Abstract
Atrial fibrillation is the most common sustained cardiac arrhythmia, affecting millions globally and significantly increasing the risk of stroke, heart failure, and mortality. Traditional detection methods often fail to identify paroxysmal or asymptomatic atrial fibrillation, leaving many patients undiagnosed until serious complications arise. The proliferation of consumer-grade wearable technology, particularly smartwatches and smartphones with advanced sensing capabilities, has created unprecedented opportunities for large-scale, cost-effective atrial fibrillation screening. This review examines the current evidence regarding the accuracy, feasibility, and clinical impact of smartphone and smartwatch-based atrial fibrillation detection technologies. We analyze various technologies, including smartphone-based electrocardiogram attachments, photoplethysmography applications, and smartwatch algorithms, evaluating their diagnostic performance, implementation challenges, and potential for integration into clinical practice. The evidence suggests these technologies demonstrate promising diagnostic accuracy with sensitivity and specificity often exceeding 95% in controlled settings, though performance may vary in real-world conditions. Successful large-scale screening initiatives demonstrate the feasibility of technology-based approaches, while ongoing research continues to address limitations related to false positives, user adherence, and clinical workflow integration. As these technologies evolve, they stand to revolutionize atrial fibrillation screening paradigms, enabling earlier intervention and potentially reducing atrial fibrillation-related morbidity and mortality.
Introduction
Atrial fibrillation (AF) represents the most prevalent sustained cardiac arrhythmia globally, characterized by chaotic electrical signals in the atria leading to irregular and often rapid heartbeats. 1 The condition affects approximately 0.51% of the world population, with prevalence increasing dramatically with age. 2 Beyond its direct cardiac effects, AF significantly increases stroke risk, with AF-related strokes typically being more severe and debilitating than those of other etiologies. 3 The economic burden of AF management and its complications is substantial, making early detection and intervention crucial for healthcare systems worldwide.
A particular challenge in AF management is that approximately one-third of affected individuals remain undiagnosed due to asymptomatic or paroxysmal presentations. 4 Traditional detection methods, including standard 12-lead electrocardiograms (ECGs), often fail to capture intermittent episodes, while Holter monitors and other continuous recording devices face limitations in terms of cost, availability, and patient compliance for long-term monitoring.
The rapid advancement and widespread adoption of consumer digital health technologies present a potential solution to these challenges. Smartwatches and smartphones, equipped with increasingly sophisticated sensors and algorithms, offer unprecedented opportunities for continuous or frequent cardiac rhythm monitoring in everyday settings. 5 These devices employ various technologies, including single-lead ECG capabilities, photoplethysmography (PPG), and advanced algorithms, to detect irregularities consistent with AF.
This review examines the current evidence regarding smartphone and smartwatch-based AF detection, analyzing their accuracy, feasibility for large-scale screening, clinical impact, and integration challenges within healthcare systems. We also discuss ongoing developments and future directions that may further enhance their utility in addressing the global burden of undiagnosed AF.
Technologies for Atrial Fibrillation Detection on Mobile Devices
Smartphone-based Electrocardiogram
Smartphone-based ECG systems typically utilize external hardware attachments or cases that function as single-lead or multi-lead ECG devices when coupled with dedicated applications. These systems generally require user initiation and active participation to record cardiac electrical activity.
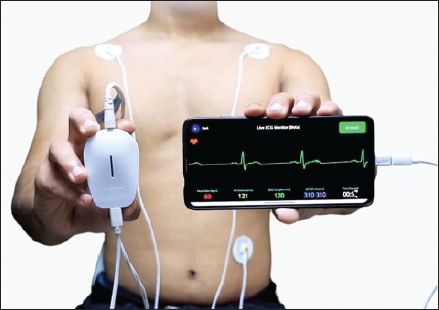
The most extensively studied smartphone ECG device is the AliveCor Kardia (formerly known as the AliveCor Heart Monitor), which consists of a portable, credit card-sized device with two electrodes that communicate wirelessly with smartphones, as shown in Figure 1. Users place their fingers on the electrodes to generate a single-lead ECG equivalent to lead I in a standard 12-lead ECG. 6 The recorded data are processed through algorithms that can automatically classify rhythms as normal sinus rhythm, possible AF, or unclassified.

