Abstract
Background
The interplay between nonalcoholic fatty liver disease and coronary artery disease is increasingly recognized, drawing attention to nonalcoholic fatty liver disease, a potential contributor to atherosclerosis. Emerging evidence indicates that nonalcoholic fatty liver disease might be associated with both traditional and novel cardiovascular risk factors. Hence, the present study aimed to determine the nonalcoholic fatty liver disease in angiographically confirmed coronary artery disease patients and to assess its association with the coronary artery disease severity.
Methods
Individuals with secondary causes of hepatic steatosis were excluded. All participants underwent a detailed cardiovascular evaluation, which comprised routine biochemical investigations, viral marker screening, abdominal ultrasonography to grade and assess the presence of nonalcoholic fatty liver disease, and coronary angiography to identify the severity of arterial involvement. Statistical analysis was carried out using Student’s
Results
A total of 531 patients were studied. A total of 405(76.2%) subjects had significant coronary artery disease, and 321(60.4%) had nonalcoholic fatty liver disease. The number of patients having nonalcoholic fatty liver disease out of coronary artery disease was 77.03%. The mean age group was 57.5 ± 11.4 years. Angiographically proven coronary artery stenosis was strongly associated with nonalcoholic fatty liver disease in a grade-dependent manner, with
Conclusions
Ultrasonographic evidence of nonalcoholic fatty liver disease independently influences both the occurrence of coronary artery disease and the extent of atherosclerotic involvement.
Introduction
Cardiovascular disease (CVD) remains the main cause of morbidity and mortality worldwide, posing a major public health challenge in both developed and developing nations. 1 The increasing burden of CVD has been attributed to several factors, which include longer life expectancy, tobacco use, sedentary lifestyles, and the widespread adoption of unhealthy dietary habits. 2 Genetic predisposition, obesity, hypertension, and dyslipidemia also play an important and critical role, alongside environmental and lifestyle influences, including stress, smoking, alcohol consumption, and high-fat diets. Although many risk factors of coronary artery disease (CAD) are well established, the threshold levels of many parameters continue to be refined across different ethnic groups. In recent years, nonalcoholic fatty liver disease (NAFLD) has gained considerable attention due to its potential role in the clinicopathogenesis of atherosclerosis. Multiple clinical findings have demonstrated the association between NAFLD and traditional/nontraditional cardiovascular pathogenic risk factors. 3
Nonalcoholic fatty liver disease is manifested by the abnormal deposition of fat within hepatocytes, resembling alcohol-induced hepatic injury but occurring in the absence of significant alcohol intake. The spectrum of NAFLD ranges from simple and classical steatosis to nonalcoholic steatohepatitis (NASH), fibrosis, and cirrhosis.4, 5 Its prevalence is estimated at 15%-40% in Western populations and 9%-40% in Asian populations. In India, NAFLD is increasingly recognized as a major cause of hepatic disorders, with epidemiological data suggesting a prevalence of 9%-32%, particularly higher among individuals who are overweight, obese, or have diabetes or prediabetes.6, 7 NAFLD, especially those with NASH, often show elevated fibrinogen and C-reactive protein (CRP) levels, which are established cardiovascular risk markers. 8
The liver also plays a pivotal role in producing inflammatory mediators and markers of endothelial dysfunction. 9 Furthermore, metabolic dysregulation and insulin resistance can modulate pathways that exacerbate disease progression. 10 It has been proposed that NAFLD is frequently regarded as the key manifestation of a dysregulated metabolic condition, a cluster of interrelated conditions including type 2 diabetes, obesity, hypertension, and dyslipidemia, all of which are strong risk factors for cardiovascular morbidity.11, 12 Given these associations, the present study was designed to investigate whether NAFLD is related to CAD and whether the grading of NAFLD correlates with the severity of CAD. By addressing these questions, we aim to clarify the potential role of NAFLD as an independent contributor to CAD.
Methods
Study Design
This observational cross-sectional study was conducted between June 2018 and May 2019. After obtaining informed consent, each patient underwent a detailed clinical evaluation, which included presenting complaints, cardiovascular risk factors, family history, and a physical examination. Anthropometric parameters such as height, weight, body mass index (BMI), and waist circumference were documented, along with electrocardiogram (ECG) and echocardiographic findings. Routine hematological and biochemical investigations were carried out, including complete blood count (CBC), renal and hepatic function tests, fasting blood glucose, cardiac biomarkers, lipid profile, and the inflammatory marker CRP.
