Abstract
Erythropoietic protoporphyria (EPP) is an ultra-rare inherited disorder with overproduction of protoporphyrin in maturating erythroblasts. This excess protoporphyrin leads to incapacitating phototoxic burns in sunlight exposed skin. Its biliary elimination causes cholestatic liver injury in 20% and terminal liver failure in 4% of EPP patients. Thereby, the risk of liver injury increases with increasing erythrocyte protoporphyrin concentrations. Afamelanotide, an α-melanocyte-stimulating hormone (MSH) analog inducing skin pigmentation, was shown to improve sunlight tolerance in EPP. Beyond this well-known effect on pigmentation, the MSHs have liver-protective effects and improve survival of maturating erythroblasts, effects described in animal or in vitro models to date only. We investigated whether afamelanotide treatment in EPP has effects on erythropoiesis, protoporphyrin concentrations, and liver injury by analyzing retrospectively our long-term safety data.
Methods:
From the 47 Swiss EPP-patients treated at our center since 2006, we included those 38 patients in the current analysis who received at least one afamelanotide dose between 2016 and 2018 and underwent regular laboratory testing before and during the treatment. We compared the means of pretreatment measurements with those during the treatment.
Results:
Protoporphyrin concentrations dropped from 21.39 ± 11.12 (mean ± SD) before afamelanotide to 16.83 ± 8.24 µmol/L (p < .0001) during treatment. Aspartate aminotransferase decreased from 26.67 ± 13.16 to 22.9 ± 7.76 IU/L (p = .0146). For both entities, patients with higher values showed a more progressive decrease, indicating a risk reduction of EPP-related liver disease. The pre-existing hypochromia and broad mean red-cell distribution width were further augmented under afamelanotide. This was more likely due to an influence of afamelanotide on maturating erythroblasts than due to an exacerbated iron deficiency, as mean zinc-protoporphyrin decreased significantly and ferritin remained unchanged. No serious afamelanotide-related adverse events were observed for a total of 240 treatment years.
Conclusion:
Our findings point to a protective effect of afamelanotide on erythroblast maturation and protoporphyrin-induced liver injury.
Plain Language summary
Patients with erythropoietic protoporphyria (EPP), an inherited metabolic disease, suffer from light-induced skin burns and liver injury elicited by the accumulated light sensitizer protoporphyrin. The excess protoporphyrin is produced in red cell precursors in the bone marrow, and it is eliminated from the body via the liver and bile. A high protoporphyrin excretion burden damages the liver cells, the risk for this increases with higher protoporphyrin concentrations. About 20% of EPP patients show some sign of liver injury and 4% develop life-threatening liver dysfunction.
Afamelanotide, closely related to natural α-melanocyte stimulating hormone (MSH), induces skin tanning. This effect protects EPP patients from light-induced skin burns as shown in previous studies. We have treated Swiss EPP patients with afamelanotide since 2006, and we regularly perform safety tests of this treatment.
Recent in vitro and animal studies demonstrated α-MSH effects other than skin tanning, including an improved synthesis of red blood cell precursors in the bone-marrow and protection of the liver from experimentally induced damage. Until now, it is unknown whether afamelanotide has similar effects in the human organism.
To study this question, we analyzed retrospectively the safety laboratory data of 38 Swiss patients, who received at least one dose of afamelanotide from 2016 to 2019. We found that both, the average protoporphyrin concentrations and aspartate aminotransferase, a test for liver function, improved during afamelanotide treatment as compared to before.
We concluded that afamelanotide applied to EPP patients to protect them from light-induced skin burns also may reduce their risk of liver injury.
Keywords
Introduction
Erythropoietic protoporphyria (EPP) (OMIM 177000) is a genetic disorder affecting heme biosynthesis in erythropoiesis. 1 In most patients, it is caused by a deficiency of ferrochelatase, the last enzyme of the pathway, and in a minority by activating mutations in the gene coding for aminolevulinate synthase 2 (ALAS 2), the first and rate-limiting enzyme of this pathway. The latter form is called X-linked erythropoietic protoporphyria (XLEPP, OMIM 300752). During late erythropoiesis, both variants lead to an accumulation of metal-free erythrocyte protoporphyrin (PPIX) in the erythroblasts which is carried into the blood stream by the maturating reticulocytes. A portion of PPIX diffuses out from the erythrocytes, binds to albumin and to endothelial cells. Due to its chemical structure, PPIX is activated by visible light around 410 nm which penetrates the skin into the subpapillary capillaries. 2 This generates oxygen radicals leading to endothelial cell photo-damage through complement system activation and mast cell degranulation, culminating into exocytosis of vasoactive mediators and acute inflammation. Clinically very painful phototoxic skin burns result within minutes of light exposure.