Studies on AliveCor Kardia have shown consistent performance across various settings. Lau et al. reported a 98% sensitivity and 97% specificity in community screening. 6 Haberman et al. demonstrated its utility across diverse clinical settings with 72%-94% sensitivity and >94% specificity among 381 participants. 7 Towhari et al. achieved 97.3% sensitivity and 99.6% specificity in a cross-sectional study of 403 patients. 8 Himmelreich et al. found 87% sensitivity and 97.9% specificity in primary care settings. 9 A newer device, Spandan (Figure 2), was evaluated by Mahajan et al. showing 81.23% sensitivity and 87.86% specificity in a single-center study. 10
Photoplethysmography Technology
Photoplethysmography represents a non-invasive optical technique that detects blood volume changes in the microvascular bed of tissue. In the context of smartphones and smartwatches, PPG typically utilizes the device’s built-in camera or light sensors to capture and analyze pulse patterns. 11
For smartphone applications, users typically place their fingertip over the phone’s camera lens and flash, which allows the application to detect subtle color changes that correspond to blood flow with each heartbeat. 12 Variations in the time intervals between successive pulses (pulse-to-pulse intervals) are analyzed to identify the irregular patterns characteristic of AF.
Several PPG applications have demonstrated strong diagnostic performance. Cardiio Rhythm achieved 92.9% sensitivity in primary-care settings with 1,013 patients. 12 Krivoshei et al. reported 95% sensitivity and specificity in a case-control study of 80 patients. 13 The deep convolutional neural network (DCNN) algorithm achieved 100% sensitivity and 99.6% specificity in real-world primary care settings. 14 FibriCheck demonstrated 96% sensitivity and 97% specificity in their 223-participant study, when compared to a single-lead ECG interpreted by cardiologists. 15 The mobile AF app (MAFA) app achieved 91.6% positive predictive value in a large-scale trial with 187,912 participants. 16 Verbrugge et al. further examined smartphone camera-based PPG for AF screening, confirming its utility in ambulatory patients with excellent diagnostic accuracy. 17
Van Der Velden et al. provided comprehensive guidance on PPG signal interpretation, addressing common artifacts and technical considerations that influence accuracy in various clinical scenarios. Their “photoplethysmography dictionary” established standardized terminology and approaches for interpreting PPG-derived data in the context of AF detection. 18
Smartwatch Algorithms and Continuous Monitoring
Modern smartwatches combine various technologies, including PPG sensors, accelerometers, and in some models, ECG capabilities, to provide more comprehensive arrhythmia detection. Smartwatches equipped with PPG sensors function similarly but offer the advantage of passive or semi-continuous monitoring without active user engagement. The sensors, typically located on the device’s underside in contact with the wrist, can perform periodic readings throughout the day or continuous monitoring during specific activities. 19 The integration of multiple sensors allows these devices to account for motion artifacts and other confounding factors that might affect single-sensor approaches.
