Abstract
Existing clinical tools that measure non-seizure outcomes lack the range and granularity needed to capture skills in developmental and epileptic encephalopathy (DEE)-affected individuals who also fall in the severe to profound range of intellectual disability. This effectively excludes those with severe impairments from clinical trials, impeding the ability of sponsors to evaluate disease-modifying therapies (DMTs). The Inchstone Project, an international, patient advocate-led collaboration, brings together leading researchers, clinicians, pharmaceutical companies, and advocates to develop an adapted, validated assessment battery within 5 years. The goal is to support trials of DMTs for the DEEs by providing sufficiently sensitive measurement tools to demonstrate therapeutic efficacy. An initial pilot study administered 7 established assessments to 10 individuals affected by SCN2A-DEE, identifying specific limitations of existing measures and areas for improvement. It was clear that most tools do not account for challenges throughout the DEE population, including vision impairments, significant motor impairments and profound intellectual disability, which need to be accounted for in creating a ‘fit-for-purpose’ battery for the DEE population. Several novel assessments, including two measures of responsivity developed for use in monitoring recovery after acquired brain injury as well as individualized Goal Attainment Scaling, showed promise in this group. The team also completed a DEE-wide survey with over 270 caregivers documenting their children’s abilities and priorities for their improvement from new treatments. The Inchstone team is using this information to evaluate how existing tools might be updated to better capture what is most important to families and measure their child’s small but important improvements over time. These efforts are building a coherent picture across multiple DEEs of what domains, or concepts of interest, have the greatest impact on most patients and families. The Inchstone team is on course to adapt non-seizure outcome measures that are (1) sufficiently sensitive to measure small increments of meaningful change (‘Inchstones’) and (2) applicable to multiple DEE conditions.
Plain language summary
Caregivers of children with DEEs understand firsthand that clinical tools intended to measure non-seizure outcomes, like communication and motor skills, were not designed for and don’t work for their children. More sensitive tools are needed to measure the small changes that occur in DEEs. The limitations of existing measurement tools for DEEs have significant consequences:
- Non-seizure responses to new therapies cannot be measured without tools designed specifically for individuals with severe to profound intellectual disability. - If a response cannot be measured in a trial, a potentially beneficial impact will be missed and a therapy, having failed to demonstrate an effect, may not gain regulatory approval. - DEE-affected individuals are less likely to benefit from the wave of new disease-modifying therapies providing hope for many other rare genetic diseases.
DEE-P Connections, a patient advocacy organization supporting families caring for those severely affected by DEEs, launched The Inchstone Project to address this problem. This team science research collaborative unites researchers, pharmaceutical companies, advocates and others around a shared vision of adapting existing tools to reliably capture the small but important changes in skills in those severely affected by DEEs.
To better understand these gaps, the Inchstone team conducted a pilot study with 10 children with SCN2A DEE. The team administered multiple assessments to explore how to adapt the tools to better capture the abilities and growth of this population. The team also completed a comprehensive DEE-wide survey with over 270 caregivers documenting their children’s abilities and priorities for their improvement from new treatments, helping to document how existing tools may be updated to better capture what’s most important to families and measure their children’s small but important improvements over time.
The Inchstone Project is on course to assure those profoundly impacted by DEEs are meaningfully included in clinical trials by establishing trusted and reliable non-seizure measurement tools.
Keywords
Background
Children diagnosed with developmental and epileptic encephalopathies (DEEs) are often profoundly impaired not only by drug-resistant seizures but also by severe developmental delays, motor impairment, and intellectual disability. DEEs, in fact, refer to a growing number of severe epilepsies characterized both by drug-resistant seizures and encephalopathy or significant developmental impairment. Caregivers of these children know firsthand the inadequacy of clinical outcome assessment (COA) tools intended to capture non-seizure outcomes, like cognition, communication, and motor functioning. While repeatedly asked to complete such standardized assessments as the Vineland Adaptive Behavior Scales, Third Edition (Vineland-3), 1 the Bayley Scales of Infant and Toddler Development, Fourth Edition (Bayley-IV), 2 or a myriad of other assessments, their child’s abilities and small but important improvements over time, are rarely if ever captured by these widely used instruments.