EPP patients often show a slight microcytic anemia to which both a frequently concomitant iron-deficiency, the impaired heme production by the defective ferrochelatase, as well as activation of the integrated stress response factor eIF2α may contribute.3–6 Nearly 20% of EPP patients exhibit liver injury, and about 4% of them develop terminal liver damage, as the excessive PPIX is eliminated from blood plasma by the liver and excreted into the bile, which leads to a toxic bile composition and cholestatic liver damage.7–9 The risk of liver disease increases with increasing erythrocytic PPIX concentrations which varies from patient to patient.7,10,11
Until recently, no effective treatment for phototoxicity in EPP was available. 12 In a first phase-II study conducted in 2006, the subcutaneous implant of a slow release formulation of afamelanotide (Nle 4 -d-Phe7-α-MSH), a nonselective α-melanocyte stimulating hormone (MSH)-analog closely related to the natural hormone α-MSH, showed for the first time a clinical benefit in these patients 13 which was later confirmed by four additional phase-II and -III studies and three long-term observational studies.14–17 The results confirmed that the afamelanotide-treated EPP patients were able to expose themselves significantly longer to light without increasing their rate or severity of phototoxic reactions. 17 Up to now, the effectiveness of afamelanotide in EPP has been considered to be due to the induced skin pigmentation, the antioxidant activities, and the immunomodulatory effects in skin cells with a reduction of the deleterious effects of light-activated PPIX in the skin.14,17
α-MSH belongs to a group of endogenous hormones, called the melanocortins. Although they are best known for their effects on skin pigmentation and steroidogenesis, 18 they have known additional effects on moderating and modulating inflammation, promotion of antioxidant defense, DNA-repair and mitochondria biogenesis, inhibition of apoptosis, regulation of food intake, thermogenesis, and sexual arousal.19,20 The melanocortins all are derivatives of the common precursor polypeptide pro-opiomelanocortin (POMC) and beside α-melanocyte-stimulating hormone (MSH) include ACTH, β-MSH, and γ-MSH. They induce their hormonal activities by binding to the melanocortin receptors (MCRs) which exist in the five different forms MC1R–MC5R. ACTH binds to all five receptors, whereas the three MSH bind to MC1R, MC3R, MC4R, and MC5R with variable affinities. The MCR are G-protein-coupled receptors and their main signaling pathway includes cAMP (Figure 1). By activating protein kinase A (PKA) cAMP stimulates the nuclear transcription factors CREB, MITF, and PGC1α. The MCR signaling and its final targets are adjusted by a complex intracellular network. 21 This network includes endogenous MCR antagonists such as agouti-signaling protein and human-β-defensin-3, the by-stander protein β-arrestin-2 that limits signaling duration by receptor desensitization, negative feedback loops by PKA-activated phospho-diesterases limiting cAMP concentrations and crosstalks to other signaling pathways such as the MAPK kinase and the phosphoinositol-3 kinase pathway with effects on cell survival and proliferation. This diversity of actors finely tune the effects of receptor-mediated signaling to the requirements and conditions of specific cell types. 21

Simplified scheme of the intracellular signaling of the MC1R including the canonical c-AMP pathway, the crosstalk to other cellular signaling pathways and the postsignaling receptor desensitization. Upon ligand binding (and displacement of the inhibitors agouti-signaling protein (ASIP) or human beta defensin 3 (HBD3)) the α-MSH-MC1R complex activates the Gs protein, followed by Gsα-dependent stimulation of adenylyl cyclase (AC), increased cAMP levels and activation of protein kinase A (PKA). PKA-dependent phosphorylation induces activation of several transcription factors including CREB, MITF and PGC1α whereby these latter regulate melanogenesis, cell cycle control, cell survival, and metabolism. A negative feed-back by PKA-dependent activation of phospho-diesterases limits on the other side the cAMP concentration. Activation of the MC1R enables also a cAMP-independent transactivation of receptor tyrosine kinase cKIT with downstream activation of the AKT as part of the phosphoinositol-3 kinase (PI3K) pathway and consecutive nuclear transcriptional activation of cell cycle control and prosurvival genes. The crosstalk to the mitogen-activated protein kinases (MAPK) pathway with effects on nuclear transcription including genes related to control of proliferation and differentiation includes a G-protein independent part. This pathway includes active ERK1/2 that in turn activates MITF followed by its degradation. This pathway is also directly addressed by a PKA-dependent phosphorylation of the MAPK p38. PKA can also phosphorylate ataxia-telangiectasia-related protein (ATR), which activates a p53-dependent DNA-repair mechanism. In the post-signaling phase, the MC1R-bound Phosphatase-and-Tensin homolog (PTEN) limits the activity of AKT by dephosphorylation. The phosphorylated MC1R binds Beta-arrestin 2 (ARRB2), which guides the receptor desensitization by endocytosis. In the endosome, the receptor is either degraded or recycled from there back to the cell membrane. This diversity of actors and the balance of activating and inhibiting factors finely tune the effects of MC1R signaling to the requirements and conditions of the specific cells. For a more in-depth description see Herraiz et al. 21 and the references therein.
Among all five MCR signaling pathways, the MC1R and its signaling in melanocytes or melanoma cells has been most extensively investigated. However, many other cells including keratinocytes, fibroblasts, leukocytes, endothelial cells, hepatocytes, erythroblasts and neurons also express MCR, although to a much lower degree. The signaling pathways in the different cell types differ. 21 This may indicate that MCR signaling in general has much broader and diverse physiological functions than appreciated so far. During erythroblast development for instance, all five MCR are sequentially expressed and activated by their appropriate ligands for the successful maturation of erythroblasts to reticulocytes. 22 Likewise, all five MCR are expressed in different cell types of the liver, including the hepatocytes, Kupffer cells and most likely also cells lining the bile ductuli and canaliculi (i.e. CK19-positive cells), and their expression is upregulated during acute-phase responses. 23 In animal models, α-MSH or afamelanotide have protective effects during liver injury provoked by lipopolysaccharides, thioacetamide, or carbon-tetrachloride and during regeneration after partial hepatectomy.23–26 Thus, isolated liver cells treated with the endogenous melanocortin agonist α-MSH or the MC4R-selective agonist THIQ inhibited the endotoxin-induced upregulation of the acute-phase cytokines interleukin (IL)-6, IL-1β, and tumor necrosis factor alpha (TNF-α) in Kupffer cells and of chemokine gene expression in isolated hepatocytes. The impact of a nonselective MCR agonist in the intact human organism on distressed erythroblast maturation or injured liver cells is currently unknown.
Based on the aforementioned in vitro effects of various MCR signaling, we hypothesized an additional impact on erythroblast maturation and liver cell protection by a the nonselective MCR agonist afamelanotide in EPP. With this objective, we retrospectively analyzed our safety data of long-term treatment with afamelanotide in our cohort of EPP patients.
