Abstract
Thromboangiitis obliterans, also known as Buerger’s disease, is a rare inflammatory vasculitis that predominantly develops in smokers and characteristically affects the small- and medium-sized peripheral arteries and veins. Patients typically present with extremity claudication, but symptoms may progress to rest pain and tissue loss, especially in those unable to abstain from tobacco use. Unfortunately, traditional medical treatments are largely ineffective and due to the small caliber of affected vessels and lack of suitable distal targets or venous conduits, endovascular and open surgical approaches are often not possible. Eventually, a significant number of patients require major amputation. For these reasons, much research effort has been made in developing techniques of therapeutic angiogenesis to improve limb perfusion, both for atherosclerotic peripheral arterial disease and the smaller subset of patients with critical limb ischemia due to Buerger’s disease. Neovascularization in response to ischemia relies on a complex interplay between the local tissue microenvironment and circulating stem and progenitor cells. To date, studies of therapeutic angiogenesis have therefore focused on exploiting known angiogenic factors and stem cells to induce neovascularization in ischemic tissues. This review summarizes the available clinical data regarding the safety and efficacy of various angiogenic therapies, notably injection of naked DNA plasmids, viral gene constructs, and cell-based preparations, and describes techniques for potentiating in vivo efficacy of gene- and cell-based therapies as well as ongoing developments in exosome-based cell-free approaches for therapeutic angiogenesis.
Plain Language Title and Summary
A review of available and emerging treatments for improving blood flow and wound healing in patients with Buerger’s disease, a rare disorder of blood vessels
Buerger’s disease is a rare disorder of the small- and medium-sized blood vessels in the arms and legs that almost exclusively develops in young smokers. Buerger’s disease causes inflammation in arteries and veins, which leads to blockage of these vessels and reduces blood flow to and from the extremities. Decreased blood flow to the arms and legs can lead to development of nonhealing wounds and infection for which some patients may eventually require amputation. Unfortunately, traditional medical and surgical treatments are not effective in Buerger’s disease, so other methods for improving blood flow are needed for these patients. There are several different ways to stimulate new blood vessel formation, both in humans and animal models. The most common treatments involve injection of DNA or viruses that express genes related to blood vessel formation or, alternatively, stem cell–based treatments that help regenerate blood vessels and repair wound tissue. This review explores how safe and effective these various treatments are and describes recent research developments that may lead to better therapies for patients with Buerger’s disease and other vascular disorders.
Keywords
Background
Epidemiology
Thromboangiitis obliterans (TAO) is a rare inflammatory vasculitis that affects the small- and medium-sized arteries and veins of the arms and legs. The disease was first described by Felix Von Winiwarter in 1879 but later named after Leo Buerger who, in 1908, provided a detailed pathological account in 11 amputated limbs. 1 Buerger’s disease predominantly develops in young to middle-aged male tobacco users,2,3 and most investigators agree that exposure to tobacco, whether firsthand or secondhand, is a prerequisite for development of the disease. 4 In a recent Japanese study, however, up to 8% of patients identified as never smokers. Never smoker patients tended to present with milder symptoms, but as objective nicotine testing was not performed, this population may represent patients with an unrecognized milder secondhand exposure to tobacco. 5 In the United States, incidence of Buerger’s disease has been declining alongside smoking rates and is now estimated as 12.6–20 per 100,000 people. 6 In parts of Asia and the Far and Middle East, however, the disease is much more prevalent. 6 In India, for example, between 45% and 63% of patients with peripheral arterial disease (PAD) are suspected of having Buerger’s disease, while in Korea and Japan, Buerger’s disease represents 16–66% of PAD cases.2,4,6
Clinical features
Buerger’s disease presents with ischemic symptoms of the distal upper and, more commonly, lower extremities. While the disease can progress proximally, involvement of large arteries is rare, especially in the absence of small vessel disease. 6 Typical symptoms are claudication of the hands, arms, feet, or legs, with severe disease leading to rest pain and ulceration. More than one limb is often affected. In a series of 112 patients with Buerger’s disease, 85 of whom had ischemic ulcerations, 28% had upper extremity, 46% lower extremity, and 26% both upper and lower extremity lesions. 7
Patients with ischemic ulcerations are at risk of developing chronic nonhealing wounds with superimposed infection that may ultimately require amputation. Compared with patients with critical limb ischemia (CLI) due to atherosclerotic PAD, patients with Buerger’s disease tend to present at younger age and with fewer comorbidities. As such, overall survival remains favorable. However, vascular events are common and represent a significant source of morbidity for this population. In a 2018 study of 224 patients with Buerger’s disease treated in France between 1970 and 2016, 5-, 10-, and 15-year vascular event-free survival was 41%, 23%, and 19% and amputation-free survival was 85%, 74%, and 66%. 8 Limb infection and nonwhite race conferred additional risk for vascular complications, and conversely, patients who quit using tobacco had lower risk of amputation that those who continued.
Pathophysiology
In contrast to atherosclerotic PAD, endarteritis in Buerger’s disease is nonatherosclerotic by definition and can affect both arteries and veins. Pathologically, Buerger’s disease progresses in three distinct phases. In the acute phase, the most diagnostic, a hypercellular inflammatory infiltrate accumulates within the vessel lumen. Vascular inflammation fosters a local prothrombotic state which leads to vaso-occlusion and then microabscess formation. Subacutely, granulomatous inflammation develops, and the thrombus is reorganized and recanalized. As the thrombus matures chronically, vascular fibrosis ensues, leaving a stenotic arterial lumen that clinically mimics lesions of atherosclerosis (Figure 1). 2

Pathogenesis of atherosclerotic PAD and Buerger’s disease. Prolonged exposure to atherosclerotic risk factors leads to plaque formation and arterial stenoses in atherosclerotic PAD. In Buerger’s disease, tobacco smoking triggers an inflammatory response in susceptible individuals leading to progressive thrombosis and fibrosis of small and medium-sized vessels. Both disease processes lead to luminal stenosis and location-dependent symptoms of tissue ischemia. Despite revascularization attempts, many patients with chronic nonhealing wounds develop gangrenous changes and superimposed infection which necessitate amputation.