Existing clinical tools lack the range and granularity needed to measure skills in DEE-affected individuals, who also fall in the severe to profound range of intellectual disability. There is good evidence that individuals with intellectual disabilities are effectively excluded from clinical trials,3–7 impeding the ability of sponsors to evaluate disease-modifying therapies.
Federal legislation provided the impetus for robust inclusion of and reliance on patient experience data when evaluating clinical benefits of new therapies in the 21st Century Cures Act. 8 In response, the Federal Drug Administration (FDA) issued the patient-focused drug development (PFDD) guidances9–13 to inform efforts by researchers and drug developers to design COAs that effectively capture a therapy’s benefits, which are important to and valued by patients and caregivers.
DEE-P (Developmental and Epileptic Encephalopathy-Project) Connections was formed in 2019 as a collaborative effort of disease-specific groups focused on providing resources and research to support the distinct needs of families with children most severely affected by DEEs. Early efforts concentrated on providing high-quality, curated resources for DEE families with webinars bringing together families and experts on a wide range of topics focused on the DEE experience. To date, DEE-P has archived resources for DEE families on over 60 topics 14 such as epilepsy surgery, rescue medications, Sudden Unexpected Death in Epilepsy (SUDEP), hypotonia, dysautonomia, Electrical Status Epilepticus in Sleep (ESES), cortical vision impairment (CVI), brain tissue donation, and hospice care.
DEE community leaders also shared an urgency to work together to tackle the inadequacy of existing non-seizure COAs for the severely affected population – recognizing that collaborative action would be both more effective and efficient in achieving accelerated progress. They understood that the success of emerging disease-modifying therapies (DMTs), a source of hope for so many families,15–17 would be at risk without tools that could capture small but important improvements in their children’s condition.
While clinical trials for the many new anti-seizure medications rely on measuring seizure reduction as a primary endpoint, 18 new DMTs, which hold promise of ameliorating the underlying cause of disease, require robust and reliable measures of the many co-occurring conditions. DEE-P Connections contracted COMBINED Brain to conduct a landscape analysis to review the adequacy of existing tools to capture the small but important ‘inchstones’ (not milestones) typically achieved by individuals with DEEs. In November 2021, DEE-P Connections hosted several workshops 19 with leading professionals who shared tools with promise for adaptation, extended scaling, or testing for further exploration with the DEE community: presentations were provided by Anne Berg, Jenny Downs, Chere Chapman, Natasha Ludwig, and Terry Jo Bichell.
Building a global collaborative team and vision
Multi-stakeholder community consensus on the merits of a broad team science collaborative effort to improve the capacity of measurement tools to capture progress in the DEE population was established in the widely attended open workshop about next steps. DEE-P Connections then launched The Inchstone Project 20 in January 2022, laying out the need, urgency, and vision in a video Call to Action. 21 The Inchstone Project’s core research team took shape, including DEE-P leadership (Conecker and Hecker) and the researchers (Berg, Chapman, Downs, and Ludwig) who helped define the problem and the way forward. The research team was soon expanded to include Mary Wojnaroski and Rebecca Hommer, adding expertise in psychological testing and CVI and Gunes Sevinc, expanding our capacity to explore Goal Attainment Scaling (GAS).