Methods
In total, 47 Swiss resident patients with EPP (including two patients with XLEPP) were treated with afamelanotide since 2006 at our outpatient clinic covering 254 patient treatment years and 330 patient observation years. The number of patients treated with afamelanotide increased with time as outlined in our previous publication 15 (Figure 2). For the current analysis, we included only those patients who were treated with at least one implant of 16 mg afamelanotide during an observation period between January 2016 and December 2018, in whom we had laboratory findings before and during treatment and who gave a written informed consent to the study (n = 38). Additional information of this study group is available. 17
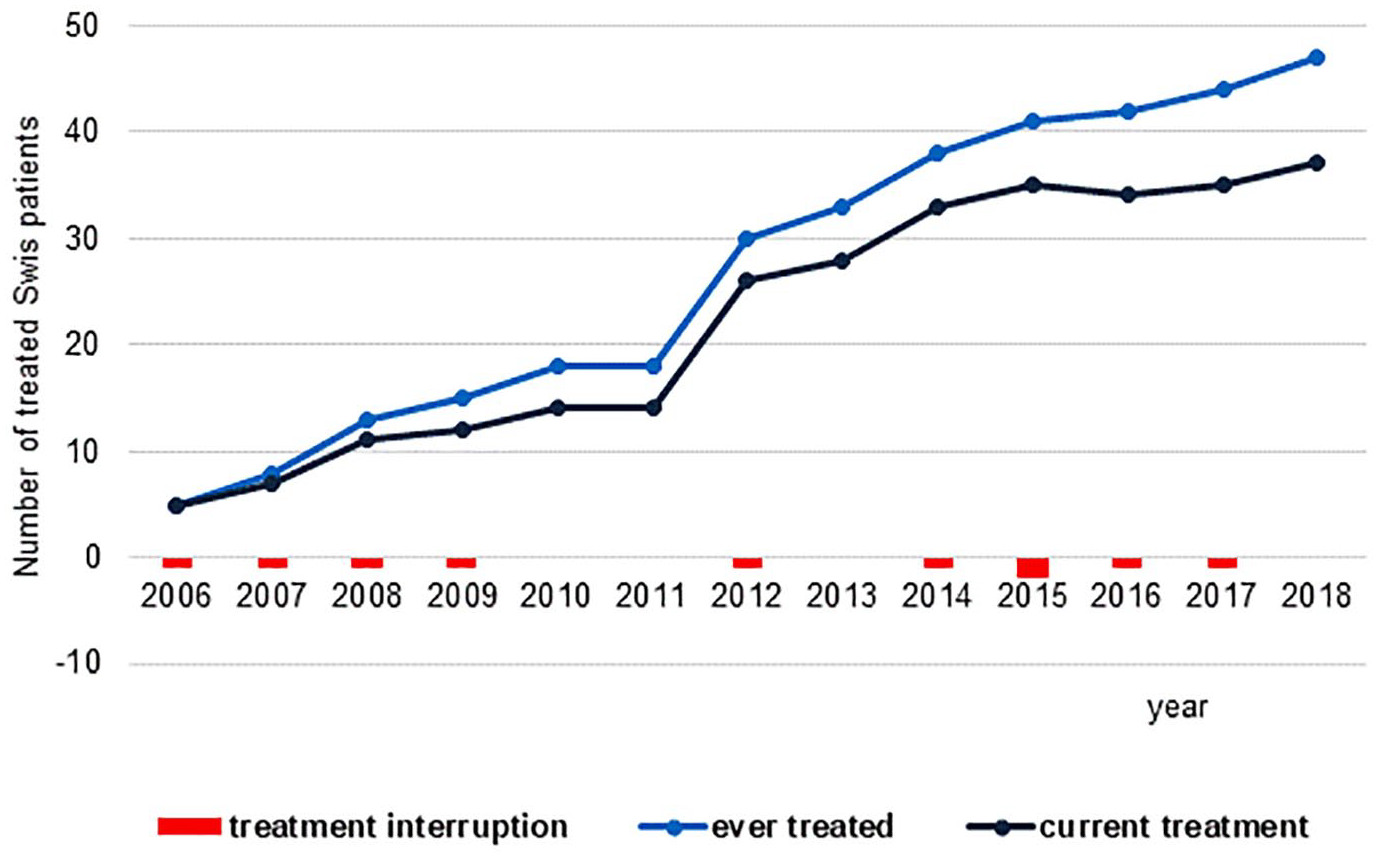
Swiss patients with EPP treated with afamelanotide between 2006 and 2018. Causes for treatment interruption: Personal decision (n = 2); pregnancy (n = 2); adverse events (AE)/co-morbidity not related to afamelanotide treatment (n = 3); lack of reimbursement (n = 1), financial restriction (n = 1); moved out of Switzerland (n = 1). No serious adverse events (SAE) or suspected unexpected serious adverse reaction (SUSAR) recorded.
The study was approved by the ethical review board of the Canton Zurich, Switzerland (BASEC 2018-80075). The data were analyzed by AEM, JB, and EIM and all authors had access to the primary clinical data.
We retrospectively analyzed laboratory safety data, which were collected from the clinical notes (including all data available from March 1993 until December 2018, 4972 in total, 1316 before and 3656 during treatment). The methods of laboratory tests were standard medical procedures and standard automated tests on the Hitachi 911 and Cobas 6000 platforms by Roche Diagnostics for chemistry and Beckman Coulter and Sysmex for hematology. The accredited laboratory ISO-Norms were 17,025 and 15,189. eGFR was calculated using age and serum-creatinine concentrations based on the CKD-EPI formula as recommended by the National Kidney foundation (accessed 16. Feb. 2020). PPIX and ZnP were assayed and quantitated by HPLC as published. 27
Prior to treatment, no standard procedures for laboratory tests were applied but chosen according to the treating physician’s judgment. During treatment with afamelanotide, complete blood count, protoporphyrin (PPIX, ZnP) in the erythrocytes, AST, and creatinine were monitored every 6 months. Other laboratory tests, such as ferritin and ALT, were only performed when suspecting an abnormality.
Statistical analyses
For the statistical analysis, the laboratory data of each patient were separated into two groups. The first included all results collected before and the second those during the treatment with afamelanotide. Mean values per patient of these laboratory test results were then calculated for those two groups.
