Abstract
A pulmonary Aspergillus nodule is a rare subtype of chronic pulmonary aspergillosis. The diagnosis is difficult and is histological. There are only a few reports on such cases. Here, we report five cases of pulmonary Aspergillus nodules confirmed by surgery and pathology in immunocompetent patient and review the literature. Among the five patients in this group, three were females and two were males, aged 44–73 years old. Two cases had hemoptysis onset, and three cases were found to have a slow disease course on chest CT during imaging, ranging from months to years. The white blood cell count, carcinoembryonic antigen, and blood Galactomannan (GM) tests in five cases were all within normal range. Four cases had normal blood C-reactive protein, and one case had an increase. On imaging, there were two cases in the upper lobe of the right lung, two cases in the lower lobe of the left lung, one case in the upper lobe of the left lung, three cases were solitary nodular shadows, and two cases were nodular shadows with cavity formation, including one case with calcification, four cases with bronchial dilation shadows, and one case with gas containing cavity shadows. Five cases were treated with surgical resection and confirmed by histopathological examination. All five patients did not receive antifungal treatment after surgery, and there was no recurrence of Aspergillus nodules during regular follow-up.
Plain language summary
Pulmonary aspergillosis nodules are a relatively rare manifestation in the spectrum of chronic pulmonary aspergillosis. This article reports five cases of pulmonary aspergillosis nodules confirmed by surgical resection and histopathological examination, all of which were patients with normal immune function, atypical clinical symptoms, varying severity, and normal Galactomannan (GM) tests. All five cases did not receive antifungal treatment after surgery, and the nodules did not recur during regular follow-up. The diagnosis of pulmonary aspergillosis nodules is difficult, and lung biopsy and bronchoalveolar lavage fluid (BALF) metagenomics next generation sequencing (mNGS) may be considered. There are various treatment methods, including surgical treatment, antifungal drug therapy, and sometimes local bronchial perfusion therapy can also be considered.
Keywords
Introduction
Chronic pulmonary aspergillosis (CPA) is a serious slowly progressing lung disease in the pulmonary aspergillosis spectrum. It often occurs in patients with underlying lung diseases or mild immunodeficiency, and even in patients with normal immune function. Among the five recognized subtypes of CPA, pulmonary Aspergillus nodules are rare. On imaging, there are one or more nodules with or without cavities. Most are <3 cm in diameter, although lesions are occasionally larger with necrotic centers. 1 They are nonspecific and difficult to distinguish from lung cancer, metastasis, cryptococcal nodules, and tuberculosis. Recently, Denning et al. reported that fewer than 10% of the cases reported to the national aspergillosis center were Aspergillus nodules. 2 They can only be diagnosed histologically based on the presence of fungal hyphae without tissue invasion.3,4 We provide five cases of pulmonary Aspergillus nodules and review the literature in the hope of improving clinicians’ understanding of the disease.
Case data

(a) Chest CT shows a mass like high-density shadow in the apical segment of the right upper lung, with a diameter of about 33 mm × 27 mm. Bronchiectasis shadow (see arrow) is seen in the periphery, lobulated change, and thin flake, and patchy exudation shadow is seen around. (b) ‘Right upper lung’ inflammatory lesion with necrosis, a large number of histiocytes and multinucleated giant cell reaction in the stroma (see arrow), and fungal flora (Hematoxylin-Eosin staining HE ×100) in the lumen of the bronchi.
Despite antibiotic treatment and hemostatic agents, the hemoptysis recurred. He had the right upper lung lobe resected under general anesthesia on 29 November 2012. At surgery, there was obvious adhesion of the right upper pleura, a 3-cm mass in the right upper lung with a distinct boundary, and no pleural invasion. Pathology showed an inflammatory right upper lung lesion accompanied by necrosis, with many tissue cells and multinucleated giant cell reactions in the stroma, fungi in the bronchial lumens, and reactive hyperplasia of four lymph nodes at the bronchial root accompanied by many dead bacteria-like objects [Figure 1(b)]. Periodic acid Schiff (PAS) staining was positive (+), which indicates fungal hyphae in the tissue sample. The diagnosis was a right upper lung Aspergillus nodule. He was not given antifungal treatment postoperatively. There was no recurrence at the 5-year follow-up.

