Abstract
Ebstein’s anomaly is an infrequent congenital malformation of the heart, in which the septal and posterior leaflets of the tricuspid valve are displaced to the apex, leading to partial “atrialization” of the right ventricle. Its clinical presentation varies widely, contingent on its severity and any co-occurring cardiac anomalies. Although most cases are identified in childhood, some individuals remain asymptomatic and may not be diagnosed until later in life. Conversely, rheumatic mitral valve disease represents one of the major causes of acquired cardiac disorders among children and young adults, particularly in developing regions such as India. Despite a significant decline in rheumatic heart disease incidence in India recently, it still poses a major public health challenge. Existing rheumatic heart disease studies report an average prevalence of 0.5 per 1,000 children aged 5-15 years. This report details the unusual co-occurrence of Ebstein’s anomaly and mitral valve stenosis rheumatic in origin in a 65-year-old man, a rare finding in an unoperated patient of this age.
Introduction
Ebstein’s anomaly (EA) is an uncommon congenital malformation of the tricuspid valve characterized by marked heterogeneity in clinical presentation. 1 It constitutes nearly 1% of all congenital cardiac anomalies, with an estimated occurrence of 1 case per 200,000 live-born infants. 2 The hallmark diagnostic criterion of EA is the downward (apical) displacement of the septal leaflet of the tricuspid valve measuring at least 8 mm/m2 of body surface area, referenced from the insertion point of the anterior mitral leaflet. The coexistence of rheumatic heart disease and EA is extremely rare.3, 4 To the best of our knowledge, this distinct combination of cardiac pathologies has not been previously reported in an elderly patient. In this article, we present the case of a 65-year-old man diagnosed with concurrent EA and rheumatic mitral valve stenosis.
Case Report
A 65-year-old male patient presented with a 5-year history of dyspnea (NYHA III), orthopnea associated with pedal edema, and intermittent palpitations. He denied any other significant medical history, and there was no relevant personal or family cardiac history. On physical examination, he was tachycardic with an irregularly irregular pulse of 92 beats per min. His blood pressure was 104/70 mmHg, and his jugular venous pressure was elevated. Examination revealed pedal edema; however, cyanosis and clubbing were not observed. The apical impulse was noted as tapping, located in the 5th intercostal space, 2 cm lateral to the midclavicular line. Hepatomegaly was noted, with the liver edge palpable 4 cm below the right costal margin in the midclavicular line. Auscultation revealed a loud S1 and a wide, variable S2 split with a normal pulmonary component. A rough, rumbling mid-diastolic murmur with a presystolic component was audible in the mitral area, accompanied by a Grade 2/6 pansystolic murmur in the tricuspid area. A chest X-ray demonstrated marked cardiomegaly. The electrocardiogram (ECG) indicated atrial fibrillation (AF), right ventricular hypertrophy, and a right bundle branch block (Figure 1).
Electrocardiogram (ECG) Suggestive of Atrial Fibrillation (AF), Right Ventricular Hypertrophy (RVH), and Right Bundle Branch Block.