Exclusion Criteria and Diagnostic Methods for Elimination Factors
Patients with potential secondary causes were excluded based on a comprehensive clinical evaluation, including detailed history (e.g., alcohol intake (>30 g/day in males and >20 g/day in females)), 13 physical examination, and relevant laboratory investigations. Specifically, patients were excluded if they had a history suggestive of heart disease other than CAD, like valvular heart disease, cardiomyopathies, congenital heart disease, rheumatic heart disease, and others, nephrotic syndrome or liver malignancy, confirmed by low serum albumin (<2.5 g/dL), urine protein excretion >3.5 g/day, and the presence of generalized edema. Viral hepatitis was excluded by testing for hepatitis B surface antigen (HBsAg) and antihepatitis C virus (HCV) antibodies. Hepatotoxic drug exposure within the past 6 months (e.g., steroids, valproate, oral contraceptive pills (OCPs), antituberculosis (TB) drugs) was elicited and documented.
Ultrasound (USG) findings were also used to support exclusion decisions. However, cases showing a contracted gallbladder on USG were not excluded solely on this basis unless accompanied by other sonographic signs or clinical indicators suggestive of liver pathology. This was done to avoid over-exclusion based on nonspecific findings. Every patient had a coronary angiography (CAG), which was carried out by a skilled cardiologist. Using the Judkins procedure, a femoral/radial approach was used to conduct CAG. 1 All patients had several images that showed the coronary arteries. A major coronary artery with at least 50% stenosis was considered to have CAD. 14 Depending on how many main coronary arteries were impacted by CAD, the subjects were divided into four groups: Group A—normal/insignificant coronary artery stenosis; Group B—one vessel disease; Group C—two vessel disease; and Group D—three vessel disease.
Under the supervision of a skilled radiosonologist (who was blind to the subjects’ personal information), all patients were evaluated for NAFLD by abdominal USG to identify fatty alterations in the liver. A high-resolution B-mode USG system with a 2.5-5 MHz linear transducer was used, with a minimum scan time of 10 min. NAFLD was diagnosed according to standard criteria recommended by the American Gastroenterological Association, including liver-to-kidney contrast (increased hepatic echogenicity compared to renal cortex); parenchymal brightness (diffuse hyperechoic appearance); deep beam attenuation; bright vessel walls/vascular blurring; and gallbladder wall definition. Fatty liver was graded as follows:
Statistical Analysis
After being coded, the data were added to a Microsoft Excel spreadsheet. Statistical Package for the Social Sciences (SPSS) version 20 (IBM SPSS Statistics Inc., Chicago, IL, USA) was the Windows software used for the analysis. Calculating percentages, means, and standard deviations were all part of descriptive statistics. All clinical indicators were quantitatively compared using the analysis of variance (ANOVA) (for quantitative data among three groups). When comparing qualitative data with two or more groups, the chi-square test was employed. A significance level of
Results
A total of 840 patients underwent angiography during the study period, but only 531 met the inclusion criteria. Out of the total patients under study, 405(76.2%) subjects had significant CAD, and 321(60.4%) had NAFLD. The number of patients having NAFLD of the CAD was 77.03%. In the present study, the mean age of the total patients was 60.7 years, and 25.4% were females. Average BMI was 27.64 kg/m2 and 59.19 years, with a minimum of 31 years and a maximum of 88 years. The detailed demographic data are given in Table 1.
General Characteristics of the Study Group-number (%).