Statistical tests (analyze-it for excel, Version 4.51) were applied and a two sided p-value of ⩽0.05 was considered as significant. In paired test results, the student’s t-test was applied, which was considered robust against violation of normality28,29 and was preferred for this analysis because it compares the mean values. In case of a violation of the normal distribution of the differences between the pairs, the Wilcoxon signed-rank test was applied as a sensitivity analysis in addition, resulting in p-values similar to the t-test. In unpaired samples the Wilcoxon–Mann–Whitney U-test was used. Normality was tested by the Shapiro–Wilk test. Correlations between tests were calculated using Spearman’s rank correlation. Retrospective power calculation was performed using Stata version 15.
Results
Patients
Among the 38 Swiss patients, no treatment interruption due to adverse events was required, and we observed no afamelanotide-related serious adverse events. Noteworthy, none of the patients developed precancerous or malignant skin lesions. The median of treatment duration calculated till the end of the study in December 2018 was 6.23 years (interquartile range (IQR): 4.08–9.28, maximum: 12). The 38 patients accumulated a total of 240 treatment years and a total of 254 observation years. Twenty-five of the 38 patients were females, 13 were males. The mean age was 44 years (range: 22–80 years) by December 2018. For this sample size, the retrospective analysis resulted in a power >0.8 for a difference of means of 3 µmol/L protoporphyrin, which was considered as clinically significant.
Safety analyses included hematology, kidney, and liver function tests, as well as measurements of PPIX and ZnP.
Protoporphyrin in the erythrocytes (Table 1, Figure 3):
We observed a highly significant decrease of mean PPIX during afamelanotide treatment (Figure 3(a)). Twenty-nine of the 38 patients had lower values during treatment compared to that before treatment. The patients with higher initial values tended to have a more pronounced decrease in PPIX than those with lower initial values. In the remaining nine patients, a slight increase of PPIX of less than 5 µmol/L was observed. No gender differences of PPIX concentrations were noted before or during afamelanotide treatment. The exclusion of the data from the XLEPP patients only marginally changed the data (see Figure 3(b)).
Laboratory results collected before and during afamelanotide treatment.

AST, aspartate aminotransferase; eGFR, estimated glomerular filtration rate; Hb, hemoglobin; PPIX, protoporphyrin; RDW, red blood cell distribution width; MCH, mean corpuscular hemoglobin, MCV, mean corpuscular volume, CKD-EPI, chronic kidney disease epidemiology collaboration, IU/L, international units per liter; SD, standard deviation; XLEPP, X-linked erythropoietic protoporphyria.
Conversion factor µmol/L × 56.18 = µg/dL.
Conversion factor µmol/L × 62.5 = µg/dL.
Number of patients with eGFR <90/<60 mL/min*1.73 m2.
The bold values represent the p values and thus indicate the significance.
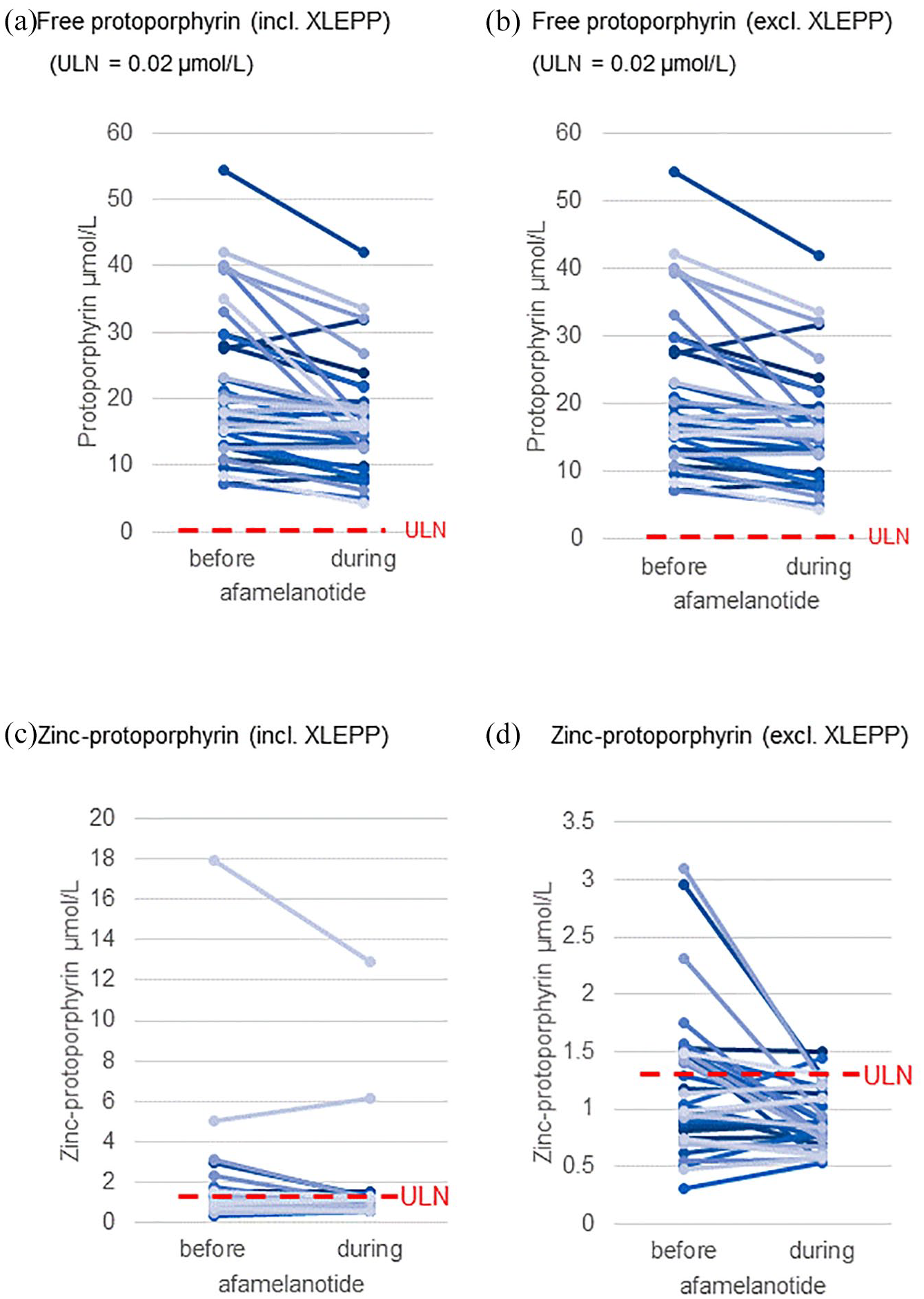
Porphyrin values. Each line represents one patient: (a) free protoporphyrin (PPIX) incl. XLEPP, (b) free protoporphyrin (PPIX) excl. XLEPP, (c) zinc protoporphyrin (ZnP) incl. XLEPP, and (d) zinc protoporphyrin (ZnP) excl. XLEPP. ULN, upper limit of normal.