The Apple Heart Study represents one of the largest evaluations of smartwatch technology for AF detection. 19 This prospective study enrolled over 400,000 participants to assess the ability of the Apple Watch’s irregular rhythm notification algorithm to identify AF. The study demonstrated that among participants who received irregular pulse notifications and subsequently wore ECG patches, 34% had confirmed AF. Importantly, 84% of notifications were concordant with AF on simultaneous ECG patch readings, suggesting reasonable accuracy for a consumer-level screening tool. Bumgarner et al. specifically evaluated the Apple Watch’s (with Kardia Band) ability to distinguish AF from normal sinus rhythm using its PPG sensor. In their study involving 100 patients, the technology achieved 93% sensitivity and 84% specificity compared to a 12-lead ECG diagnosis. 20 However, Gala et al. reported only 67.4% sensitivity and 74.6% specificity in the detection of AF with the Apple Watch 6, which is much lower than the previous studies. 21 More recently, Fabritz et al. conducted a fully digital European case-finding study examining smartphone and wearable-detected atrial arrhythmias in older adults. Their findings supported the feasibility of digital case-finding approaches, while highlighting the importance of clinical confirmation for device-detected irregularities. 22
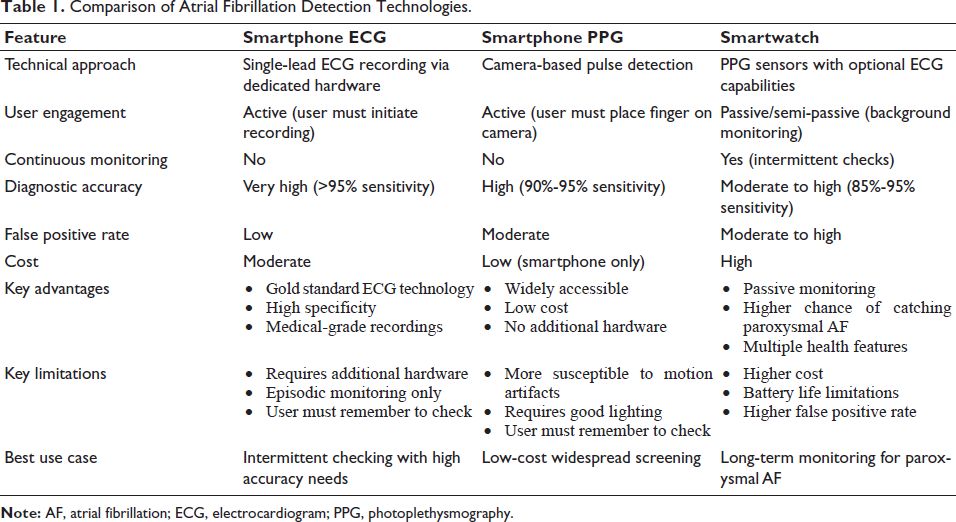
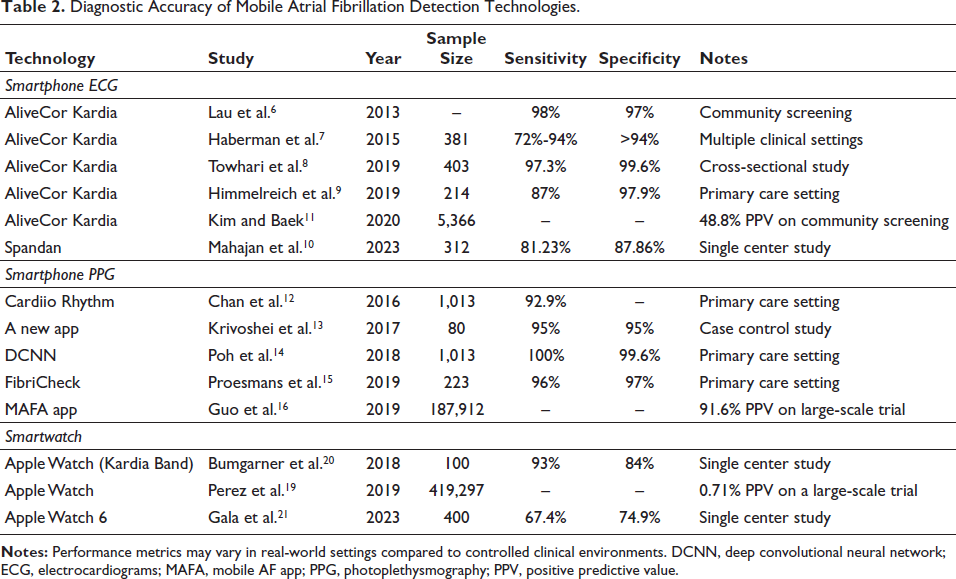
Table 1 gives a clear view of the different technologies used in the detection of AF, and Table 2 summarizes the diagnostic accuracy of various mobile AF detection technologies.
Comparison of Atrial Fibrillation Detection Technologies.
Diagnostic Accuracy of Mobile Atrial Fibrillation Detection Technologies.
Diagnostic Accuracy and Performance
Sensitivity and Specificity in Controlled Settings
The diagnostic performance of mobile AF detection technologies has been extensively evaluated in controlled clinical environments, generally demonstrating favorable results when compared to gold standard methods.