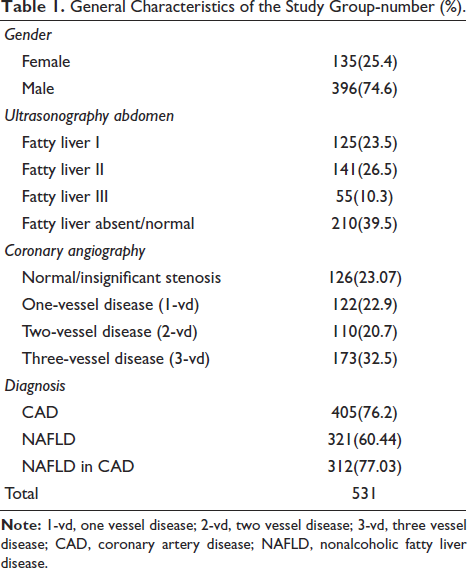
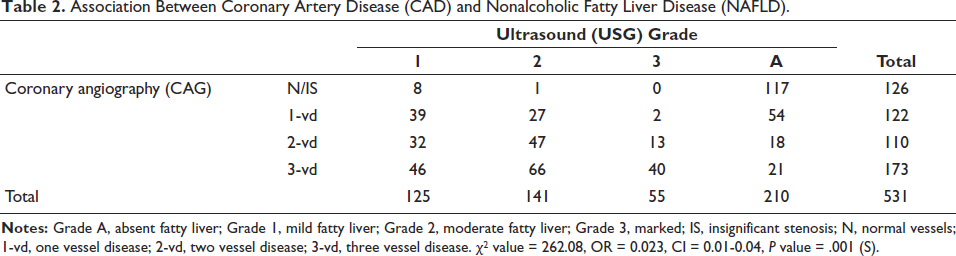
Table 2 and its corresponding Figure 1 depict the association of CAD and NAFLD. NAFLD was significantly correlated with angiographically shown coronary artery stenosis in a grade-dependent manner (
Association Between Coronary Artery Disease (CAD) and Nonalcoholic Fatty Liver Disease (NAFLD).
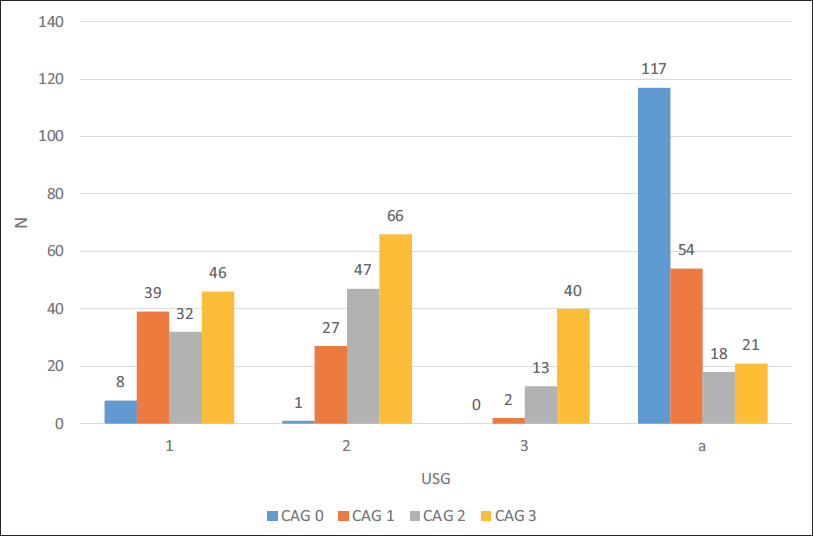
Coronary Artery Disease (CAD), Regression Analysis.
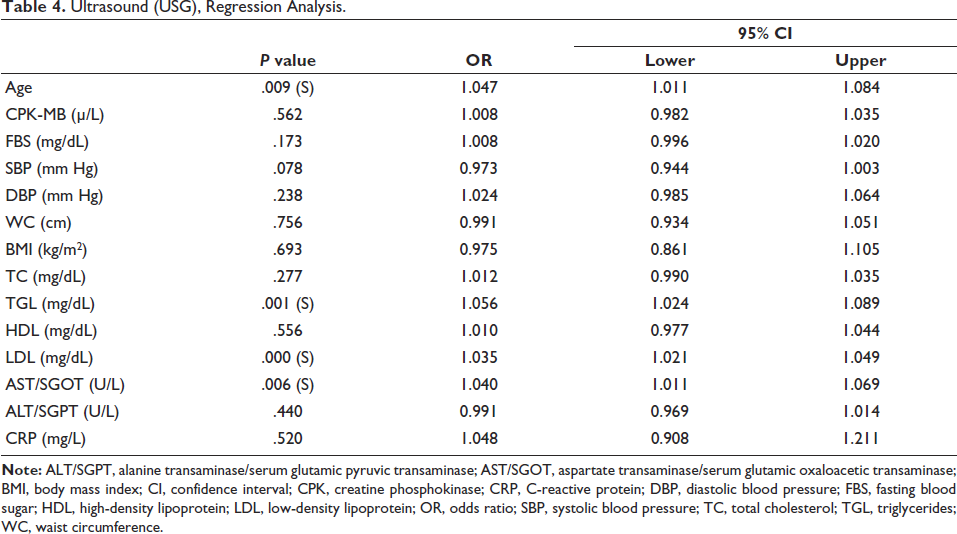
Ultrasound (USG), Regression Analysis.