In contrast to EPP, patients with XLEPP showed not only an elevated PPIX, but also a substantially elevated ZnP, as previously published. 30 Figure 3(c) shows an overall decrease in the mean ZnP in the entire cohort during treatment. After excluding two XLEPP patients, changes in the mean ZnP among the remaining EPP patients (n = 36) was more significant statistically (Figure 3(d), Table 1).
Blood chemistry (Table 1, Figures 4 and 5):
As depicted in Figure 4(a) and (b), the liver transaminase AST values decreased significantly during treatment (p = 0.0146), irrespective of the type of protoporphyria. Three (7.9%) patients had increased AST values before treatment, none of them suffered from XLEPP, which is known for a high risk of liver damage (Figure 4(b)). Of those patients with increased values prior to treatment, the patient with the highest value had a 32% decrease in AST to the initial value and resulted in a value of less than two times the upper limit of normal. In the two other patients, the AST values dropped back to the normal range during treatment. Fourteen of 38 patients showed an increase in the AST concentrations, whereby the increase was always ⩽4 IU/L. Twenty-four of the 38 patients showed a decrease of AST, whereby two patients had a decrease of more than 20 IU/L, two between 10 and 20 IU/L, and six between 5 and 10 IU/L. AST values did not differ between the genders.

Liver function. Each line or point represents one patient. (a) AST (incl. XLEPP) before and during treatment and (b) correlation of PPIX and AST (circles before treatment, triangles during treatment). AST, aspartate aminotransferase; PPIX, protoporphyrin; ULN, upper limit of normal.
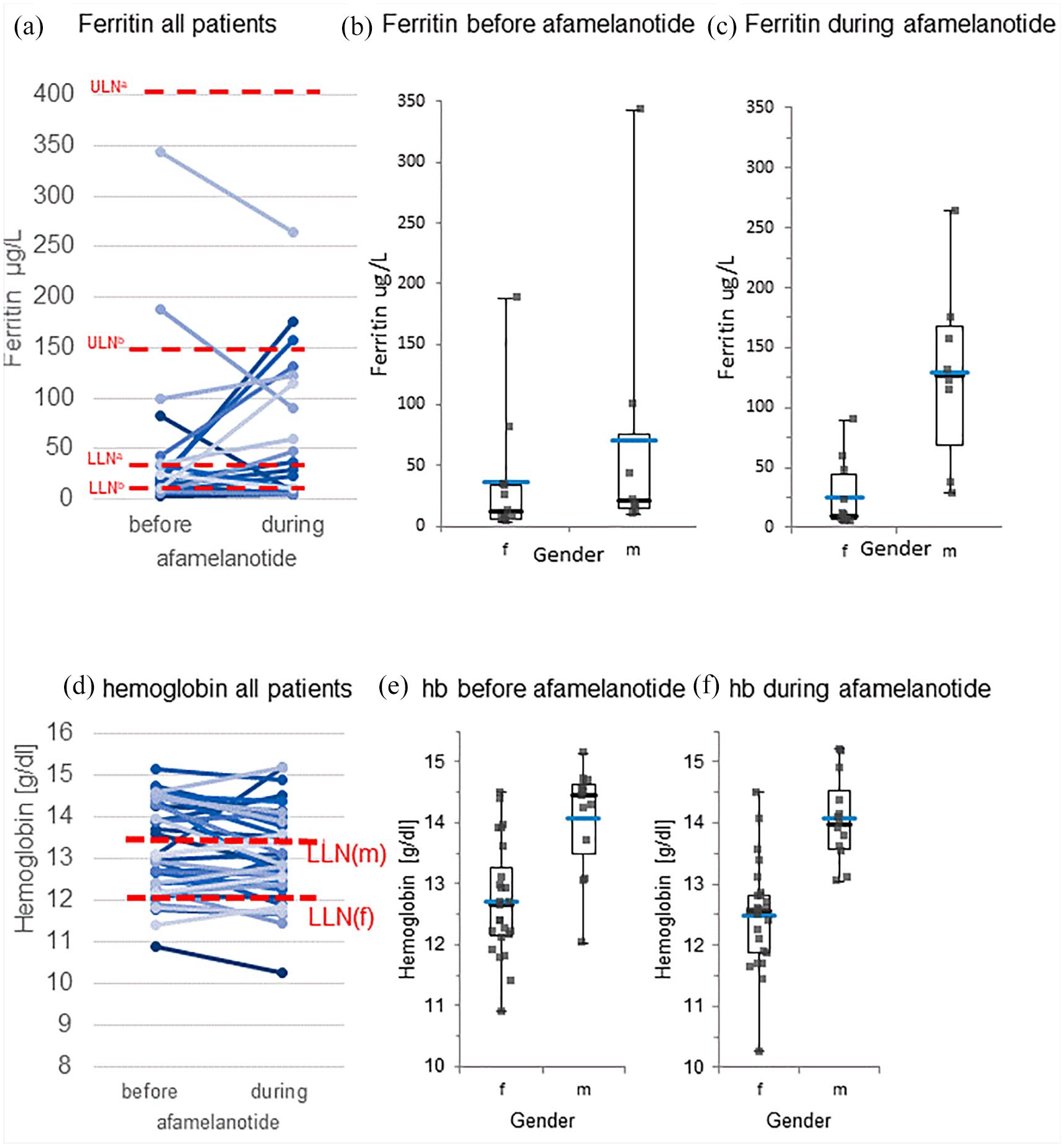
Ferritin and hemoglobin. Each line or point represents one patient: (a) ferritin before and during treatment, (b) ferritin before treatment according to gender, box plot representing IQR, black line median, blue line mean, (c) ferritin during treatment according to gender, box plot representing IQR, black line median, blue line mean, (d) hemoglobin before and during afamelanotide, (e) hemoglobin before treatment according to gender, box plot representing IQR, black line median, blue line mean, and (f) hemoglobin during treatment according to gender, box plot representing IQR, black line median, blue line mean. a, male or postmenopausal female; b, premenopausal female; f, female; m, male; LLN, lower limit of normal; ULN, upper limit of normal.