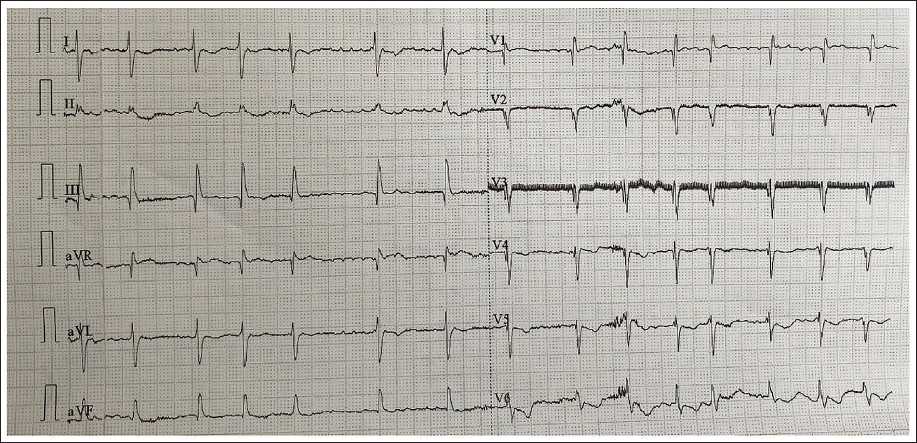
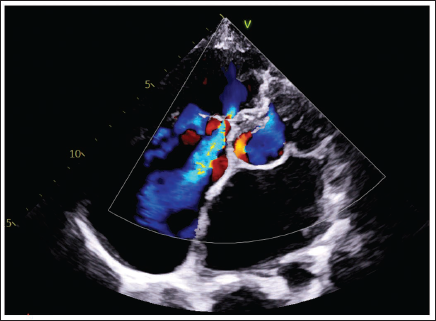
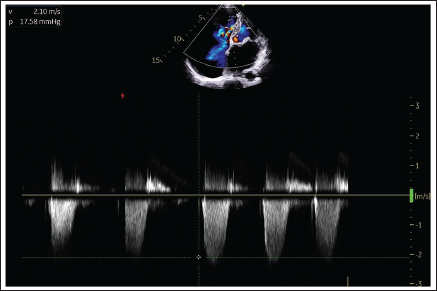
Two-dimensional echocardiography, utilizing the apical four-chamber view, demonstrated that apically displaced septal leaflet of tricuspid valve is around 43 mm, along with a prominent atrialized right ventricle (Figure 2), findings consistent with EA. Both the interatrial and interventricular septa were intact, and the ejection fraction was 60%. The mitral leaflets appeared thickened, exhibiting restricted posterior mitral leaflet movement and diastolic doming of the anterior mitral leaflet, though no mitral regurgitation was present (Figure 3). Planimetry measured the mitral valve orifice area at 0.7 cm2, with a mean mitral valve gradient of 16 mmHg, indicating severe stenosis (Figure 4). Additionally, mild aortic and pulmonary valve regurgitation were noted. Color Doppler across the tricuspid valve showed severe tricuspid regurgitation with a pressure gradient of 17.58 mmHg (Figures 5 and 6).
Two-dimensional Echocardiography (2D-ECHO) Shows a Giant Right Atrium, Atrialization of the Right Ventricle, Apical Displaced Septal Tricuspid Leaflet by 43 mm.
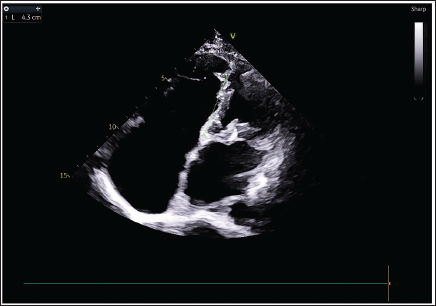
Transthoracic Echocardiogram PLAX View Shows Thickening and Doming of the Anterior Mitral Leaflet (AML) and Fixed Posterior Mitral Valve Leaflet (PML), Suggestive of Rheumatic Involvement.
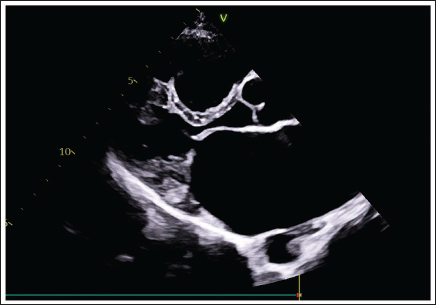
Planimetric Mitral Valve Area—0.7 cm2.
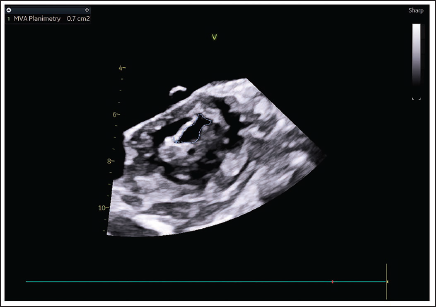
A4C View Shows Severe Tricuspid Regurgitation.
Continuous Wave Doppler Estimation of Gradient Across the Tricuspid Regurgitation Jet.