Discussion
Coronary artery disease remains the foremost cause of mortality globally and contributes substantially to premature morbidity. 16 Extensive epidemiological studies have identified diabetes, obesity, dyslipidemia, hypertension, and smoking as well-established risk factors for CAD.1, 13 The interplay between metabolic and cardiovascular disorders has gained increasing attention over the past decade, with NAFLD emerging as an important systemic condition extending beyond the liver. Nowadays, NAFLD is considered a manifestation of metabolic syndrome along with a potential risk factor for CAD. 12 Several noninvasive techniques have been developed to assess hepatic fibrosis, including the NAFLD fibrosis score, Fibrosis-4 index, and the aspartate aminotransferase-to-platelet ratio index, alongside imaging techniques such as transient and/or magnetic resonance elastography. 15
A noteworthy association has been observed between NAFLD and CAD, with 43.9% of CAD patients in the present study also exhibiting NAFLD. Supporting this, Wild et al., in a large retrospective cohort involving 134,368 individuals, reported that the presence of NAFLD along with diabetes was significantly associated with increased risks of CVD, malignancy, and all-cause mortality. Similarly, Vilar et al., in their study of 244 Brazilian participants, found CAD in 63.5% and NAFLD in 42.2% of cases, further reinforcing the link between these two conditions. 16 A meta-analysis by Mahfood et al. of 25,837 individuals from six trials demonstrated that people with NAFLD were 77% more likely to experience severe cardiovascular events, such as a higher risk of CAD (126%), ischemic stroke (109%), and CVD mortality (46%). 17
The overall prevalence of NAFLD in the normal population ranges from 15% to 40%. 18 In our study, 531 patients were studied, and the prevalence rate of NAFLD was 60.4%. Recently, Duseja et al. 15 also reported a prevalence of 53.5% in North India. A similar study by Agarwal et al. 6 had reported a prevalence of 57.2%. In the Western world, NAFLD is now the most common and major chronic liver disease in high-income countries, and it is estimated to affect at least 25%-30% of the general population. 19 Most individuals with NAFLD are obese or overweight, and this is associated with nearly a fivefold higher likelihood of developing hepatic steatosis. 20 In the present study, participants had an average waist circumference of 90.62 ± 7.6 cm and a mean BMI of 27.64 ± 2.9 kg/m2, reflecting the contribution of central obesity. Central obesity with raised waist circumference and increased BMI also underscores the importance of insulin resistance in the linking mechanism between CAD and NAFLD.
The joint clinical practice guidelines of the European Association for the Study of the Liver (EASL) [Ref], the European Association for the Study of Diabetes (EASD) [Ref], and the European Association for the Study of Obesity (EASO) recommend that individuals with obesity or other metabolic syndrome components undergo evaluation of serum liver enzymes and steatosis markers, with ultrasonography as the primary screening modality.
21
Consistent with this, our findings demonstrate a significant correlation between BMI, fatty liver, and CAD (
People with impaired fasting glucose, type 2 diabetes, dyslipidemia, and hypertension are at a higher risk of developing NAFLD.1, 7 Moreover, obesity and an excess of free fatty acids cause liver insulin resistance, lower insulin clearance, and muscular insulin resistance.
23
This study’s findings, which demonstrated a clinically significant (
Nonalcoholic fatty liver disease is reported to be associated with CVD, and the two disorders share cardiometabolic risk factors.
20
Our findings demonstrated that NAFLD is strongly associated with CAD in a grade-dependent manner. Regression analysis and statistical association show that the grading of fatty liver was directly proportional to the number of vessels involved in CAG. In the current study, 77.03% of CAD patients had NAFLD. Our data can be substantiated by the following studies, in which the number of patients with NAFLD was higher with existing CAD. Ajmal et al.,
22
in their study found a high prevalence (69.2%) in patients with CVD, further supporting the association between the two disease entities. A study by Boddi et al.,
23
showed that in non-diabetic patients admitted for ST-elevation myocardial infarction (STEMI), NAFLD prevalence was very high (87%) and also found that severe NAFLD independently increases the risk for multivessel CAD and was subsequently associated with CV events. Another study by Friedrich-Rust et al.,
24
detected a high incidence of NAFLD (72.7%) in patients with any grade of CAD, and even higher rates in individuals with severe CAD. Wong et al.