While tobacco exposure is central to disease onset and continuation, the precise mechanisms of disease initiation remain unknown. Genetic predisposition certainly plays a role as several polymerase chain reaction (PCR)-based studies have identified specific human leukocyte antigen (HLA) class I and II alleles associated with increased odds of developing Buerger’s disease.9 –11 In susceptible individuals, exposure to tobacco may incite an aberrant immune reaction toward endothelium via toll-like receptor (TLR) signaling and the TLR-MyD88-NFκB pathway. 12 Serologic analyses have also found increased levels of circulating pro-inflammatory (IL-1β, TNF-α, and IL-6), type 1 helper cell (IFN-γ and IL-12), and type 2 helper cell cytokine profiles (IL-4, IL-5, and IL-13) in patients with Buerger’s disease, as well as those of type 17 helper cells (IL-17 and IL-23) which are strongly associated with autoimmunity. 13
Diagnosis
Careful history-taking and physical examination are critical for identifying Buerger’s disease. Diagnostic criteria by Shionoya 14 and Olin 4 and a scoring system by Papa et al. 15 all utilize common elements including history of tobacco use, onset before the age of 45–50 years, documentation of peripheral vascular disease by noninvasive or angiographic imaging, and exclusion of other etiologies, such as atherosclerosis, proximal embolic source, hypercoagulable state, diabetes, and autoimmune disorders.4,14 –17 Workup for Buerger’s disease should focus on ruling out other disease processes with similar presenting signs and symptoms. Routine laboratory tests, including complete blood count with differential, renal and liver function tests, erythrocyte sedimentation rate, and C-reactive protein levels are indicated, as well as markers of certain autoimmune conditions, notably rheumatoid factor (rheumatoid arthritis and vasculitis), antinuclear antibody (systemic lupus erythematosus, SLE), anticentromere antibody (CREST syndrome: calcinosis, Raynaud’s phenomenon, esophageal dysmotility, sclerodactyly, telangiectasia), and serum antitopoisomerase (scleroderma). Hypercoagulability screening is also indicated given the pathogenic role of thrombosis in this disease.
Angiographic evaluation can provide both diagnostic information and, in select cases, opportunity for therapeutic intervention. Characteristic angiographic findings include segmental stenoses or occlusions of the small- and medium-sized arteries distal to the brachial and popliteal arteries in the arms and legs, respectively. As such, the vessels most commonly involved are the palmar, radial, ulnar, and digital arteries in the upper extremities and the plantar, tibial, and peroneal arteries in the lower extremities. Corkscrew collaterals, known as Martorell’s sign, can reflect compensatory changes in the vasa vasorum, although similar changes may be observed in other autoimmune vasculitides and connective tissue disorders.4,18
Burden of disease
Failure of medical and surgical treatment in Buerger’s disease is multifactorial. Smoking cessation remains the mainstay of treatment, and those unable to do so are at increased risk of persistent disease and complications from surgical interventions. In addition, some patients present with late-stage, irreversible features such as digital gangrene which cannot be ameliorated by limb reperfusion. Given the autoimmune nature of this disease, individual variations in genetic and environmental background may also predispose certain patients to recurrent disease. In some cases, tissue loss may be severe enough to require amputation of the affected digit or limb. The risk of any amputation has been reported as 25% at 5 years, 38% at 10 years, and 46% at 20 years, and that of major amputation (above-the-knee, below-the-knee, or hand amputation) as 11% at 5 years, 21% at 10 years, and 23% at 20 years. 19 Patients undergoing amputation for gangrene or infection often face impaired mobility, reduced quality of life and psychosocial well-being, and increased healthcare costs. One study reported that 85% of patients with Buerger’s disease who underwent major lower limb amputation lost their jobs compared with 9% of those who did not require amputation. 20 Compared with a matched US population, patients with Buerger’s disease also had worse overall survival with average lifespan of 52.2 years. These findings highlight that despite earlier age and fewer comorbidities at presentation compared with patients with atherosclerotic PAD, patients with Buerger’s disease still face significant disease-related morbidity.
Current medical therapy
Cessation of all tobacco use remains the most important intervention in Buerger’s disease but substituting for smokeless tobacco does not appear to prevent limb loss. 21 Various pharmacological treatments have been used in the care of patients with Buerger’s disease, including anticoagulants, thrombolytics, vasodilators, and anti-inflammatory drugs; however, most of these therapies are ineffective. A recent Cochrane meta-analysis of five randomized controlled trials (RCTs) with a total of 602 patients evaluated the efficacy of several intravenous and oral therapies in treating Buerger’s disease. 22 Compared with aspirin, intravenous iloprost, a prostacyclin (PGI2) analogue, was effective in healing ulcers and eradicating rest pain after 28 days of treatment but had no effect on amputation rate 6 months after treatment.22,23 In addition, there was no difference in ulcer healing rate between prostacyclin (iloprost and clinprost) and prostaglandin (alprostadil) analogues, and oral iloprost was no more effective than placebo for healing of ischemic ulcers and resolution of rest pain.22,24 Outcomes data from these studies remain limited as none of the trials evaluated amputation-free survival, pain-free walking distance, or ankle-brachial indices (ABI).
Current surgical therapy
Surgical treatment of Buerger’s disease is limited by the small caliber of peripheral vessels typically involved. While angiography may be performed for diagnostic purposes, the affected arteries are often too small to access for angioplasty. In recent series, however, outcomes following percutaneous transluminal angioplasty (PTA) have been more favorable with technical success and limb salvage rates as high as 92–96% and 92–100%, respectively.25 –28 Although reintervention is required in approximately 30% of endovascular cases, 25 outcomes following open vascular reconstruction are significantly worse, partially due to lack of suitable venous conduits. In a retrospective study of 110 patients with Buerger’s disease, primary graft patency was 41% at 1 year, 32% at 5 years, and 30% at 10 years for 46 bypasses performed. 20 Secondary graft patency was 54% at 1 year, 47% at 5 years, and 39% at 10 years. All bypasses with prosthetic, bovine, or composite grafts were occluded within 10 months and, in patients with failed grafts, amputation was required in 5 of 35 limbs (14%). In inoperable cases, lumbar sympathectomy is sometimes offered to help reduce pain and increase perfusion via vasodilatory effect. However, the only RCT evaluating sympathectomy in Buerger’s disease found inferior benefit for pain relief and wound healing compared with intravenous iloprost. 29
Therapeutic angiogenesis
Other than smoking cessation, standard medical and surgical therapies for Buerger’s disease remain limited. In parallel, as many as 25–40% of patients with CLI due to atherosclerotic PAD are no longer eligible for or have failed prior revascularization attempts. 30 As such, both for Buerger’s disease and no-option CLI patients, alternative methods of improving limb perfusion are needed. With this goal in mind, significant research efforts have been made in developing gene and cell-based therapies to promote neovascularization in ischemic tissues, otherwise referred to as therapeutic angiogenesis. The term neovascularization encompasses all types of new blood vessel formation. In response to tissue hypoxia, endothelial cells (ECs) may be stimulated to sprout capillaries from pre-existing vessels (angiogenesis) or recruited from a pool of circulating stem and progenitor cells (vasculogenesis) to areas in need of new blood vessels (Figure 2). 31 In contrast, arteriogenesis refers to collateral formation in response to arterial occlusion and resulting shear stress forces. Arteriogenesis therefore plays an important role in compensating for large vessel occlusion seen in atherosclerotic PAD, but vascular remodeling secondary to small vessel disease, such as that in Buerger’s disease, relies primarily on angiogenesis and vasculogenesis.