The team developed a schemata to reflect the overall plan for moving forward. Building on the landscape analysis and the significant body of work capturing abilities among multiple DEE groups (Berg),22–25 the team evaluated a variety of measurement scales, including broad measures, domain-specific tools, and individualized approaches. Specific assessments evaluated included several widely used standardized and norm-referenced assessments (e.g. Vineland and Bayley), existing neurorehabilitation measures of responsivity (i.e. alertness, awareness, and responsiveness to the environment) developed for monitoring recovery after severe acquired brain injury (used clinically by Ludwig), the Quality of Life – Disability scale (developed by Downs)26,27 and adaptation of GAS (widely applied by Chapman)28–30 to use within the DEE population. The focus would be on collaborating to develop or adapt outcome measures suitable to create an Inchstone COA battery for use among those most severely affected by DEEs. While the team has an ambitious timeline, we have a continuous commitment to rigor, building a process that would encompass every core element of validation outlined in FDA’s PFDD guidances – being ‘fit-for-purpose’ (FFP), 11 reliable, key structural characteristics, validation and responsiveness, and clinical testing (Figure 1).

Inchstone project in overview schematic.
Planning and vision
The Inchstone team created a plan to develop an adapted, validated Inchstone COA battery sufficiently sensitive to capture small yet important improvements for severely affected individuals with DEEs within 5 years. This approach included three major phases: laying the foundation, gathering the evidence to design a core Inchstone COA battery, and completing a rigorous validation phase (Figure 2).

Three phases of the 5-year plan for the Inchstone project.
The foundation phase, year 1, confirmed the need and informed the approach for the work by piloting several tools of interest in the SCN2A community. The team partnered with six industry leaders who recognized the importance of this gap and the value of the broad collaborative strategy. 31 A governance structure for the project was established, including quarterly consultation with a range of experts, Patient Advocacy Group (PAG) leaders and subject matter experts, including clinicians, researchers, and government thought leaders who sit on a Steering Committee 32 and an Industry Advisory Panel. 31 At these meetings, we present progress, methodology and new concepts and then get feedback/guidance on our process and next steps. Years 2 and 3 would be the design phase, focusing on confirming caregiver priorities, studying priority tools to adapt, specifying an initial Inchstone COA battery, and designing a comprehensive validation plan. Years 4 and 5 would be the validation phase, including using test/retest methodology, performing psychometric evaluations, piloting the battery, and releasing the completed Inchstone COA battery for use in clinical trials.
The team prepared an updated overview of the work, better representing that the core of our analytical work – to adapt outcome measures that were clinically meaningful, evidence-informed, and fit-for-purpose – would all be patient-centered. See Figure 3.
Project framework highlighting that all work be patient-centered.
As reviewed below, the Inchstone project has already been productive, engaged a large number of rare disease communities, and is positioned to make continuous progress toward its goal of patient-centered FFP COAs.
How to capture an Inchstone: A pilot study
Members of the Inchstone team identified several tools with the potential to be included in an FFP COA battery based on their previous research and experience. However, more applied investigation in the DEE community was needed to explore possible adaptations of existing tools, including modifications to administration and the actual skills measured. FamilieSCN2A Foundation is a DEE-P Connections partner and was already working with Anne Berg to assess the SCN2A community’s clinical trial readiness. Interested in how new tools might measure their children’s progress compared to the other tools in the clinical trial readiness study (CTRS), FamilieSCN2A allowed the team to attend a family meeting to pilot a range of measures on 10 individuals with SCN2A-DEE.
There were three main assessment areas included in the pilot: clinical measures, capturing ‘meaningful change’, and GAS.
Standardized and norm-referenced clinical measures
Clinicians administered direct clinical assessments and parents completed multiple parent-reported assessments. Measures included the Developmental Profile, Fourth Edition (DP-4), 33 a parent report measure of five domains of development: cognitive, communication, physical, social-emotional, and adaptive behavior. We also included the Bayley-4, Cognitive scale, a standardized direct assessment of child cognitive ability. Note that the Bayley-4 is intended for children 16 days to 42 months old and all children included in the pilot study were older; however, abilities assessed were thought to be consistent with the developmental level of participants. Results from the Vineland-3 comprehensive interview, a standardized caregiver interview of adaptive functioning, were available through the SCN2A-sponsored CTRS. In this small sample, the team observed a similar frequency of floor effects and high correlations between the DP-4, Bayley-4 and Vineland-3.34,35
During the direct assessments, the Inchstone team recognized that some of the children in the pilot had cortical visual impairment (CVI) and this seemed to be a barrier to demonstrating the full extent of their skills. With the help of parents and caretakers in the room during the examination, the Inchstone team piloted some adaptations of the activities with different colored and lit stimuli with an overall positive response from the participants. This confirmed the need for any Inchstone COA battery to provide systematic, consistent adaptations for various levels of CVI.