Smartphone-based ECG systems consistently show sensitivity of 72%-98% and specificity of 87%-99%. Photoplethysmography applications demonstrate comparable or slightly better performance with a sensitivity of 93%-100% and specificity of 95%-99%. Smartwatch algorithms show promising but slightly lower metrics with a sensitivity of 93% and a specificity of 84% in controlled studies.
Performance in Real-world Conditions
While controlled clinical studies provide valuable insights into diagnostic capabilities, real-world performance often differs due to various factors, including user technique, environmental conditions, motion artifacts, and population characteristics.
Rizas et al. conducted a pragmatic randomized clinical trial of smartphone-based screening for AF in real-world conditions. Their study demonstrated that while the technology remained clinically valuable, performance metrics were somewhat lower than those observed in highly controlled environments. 23 Similarly, Svennberg et al. in the STROKESTOP study found that implementation factors significantly impacted the effectiveness of screening programs despite high technological accuracy. 24
A particular challenge in real-world settings is the rate of uninterpretable or inconclusive results. Vandenberk et al. evaluated heart rate apps in mixed-methods studies, finding that user technique significantly influenced the quality of recordings and subsequent diagnostic yield. Their work emphasized the importance of user training and intuitive interfaces to maximize real-world effectiveness. 25
Reliability Challenges and False Positives
Real-world implementation of mobile AF detection technologies faces substantial reliability challenges that extend beyond controlled study findings. Isenegger et al. 26 conducted a comprehensive analysis, revealing that patients with baseline ECG abnormalities, including bundle branch blocks and frequent premature beats, experience up to 40% higher rates of inconclusive smart device readings compared to those with normal baseline ECGs (Table 3).
Diagnostic Accuracy of Five Smart Devices in Identifying Atrial Fibrillation Using Validation Studies.

Technical limitations significantly impact practical performance. Motion artifacts, poor electrode contact, and environmental interference contribute to false positive rates that often exceed those reported in controlled clinical trials. Medina-Avelino et al. emphasized particular challenges in hospital-at-home applications due to increased artifact rates and reduced supervision. 27
Studies demonstrate that 15%-25% of recordings may be technically inadequate or yield inconclusive results, especially in elderly populations with multiple comorbidities. The BASEL Wearable Study by Mannhart et al. showed inconclusive result rates varying from 8% to 23% across different device types and manufacturers. 28
The Apple Heart Study found that only 34% of participants who received irregular pulse notifications had AF confirmed on subsequent ECG monitoring. 19
These limitations create substantial healthcare system burdens through unnecessary specialist referrals, additional testing, and patient anxiety, emphasizing the need for realistic implementation expectations and comprehensive clinical workflow planning.
Factors Affecting Accuracy
Multiple factors influence the diagnostic performance of mobile AF detection technologies, including:
User characteristics: Age, skin tone, presence of tremor or movement disorders, and anatomical factors such as wrist size for smartwatches can all impact recording quality and algorithm performance.
25
Environmental factors: Ambient light conditions affect PPG performance, while electromagnetic interference may influence ECG-based systems.
18
Comorbid conditions: Other arrhythmias, particularly those causing irregular rhythms, can trigger false positive AF detections. Similarly, conditions affecting peripheral perfusion may impact PPG signal quality.
24
Algorithm design: Different computational approaches to rhythm analysis yield varying performance characteristics, with some optimized for specificity and others for sensitivity.
25
User technique: Proper placement of fingers on smartphone cameras or ECG electrodes significantly impacts signal quality. Similarly, appropriate smartwatch positioning and strap tightness affect PPG signal acquisition.
18
Large-scale Screening Initiatives
Community-based Screening Programs
Multiple large-scale community-based AF screening initiatives using mobile technologies have demonstrated feasibility and clinical impact.