25
in their study showed that 84.6% of patients with NAFLD were found to have >50% stenosis in one or more coronary arteries. Choi et al.
26
investigated that in cases of CAD in Koreans, 80.4% of patients had NAFLD, and it was strongly associated with NAFLD in a grade-dependent manner. A study by Agac et al.,
27
had NAFLD prevalence of 81.2% in CAD patients, and those with NAFLD have more complex CAD as assessed by syntax scoring (SS). In the present study, we found that NAFLD significantly elevated the risk of CAD (OR = 0.023, CI = 0.01-0.04,
Fatty liver is linked to CAD in individuals with clinical grounds for CAG, regardless of other metabolic variables, as shown by Wong et al. 31 The connection between NAFLD and CAD has several reasons. Numerous researchers have looked at potential noninflammatory processes of NAFLD, which are corroborated by the levels of different biomarkers, including reactive oxygen species, adipocytokines (adiponectin and leptin), CRP, and caspase-generated cytokeratin. 32 Two recently published studies by Wong 33 and Lonardo 34 postulated that uncomplicated simple steatosis leads to subclinical atherosclerotic burden via traditional and nontraditional cardiovascular risk factors, including type 2 diabetes, hypertension, dyslipidemia, hyperuricemia, chronic kidney disease, chronic inflammation, or expanded adipose tissue in the epicardial region. These studies also mentioned an enhanced risk of major adverse cardiovascular events (MACE) via increased hepatic production of multiple pro-thrombotic mediators such as hypoxia-inducible factor, vascular endothelial growth factor, and intercellular adhesion molecule-1, among others, as commonalities shared by NAFLD and unstable atherosclerotic plaques of coronary arteries.
Beyond traditional metabolic markers, adipocytokines such as leptin and adiponectin, inflammatory mediators like CRP and tumor necrosis factor-alpha (TNF-α), and hepatokines such as fetuin-A and fibroblast growth factor-21 (FGF-21) have been shown to influence both hepatic steatosis and vascular inflammation. These markers play a key role in endothelial dysfunction, insulin resistance, and pro-atherogenic states. Incorporating such molecular profiling into future studies may enhance the risk stratification of patients with NAFLD for cardiovascular events and help in identifying potential therapeutic targets. 35 Given the resource constraints and lack of funding in the current study, we could not perform molecular testing, but we emphasize its academic importance and recommend it for future research.
Study Limitations
Liver biopsy and magnetic resonance imaging (MRI), which are considered the gold standard for diagnosing NAFLD, are not being used as they are invasive processes, and noninvasive tests have been shown to be reliable in detecting advanced fibrosis or cirrhosis and in diagnosing many liver diseases and gauging long-term clinical outcomes. Furthermore, these tests are safer and cheaper than biopsy and MRI, and they can be repeated over time to monitor disease progression.
Conclusion
Among patients undergoing CAG, NAFLD is associated with coronary artery stenosis, and higher grades of NAFLD correlate with multivessel coronary involvement. NAFLD with CAD shares overlapping metabolic and cardiovascular risk factors, and the complexity of CAD remains an important determinant of future adverse cardiac events, as well as a guide for selecting optimal revascularization strategies, either surgical or percutaneous. While the precise causal relationship between NAFLD and CAD is not yet fully understood, the heterogeneity of both emphasizes the unmet need for further research to elucidate the mechanistic pathways through which NAFLD may influence the progression and severity of CAD.
Footnotes
Acknowledgments
The authors would like to thank hospital staff, technicians, and the radiology department. Authors are also thankful to Dr. Nishant Dalal (Intas Pharmaceutical Ltd., Gujarat, India) for critically reviewing the manuscript and fruitful suggestions. Authors also acknowledge Ms. Sakshi Srivastava and Dr. Mehul R. Chorawala, Intas Pharmaceutical Ltd., Gujarat, India, for medical writing assistance and additional editorial communication. The authors are also grateful to the study patients for their participation in this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Ethics Committee of All India Heart Foundation.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has provided informed consent for the submission of the article to the journal.