Sequence of events in angiogenesis and points of action of various angiogenic therapies. Tissue hypoxia, ischemia, and injury lead to the release of pro-inflammatory cytokines from stromal cells. Activated endothelium expresses a variety of CAMs via HIF-1 and other response pathways to recruit EPCs necessary for vasculogenesis and MSCs that support the local tissue microenvironment. Stromal cells secrete VEGF and other growth factors to stimulate tip cell selection and migration, key steps in angiogenesis.
Stem and progenitor cells include endothelial progenitor cells (EPCs) that directly contribute to blood vessel formation and mesenchymal stem cells (MSCs) that influence the local stromal environment and support angiogenesis via paracrine secretion of growth factors and cytokines/chemokines. In 1997, putative EPCs were first characterized as CD34+/KDR+ (CD34, marker of stemness/immaturity; KDR, vascular endothelial growth factor receptor-2 (VEGFR2), marker of endothelial commitment). 32 EPCs arise from a heterogenic pool of CD133+ hemangioblasts that may differentiate into endothelial or myeloid lineages. 33 MSCs, however, are multipotent cells present in bone marrow, adipose tissue, and the umbilical cord that may differentiate into various connective tissue cells, including adipocytes, osteoblasts, myocytes, and chondrocytes.
In Buerger’s disease, angiogenic potential is uniquely altered compared with otherwise healthy smoker and nonsmoker patients. In a study comparing circulating progenitor cells and angiogenic factor levels in these groups, both smokers and patients with Buerger’s disease exhibited a decrease in the heterogenic pool of circulating CD45+/CD34+ progenitor cells. In Buerger’s disease only, however, the number of endothelial-specific progenitor cells remained comparable with healthy nonsmokers. Interestingly, despite similar levels of angiogenic factors (vascular endothelial growth factor (VEGF), angiopoietin-1 (ANG-1), endoglin, endostatin, matrix metalloproteinase-8 (MMP-8)), only serum from patients with Buerger’s disease impaired migration, sprouting, and proliferation of human umbilical vein endothelial cells (HUVECs) in vitro. 34 In contrast, another study found reduced number of EPCs in patients with Buerger’s disease and smokers, but no differences in endothelial cell function compared with healthy nonsmokers. 35 These contradictory findings highlight the poorly understood relationship between tobacco use, endothelial cell function, and EPC viability, but do suggest unique differences in angiogenic potential in patients with Buerger’s disease compared with smoker and nonsmoker controls.
Gene therapies
In response to tissue hypoperfusion, angiogenic factors such as VEGF, ANG-1, fibroblast growth factor (FGF), and various cytokines/chemokines are released by inflammatory cells, thereby initiating a paracrine signaling cascade that leads to pericyte detachment, endothelial permeabilization, and endothelial cell migration. 36 Given their pivotal role in angiogenesis, inducing overexpression of such factors in ischemic tissue is a logical therapeutic approach. To date, angiogenic factors have primarily been administered as naked DNA plasmids encoding the respective gene of interest or DNA packaged with a viral gene delivery vector. Given the higher prevalence of CLI due to atherosclerosis rather than Buerger’s disease, many clinical trials of therapeutic angiogenesis have primarily enrolled patients with atherosclerotic PAD. Nevertheless, results from these studies are valuable and, in several cases, patients with Buerger’s disease are specifically recruited in trials and included in analyses (Table 1).
Clinical trials of gene therapy in Buerger’s disease and atherosclerotic PAD/CLI.
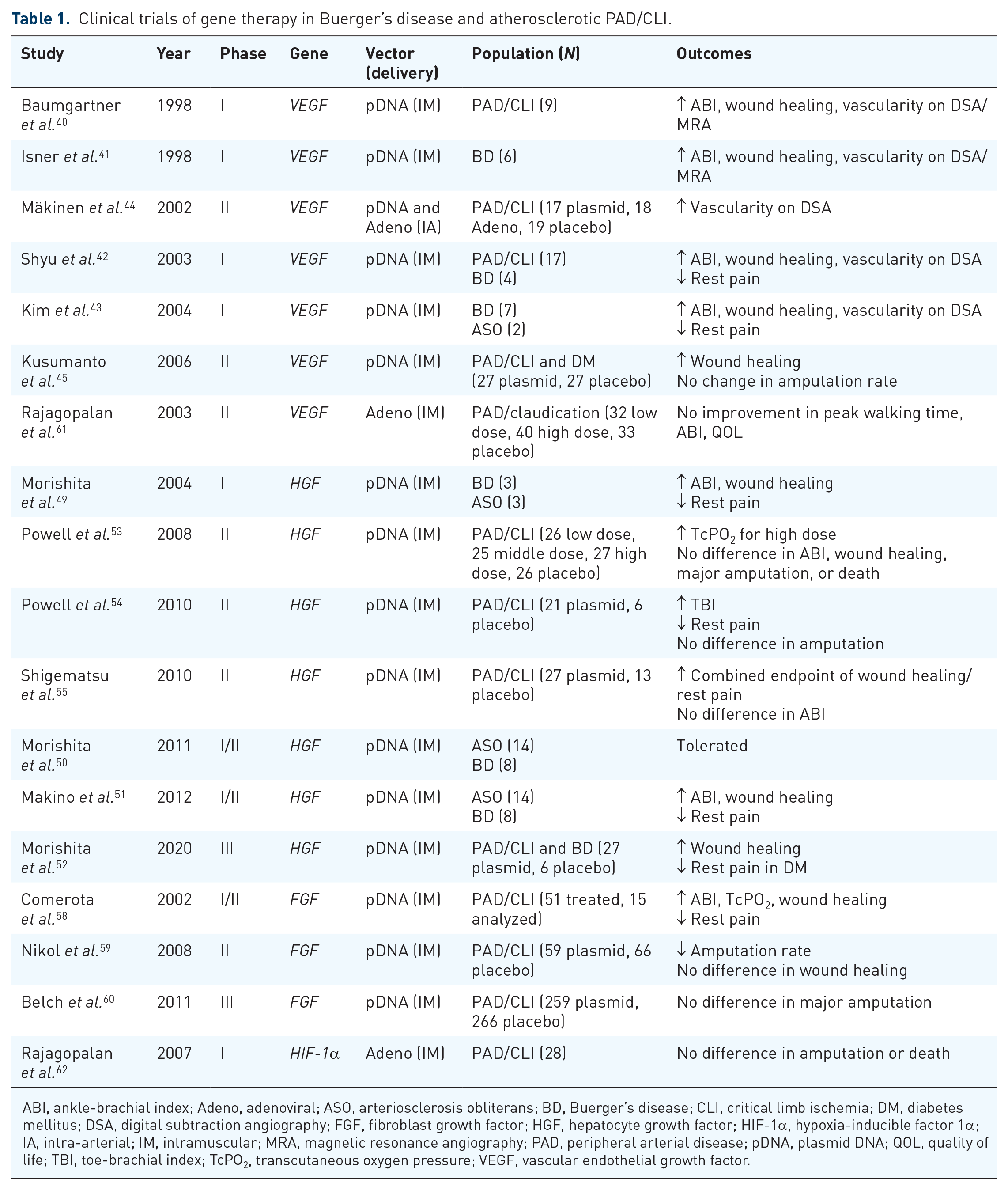
ABI, ankle-brachial index; Adeno, adenoviral; ASO, arteriosclerosis obliterans; BD, Buerger’s disease; CLI, critical limb ischemia; DM, diabetes mellitus; DSA, digital subtraction angiography; FGF, fibroblast growth factor; HGF, hepatocyte growth factor; HIF-1α, hypoxia-inducible factor 1α; IA, intra-arterial; IM, intramuscular; MRA, magnetic resonance angiography; PAD, peripheral arterial disease; pDNA, plasmid DNA; QOL, quality of life; TBI, toe-brachial index; TcPO2, transcutaneous oxygen pressure; VEGF, vascular endothelial growth factor.