Lowres et al. conducted the SEARCH-AF study, implementing smartphone-based ECG screening in community pharmacies. Among 1,000 participants aged ≥65 years, previously undiagnosed AF was identified in 1.5%, with the technology demonstrating 98.5% sensitivity and 91.4% specificity. The study also established cost-effectiveness, estimating an incremental cost-effectiveness ratio (ICER) of AU$4,066 per quality-adjusted life year gained for preventing strokes through early AF detection and anticoagulation. 29
Chan et al. reported on a large-scale community AF screening program using smartphone ECG technology implemented by a non-governmental organization in Hong Kong. Among 13,122 participants, the screening identified AF in 244 (1.8%) individuals, with a higher prevalence in those aged ≥75 years (3.7%). Importantly, 74% of the AF cases detected were previously undiagnosed, highlighting the value of community-based screening approaches. 30
Gruwez et al. conducted a fully digital European case-finding study using smartphone-based screening in the general population. Their work demonstrated not only technical feasibility but also significant clinical impact, with meaningful changes in medical treatment resulting from newly diagnosed AF cases. 31
Primary Care Implementation
Primary care settings represent optimal environments for systematic AF screening due to their accessibility and continuity of care advantages. Several studies have evaluated the implementation of mobile AF detection technologies in these settings.
Fitzmaurice et al. conducted a cluster randomized controlled trial comparing systematic screening to routine practice for AF detection in primary care. They found that systematic screening identified substantially more cases of AF than routine care, particularly among asymptomatic individuals who would otherwise remain undiagnosed. 32
Proesmans et al. specifically evaluated smartphone PPG technology in primary care settings, demonstrating high diagnostic accuracy and a favorable user experience among both patients and healthcare providers. Their study highlighted the technology’s potential to enhance routine clinical workflows without imposing substantial additional burdens on practice resources. 15
Willits et al. examined the WatchBP Home A technology for opportunistically detecting AF during hypertension management in primary care. Their analysis, which informed the UK National Institute for Health and Care Excellence (NICE) guidance, demonstrated cost-effectiveness and practical implementation advantages of integrating AF screening into routine primary care processes. 33
Clinical Outcomes and Cost-effectiveness
The ultimate value of mobile AF detection technologies lies in their ability to improve clinical outcomes while maintaining reasonable cost-effectiveness. Evidence in this domain continues to evolve, but shows promising trends.
Steinhubl et al. conducted the mSToPS randomized clinical trial, which demonstrated that immediate monitoring with a wearable ECG patch led to a significantly higher rate of AF diagnosis at 4 months compared with delayed monitoring (3.9% vs. 0.9%). Importantly, this higher diagnosis rate translated into increased initiation of anticoagulation therapy among those with diagnosed AF. 34
Mahajan et al. conducted a systematic review and meta-analysis examining the relationship between device-detected subclinical AF and stroke risk. Their analysis of 11 studies including 36,965 patients found that device-detected subclinical AF was associated with a significantly increased risk of stroke or systemic embolism (hazard ratio: 2.4). This finding supports the clinical relevance of AF detected through mobile technologies, even when episodes are brief or asymptomatic. 35
From a cost perspective, Lowres et al. demonstrated the cost-effectiveness of community pharmacy-based AF screening using smartphone ECG technology. Their economic analysis estimated that screening would prevent 8.8 strokes and gain 137.9 quality-adjusted life years per 1,000 patients screened, with an ICER within commonly accepted thresholds for healthcare interventions. 29
Mobile AF detection implementation costs extend beyond device expenses to encompass broader healthcare system impacts. False positive rates exceeding 10% significantly affect cost-effectiveness through unnecessary specialist consultations, repeat testing, and patient anxiety management. Infrastructure costs include staff training, electronic health record integration, and workflow modifications often underestimated in initial evaluations.
Implementation Challenges in the Indian Subcontinent
Mobile AF detection implementation in India faces unique challenges requiring tailored approaches. The SMART-India study revealed a 1.6% AF prevalence in rural Western India, but comprehensive validation studies for mobile detection technologies remain limited in Indian populations. 36 Population-specific considerations include genetic variations affecting PPG signal quality, a higher prevalence of rheumatic heart disease, potentially increasing false positives, and varying skin tones requiring specific validation studies. Infrastructure challenges encompass limited internet connectivity in rural areas, inconsistent power supply affecting device charging, and variable smartphone penetration across socio-economic groups.