(a) Chest CT: the lower lobe of the left lung is patchy and surrounded by fuzzy flocculent shadow (see arrow). (b) Chest CT: left lower pulmonary nodule with cavity formation with a diameter of about 16 mm × 32 mm (see arrow). (c) ‘Left lower lung’ mycosis with bronchiectasis, a large number of inflammatory cells and multinucleated giant cell reaction in the lung stroma, and a large number of fungal flora can be seen in the bronchial lumen (see arrow) (HE ×100). (d)
After 10 days of antibiotic treatment, repeat chest CT showed a left lower lobe nodule with cavity formation [Figure 2(b)]. She underwent a left lower lobectomy under general anesthesia on 20 May 2013. During the operation, a 1.5-cm mass with an indistinct boundary was found in the left lower lobe. Pathology showed left lower lung mycosis with bronchiectasis, many inflammatory cells and a multinuclear giant cell reaction in the lung stroma, and many fungi in the bronchial lumen [Figure 2(c)]. After magnification, hyaline hyphae with acute branching can be seen [Figure 2(d)]. The diagnosis was a left lower lung Aspergillus nodule. There was no postoperative antifungal treatment and no recurrence at the 5-year follow-up.

(a) Chest CT: a high-density nodular shadow with a diameter of about 11 mm × 13 mm can be seen in the right upper lung apex, the surface is not smooth, and bronchiectasis shadow can be seen around it (see arrow). (b) The bronchiectasis of the right upper lung is accompanied by fibrosis of surrounding lung tissue, a large number of lymphocytes and plasma cells infiltrate, a large number of foam like tissue cells in the alveolar cavity, and a fungus group (see arrow) in the expanded bronchoalveolar cavity (HE ×100).
After treatment with antibiotics and hemostatic agents, the hemoptysis recurred. A wedge resection of the right upper lung was performed under general anesthesia on 26 October 2011. At surgery, dense adhesions were seen at the top of the right lung, and the right upper lobe contained a medium-hard 1.5 cm × 1.5 cm nodule, with a distinct boundary. Pathology showed right upper lung bronchiectasis with peripheral lung tissue fibrosis, many infiltrating lymphocytes and plasma cells, many foam-like tissue cells in the alveolar cavity, and fungi in the expanded bronchi cavity [Figure 3(b)]. The tissue was PAS (+), which indicates fungal hyphae in the tissue sample. The diagnosis was a right upper pulmonary aspergillosis nodule. There was no postoperative antifungal treatment and no recurrence at the 5-year follow-up.

(a) Chest CT lung window: a high-density nodule with a diameter of about 11 mm × 13 mm with calcification can be seen in the left upper lung lingula segment, the surface is not bright, and bronchiectasis shadow can be seen around (see arrow). (b) Fungal mass resembling Aspergillus (see arrow) in the lumen of the ‘left upper lung tongue’ branch, accompanied by inflammatory changes in the surrounding lung tissue and local regional lymphoid tissue hyperplasia (HE ×100).
On 8 November 2017, the patient underwent thoracoscopic resection of the left upper lung lingual segment under general anesthesia. At surgery, there were no chest adhesions, and no obvious mass was detected in the left upper lung. Pathology showed fungal hyphae in the bronchi of the left upper lung lingual segment accompanied by inflammatory changes in the surrounding lung tissue and local regional lymphoid tissue hyperplasia [Figure 4(b)]. The diagnosis was an Aspergillus nodule in the left upper lung. There was no postoperative antifungal treatment and no recurrence at the 4-year follow-up.
