WFE was an early and active participant in the Rare Epilepsy Network (REN) and played an active role in helping it become an independent organization. WFE/Alliance has been an active participant in the Epilepsy Leadership Council supporting efforts to maximize its impact and role in being a collective voice of shared interests across all the epilepsies. The Alliance continues to play a role in the Curing the Epilepsies (CTE) process, which conducts periodic assessments of the progress in achieving defined benchmarks and updates benchmarks for future research on the epilepsies, hosted by both the AES and the National Institute of Neurological Disorders and Stroke. We participated in developing an Editorial 33 published by the team of advocates on the 2021 CTE session calling for transformative change to accelerate translation of epilepsy research into improved outcomes, drawing lessons learned 34 decades earlier in the pediatric oncology space.
In parallel, to address the many shared challenges of more severe DEEs, DEE-P was formed in 2019 in partnership with seven major patient organizations (with membership since growing to 45 partners). The collaborative effort came together to fill a gap and provide efficiencies for patient advocacy groups to better serve the distinct needs of the most severely affected DEE populations, cutting across many etiologies. The focus is on empowering DEE families to be more informed and effective advocates for their children. Working with its partners, DEE-P provides DEE families access to cutting edge research on a wide range of medical, care, and research topics relevant to the treatment of children severely affected by DEEs. DEE-P amplifies the voices of DEE families’ shared priorities and unmet needs to federal regulators including with a recent DEE Listening Session with the FDA on November 22, 2024 highlighting challenges getting access to new treatments (and the need for increased use of basket trials) and inadequate clinical outcome assessments to measure change in those profoundly impacted by DEEs.
In 2022, The Inchstone Project (Inchstone) was formed as a project of DEE-P to fill the void in the availability of clinical outcome assessment tools 35 which are sufficiently sensitive to capture the small but meaningful outcomes achieved by individuals most severely affected by DEEs. Inchstone is a global partnership of patient advocacy organizations, clinical outcomes researchers, and innovative industry partners, dedicated to accelerating the adaptation of outcome measures capable of capturing the “inchstones” achieved by those most severely affected by DEEs. The project seeks to improve the equity and potential success of emerging transformative clinical trials. Improved tools are needed to enable those with a DEE to participate in drug trials that otherwise might exclude them due to a lack of suitable evaluation tools, ultimately broadening access to potentially life-changing treatments.
Coordinate global community engagement
The Alliance has also worked internationally to foster community engagement in research, advocacy, and community initiatives. A unique program within this framework is the Citizen Scientist bidirectional family meetings, during which families support one another, learn from recent research and outcomes derived from data in the International SCN8A Registry and contribute valuable real world experiences, thus enriching the understanding of and investigations into the disorder. An example of SCN8A family experience informing research was families reporting multiple cases of precocious puberty which led to the addition of the topic to the Registry and the ongoing evaluation of impacts. Meetings are tailored to various phenotypes and are conducted in multiple languages, ensuring engagement from families across the globe. Another initiative, SCN8A Unraveled, provides accessible discussions with authors of recent scholarly articles to help families understand emerging literature, how it applies to them and the importance of a robust SCN8A research community.
Additionally, our team proudly serves as founding partners of the Epilepsies Action Network (EAN), a collaborative initiative dedicated to uniting advocacy efforts across the epilepsy community. The aim is to garner increased investment in all forms of epilepsy and improve outcomes for the 1 in 26 Americans grappling with various forms of epilepsy. EAN is led by advocates associated with several of the nation’s top epilepsy organizations including CURE Epilepsy, the Epilepsy Foundation, the REN, and DEE-P, along with many others aligned with its mission to increase research toward improving outcomes for patients and families.
The EAN is advised by a group of widely recognized thought leaders including former HHS Secretary Dr. Howard Koh, former Member of Congress Tony Coehlo, plus leading researchers, clinicians, advocates, and public figures/actors. This important initiative is a valuable complement to the work of the Alliance and other epilepsy organizations. Progress in certain areas, such as national policy, necessitates a collective effort, where the community comes together with a unified voice to advocate for improvements in national policies and funding for all epilepsies. Recent achievements include formation of bi-partisan House and Senate caucuses on the epilepsies (led by the National Epilepsy Foundation) and legislation introduced to direct HHS to create a National Plan for Epilepsy in partnership with the community.
Build and host Global SCN8A Leaders’ Alliance
The Alliance also convenes and collaborates with the Global SCN8A Leaders Alliance (Leaders Alliance), which includes SCN8A organizations representing 5 continents and 10 countries including Australia, Brazil, China, France, Italy, Netherlands, Portugal, Spain/Latin America, U.K./Ireland and the U.S. as well as the SCN8A European Federation, as illustrated in Figure 6. Coordination efforts include facilitating education, resource sharing, data collection, and research efforts. The Leaders Alliance meets quarterly to share successes, challenges, and find additional ways to coordinate and amplify each other’s efforts. For example, the Alliance collaborates on an annual event marking International SCN8A Awareness Day and uses it as an opportunity to highlight the work of all global partners. The Leaders Alliance is also working toward the development of a Global Scientific Advisory Board to jointly prioritize and fund the highest impact research.