Clinical Integration and Workflow Considerations
From Detection to Diagnosis
While mobile technologies offer valuable screening capabilities, the pathway from initial detection to formal diagnosis requires careful consideration. Svennberg et al. provided comprehensive guidance on using digital devices to detect and manage arrhythmias, emphasizing the importance of clinical verification of device-detected irregularities. 24 The consensus workflow of AF detection to management is shown in Figure 3.
Clinical Workflow Diagram for Atrial Fibrillation Management, Illustrating the Pathway from Initial Screening with Mobile Devices Through Clinical Review, Diagnostic Confirmation with 12-lead Electrocardiogram, and Treatment Decision-making Including Anticoagulation, Rate/Rhythm Control, and Lifestyle Modifications.
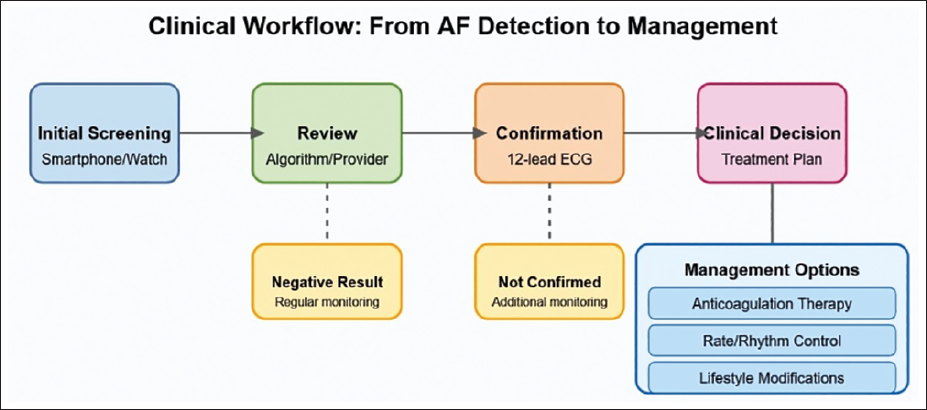
Perez et al. highlighted this pathway in the Apple Heart Study, where participants with irregular pulse notifications underwent additional monitoring with ECG patches to confirm the presence of AF before clinical interventions were considered. 19
Clinical Practice Integration
Successful integration of mobile AF detection technologies into clinical practice requires addressing several practical considerations:
Healthcare provider education: Clinicians require training in interpreting results from these technologies and understanding their limitations. Vandenberk et al. emphasized the importance of provider education for effective implementation.
25
Electronic health record integration: Seamless data flow between mobile devices and clinical systems enhances usability and reduces administrative burden. Sridhar et al. discussed ongoing challenges and opportunities in this domain.
5
Reimbursement models: Sustainable implementation requires appropriate compensation mechanisms for both technology costs and clinician time spent reviewing results. Current reimbursement structures in many healthcare systems do not adequately account for these activities. Liability considerations: Clear guidelines regarding responsibility for reviewing and acting upon device-generated alerts are essential. Svennberg et al. addressed various legal and ethical aspects of digital arrhythmia detection.
24
Proietti et al. reported on the Belgian Heart Rhythm Week screening program, highlighting practical aspects of integrating mobile AF detection technologies into existing healthcare frameworks. Their experience demonstrated the importance of clear communication channels between screening initiatives and primary care providers to ensure appropriate follow-up of positive findings. 37
Patient Engagement and Adherence
The effectiveness of mobile AF detection technologies ultimately depends on patient engagement and adherence to monitoring protocols. Steinhubl et al. noted in the mSToPS trial that adherence to wearing the ECG patch decreased over time, highlighting the importance of user-friendly designs and minimizing monitoring burden. 34
Strategies to enhance engagement include:
Educational interventions: Informing users about AF risks and the benefits of early detection improves motivation for consistent monitoring. User experience optimization: Intuitive interfaces and minimal disruption to daily activities enhance long-term adherence. Feedback mechanisms: Providing users with meaningful insights from their monitoring data maintains engagement beyond the novelty period. Integration with existing health behaviors: Embedding AF monitoring within broader health and wellness routines increases sustainability.