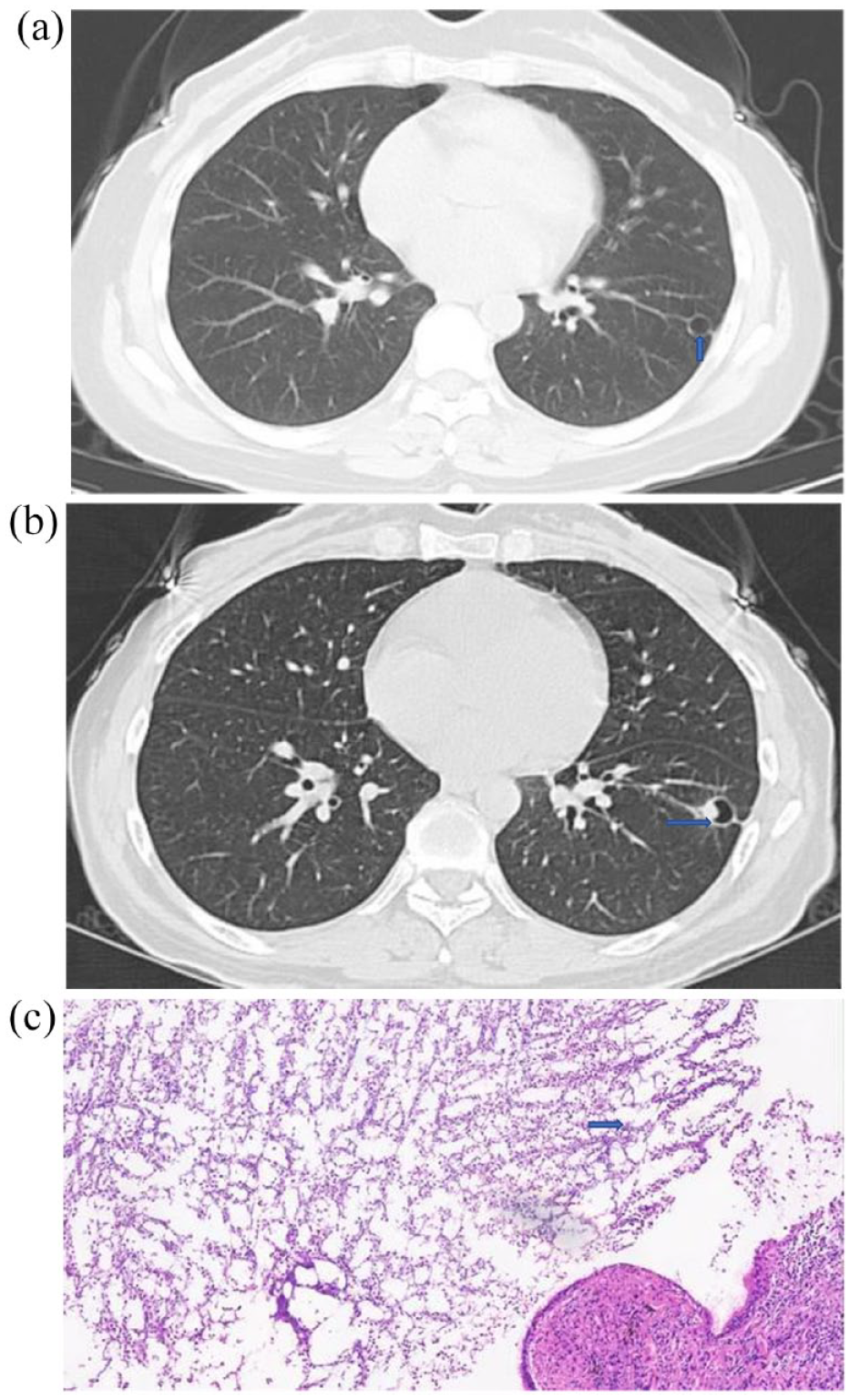
(a) Chest CT in 2018 shows 8-mm circular translucent shadow with surrounding fibrous strip shadow in the left lower lung (see arrow). (b) 2021-7-30 chest CT: the lower lobe of the left lung is like a circular high transparency shadow, with a size of about 12 mm × 13 mm. There is a nodular shadow protruding into the cavity on the inner wall (see arrow). (c) ‘Wedge-shaped specimen of left lower lung’ (HE × 100): cystic bronchiectasis, pus in the cystic cavity, containing fungal mass resembling Aspergillus (see arrow), peripheral fibrosis, infiltration of a large number of lymphocytes, plasma cells, and neutrophils, with multinuclear giant cell reaction and lymphoid tissue hyperplasia.