The patient received conservative medical therapy for AF and heart failure, along with prophylactic anticoagulation for thromboembolic risk reduction. Over 4 days of close observation and treatment, his condition improved, and vital signs normalized. He was discharged on furosemide, metoprolol, digoxin, and oral anticoagulants. During a 2-week follow-up, surgical treatment options were discussed, but the patient preferred to continue with medical management only, citing financial constraints. He remains on his prescribed medications and attends regular monthly follow-up appointments at our hospital. His condition has neither improved nor deteriorated.
Discussion
Ebstein’s anomaly is a rare congenital cardiac abnormality in which the septal and posterior tricuspid valve leaflets are abnormally adherent to the myocardium, resulting in downward (apical) displacement of the functional tricuspid annulus within the right ventricle.1, 2 Consequently, a portion of the right ventricle becomes “atrialized” and often exhibits varying degrees of hypertrophy and wall thinning.1, 2 The impact can be elaborated in the given subcategaries.
Diagnostic Criteria and Physiological Impact
A defining diagnostic criterion for EA is apical displacement of the septal leaflet of the tricuspid valve by at least 8 mm/m2 of body surface area when measured from the anterior mitral leaflet insertion point. 2 This structural anomaly leads to a reduced functional right ventricle and concomitant dilation of the right atrium. These changes frequently predispose patients to both atrial and ventricular arrhythmias.
Clinical Presentation and Long-term Outcomes
While EA is present from birth, most isolated cases are typically diagnosed and present in early childhood. 5 However, as observed in our patient, it can, in rare instances, remain asymptomatic until adulthood. The prognosis of untreated EA is generally poor, with fewer than 5% of patients surviving beyond the age of 50 years in the absence of surgical intervention. 6
Associated Abnormalities and Disease Progression
Reported mitral valve abnormalities associated with EA are diverse, encompassing congenital valvular stenosis, parachute mitral valve, mitral valve prolapse, supravalvular rings, and cleft mitral leaflets. 7 These are often found in conjunction with other congenital anomalies such as endocardial cushion defects and coarctation of the aorta. 7 The presence of an acquired rheumatic mitral valve disease in association with EA is highly unusual. This particular combination, however, can significantly alter the disease course, leading to rapid progression of symptoms, including heart failure, pulmonary hypertension, and AF.3, 4
Therapeutic Considerations
Therapeutic options for EA are limited, with medical management focused mainly on arrhythmia suppression and heart failure control. We have observed that repairable tricuspid valves are uncommon, particularly when marked tricuspid regurgitation is present. When repair is possible, an anterior leaflet-based approach is favored over valve replacement to minimize prosthetic valve-related complications and the need for anticoagulation.
Unique Case Presentation
The present case is noteworthy, as the patient manifested symptoms of arrhythmia and heart failure in his sixth decade of life. This specific combination of rare congenital and acquired diseases, coupled with survival into late adulthood without prior surgical intervention, represents a unique presentation not previously reported in the literature, to the best of our knowledge.
Learning Points
Rarity and epidemiology: EA of the tricuspid valve represents an exceptionally uncommon congenital cardiac defect, with an estimated incidence of approximately one per 200,000 live births.
Unique association: The concurrent presence of acquired rheumatic mitral stenosis alongside EA is exceedingly rare and has been infrequently documented in medical literature.
Clinical implications: This unusual coexistence may accelerate the clinical progression of EA, leading to earlier manifestation of serious complications such as AF, pulmonary hypertension, and congestive heart failure.
Management considerations: Surgical intervention should be contemplated in patients demonstrating clinical decline, as indicated by progressive right-sided cardiac enlargement, recurrent paradoxical embolic events, cyanosis, right heart failure, arrhythmias, or worsening functional symptoms.
Footnotes
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
The authors confirm that all required patient consent forms have been obtained. The patients have provided explicit consent for the publication of their clinical information and images in the journal. They understand that identifying details, including names and initials, will not be disclosed, and that every effort will be made to preserve anonymity; however, complete anonymity cannot be assured.