Recent developments in smartwatch technologies address many of these considerations by integrating AF detection capabilities into devices that provide multiple other functions, thereby encouraging regular use without specific focus on cardiac monitoring.
Limitations and Challenges
Technical Limitations
Despite promising advances, mobile AF detection technologies face several technical challenges:
Signal quality variability: Environmental conditions and user factors significantly influence recording quality, particularly for PPG-based methods.
18
Algorithm performance in specific populations: Most validation studies have been conducted in predominantly White, middle-aged, or older populations. Performance characteristics may differ in other demographic groups, particularly those with darker skin tones, affecting PPG accuracy.
25
Differentiation between AF and other arrhythmias: Some technologies struggle to distinguish AF from other irregular rhythms, such as frequent premature atrial or ventricular contractions, potentially leading to false positive results.
24
Battery life and processing constraints: Continuous monitoring capabilities remain limited by device battery capacity and processing power, particularly for smartwatches attempting to detect rare, intermittent events.
Clinical and Ethical Considerations
Beyond technical challenges, several clinical and ethical considerations warrant attention:
Management of subclinical AF: The optimal treatment approach for brief, device-detected AF episodes remains uncertain, particularly regarding anticoagulation decisions. Health anxiety: Frequent alerts or false positive notifications may induce unnecessary anxiety among users. Balancing sensitivity for detecting clinically significant arrhythmias against the psychological impact of false alarms remains challenging. Digital divide: Access to and familiarity with mobile technologies varies across populations, potentially exacerbating healthcare disparities if screening programs rely heavily on these approaches. Privacy and data security: Continuous heart rhythm monitoring generates sensitive health data, raising concerns about privacy protections and appropriate data governance.
Regulatory and Validation Standards
The rapid evolution of mobile health technologies has outpaced regulatory frameworks, creating challenges for both manufacturers and healthcare providers:
Inconsistent validation requirements: Standards for validating AF detection algorithms vary across jurisdictions, complicating international implementation and comparison. Software as a medical device: Regulatory approaches to software-based diagnostic tools continue to evolve, with varying requirements for clinical validation before market approval. Post-market surveillance: Mechanisms for monitoring real-world performance and safety of these technologies after regulatory approval remain underdeveloped in many regions. Liability distribution: Unclear delineation of responsibility between technology developers, healthcare providers, and users creates liability concerns that may impede adoption.
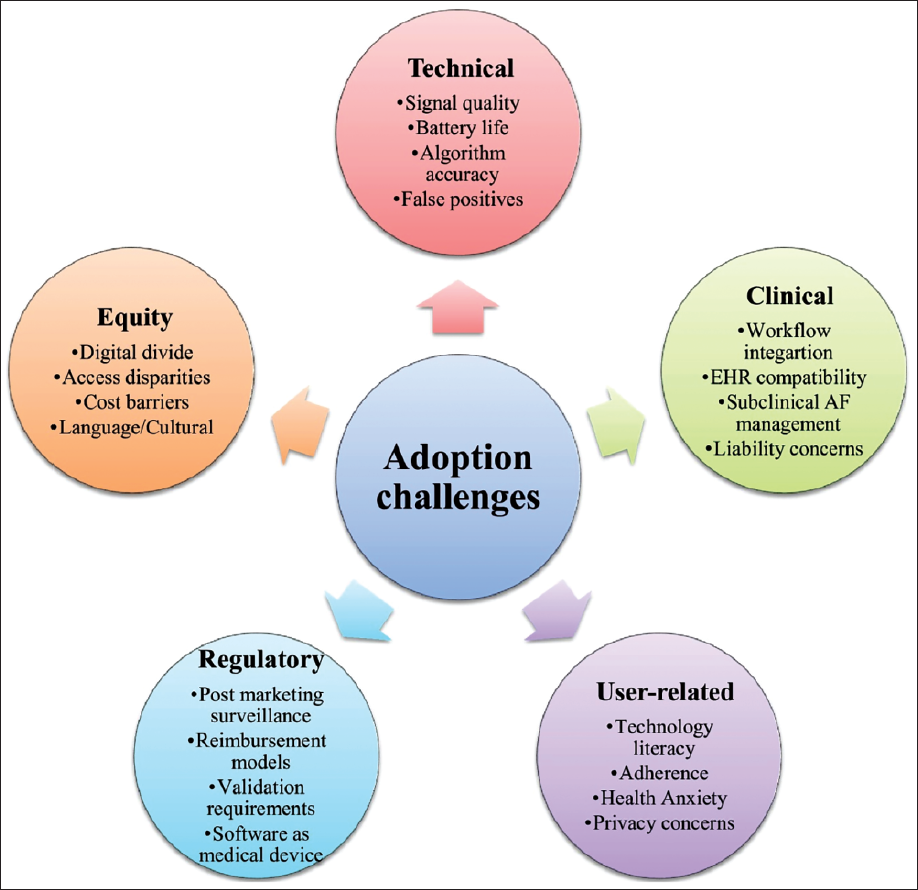
Sridhar et al. 5 discussed these regulatory challenges in their review of mobile health technologies in clinical arrhythmia care, highlighting the need for balanced approaches that ensure safety while enabling innovation. 5 Various obstacles faced during the adoption of mobile AF detection technologies have been summarized in Figure 4.