On 24 August 2021, she underwent thoracoscopic wedge resection of the left lower lung under general anesthesia. During the operation, some adhesions were seen in the chest. After cutting the specimen, a spherical 5-mm-diameter lesion was found in the cavity. The postoperative pathology showed cystic bronchiectasis, pus in the cystic cavity, including fungal hyphae, peripheral fibrosis, and many lymphocytes, plasma cells, and neutrophils, accompanied by multinucleated giant cell reaction and lymphoid tissue hyperplasia [Figure 5(c)]. The staining was PAS (+), silver hexamine (+), and mycobacterium acid fast staining (–). The diagnosis was a left lower lobe Aspergillus nodule. There was no postoperative antifungal treatment and no recurrence at the 1-year follow-up.
Discussion
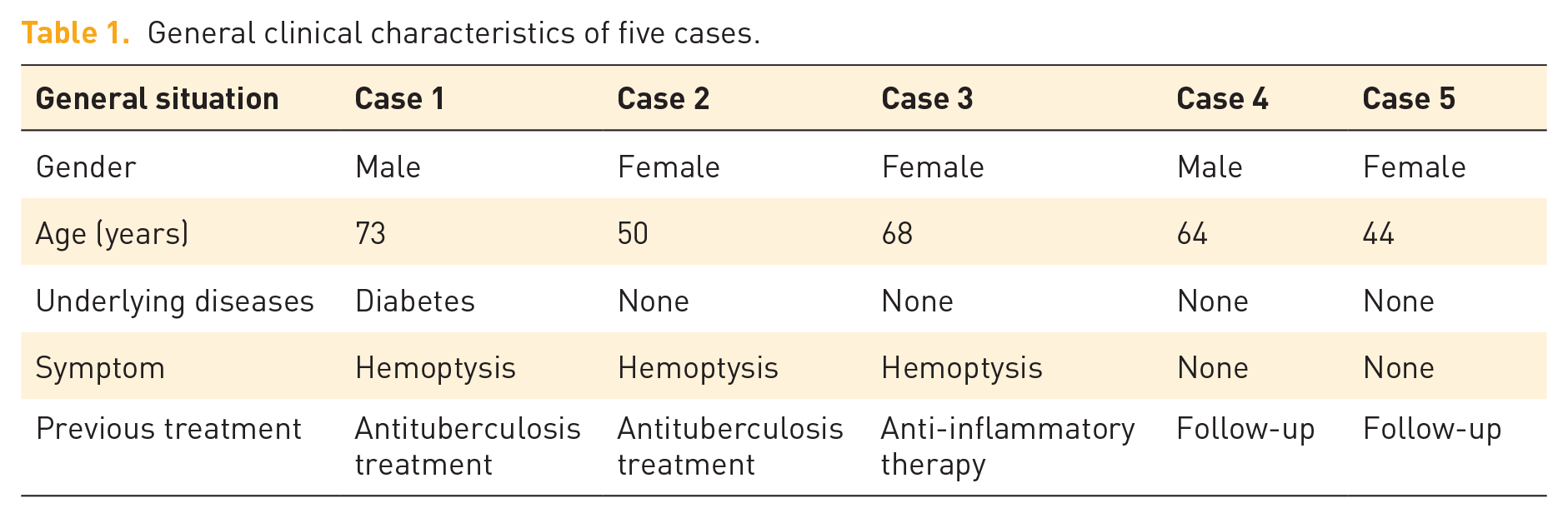
The majority of patients with Aspergillus nodules have underlying diseases.2,5 In our series, one patient had diabetes, while the remaining four had no underlying disease. They had no history of immune deficiency such as tumor, hormone use and other immunosuppressant diseases (Table 1). The incidence rate of the disease in men and women is not clear. In a retrospective study of pulmonary aspergillosis, Denning et al. reported the incidence was slightly higher (54.5%) in males, with an average age of 58 years. 2 By contrast, Kang et al. found 46 (58%) of 80 aspergillosis nodules in women. 5 The natural course of the disease is not clear, but it is chronic, which may be related to the immune state, ranging from months to years. 2 Our patients’ age range is between 44 and 73 years old. The clinical symptoms and signs are nonspecific, such as cough, dyspnea, hemoptysis, and weight loss.2,5 Many cases are found incidentally on thoracic CT. Three patients had recurrent hemoptysis, while two were asymptomatic. Pulmonary nodules were present in all cases. With the popularization of low-dose chest CT in physical examinations, the diagnostic rate of asymptomatic pulmonary Aspergillus nodules may increase.