Vascular endothelial growth factor
In the 1970s, Folkman hypothesized the existence of a soluble mediator of tumor angiogenesis, 37 and in 1989, Ferrara and Henzel isolated the factor now known as VEGF in media conditioned by pituitary follicular cells. 38 VEGF expression is critical for embryonic development and postnatal neovascularization. Preclinical studies in rabbits provided early evidence that VEGF gene therapy can enhance angiogenesis following induction of hindlimb ischemia. 39 In 1998, Baumgartner et al. 40 subsequently published a phase I trial of intramuscular (IM) administration of naked DNA plasmid encoding the 165-amino-acid soluble isoform of human VEGF (phVEGF165) in 10 limbs of nine patients with CLI and nonhealing ulcers. Mean ABI improved from 0.33 to 0.48, ischemic ulcers improved in four of seven limbs, and three patients originally recommended for below-knee amputation achieved limb salvage. An additional seven limbs in six patients with Buerger’s disease were treated in a separate study with similar results: an increase in ABI of 0.10 in four of five patients with baseline ABI <0.60, improved angiographic findings, and salvage of six of seven limbs. 41
Following Isner and Baumgartner’s pioneering work, several other studies of VEGF-based gene therapy emerged. In 2003, Shyu et al. 42 published their experience with phVEGF165 in 24 limbs of 21 patients with rest pain, 16 of whom also had nonhealing ulcers. Mean ABI improved from 0.58 to 0.72, ischemic ulcers improved in 75% of cases, and rest pain was relieved in 83% of patients. In a 2004 dose-finding study of IM VEGF165 in nine patients with debilitating PAD, seven of whom had Buerger’s disease, mean ABI improved from 0.52 at baseline to 0.71 and 0.69 at 3 and 9 months posttreatment. 43 Of the seven patients with ischemic ulcers, two had complete resolution and another two had improvement in ulcer size. Digital subtraction angiography (DSA) also demonstrated increased collateral density in six of nine patients at 3 months posttreatment and ischemic pain improved in most patients. Of note, both studies reported transient leg edema in six of 24 limbs and three of nine patients, respectively.
In randomized studies, VEGF-based plasmid therapy has yielded more modest benefit. In 2002, Mäkinen et al. 44 published a trial of intra-arterial (IA) VEGF gene therapy administered at time of angioplasty either as naked plasmid or adenoviral vector. Three months after treatment, DSA revealed improved vascularity in both VEGF-treated groups compared with placebo, but there were no significant difference in hemodynamic parameters or amputation rate. In a RCT of phVEGF165 in 54 diabetic patients with CLI, Kusumanto et al. 45 found that 33% of patients in the treatment group had a >60% decrease in ulcer surface compared with none of the patients in the control group; however, there was no significant difference in major amputation rate at 100 days. Altogether, these results indicate that VEGF therapy may improve wound healing rate with increase in ABI on the order of 0.1 but no definitive clinical efficacy with regard to amputation rate.
Hepatocyte growth factor
Hepatocyte growth factor (HGF) is secreted from stromal cells and acts via its target receptor, cMet, on smooth muscle cells, fibroblasts, and ECs. 46 As a potent mitogen, HGF stimulates proliferation, motility, and morphogenesis of these cells via paracrine and autocrine effects. In contrast to VEGF and FGF, HGF also exerts anti-inflammatory and antifibrotic effects by means of epidermal growth factor receptor (EGFR) downregulation.46,47 Interest in HGF-based angiogenic therapies stemmed from preclinical studies of HGF plasmid transfection in rabbit and rat ischemia models that showed dose-dependent increases in blood flow following treatment. 48 In a subsequent trial of HGF gene therapy in six patients with CLI secondary to arteriosclerosis obliterans (ASO) and Buerger’s disease who were not candidates for surgical revascularization, 49 ABI improved from 0.43 at baseline to 0.63 at 4 weeks and 0.60 at 8 weeks.
In 2011, Morishita et al. 50 published their initial findings of HGF gene transfer in 22 patients with CLI. ABI increased from 0.44 at baseline to 0.59 at 2 months and 0.65 at 6 months. Walking distance also improved in all seven patients with severe claudication and ulcer size decreased in 63.6% of patients with wounds. Treatment effect was sustained in the 14 patients evaluated at 2-year follow-up as ABI remained significantly elevated to 0.61. 51 Importantly, no adverse events related to the treatment were reported. In a follow-up analysis of all Japanese trials, Morishita et al. reported that HGF therapy was associated with improved wound healing, including complete ulcer resolution at 12 weeks in 48% of treated patients compared with 17% for placebo group. Rest pain was also significantly improved but only in patients with Buerger’s disease and diabetes. 52
Placebo-controlled studies have confirmed some positive effects of HGF therapy but failed to show benefit for other important clinical endpoints. In 2008, Powell et al. 53 reported that high-dose (4 mg IM at 0, 14, and 28 days) HGF plasmid therapy increased transcutaneous oxygen pressure (TcPO2) in 100 patients with CLI and screening TcPO2 < 40 mmHg. However, there were no differences in ABI, toe-brachial index (TBI), ulcer healing rate, or major amputation rate at 6 months. In a follow-up phase II study, the same group showed that TBI and pain were significantly improved after HGF therapy in 21 patients with CLI compared with six subjects assigned to placebo. 54 However, wound healing, amputation, and mortality rates were no different between the two groups. Another randomized study of HGF plasmid injection in CLI found improved ulcer size or rest pain in 70.4% of treated patients compared with 30.8% in placebo group but no significant difference in change of ABI at 12 weeks between the two groups. 55 In all trials, HGF therapy demonstrated favorable safety profile without significant treatment-related adverse events or cases of peripheral edema as seen with VEGF therapy.