We often ask our children to use their vision to demonstrate understanding and cognition, especially during standardized testing. However, when the materials being provided throughout the assessment process do not meet the prerequisites needed to process visual information, we are measuring the child’s inability to visually interpret information and not their ability to demonstrate what they understand and know. As clinicians we must ensure that we are measuring the child’s ability/skills based on their knowledge. We cannot do that if the materials are not presented in a way that is visually accessible. RH
The pilot also included administration of two neurorehabilitation tools developed to measure changes in responsivity (i.e. alertness, awareness, and responsiveness to the environment) after severe acquired brain injury. Many individuals with DEEs and severe to profound impairment are not consistently alert and responsive to their environment; however, these skills are critical for explicit learning and development. As such, Dr. Ludwig hypothesized these measures of responsivity might offer the ability to capture change in an aspect of cognition that is critical and meaningful to ongoing development in this population.
Initial results indicated high correlations between the measures of responsivity and the Bayley-4, DP-4, and Vineland-3, but greater range and variability in scores, especially for those who were most severely impacted (i.e. mental age below a 12-month level); however, ideas for measure modification to further improve psychometric properties of these tools for use in this population also emerged. This provides support for further exploration of these tools in individuals with DEE.
Parents often tell me during clinical evaluations that they know their child is having a good day when they are awake and responsive. This small but meaningful change increases their child’s ability to engage in, learn from, and enjoy their environment, whether at school, in therapies, in play, or in social interaction with family and peers. Measuring responsivity may provide useful information both for clinical purposes and for measuring the effects of intervention in clinical trials. MW
Meaningful change
Qualitative interviews were conducted with parents of all 10 children after the pilot study. The primary goal was to explore what ‘better’ looks like for SCN2A-DEE, as understood by their parents, in the functional domains of gross motor, fine motor, communication, and activities of daily living. Parents were asked: ‘What difference in function would be just enough for you to enter a clinical trial?’ and ‘What difference in function would be just enough for you to enter a gene therapy trial – where risks could be greater?’
The information in the interviews was coded for children with severe functional impairments and for children with more profound functional impairments for the different clinical trial scenarios. Parents described meaningful changes that seemed to describe smaller developmental steps for children with more functional impairment and more complex developmental steps for children with less functional impairment. Parents also described how potential risks associated with the trial would affect their views of what changes would be important to achieve. For example, greater changes were described as important when the potential risks associated with the trial were greater, such as in gene therapy trials.
Each of the meaningful and important differences, irrespective of being a small or larger change, was described as important because of their capacity to assist the child to live well, such as eating during mealtimes or playing with siblings. Our findings highlight that it is critical to ensure that the adapted tools in the Inchstone COA battery are sensitive enough to capture these meaningful changes. The Inchstone team recognizes that every individual with a DEE has their own specific set of challenges, and therefore, meaningful differences should be tailored to each individual.
Parents told us that new treatments are desperately needed to enable the child to engage and participate in life in their home and community and for care regimens to be easier for parents, such as understanding if the child is in pain. Our Inchstone work aims that these small changes in children with severe developmental impairments that would be meaningful to both child and family quality of life can be measured. JD
Personalized assessment – GAS
GAS is a patient-centric outcome that captures meaningful change by measuring progress towards personalized goals. 36 Goal setting starts with an interview, where a clinician or a healthcare professional trained in GAS helps the patient (and/or caregiver) identify those goals that are most important to each individual patient. For each unique goal, the patient’s baseline status is described, and a scale with five levels, ranging from +2 (best-expected outcome) to −2 (worst-expected outcome), is developed. Then, a post-intervention assessment is performed where the goal rater (e.g. the patient or caregiver and/or clinician) describes the level of attainment achieved for each goal.