Key Adoption Challenges for Mobile Atrial Fibrillation Detection Technologies.
Future Directions
Technological Advancements
Several technological developments promise to enhance the capabilities of mobile AF detection systems:
Advanced sensor fusion: Integration of multiple sensor types (ECG, PPG, accelerometer, and temperature) with sophisticated algorithms will likely improve accuracy and reduce false positives.
5
Artificial intelligence and machine learning: Continued refinement of algorithms through machine learning approaches using larger and more diverse training datasets may enhance performance across different populations and conditions.
24
Extended battery life: Improvements in battery technology and power-efficient sensors will enable longer continuous monitoring periods, increasing the likelihood of capturing paroxysmal AF episodes. Miniaturization and form factor innovation: Development of smaller, more comfortable, and less obtrusive monitoring devices will improve user adherence for long-term monitoring.
Research Priorities
Key research priorities to advance the field include:
Comparative effectiveness studies: Head-to-head comparisons of different mobile technologies in diverse real-world settings are needed to guide implementation decisions. Long-term outcome studies: Research examining the impact of mobile AF detection on hard clinical endpoints such as stroke reduction, rather than simply AF diagnosis rates, will clarify the ultimate value of these approaches. Optimal screening protocols: Studies to determine the ideal frequency, duration, and targeting of screening efforts will maximize cost-effectiveness and clinical impact. Integration with predictive models: Research combining device-detected parameters with clinical risk factors may enhance risk stratification and personalize screening and treatment approaches.
Clinical Implementation Models
Evolving models for clinical implementation may include:
Hub-and-systems: Centralized interpretation centers receiving data from distributed monitoring devices may optimize specialist resource utilization while maintaining quality. Risk-based screening approaches: Targeting higher-intensity monitoring to those at greatest AF risk based on clinical factors may improve efficiency. Integration with comprehensive cardiovascular monitoring: Combining AF detection with monitoring of other parameters, such as blood pressure and physical activity, may provide a more holistic cardiovascular risk assessment. Hybrid care models: Blending traditional clinical care with remote monitoring approaches may optimize resource utilization while maintaining necessary patient-provider relationships.
Conclusion
Smartphone and smartwatch technologies offer promising advantages for AF detection through accessibility, affordability, and sophisticated monitoring capabilities. These devices can detect paroxysmal AF by monitoring over extended periods, potentially capturing episodes missed by traditional methods. Their widespread availability enables large-scale screening initiatives previously impossible with conventional equipment. Implementation challenges include determining optimal strategies, managing device-detected AF, and ensuring equitable access. The evolving technology landscape requires balanced regulatory approaches. As these technologies integrate into clinical workflows, they may significantly reduce the burden of undiagnosed AF, enabling timely intervention with anticoagulation therapy to reduce stroke risk and improve patient outcomes.
Footnotes
Declaration of Conflict of Interests
The author declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Ethical Approval and Patient Consent
Patient consent along with ethical approval from the Institutional Review Board was not applicable to this manuscript.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