General clinical characteristics of five cases.
The laboratory examinations of our five cases included normal routine liver and kidney function, blood parameters, coagulation function, and tumor markers carcinoembryonic antigen. Three patients underwent tracheoscopy: two had no abnormalities and one showed congestion, edema, and bronchial mucosa stenosis. Five cases that had blood GM tests were all normal (Table 2). Although the serum GM test is widely used to diagnose invasive pulmonary aspergillosis, its positive rate in CPA is low. 6 The bronchoalveolar lavage fluid (BALF) GM test is more sensitive than the serum test and can be used in the diagnosis of CPA. 7 However, there are limitations of the test accuracy depending on the GM cutoff value used and should not be used as a single test to determine CPA. 8 But in this group of cases, one case was sent for BALF and the GM test was normal . For CPA, Aspergillus IgG antibody is usually a confirmatory test, which can help monitor the treatment response and CPA recurrence. Its sensitivity is 80–96% and specificity 85%. 9 Aspergillus IgG antibody is not CPA-specific, and it can be positive in allergic bronchopulmonary aspergillosis and Aspergillus bronchitis and sinusitis. According to the latest guidelines, Aspergillus PCR is a very useful for diagnosing CPA and is used to confirm the presence of Aspergillus in sputum. With negative cultures, the positive rate of samples can reach 50%. 10 The positive rates of these two laboratory tests are lower in pulmonary Aspergillus nodules. In Denning et al., 32 patients underwent Aspergillus IgG antibody testing, with a positive rate of 69% and sputum samples from 22 patients were subject to Aspergillus PCR analysis, with a positive rate of 45%. 2 Since Wilson et al. 11 reported a case of leptospira diagnosed through cerebrospinal fluid metagenomic sequencing in 2014, mNGS has attracted attention and research. mNGS has higher sensitivity than traditional culture methods and is more accurate in diagnosing tuberculosis, fungi, viruses and anaerobes.12,13 The diagnostic accuracy of BALF mNGS positive for Aspergillus in immunocompromised IPA patients reached 82.3%, 14 but its diagnostic value in CPA lacks relevant research data. Due to limitations in conditions and high costs, our group of cases were unable to undergo preoperative BALF mNGS testing.
Laboratory examination of five cases.

The imaging features of pulmonary Aspergillus nodules are one or more nodules, often in the upper lungs, which may or may not be accompanied by cavity formation.2–3 Of our five cases, three had solitary upper lung lobe nodules without cavity formation (two in the right upper lobe and one in the left); the other two cases were in the lower left lobe, including one in the dorsal segment of the lower left lung (Table 3). While calcification is rare in pulmonary Aspergillus nodules, one of our patients had calcified Aspergillus nodules on chest CT.2,5 Bronchiectasis was seen in cases 1–4 on chest CT, and case 5 had air-containing cavities. Based on the pathology, it is thought that this was a secondary change caused by Aspergillus blocking the bronchial lumen. Studies need to examine whether imaging combined with bronchiectasis has diagnostic significance in pulmonary Aspergillus nodules. PET-CT of most pulmonary Aspergillus nodules shows low to medium fluorodeoxyglucose (FDG) uptake.2,15 Case 5 underwent PET-CT, which showed increased FDG uptake. Aspergillus nodules are easily misdiagnosed as lung malignancies.
Chest imaging of five cases.