As an individualized approach to the assessment of treatment response, the process of constructing goal scales may be prone to inconsistencies. As part of this pilot, we standardized the process of eliciting personal goals using (i) clinician training, (ii) caregiver orientation, where the patients are introduced to the goal-setting process and a potential list of goals before the interview, and (iii) an electronic data capture platform, GoalNAV®, specifically developed to capture GAS data. Investigation of goal scale quality, data completeness, and interview time suggested that GAS is feasible.
GAS interviewers reported that these interviews offered insights into the challenges and priorities of people living with SCN2A-DEE and their caregivers. One interviewer noted that ‘Parents [were] very engaged and willing to share some tough stuff in order to identify a way forward for their children’. Interviewers also indicated that the overall GAS process was a positive experience, with one interviewer stating, ‘Overall, the experience was positive, and I think there is a lot of potential with GAS in this population’.
Additional feedback indicated that conducting a feasibility study without intervention was difficult for the caregivers to understand and, at times, made the goal-setting process more difficult. Nevertheless, the high prevalence of non-seizure goals and the distribution of goals across several other domains, such as communication, eating, and toileting, underlined the need for personalized endpoints that capture within-patient meaningful change.
DEE parents speak: Beyond seizures, what really matters?
While the pilot with FamilieSCN2A provided in-depth experience with several important COAs in a small number of DEE-affected individuals, information about a broader range of DEEs was essential.
In 2023, with the PAG partnerships from DEE-P Connections, the Inchstone project launched the parents’ priorities survey to elicit structured information, both quantitative and qualitative, from parents of children with DEEs who had severe and profound impairments secondary to their disorders. In doing this, the team’s work was informed by and followed closely the PFDD guidance. The survey forms were reviewed for content, structure, and clarity with parent volunteers and modified based on their feedback. Over 270 families participated in the project and provided a wealth of information about the severity of their children’s difficulties in communication, mobility, hand use, eating, behavior, vision, and other areas. Parents assessed the impact of each of a large number of domains both on their children and on themselves and the rest of the family. They also identified the top three areas in which they would most like to see improvement. Based on the preliminary analyses, 37 communication was consistently identified as the area with greatest impact across all high-reporting disease groups and, regardless of the presence of other severe impairments and disabilities, the one that parents prioritized as most important for improvement (Figure 4). These findings, combined with a quality of life measure 38 that was also included in the project, The Developmental Profile-4, a neuropsychological assessment already trialed in the initial SCN2A-CTRS pilot project, and novel caregiver-report measures of both CVI and responsivity are helping to build a coherent picture across multiple different DEE diseases of what areas have the greatest impact for the most patients and families, guiding us toward adequate, FFP instruments for assessing these clinical outcome concepts in clinical trials.