SCN8A Global Leaders Alliance Partners.
Advance global SCN8A research roadmap
The Alliance, in collaboration with 10 Leaders Alliance partners, has undertaken efforts to develop the first-ever SCN8A Research Roadmap. This work is motivated, in part, by the evident gaps in knowledge that emerged so clearly from the first global Consensus on SCN8A. The effort brought together families and advocacy leaders to meet with leading basic researchers, clinicians, and industry professionals, in August 2024. A publication identifying the most critical and promising research efforts to improve treatments and outcomes for every individual affected by SCN8A is under review. The roadmap will not only facilitate collaboration across SCN8A family organizations to fund the highest payoff areas, but also serve as a guide for those choosing research directions in and/or developing new therapies targeting SCN8A.
Discussion
The International SCN8A Alliance remains dedicated to advancing and expanding the foundation for accelerating scientific progress in SCN8A and other DEEs in order to advance treatments addressing the underlying causes of the disease, not just the symptoms. The Alliance also operates under the assumption that progress is built upon an ever-expanding foundation of high quality research and collaboration.
Figure 7 charts an overview of the growth in scholarly publications about SCN8A over the last 12 years mapped against Alliance milestones as well as major research and related SCN8A community highlights.

Growth in SCN8A Publications and SCN8A Alliance & Related Milestones.
Assessment of the actual impact of the work on either the pace or focus of SCN8A research and collaboration is complex. To better understand WFE/Alliance’s impact, on the 7 year anniversary, the Alliance asked for reflections from several of the leading researchers and early investigators. Personal statements by these partners in research are available for viewing.
The impact of broad collaboration on accelerating progress in SCN8A has been recently recognized in a study examining the advances and challenges in the field of precision therapies for rare genetic epilepsies.
36
“We have described tremendous progress at each stage of the epilepsy PM (Precision Medicine) pipeline, including gene discovery, diagnostics, natural history studies, therapeutic strategies, preclinical models, and clinical trials, but with formidable challenges remaining to translate this progress into precision therapies and cures. SCN8A-related developmental and epileptic encephalopathy is a genetic epilepsy progressing in this pipeline with relative efficiency. It has been ~10 years since the discovery of SCN8A as a disease-causing gene, with significant advancement toward PM in that time. . .The examples of SCN8A-related epileptic encephalopathy are proof that teams with the necessary combination of expertise (clinicians, scientists, patient advocates, federal resources, and regulatory bodies) can efficiently shepherd particular therapeutics through the pipeline.”
Emerging directions for the SCN8A Alliance
The SCN8A Alliance is poised to continue pursuing collaborative patient-centered and patient-led efforts to drive therapeutic advances. Key planned initiatives include:
• Comprehensive dissemination, implementation, and monitoring of the new global consensus on the diagnosis and treatment of SCN8A
• Publication and dissemination of the first global SCN8A Research Roadmap. We anticipate this will facilitate: ○ Increased funding to enable competitive direct research grants sponsored by SCN8A patient organizations collaborating to support highest priority areas ○ Use of the priorities among research funding organizations and among senior and emerging researchers to align their research with identified gaps.
• Initiation of a prospective arm of the International SCN8A Registry, improving the granularity of data on the heterogeneity of SCN8A and the natural history or course of the disease on a cohort of individuals over time
• Formation of a Global Science Advisory Board to facilitate continued expansion of collaboration among the Leaders Alliance on research priorities and optimize progress toward improved treatment and outcomes for everyone affected by SCN8A.
The SCN8A Alliance, supported by families sharing their experiences and drive for better answers, has played a pivotal role in the remarkable success of the SCN8A community to date. Caregivers from across the globe have helped document the course of this complex and highly heterogeneous disorder by contributing to the Registry. Targeted efforts to facilitate and support active coordination and collaboration across many stakeholders have been core to the rapid pace of progress. Seminal signs of progress include the milestone of the first Consensus on SCN8A and two clinical trials for the disorder within a decade of the gene’s discovery.
As leaders of a global community, the SCN8A Alliance is focused on bringing patient and caregiver voices to the forefront and building a working alliance to methodically address issues standing in the way of better care, treatments and quality of life for those with SCN8A and other DEEs. Together, the SCN8A Alliance, the Global Leaders Alliance, and all our partners are collaborating for a cure.
Lessons learned
The SCN8A Alliance has experienced challenges as well as success in the cousrse of pursuing these broad efforts involving diverse stakeholders.
The journey began as a disease-specific effort, but as a result of the co-founders’ experience in global public health, population genetics, program evaluation, and national public policy, they concluded early on that with limited resources and capacity, focusing on building partnerships – including beyond SCN8A – would increase the potential for broad and sustainable impact in accelerating scientific progress.