The clinical, laboratory, and imaging features of pulmonary Aspergillus nodules are nonspecific. The diagnosis depends on a histopathological examination. When there is only a small amount of mycelium, hexamine silver and PAS staining can be used for pathogen detection. But this method has no specificity. Hyaline hyphae with regular intervals visible under the microscope, accompanied by acute angle as well as dichotomous branching, without vascular invasion, can distinguish the genus Aspergillus. 16 All five resected specimens in our group showed bronchiectasis with fungal hyphae, inflammatory cell infiltration, exudation, and tissue cells or giant cell reaction in the lung stroma, without vascular invasion, consistent with the pathological characteristics of Aspergillus nodules (Table 4). Unfortunately, the identification of specific Aspergillus subspecies was not carried out. PAS staining was positive in three cases, and dead bacteria-like matter was seen in the peribronchial lymph nodes of case 1. It is difficult to obtain pathological specimens on bronchoscopic biopsy and the positive rate is low, while patients often refuse highly invasive thoracoscopic or thoracotomy lung biopsies, delaying the diagnosis.2,3
Pathological results of five cases.

At present, the treatment options for pulmonary aspergillosis nodules include drug therapy such as voriconazole, itraconazole systemic antifungal therapy, and surgical resection, and local bronchial perfusion therapy.1,3 Scholars have studied the use of local infusion of amphotericin B under bronchoscopy to treat pulmonary mycosis. The effective rate of treatment for pulmonary aspergillosis is 73.81% (imaging safety or partial absorption), with some cases showing nodular shadows on imaging. The disadvantage of endoscopic injection is that it has poor therapeutic effect on multiple lesions and is invasive. Some patients may experience pain and poor compliance after medication injection and may also experience intraoperative and postoperative bleeding. 17 It is not clear whether asymptomatic Aspergillus nodules need only CT follow-up, without drug or surgical treatment.1,18 When deciding on treatment, clinicians must consider the patient’s symptoms, serum Aspergillus IgG, dynamic imaging changes, and Aspergillus culture or PCR-positive airway samples.2,19 Surgical treatment of pulmonary Aspergillus nodules is effective and the recurrence rate is low.2,15 The role of perioperative antifungal therapy needs further study. None of our patients were given postoperative antifungal treatment. The prognoses were good and there were no recurrences of nodules. If pulmonary aspergillosis can be diagnosed before surgery, local infusion of amphotericin B under bronchoscopy or systemic/oral antifungal therapies can be considered to avoid surgical treatment.
Conclusion
When clinicians see healthy patients with normal immune function, and single or multiple nodule shadows on imaging, in addition to considering pulmonary tuberculosis, lung cancer, and other diseases, they need to consider pulmonary Aspergillus nodules. Tracheoscopy examination should be performed, and pathogen examination including BALF mNGS. After definite diagnosis, surgical resection, the first-line treatment of pulmonary Aspergillus nodules, and drugs are sometimes used; also local bronchoscopic lavage with amphotericin B can be attempted.
Supplemental Material
sj-doc-2-trd-10.1177_26330040241252446 – for Five cases of pulmonary Aspergillus nodules diagnosed at surgery and by pathology in immunocompetent patients, with a literature review
sj-doc-2-trd-10.1177_26330040241252446 for Five cases of pulmonary Aspergillus nodules diagnosed at surgery and by pathology in immunocompetent patients, with a literature review by Shuangxia Dong, Fengxiang Wang, Haizhen Jin and Xinjian Dai in Therapeutic Advances in Rare Disease
Supplemental Material
sj-docx-1-trd-10.1177_26330040241252446 – for Five cases of pulmonary Aspergillus nodules diagnosed at surgery and by pathology in immunocompetent patients, with a literature review
sj-docx-1-trd-10.1177_26330040241252446 for Five cases of pulmonary Aspergillus nodules diagnosed at surgery and by pathology in immunocompetent patients, with a literature review by Shuangxia Dong, Fengxiang Wang, Haizhen Jin and Xinjian Dai in Therapeutic Advances in Rare Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