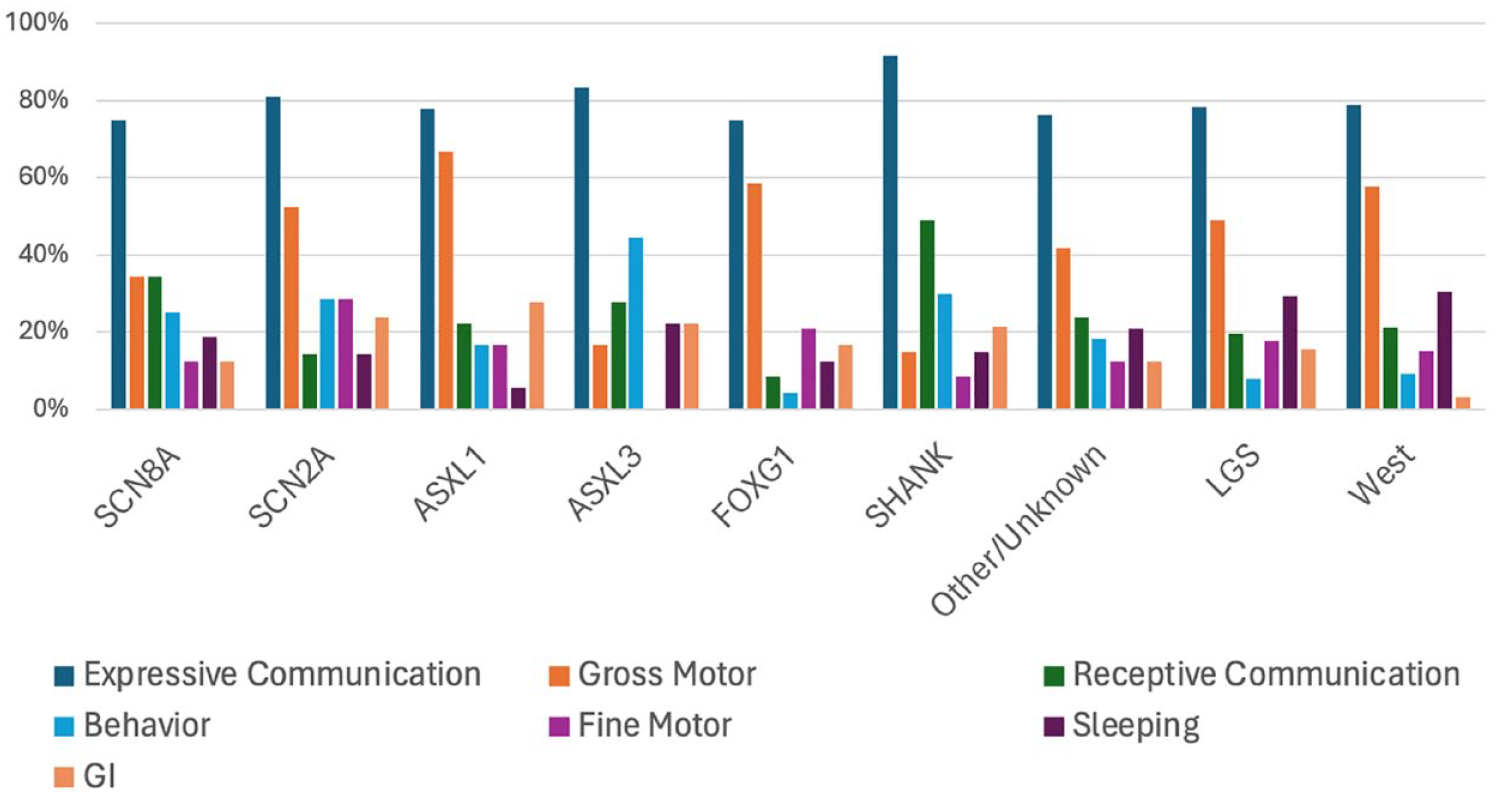
Preliminary data on parents’ top three priorities for non-seizure improvements from treatments.
Active engagement with broad DEE community – Families and scientists
The team recognizes that the success of this accelerated and complex effort requires that we stay in active communication with the broader range of community stakeholders. A major connection with the community is achieved through both the Industry Advisory Panel, with active participants from each of our industry sponsors/partners, and the Inchstone Steering Committee, with multiple PAG leaders and diverse professional experts. Each of these groups meets quarterly; meetings include updates on the progress of the work in addition to focused discussions of various strategic and methodological issues. These sessions are integral to the direction and progress of the overall project.