AST correlated significantly with PPIX concentrations both before and during treatment: Spearman’s rank correlation ρ = 0.373, p = 0.0209 and ρ = 0.513, p = 0.0010, respectively (n = 38, Figure 4(c)). These statistical data make obvious that the relation of PPIX and AST persists and is even reinforced under treatment. The graph illustrates that the data points before and during treatment cluster together. Indeed, if a linear regression is calculated, assuming PPIX as independent and AST as dependent variable, the slopes are similar (r = 0.349 before and r = 0.483 during afamelanotide).
ALT was measured only in half of our patients and only intermittently in suspicion of liver injury and therefore likely is subject to sampling bias. The values did not change significantly during treatment.
The creatinine values were significantly different between the genders, as expected, and were not influenced by the treatment (Table 1). The estimated glomerular filtration rate (eGFR), however, decreased significantly under treatment (p = .0008). The eGFR during treatment linearly correlated with the eGFR before treatment (p < .0001). We further calculated the change of eGFR over time. The yearly decrease in eGFR (0.95 mL/min) was comparable to the age-dependent decrease in normal populations. 31
A considerable portion (63%) of study participants had ferritin values below the lower limit of normal, in premenopausal women even 71% (Figure 5(a)). We found no significant change during treatment in the whole group (p = .279). There was also no significant difference between the genders before treatment (Figure 5(b), p = .238). During treatment, though, we observed a significant increase of ferritin in men compared to women (Figure 5(c), p = .008). Overall, the number of patients with values outside the reference range changed from 13 to 8 (Table 1).
Hematology (Table 1, Figures 5 and 6)
Mean hemoglobin (Hb) values before and during treatment remained stable (Figure 5(d)). As expected, we found significantly lower mean Hb values in females than in males both before and during the treatment with afamelanotide (p < .0001, p < .0001; Figure 5(e) and (f)).
The mean red blood cell distribution width (RDW) was above the upper limit of normal before treatment and further increased significantly during treatment (Figure 6(a)). Also, the mean percentage of microcytes and hypochromic red blood cells were above the upper limit of normal prior to treatment. The percentage of hypochromic cells further increased significantly with treatment (Figure 6(b)), while the percentage of microcytes remained unchanged. Although the mean percentage of hyperchromic cells was within the reference range, it decreased within the normal range under treatment, whereas the percentage of macrocytes remained unchanged. The leukocyte count was normal before treatment and remained unchanged throughout the treatment. The mean count of thrombocytes was within the normal range and decreased slightly under treatment, although the lowest value increased from 101 to 114 ×103/µL.

Additional hematological values. Each line or point represents one patient: (a) red blood cell distribution width (RDW) before and during treatment, (b) percentage of hypochromic cells before and during treatment, and (c) correlation of PPIX with red cell distribution width. ULN, upper limit of normal.
RDW increased significantly with increasing PPIX concentrations both before and during treatment (Spearman’s ρ = .483, p = .0022 and ρ = .424, p = .0080, respectively; n = 38, Figure 6(c)). Also, the percentage of microcytes correlated positively with PPIX concentrations before and during treatment (Spearman’s ρ = .350, p = .0498, and ρ = .390, p = .0272, respectively; n = 32, data not shown).
Discussion
The MCR agonist afamelanotide is the only approved symptomatic therapy for EPP.12,32 It functions through its effect on skin pigmentation, and its anti-oxidative and anti-inflammatory properties. As this peptide is closely related to the natural hormone α-MSH, it binds orthosterically to the MCR. 33 In contrast to the signaling induced by allosteric agonists, orthosteric agonist signaling is controlled by physiological processes, which might reduce the risk of long-term adverse effects.21,33 EPP requires a lifelong treatment as long as its genetic condition is not normalized by therapeutic means, such as bone marrow transplantation. Therefore, the long-term safety of any treatment is essential. In our cohort, laboratory tests to monitor the safety of afamelanotide for a total duration of 240 treatment years did not show any undesired development in the parameters. The only exception was a slight reduction in eGFR, which likely is explained by the natural aging process. Thus, this observational study demonstrates that afamelanotide is a safe drug for long-term treatment of EPP for up to 12 years.