Fibroblast growth factor
FGF comprises a family of growth factors and signaling proteins that exert mitogenic effects on a variety of cell types, thereby playing a role in organogenesis and angiogenesis. FGF exists in two major forms, acidic FGF (FGF-1) and basic FGF (FGF-2). In preclinical studies, both FGF-1 and FGF-2 were able to improve limb revascularization in hamster and rabbit models of hindlimb ischemia, respectively.56,57 An early pilot study of nonviral FGF-1 (NV1FGF) in 51 patients with inoperable CLI found that therapy was well-tolerated and associated with reduced pain and ulcer size and increased TcPO2 and ABI compared with pretreatment values. 58 Subsequently, the phase 2 TALISMAN trial randomized 107 patients with no-option CLI and nonhealing ulcers to NV1FGF or placebo. While ulcer healing rate, the primary endpoint, was no different between the two groups, NV1FGF did reduce risk of major and any amputation (hazard ratio (HR) = 0.371 and 0.498, respectively). 59 In contrast, the phase 3 TAMARIS study randomized 525 patients with inoperable CLI to NV1FGF or placebo and showed no difference in the combined primary endpoint, time to major amputation on the treated leg or death. 60 This discrepancy regarding benefit of NV1FGF therapy on amputation rate may be explained by unequal amputation rates in the placebo groups of these studies, which was more than 50% in the initial TALISMAN study compared with 33% in the subsequent TAMARIS trial.
Cell therapies
While angiogenic factors are logical targets for therapeutic angiogenesis, cell-based approaches provide another means of improving perfusion and wound healing in ischemic tissue. Mononuclear cell (MNC) fractions derived from bone marrow or peripheral blood contain cells of monocyte/macrophage lineage, as well as EPCs that can directly participate in vasculogenesis. MSCs, however, are multipotent stromal precursors that may differentiate into cells required for tissue regeneration, such as ECs for angiogenesis and fibroblasts for wound healing, but primarily function via paracrine mechanisms to prime the engrafted tissue for cell recruitment, migration, and differentiation. In preclinical studies, both MNCs and MSCs have been shown to induce neovascularization. For example, in a rat model of hindlimb ischemia, perfusion index was significantly increased 3 weeks after IM injection of either 5 × 106 MSCs or MNCs compared with vehicle. 63 Compared with MNCs, however, MSCs were better able to improve perfusion index and capillary density and were the only cell type capable of differentiation into vascular smooth muscle cells and ECs.
MNC therapy
Findings from the above and other preclinical studies paved the way for clinical trials of cell-based therapeutics for patients with CLI due to atherosclerotic PAD and Buerger’s disease (Table 2). In the Therapeutic Angiogenesis by Cell Transplantation (TACT) trial, 45 patients with bilateral lower limb ischemia received injection of bone marrow–derived MNCs (BM-MNCs) into the left or right gastrocnemius muscles and saline or peripheral blood MNCs (PB-MNCs) in the other limb as control treatment in a within-patient trial design. 64 Compared with those receiving PB-MNCs, limbs treated with BM-MNCs demonstrated mean ABI increase of 0.09. Improvements were also noted in TcPO2 measurements, rest pain, new collateral formation, and pain-free walking time at 4 and 24 weeks posttreatment. Long-term follow-up of 115 patients from TACT showed that beneficial effects on pain, ulcer size, and pain-free walking distance were durable up to 2 years after therapy. 65 Importantly, patients with Buerger’s disease fared better than those with atherosclerotic PAD: 3-year amputation-free survival was 91% in patients with Buerger’s disease compared with 60% in those with atherosclerotic PAD. Idei et al. 66 found similar differences in their nonrandomized study of BM-MNC transplantation in 51 patients with CLI, 26 of whom had Buerger’s disease. Four-year amputation-free survival for patients with Buerger’s disease was 95% after BM-MNC treatment and 6% in the control group, compared with 48% and 0%, respectively, for patients with atherosclerotic PAD. Consistently, in Buerger’s disease patients, ABI and TcPO2 increased 1 month after treatment and remained elevated at 3-year follow-up, while in PAD patients, improvements in ABI and TcPO2 observed at 1 month regressed to baseline on long-term follow-up. The more durable benefits observed in Buerger’s disease compared with atherosclerotic PAD may reflect the potentially reversible autoimmune nature of the former compared with the cumulative, and more permanent, plaque burden characteristic of the latter.
Clinical trials of cell-based therapeutic angiogenesis in Buerger’s disease and atherosclerotic PAD/CLI.

ABI, ankle-brachial index; AD-MSC, adipose-derived mesenchymal stem cell; ADRC, adipose-derived regenerative cell; BD, Buerger’s disease; BM-MNC, bone marrow-derived mononuclear cell; BM-MSC, bone marrow-derived mesenchymal stem cell; CLI, critical limb ischemia; DM, diabetes mellitus; DSA, digital subtraction angiography; PAD, peripheral arterial disease; PB-MNC, peripheral blood mononuclear cell; QOL, quality of life; TcPO2, transcutaneous oxygen pressure.
Studies of cell-based therapy specifically enrolling patients with Buerger’s disease have also yielded encouraging results. In 2006, Durdu et al. 67 reported on their experience with autologous IM injection of BM-MNCs in 28 patients with unilateral CLI secondary to Buerger’s disease. By 6 months posttreatment, patients demonstrated significant improvements in rest pain, peak walking time, and quality of life. Complete ulcer healing was achieved in 83% of patients with wounds, while half of patients had an increase in ABI >0.15 and 78.5% demonstrated collateral formation on DSA. In 2007, Saito et al. 68 published their results of a nonrandomized study of autologous BM-MNC transplantation in 14 patients with ischemic rest pain or ulceration due to Buerger’s disease. Pain scores improved from 5.4 at baseline to 2.5 at 4 weeks and 1.1 at 24 weeks. Of the 19 ulcers in nine patients, 12 completely healed, two diminished in size, and four increased in size by 24 weeks. In 2008, Motukuru et al. 69 reported on 36 patients with inoperable Buerger’s disease treated with IM injection of autologous BM-MNCs. Three patients (12%) required major amputation by 6 months posttreatment. ABI and TcPO2 significantly improved by 0.14 and 52 mmHg, respectively, as did pain measured on a visual analogue scale. In all studies, no treatment-related complications were reported.