The Inchstone team has been active in engaging professionals through presentations at national professional meetings. In April 2023, the full team presented a symposium at the Gatlinburg Conference on Research and Theory in Intellectual and Developmental Disabilities, with an emphasis on measurement. In both 2023 and 2024, team members participated in the prestigious and action-oriented Epilepsy Foundation’s Research Roundtable and the Epilepsy Therapies & Diagnostics Development XVII symposium in 2023. We presented an Investigator Workshop at the 2022 American Epilepsy Society annual meeting, a panel of presentations at the 2023 Lennox-Gastaut Syndrome Foundation Meeting of the Minds Conference, a webinar for the Child Neurology Society, and presented posters at many professional meetings.
The team also has a commitment to publishing results throughout the course of our work to ensure dissemination of our findings to the scientific, regulatory, and advocacy communities. The first collaborative reporting by the group was a detailed filing with the FDA in response to a request for comments on the draft of the third PFDD guidance – Patient-Focused Drug Development: Selecting, Developing, or Modifying Fit-for-Purpose Clinical Outcome Assessments. While we had just begun collaborating, the draft guidance spoke directly to the entire mission of the Inchstone Project, providing a unique opportunity for the team. Our first intensive collaboration focused on a studious review of the draft guidance. We filed a comprehensive and fully referenced input on the draft guidance highlighting the distinct and challenging experience of applying the many elements of the guidance to the measurement of the most severely impacted DEEs and commended the FDA for the flexibility built into the guidance. We copied the Commissioner and alerted him to our work at a community event and were pleased to receive a direct response acknowledging the contribution to this complex area. Our comments 39 are part of the public record.
Jenny Downs led the team documenting the minimal clinically important difference (MCID) study in the pilot with children with SCN2A discussed above. This was a descriptive qualitative study to improve understanding of meaningful change for children with DEEs, 40 given the limited psychometric data on outcome measures for children with DEEs beyond measuring seizures and the absence of data to describe meaningful change. Establishing meaningful change thresholds for score interpretation is a critical component of outcome measure validation. This study explored caregiver perceptions of important differences in functional abilities that would guide their participation in clinical trials. Caregivers describe smaller developmental steps as meaningful for children with more limited developmental skills and more complex developmental steps for children with less limited skills. Caregivers also looked for ‘larger’ achievement from gene therapy trials relative to traditional trials, reflecting the potential larger benefits and harms of gene therapy trials. 39
Anne Berg led the team in a recent publication examining FDA’s PFDD guidances. 41 The paper discusses the challenges of adopting available COAs in severely impaired patient groups and strategies to adapt or modify existing COAs to be FFP for this population. Berg explores a range of potential adaptations, including alternative scoring, use of assessments in out-of-intended age ranges, and modifications for individuals with sensory or motor impairments. Berg specifically discusses the merits of ability versus disease focus, in particular where there are many commonalities across over a hundred severe DEEs.
COAs are often developed and validated for individual rare diseases. Although some disease-distinctive features encourage this practice, many outcome domains that might be targeted in a trial are common across several rare diseases. Rather than pursuing separate COAs for each disease, measures that are ability- or impairment-specific and capture the full range of function across multiple diseases, especially for functioning >3 SD below the mean, could provide great efficiencies for trial readiness in the rare DEE space. Further, as work is completed analyzing the major findings from the DEE Parents Speak survey and parent assessments, the team will prepare articles to present the results and their implications for further Inchstone work.
Future planned research by the Inchstone project
The Inchstone team has laid a solid foundation with the SCN2A pilot and advanced the design phase with completion of a comprehensive DEE-wide survey on preferences and several standardized instruments. The team is entering the second year of the design phase where much work remains to be done. Major tasks to complete the design phase include confirming the concept of interest, completing expanded clinical assessments, developing a standardized goal inventory for GAS, and identifying the initial Inchstone COA battery, including a comprehensive validation plan. See Figure 5, which outlines key tasks to complete design of the Inchstone COA battery.

Key tasks to complete design of the Inchstone COA battery.