In our cohort, we observed some specific hematological abnormalities prior to treatment with afamelanotide in the EPP patients, which are reported here for the first time. These include a broader than normal RDW, most likely due to a concomitant higher than normal percentage of microcytes, as well as an elevated percentage of hypochromic cells. Also, EPP patients frequently show signs of iron deficiency with ferritin values in the lower range of normal or even below the normal range,3,4 which was confirmed by this study. However, our data show that irrespective of treatment, the ZnP concentrations, an indicator for iron deficiency, 34 did not correlate with RDW, % microcytes or % hypochromic cells (Spearman’s rank correlation all nonsignificant), which renders iron deficiency a very unlikely cause of those abnormalities. However, an upregulated heme-related inhibitor (HRI), as described in the erythroblasts of an EPP mouse model, would explain our findings 6 (see Figure 7). HRI is predominantly expressed in the erythropoietic tissue and balances the production of heme and globin chains and protects the erythroblasts from oxidative damage. 35 HRI is one of four kinases activated as an integrated stress response in vertebrates. 36 Heme deficiency, as well as other stressors, 37 cause its activation by phosphorylation. The phosphorylated HRI inactivates the eukaryotic translation initiation factor 2 (EIF2) through phosphorylation. This consecutively down-regulates the production of RNA and, thus, reduces protein synthesis, including the synthesis of globin. In the erythroblasts, this results in microcytosis35,38,39 and hypochromia. According to our data, PPIX may well be one of those stressors causing an activation of HRI. Indeed, we found a correlation between PPIX concentrations and both RDW and microcytosis, but no correlation between hemoglobin and RDW or microcytosis. In addition, the phosphorylated EIF2 activates the expression of stress response genes, such as ATF4. 39 ATF4 inhibits mTORC1 signaling, which results in antiapoptosis and reduces ineffective erythropoiesis but induces hypochromia and microcytosis. 39

A model of pathophysiological processes in erythropoiesis in EPP and proposed effects of afamelanotide on the maturating erythroblast carrying an EPP-metabolic defect. The erythropoietin receptor is expressed and can be activated by erythropoietin (EPO) during the whole maturation process from basophile to orthochromatic erythroblasts. In the basophile erythroblast, melanocortin 2 receptor (MC2R) is expressed and activated exclusively by ACTH. It induces the activation of MAPK (mitogen-activated protein kinase) and ERK (extracellular signal-related kinase) by phosphorylation leading to the proliferation of erythroblasts. 22 During the maturation process, the erythroblasts become polychromatic and melanocortin 1 receptor (MC1R) is expressed. The combined EPO-receptor and MC1R signaling activates JAK2 (Janus kinase 2) and, consecutively, STAT5 (Signal transducer and activator of transcription 5). This in turn suppresses ALAS2 activity by an increased expression of iron-responsive element-binding protein 2 (IRP2). As ALAS2 is the rate-limiting enzyme for heme synthesis pathway, its inhibition likely reduces heme synthesis. Therefore, excess PPIX formation may be reduced. Also, hypochromia may increase due to reduced synthesis of the end product heme. Moreover, STAT5 induces the expression of transferrin receptor 1 (TR1), which in turn may contribute to improved intracellular iron availability and therefore could reduce the ZnP formation. In addition, STAT5 causes antiapoptosis by increasing the expression of antiapoptotic factors Bcl-xL (BCL2-related gene) and MCL1 (myeloid cell leukemia sequence 1). The further maturation of the erythroblast leads to the formation of an orthochromatic cell that expresses the melanocortin 5 receptor (MC5R), which is directly involved in the enucleation process leading to the formation of reticulocytes. The heme related inhibitor (HRI) is progressively expressed throughout the whole maturation process of erythroblasts. 40 In heme deficiency or by other stressors it is activated by phosphorylation. 35 The phosphorylated HRI inactivates the eukaryotic translation initiation factor 2 (EIF2) by phosphorylation. This leads to a reduced RNA synthesis and consecutively reduced protein synthesis, including the synthesis of globin, resulting in microcytosis and hypochromia. Also, the phosphorylated EIF2 induces the expression of stress-related genes, such as ATF4. 39 In addition, ATF4 inhibits mTORC1 signaling, which causes antiapoptosis and therefore, reduces stress-related ineffective erythropoiesis. The other two MCRs (MC3R and MC4R) are not known to have an impact on erythroblast maturation.
Liver injury is a life-threatening complication of EPP. Thereby, a vicious cycle between increased protoporphyrin concentrations and liver injury exists: Hepatic elimination of a high protoporphyrin burden leads to a toxic bile composition, as the biliary route is the only elimination route for the excess protoporphyrin.7,9 The toxic bile composition results in damage to bile ductules and in cholestasis, which further reduces elimination of the excess protoporphyrin by the bile and leads to a further increase in blood protoporphyrin concentration. In our cohort, only 7.9% of the patients exhibited increased liver enzyme levels before treatment with afamelanotide, which is lower than that published for other cohorts. 41 The regularly performed clinical visits with the advice to avoid hepatotoxins including alcohol and hepatotoxic drugs, as well as the avoidance of iron substitution 42 may explain this relatively low percentage of patients with signs of liver injury in our cohort. During treatment with afamelanotide, the mean AST decreased from 26.7 to 22.9 IU/L, with the highest values decreasing most. The proportion of patients with increased AST as a sign of liver injury decreased to 1/38 (2.6%) and even within normal ranges the majority of patients showed a decrease in their AST value. We also found a decrease in the PPIX concentrations under treatment. In fact, five out of 11 patients with PPIX values >20 µmol/L before treatment showed a decrease below 20 µmol/l, which has been considered as a threshold for liver damage. 10 As mentioned above, in our EPP population AST and PPIX concentrations correlated significantly both before and during treatment. This observed correlation of AST with PPIX suggests that the PPIX concentrations may be a biomarker for the risk of liver injury, as suggested previously,7,10,11 and a treatment-induced reduction of PPIX may reduce AST concentrations. As can be seen in Figure 4(a), those patients with increased AST concentrations before treatment, however, exhibited a more pronounced decrease of AST than the rest of the cohort. This suggests that there may not only be an indirect effect of afamelanotide on AST due to the reduction of PPIX levels, but there may be an additional direct effect of afamelanotide on liver protection. Such an interpretation is supported by the observation in a rat model of acute liver injury showing an upregulated expression of all five MCR in the liver tissue. Their activation by an agonist has cytoprotective effects by inhibiting pro-inflammatory chemokines and cytokines. 23
The limitation of this study lies in its retrospective observational nature. However, the fact that the results were able to detect the slight, expected, age-related reduction in eGFR suggests the reliability of our study data despite its design being of retrospective and observational nature. Nevertheless, we considered other potential reasons for a decrease in PPIX apart from afamelanotide. First, technical biases are always possible; however, a technical bias is unlikely to cause a decrease in PPIX for several reasons: (a) a time-dependent shift in the analytical technique is implausible, as the patients were treated sequentially and, therefore, on a given day samples from different patients at different treatment stages were analyzed. (b) To minimize inter-assay variation, PPIX standards used for the quantification were controlled by absorbance measurements immediately before each assay. (c) The extent of the treatment-related PPIX change was independent of the date of the first dose of afamelanotide.