MSC therapy
In 2009, Dash et al. published the only randomized study of autologous bone marrow–derived MSC (BM-MSC) administration in 24 patients with nonhealing ulcers of the lower extremities.70,71 In the 18 patients with a diagnosis of Buerger’s disease, ulcer area decreased by 71% in the treatment group compared with 23% in the control group by 12-week follow-up. Pain-free walking distance also improved from 38.3 m to 284.4 in the treatment group compared with an increase from 35.7 to 78.2 m in the control group. In 2017, a nonrandomized dose-finding study of allogenic BM-MSC treatment in 72 patients with Buerger’s disease found that ulcer healing and rest pain reduction rates were significantly better in the 2 million cells/kg group compared with 1 million cells/kg and standard of care groups; however, there was no significant difference in collateral formation on magnetic resonance angiography (MRA) or amputation-free survival. 72
Adipose tissue is another source of MSCs for autologous transplantation and early pilot studies demonstrated feasibility and safety of this technique. 73 In one series, seven of 12 patients with Buerger’s disease noted improved ulcer appearance after AD-MSC treatment, 74 while in another, walking distance increased from 300 m at baseline to 397 m 24 weeks after treatment in 17 patients with severe Buerger’s disease. 75 In TACT-ADRC, five patients with CLI received autologous IM injection of adipose-derived regenerative cells (ADRCs) and noted significant improvements in pain and 6-min walking distance with no amputations during the 6-month study period. 76 Interestingly, ADRCs increased the number of circulating progenitor cells (CD34+/CD133+) and decreased the relative expression of VEGF-A165b, an antiangiogenic alternative splice variant of VEGF-A. In comparing different cell preparations, a Cochrane review pooling results from seven RCTs (359 patients) of autologous cell-based therapy in no-option CLI found no difference in outcomes between BM-MNC and mobilized peripheral blood stem cell (mPBSC) treatment, or between IM or IA injection. 30 However, in one RCT comparing BM-MSCs and BM-MNCs in 41 patients with diabetes and CLI, BM-MSCs were better able to induce limb reperfusion and ulcer healing with significantly greater increase in ABI, TcPO2, and pain-free walking time. 77
Adverse effects and limitations of current angiogenic therapies
In all examined studies, the most prevalent treatment-related side effects have been limited to injection site reactions which are generally self-limited and managed conservatively. In the case of VEGF gene therapy, specifically, there are several reports of peripheral edema as a complication of both naked plasmid and adenoviral delivery mechanisms. This complication has not been observed with other gene therapies. Potential adverse effects of angiogenic therapy include proliferative retinopathy, arteriovenous malformation, and neoplasia; however, none of the available studies reported significant associations between either gene or cell therapy and such complications. Nevertheless, patients should be screened for malignancy and retinopathy and counseled on these potential risks prior to initiating angiogenic therapy.
As research into therapeutic angiogenesis has progressed from small observational studies to larger prospective cohort and randomized trials, several meta-analyses have collectively analyzed safety and efficacy data for gene and cell-based therapeutics in CLI. Recently, a Cochrane meta-analysis of 17 RCTs evaluated efficacy of various gene therapies for intermittent claudication and CLI in 1988 participants. 78 Pooled data showed no clear difference between gene therapy and placebo with regard to amputation-free survival and all-cause mortality. Although there was limited evidence for improved ulcer healing with gene therapy, the authors found no difference in pain scores or change in ABI between treatment and control groups. In subgroup analyses, there was no difference in outcomes between gene therapy and placebo according to PAD severity (claudication versus CLI), vector type (plasmid versus viral), or gene encoded.
Results from meta-analyses of cell-based therapies have also been mixed. Multiple studies using data from randomized as well as nonrandomized and uncontrolled trials have reported favorable effects on amputation and ulcer healing rates with cell therapy.79 –82 However, when only randomized studies are included, the benefits are more marginal. For example, Peeters Weem et al. 83 found significantly improved ABI (mean difference 0.11), TcPO2, and pain scores with cell therapy, but no significant differences in primary outcome measures of amputation, survival, and amputation-free survival in 499 CLI patients from 10 studies. Another meta-analysis of 12 randomized trials including 510 CLI patients initially found reduced major amputation risk (risk ratio (RR) = 0.58) with cell therapy, but the results became nonsignificant when only placebo-controlled studies were analyzed. 84 Similarly, an analysis of 774 CLI patients from 16 RCTs found reduced odds of major amputation (odds ratio (OR) = 0.54) and improved ulcer healing (OR = 2.90) with cell therapy, but again, all differences became nonsignificant when only placebo-controlled trials were reanalayzed. 85
Emerging trends in therapeutic angiogenesis
To date, clinical trials of therapeutic angiogenesis have primarily focused on DNA plasmids encoding known angiogenic factors or cell-based treatments using MNCs or MSCs. Improving upon these treatments and developing novel therapies for CLI hinges on an understanding of the precise mechanisms involved in vasculogenesis and angiogenesis. The recruitment of EPCs and MSCs from the bone marrow to areas of ischemia and wound healing is known as the process of homing. Homing of EPCs and MSCs is subject to regulation by many factors and mediated by a panel of cell adhesion molecules (CAMs). Under pathological conditions, such as tissue injury, ischemia, and inflammation, the normally quiescent endothelium is stimulated to express a variety of CAMs and secrete cytokines/chemokines to recruit the necessary immune-modulatory and repair cells. Importantly, hypoxia-inducible factor 1α (HIF-1α) expression induces secretion of various cytokines/chemokines, including stromal cell-derived factor 1 alpha (SDF-1α&x41;, TNF-α, and IL-1.86,87 Consistently, in preclinical studies, increasing tissue microenvironment levels of SDF-1α in diabetic mice has been shown to improve wound healing rates. 86
E-selectin for therapeutic angiogenesis
In particular, SDF-1α upregulates expression of E-selectin and its ligands on ECs.86,88 E-selectin is an inducible CAM expressed on endothelium which binds P-selectin glycoproteins-1 (PSGL-1/CD162), CD44, and E-selectin ligand (ESL-1) presented on the surface of circulating cells. In this way, E-selectin serves as endothelial docking sites to anchor circulating stem and progenitor cells, such as EPCs, that reciprocally express E-selectin ligands. In murine models, levels of membrane-bound E-selectin peak 12 h after ischemic insult, while serum levels of soluble E-selectin remain elevated for up to 7 days. 89 Homing of EPCs is impaired in E-selectin knock-out mice (Esel−/−) but can be rescued by IV administration of sE-selectin. 89 In addition, Esel−/− mice demonstrate impaired EPC homing to wound tissue, slower wound closure rate, and delayed recovery of hindlimb perfusion following femoral artery ligation. 88 E-selectin-mediated cell–cell interactions between MSCs and ECs are also critical events for MSC-induced angiogenesis and tissue repair.86,90,91 While the above effects of E-selectin reflect cell–cell interactions mediated by the protein’s extracellular domain, we have unpublished gene profiling data from ischemic hindlimbs treated with IM E-selectin gene therapy indicating that, compared with control vector, E-selectin overexpression leads to intense upregulation of dozens of pro-angiogenesis genes while having an overall inhibitory or cooling effect on the majority of pro-inflammatory genes. Altogether, these findings illustrate the role of E-selectin in the tissue response to ischemia and present a novel target for therapeutic angiogenesis in CLI and Buerger’s disease.