In addition, the team will continue to engage with the broad community. We are requesting a Critical Path Innovation Meeting meeting with the FDA to review the direction of the research. In addition, we will continue to engage with the broad stakeholder communities, disseminate our work through webinars, meetings, and publications as well as seek new industry partners and sponsors, and actively engage with other potentially relevant disease areas. Progress and maintaining the 5-year goal remains heavily dependent on receiving sufficient funding – sustaining the expert global team and providing each with analytical and operational support is critical to success. While there are benefits of a patient-led effort, the team is addressing challenges in rapidly building the capacity to coordinate multi-site research, manage the breadth of operational issues, and finding efficiencies in the management of such a complex effort.
Conclusion – Impact of patient-led broadly collaborative effort
The severe limitations of existing tools to measure the small but important improvements achieved by those severely affected by DEEs are impediments to progress in therapeutic development for rare diseases by all stakeholders:
- Families experience the limitations every time they are involved in assessments, which are unsuitable for capturing what their children actually CAN do and only continue to document what they canNOT do. They recognize this effectively excludes their children from meaningful participation in clinical trials – especially when improvement in all important non-seizure outcomes is anticipated.
- Multiple dedicated researchers are independently studying and documenting the challenges and characterizations of individual DEEs on affected individuals, but progress toward needed systemic improvements has been limited.
- Clinicians are experiencing limitations of tools to measure important changes in the well-being or quality of life of their DEE-affected patients. Physical, occupational, and speech therapists, along with neuropsychologists, are individually seeking to adapt tools to approximate meaningful measures of improvements or regression.
- Pharmaceutical companies are struggling to improvise COAs for each new clinical trial in an effort to capture critical non-seizure outcomes important to participants and their families. Yet, there has been no coordinated effort to systematically address the gaps in measurement that could threaten approval for potentially transformative new treatments.
However, a coordinated effort to resolve the problems has been lacking. The Inchstone Project – a patient-led initiative – was created to address this challenge and, in just 2 years, has made significant inroads in laying an evidentiary basis for adaptation of existing instruments into an Inchstone COA battery, validated to capture small but meaningful improvement in the severely affected DEE population.
It is both depressing and demoralizing as a parent to be asked again and again to fill out assessments that cannot capture the small but meaningful abilities of my child. Every inchstone of progress should be measured and counted, especially when there are potential treatments on the line. Working with the Inchstone Project’s global group of leading, innovative researchers, Industry leaders, and experts who are committed to collaboratively pioneering this work is a testament to how far we can go when we tackle big problems as a community. GAC
The Inchstone Project has built a patient-led collaboration to address the problem across DEEs with potential applicability to a wide range of disease-modifying therapies in the pipeline. The project brings the passion, urgency, and deep insight of families to the collaborative effort of leading researchers, pharmaceutical companies, clinicians, experts, and government specialists. The team is driven by the urgency of families to tackle the critical need to measure every inchstone but is informed and disciplined by the FDA guidances and methodological rigor.
While the vision, urgency, and leadership for the Inchstone Project emerged from a cross-DEE collaborative, the project’s success reflects the shared vision and commitment by a wide range of other essential stakeholders. COA researchers, pharmaceutical partners, and other experts each recognized the clear benefits of a broad multi-stakeholder collaborative working in a pre-competitive space to rigorously develop/adapt measurement tools suitable for capturing smaller but no less significant changes in the DEE population. Precision medicine requires precision measurement – and the Inchstone Project is on track to create validated tools to ensure that the small but meaningful improvements in those severely affected by disease will be measured in trials. This, we believe, will help bring more transformative DMTs to the DEE community.
Footnotes
Appendix
Acknowledgements
We acknowledge the consistent support and interest by FamilieSCN2A, Leah Shust, their Executive Director, other DEE families and leaders of participating DEE-P partners and other PAGs, Industry Advisory Panel, Steering Committee and collaborators. We acknowledge the support of Charles Anderson, who provided extensive support on technical issues and required formatting of the manuscript.