But partnerships and collaboration do not always come together easily. Reflecting on 10 years of working with limited resources and ever growing urgency, we share three broad lessons learned:
Recognize the significant potential of the power of convening around patient priorities
Patient advocates can be effective agents in accelerating new therapeutics through maximizing their power to convene diverse stakeholders around a shared vision grounded in patient/caregiver priorities. By creating forums that bring various groups together to collaboratively seek solutions to address major challenges facing the community, advocates deepen relationships and partnerships. The impact of the Alliance has been maximized by making extensive use of the power to convene: hosting the seminal SCN8A scientific gatherings, convening clinicians to develop the SCN8A Consensus, building a platform for global leaders to collaborate and identify shared priorities, collaborating to develop coordinated advocacy for all epilepsies, and convening researchers and industry to fill the major gap in outcome measures for profoundly impacted individuals.
Maintain a constant focus on improving outcomes
• An alarming number of children with rare epilepsies are dying. There is often a limited evidence-basis for treatment decisions and children’s quality of life is often impaired not just by scientific uncertainty, but by uneven access to informed care. The constant focus on improving outcomes has helped in building coalitions with those sharing a commitment to collective strategies to move the needle.
• While it may require more up-front work and investment, prioritizing outcomes early has led to more impactful results. For example, when Hammer sought to find other families struggling with SCN8A, instead of launching a basic SCN8A contact registry, he designed a comprehensive instrument that while time-consuming for reporting caregivers, has accelerated insights into the complexity of SCN8A and yielded benefits for families, clinicians, researchers, and industry alike.
Be bold and think big
• The SCN8A Alliance has learned to be more efficient and achieve more by being bold, thinking big, and investing time and effort into partnerships of all kinds with diverse stakeholders.
• The boldest action – convening a global modified Delphi on SCN8A – has the potential to yield our highest impact. Despite limited financial resources or a Science Director, the Alliance launched and hosted a rigorous and complex global effort to develop the first Consensus on SCN8A. The compelling need and benefit helped to mobilize leading clinicians and family advocates from across the globe – engaging in a seminal effort to make evidence-based SCN8A care available to every clinician and family, everywhere.
• Even though the core of the SCN8A Alliance’s advocacy is rooted in the experience of the rare epilepsies, it sees the importance of partnering with diverse patient advocates and leading practitioners from across the spectrum, joining forces to advocate for effective federal investment in improving outcomes for all epilepsies. Research on the epilepsies is critical not only for children, but young adults, the elderly, veterans and those impacted by traumatic brain injury and more. Through the EAN, advocacy groups are moving into the gap, bringing together diverse voices to speak up for increased and outcome-focused federal investment in the epilepsies.
Big thinking and broad actions can be complex, time-consuming, and make progress dependent on the commitment and action of others – but as Helen Keller observed: “alone we can do so little; together we can do so much.”
Conclusion
Parent-led organizations are uniquely driven and positioned to provide leadership and focus in the pursuit of therapeutic advances in the panoply of rare epilepsies. While the reach and scope of activities vary, patient/caregiver-led organizations bring an urgency and ability to convene and collaborate across traditional boundaries to accelerate scientific progress.
The Alliance’s advocacy for SCN8A, the DEEs, and all the epilepsies has reflected a core belief in collaboration and partnerships. Three overriding strategies we have used may provide insights for other patient advocacy organizations looking to effectively tap promising but underutilized approaches:
• Recognize the significant potential of the power of convening various stakeholders around patient priorities.
• Maintain a constant focus on improving outcomes.
• Be bold and think big – explore common challenges and pathways for collaboration, cutting across ever expanding monogenic disorders.
Supplemental Material
sj-docx-1-trd-10.1177_26330040241252449 – Supplemental material for Patient leadership and partnerships accelerate therapies for SCN8A and other developmental and epileptic encephalopathies
Supplemental material, sj-docx-1-trd-10.1177_26330040241252449 for Patient leadership and partnerships accelerate therapies for SCN8A and other developmental and epileptic encephalopathies by Gabrielle Conecker, JayEtta Hecker and Michael F. Hammer in Therapeutic Advances in Rare Disease
Footnotes
Acknowledgements
We acknowledge the input from Dr. Miriam Meisler and the technical support of Dr. Kelly Muzyczka Mitchell. Charles Anderson was responsible for developing all figures included in this article. We would like to acknowledge the contributions of Dianne Mitchell when preparing the initial draft of the manuscript. We acknowledge the consistent support and interest by SCN8A families, as well as the many partners and collaborators of the Alliance. Dedicated SCN8A Ambassadors, Tammy Kooistra, Megan Hilton, Mary Tone, and Kristin Bejarano, play a vital role in providing close personal support for families, especially those who are newly diagnosed or in crisis - their efforts are critical to the success of our community.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