Second, a reduced iron availability inhibiting the rate-limiting enzyme ALAS2 could be another reason for a decreasing PPIX. 42 However, we found no evidence to support a decrease in iron availability during treatment, as the concentration of ferritin as well as the percentage of microcytes, MCV and MCH remained unaffected. We only observed an increase in the percentage of hypochromic cells. However, ZnP decreased during treatment. ZnP is a well-established indicator for iron availability in the erythropoietic tissue, with increased levels pointing to reduced iron availability. 34 This suggests an improved rather than a reduced intracellular iron availability in the heme synthesizing erythroblasts. As XLEPP is characterized by a substantial increase of ZnP, 30 and the XLEPP patients were also closely monitored for iron deficiency and, if indicated, supplemented with iron in order to prevent hepatic complications, 43 their data may confound the results of ZnP in our cohort. Thus, we excluded the two XLEPP patients in a second analysis, which resulted in an even more significant reduction of ZnP in the treated EPP patients. Therefore, we found no signs for exacerbated iron deficiency as a reason for a decrease in PPIX under treatment.
Third, a longer and more intense light exposure leading to PPIX destruction by photobleaching 44 may contribute, as previously published data indicate that EPP patients during treatment expose themselves to a more intensive light for a longer period of time than before treatment.16,17 However, to our knowledge it is unknown, whether the phenomenon ‘photobleaching’ has any detectable effect in EPP patients in vivo.
Hence, we propose another hypothetical explanation for the decrease of PPIX under treatment and suggest that afamelanotide itself could have induced the PPIX decrease and the observed hematological changes, the increase in RDW and hypochromia, via specific MCR (see Figure 7). Indeed, Simamura et al. showed that the human umbilical erythroblasts express all five MCR in a consecutive time order during maturation. 22 Their activation by specific ligands is required for the normal maturation process. Like α-MSH, but with a higher affinity and prolonged half-life, afamelanotide binds to and activates 4 of the 5 MCR, as stated before. The activation of MC1R leads to an activation of JAK2 and, consecutively, STAT5, which in turn induces antiapoptosis by increasing the antiapoptotic factors Bcl-xL and MCL1.22,45 Antiapoptosis may well reduce an ineffective erythropoiesis and, thus, the efflux of accumulated PPIX from apoptotic erythroblasts in EPP. Also STAT5 stimulates the expression of the iron-responsive element binding protein 2 (IRP2), which in turn inhibits ALAS2-mRNA translation into active enzyme.42,46 As ALAS2 is the rate-limiting step in the erythropoietic heme synthesis, a reduction of ALAS2 activity leads to a decreased accumulation of excess PPIX in these maturating erythroblasts. 47 STAT5 also increases the expression of transferrin receptor 1 (TR1), which likely leads to increased rate of iron import into erythroblasts. 46 The imported iron is used in the final step in heme synthesis, the chelation of iron into protoporphyrin in order to form heme, 48 whereas ZnP is formed in case of iron deficiency. 49 The observed decrease in ZnP without a change of ferritin during afamelanotide treatment supports an improved iron uptake into the erythroblasts by the increased expression of TR1, which may be considered as an improved utilization of the systemically available iron. Finally, the MC5R is directly involved in the enucleation process 22 and, hence, leads to the final maturation of erythroblasts to reticulocytes.
These suggested multiple processes under afamelanotide treatment leading to an improvement of the metabolically stressed erythropoiesis including antiapoptosis, improved iron utilization, reduction of PPIX formation, and improved erythroblast maturation – as illustrated in Figure 7 – likely result in a reduced production of excess PPIX and, therefore, supports the hypothesis of a causative treatment effect of afamelanotide on the metabolic disturbances in EPP. Indeed, the ability of the MSHs to restore homeostasis in organisms exposed to a range of heterogeneous distresses has been emphasized recently. 20
Conclusion
To conclude, we found a reduction of AST as a marker of liver injury in our cohort of EPP patients during treatment with the nonselective MCR agonist afamelanotide. Moreover, our data suggest a positive treatment effect of afamelanotide on erythropoiesis, iron utilization, and the PPIX levels. No adverse effects of long-term treatment with afamelanotide on safety laboratory tests were observed under a long-term treatment of EPP for up to 12 years.
Footnotes
Acknowledgements
The authors thank all the patients willing to share their data with them.
Authors’ contributions
Conceptualization: AEM, JB, and EIM.
Methodology: AEM, JB, and EIM.
Formal analysis: AEM, JB, EIM, and CEM.
Investigation: AEM, JB, EIM, MS, HZ, CEM, and XS.
Writing – original draft preparation: AEM.
Writing – review and editing: MS, JB, AEM, EIM, HZ, CEM, and XS.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AEM received an unrestricted research grant by Clinuvel Pharmaceuticals. EIM is a consultant to Alnylam and Clinuvel. The other authors declare no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
Ethics statement
The study was approved by the ethical review board of the Canton Zurich, Switzerland (BASEC 2018-80075). All patients provided written informed consent.