Viral vectors for gene delivery
To date, most growth factor–based therapeutics have been formulated as naked DNA-encoding plasmids. Viral vectors provide another means of increasing local expression of angiogenic factors, and adenovirus, specifically, has been used in studies of therapeutic angiogenesis. A randomized, double-blind, placebo-controlled trial assessed the efficacy of Ad2/HIF-1α/VP16, an adenoviral construct encoding a constitutively active form of HIF-1α, in 34 no-option CLI patients. 62 Injection site reactions and peripheral edema were the most common side effects. After 1 year, complete resolution of rest pain occurred in 14 of 21 patients and complete ulcer healing in 5 of 8 patients alive with the index limb. Another RCT evaluated adenoviral delivery of VEGF but failed to show benefit in 105 patients with unilateral exercise-limiting claudication. 61 Specifically, there was no significant difference between treatment and placebo groups with regards to change in peak walking time, the primary endpoint, or secondary endpoints of ABI, claudication onset time, and quality of life. In addition, AdVEGF121 therapy was associated with peripheral edema in the treated limb. Recently, the Food and Drug Administration (FDA) approved two gene therapy products, Luxturna® (voretigene neparvovec) and Zolgensma® (onasemnogene abeparvovec-xioi), which are now available for the treatment of Leber congenital amaurosis and spinal muscular atrophy, respectively. These treatments employ another gene delivery vector, adeno-associated virus (AAV), which has a more favorable safety profile than adenovirus. In comparison with adenoviral vectors, which activate the NF-κB pathway, AAV does not promote a strong immune response to virally encoded proteins and provides long-lasting stable tissue transduction up to 2 years. 92 These features make AAV an interesting gene delivery platform for future clinical trials of therapeutic angiogenesis.
E-selectin/adeno-associated virus gene therapy
As discussed, the adhesion molecule E-selectin and its ligands play a crucial role in the response to tissue ischemia and injury by priming the endothelium to recruit necessary repair cells from the circulation. E-selectin overexpression in ischemic hindlimb tissue also causes activation of secondary paracrine signaling cascades that augment tissue angiogenesis and downregulate inflammation. With the goal of leveraging these actions of E-selectin for therapeutic benefit, we have tested the feasibility, safety, and efficacy of IM E-selectin/AAV gene therapy in murine hindlimb ischemia and gangrene models (Table 3).93,94 In summary, treating hindlimb muscle with E-selectin/AAV results in significantly improved recovery of hindlimb perfusion following ischemic insult. Clinically, enhanced blood flow to the hindlimb translates into more rapid wound healing, reduced gangrene severity, and increased exercise capacity. On DiI perfusion imaging, mice treated with E-selectin/AAV demonstrate plusher capillary networks surrounding the wound bed and murine footpad. Importantly, systemic D-dimer levels are not increased from baseline and no microvascular thrombosis or T cell infiltration is noted on histological analysis, indicating that local IM E-selectin gene therapy does not have a prothrombotic effect or cause a lymphocytic reaction. Altogether, these preclinical results pave the way for development of E-selectin-based angiogenic therapies.
Emerging modalities for therapeutic angiogenesis in preclinical stages.
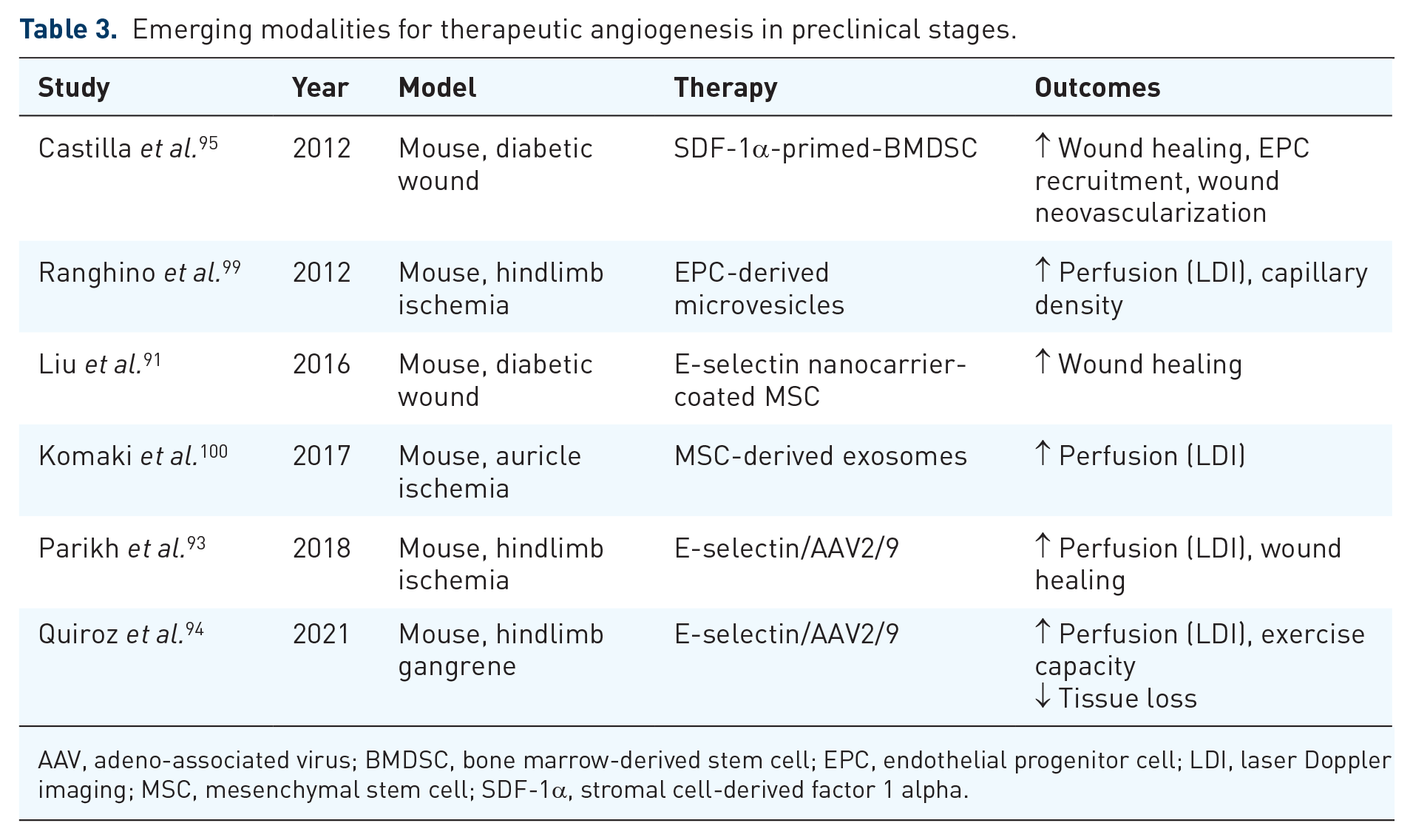
AAV, adeno-associated virus; BMDSC, bone marrow-derived stem cell; EPC, endothelial progenitor cell; LDI, laser Doppler imaging; MSC, mesenchymal stem cell; SDF-1α, stromal cell-derived factor 1 alpha.
Enhanced cell-based therapies
In clinical studies, cell-based therapies have shown potential for improving ulcer healing, pain, and walking capacity, but placebo-controlled trials have reported modest to no benefit on hemodynamic parameters and amputation rate. Importantly, these studies have only employed unmodified, unstimulated MSCs or MNCs isolated from the circulation, bone marrow, or adipose tissue. One potential avenue for increasing the potency of these therapies involves priming stem cells with various growth or mitogenic factors in culture prior to transplantation. For example, in mice, priming bone marrow–derived stem cells (BMDSCs) with SDF-1α prior to intradermal injection increases wound healing potential compared with unstimulated BMDSCs. 95 There is also evidence that AAV-induced expression of E-selectin in MSCs, which do not normally express this adhesion molecule, can increase proliferative and anti-inflammatory properties of these cells, translating to improved recovery of limb perfusion and function in murine hindlimb ischemia models. Similarly, in rat myocardial infarction models, HGF gene-engineered myoblasts exhibit improved engraftment and ultimately lead to better recovery of cardiac function. 96 Rather than directly administering modified stem cells, BM-MSCs can also be primed in co-culture with genetically modified HGF-expressing MSCs. 97 In the form of a cardiac patch, conditioned BM-MSC demonstrated improved repair of infarcted myocardium compared with unstimulated BM-MSC. In another approach, coating MSCs with E-selectin/dendrimer nanocarrier can specifically direct these cells from the circulation to activated endothelium in ischemic or inflamed tissue, thereby improving wound healing rate. 91
Exosomes and cell-free approaches
Despite promising preclinical and clinical results, there remain several inherent limitations to cell-based therapies. Autologous stem cell treatments raise the theoretical risk of unmitigated cell proliferation and potential malignant transformation, while allogeneic transplantation carries additional risk of immune rejection. In addition, there is evidence that cells isolated from patients with Buerger’s disease, diabetes, or other forms of PAD and CLI have poorer angiogenic potential compared with cells from healthy subjects, which further limits in vivo efficacy. 74 With this in mind, multiple studies have observed that conditioned media, or the supernatant collected from cell culture plates after incubation for a period of time, can alone exert angiogenic and antiapoptotic effects in vitro. 98 In particular, microvesicles derived from EPCs have been shown to improve neovascularization in a murine hindlimb ischemia model, 99 whereas exosomes isolated from MSC cultures have been shown to improve neovascularization in a murine model of auricle ischemia 100 and fracture healing in a rat model of nonunion. 101 Exosomes are small vesicles (30–150 nm) secreted by MSCs and other cell types to deliver protein, RNA, and cytokines/chemokines that stimulate biological functions by paracrine mechanisms. Exosomes lack major histocompatibility complex (MHC) and are therefore nonimmunogenic. This is advantageous as exosome extracted from cells isolated from unrelated healthy donors could be administered to patients in an allogeneic manner without concern for immune reaction. In addition, exosomes can be concentrated and then stored or transported with greater logistical ease than the cell cultures they are derived from.
Conclusion
While increasingly rare in the West, Buerger’s disease represents an important form of PAD worldwide with significant morbidity for patients afflicted with the disease. Despite remarkable advances in drug development and surgical technology over the past century, the treatment landscape for Buerger’s disease remains limited. Current medical therapies have little to no benefit in randomized studies, and surgical revascularization, both open and endovascular, is not a viable option for many patients. As such, noninvasive regenerative approaches are attractive avenues for clinical research. Multiple prospective studies have reported positive effects of gene and cell therapies on limb pain, walking distance, and ulcer healing rates in patients with CLI and Buerger’s disease. However, there is still a lack of strong evidence for improvement in hemodynamic parameters or amputation-free survival in randomized trials. For these reasons, optimizing existing technologies and exploring new targets for therapeutic angiogenesis are important strategies for helping patients with Buerger’s disease, and the larger population of no-option CLI patients. E-selectin is one such possible target, with multiple preclinical studies having elucidated its role in angiogenesis and efficacy in diabetic wound healing and reversal of hindlimb ischemia. Inducing cellular expression of E-selectin, or other CAMs and growth factors, may be used to potentiate in vivo angiogenic effect of some cell-based therapies. Alternatively, cell-free preparations of concentrated MSC-derived exosomes may deliver angiogenic effect while avoiding complications of naked plasmids, viral vectors, and cell-based therapies. In summary, the treatments reviewed herein represent some of the most promising emerging modalities for therapeutic angiogenesis that may someday be readily available to relieve ischemic pain, heal ulcers, prevent major amputation, and improve quality of life in patients with Buerger’s disease and no-option CLI.
Footnotes
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The E-selectin gene modification technologies described were developed in our research laboratory and patented/licensed by the University of Miami. This technology is currently under preclinical development by Ambulero Inc., a new start-up company out of the University of Miami that focuses on developing new vascular treatments for ischemic conditions. Co-authors, Z.J.L. and O.C.V., serve as consultants and scientific and medical advisory officers.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics statement
Animal experiments performed at the University of Miami were approved by the University of Miami Institutional Animal Care and Use Committee (IACUC) under protocol 19-163. Ethical approval and informed consent were not required for the remainder of this review.
