Abstract
Tricuspid valve, once considered a “forgotten valve,” is experiencing a remarkable resurgence in clinical practice. Previously viewed as a secondary consequence of left-sided heart disease, tricuspid regurgitation is now recognized as an independent driver of morbidity and mortality. This recognition has led to the development of a diverse range of minimally invasive transcatheter tricuspid valve interventions, tailored to treat a high-risk patient population that often presents with inoperable conditions. The therapeutic landscape now includes multiple modalities, each with distinct mechanisms and indications: tricuspid transcatheter edge-to-edge repair, direct and indirect annuloplasty, orthotopic transcatheter tricuspid valve replacement, and heterotopic caval valve implantation. With pivotal randomized trial data available for both tricuspid transcatheter edge-to-edge repair and transcatheter valve replacement, evidence has solidified their roles in improving quality of life and, in the case of transcatheter valve replacement, achieving near-complete tricuspid regurgitation elimination. This comprehensive review summarizes the current state of all major transcatheter tricuspid valve interventions, discussing the complex anatomy, pathophysiology, diagnostic advancements, key technologies, landmark clinical evidence, and the specific challenges inherent to each approach. It aims to serve as a reference for clinicians navigating the rapidly evolving and increasingly personalized options for managing this complex and no-longer-forgotten disease.
Keywords
Introduction: The “Forgotten Valve” No More
For decades, the tricuspid valve (TV) was overshadowed by its left-sided counterparts, with tricuspid regurgitation (TR) often dismissed as a secondary issue that seldom required direct intervention. 1 This perception was sustained by the perceived lower physiological impact of the right heart and the initial efficacy of diuretics in controlling symptoms.2, 3 Consequently, surgical intervention for isolated TR was often delayed until the onset of end-stage right heart failure, a point at which high operative mortality rates discouraged referrals. 4
In the last decade, this dogma has been decisively challenged. A wealth of epidemiological data has re-cast significant TR not merely as a marker of advanced disease but as a direct contributor to poor prognosis, with the mortality of untreated severe TR reaching approximately 50% at 2-year follow-up.5, 6 This revised understanding, coupled with the success of transcatheter therapies for other valve diseases, has spurred unprecedented innovation. The emergence of a wide spectrum of transcatheter tricuspid valve intervention (TTVI)—from repair to replacement—offers new hope for a large, undertreated population of patients previously deemed inoperable.
The Burden and Prognostic Impact of Tricuspid Regurgitation
The assertion that TR is a benign condition has been refuted by numerous large-scale studies. The cumulative mortality of untreated severe TR is substantial, and its prevalence increases with age, affecting up to 4.0% of individuals beyond the age of 75 years.5, 7 Tricuspid regurgitation is an independent predictor of morbidity and mortality across various populations, including those with heart failure (both preserved and reduced ejection fraction), multivalvular disease, and after cardiac implantable electronic device (CIED) placement.8, 9 The presence of significant residual TR after interventions like tricuspid transcatheter edge-to-edge repair (T-TEER) has also been linked to worse prognosis, highlighting the importance of achieving substantial and durable TR reduction.10, 11
Etiology of Tricuspid Regurgitation: A Spectrum of Disease
Understanding the underlying cause of TR is paramount for determining the optimal therapeutic strategy. The etiologies are broadly categorized into primary (or organic) and secondary (or functional) TR, with secondary TR being far more prevalent and itself composed of distinct phenotypes.
Primary TR results from intrinsic abnormalities of the valve leaflets, chordae, or papillary muscles. This category includes a variety of conditions, such as rheumatic heart disease (RHD), which leads to leaflet thickening and retraction; infective endocarditis causing vegetations and leaflet destruction; myxomatous degeneration resulting in leaflet prolapse or flail; carcinoid syndrome, which deposits fibrous plaques on the valve; and traumatic injury leading to chordal or papillary muscle rupture. While less common than secondary TR, primary TR often requires surgical or replacement therapies due to the structural damage to the valve apparatus.1, 12
Secondary TR is the most common form, accounting for over 90% of cases. Here, the valve leaflets are structurally normal, but regurgitation occurs due to geometric distortion of the right heart. A critical advancement in the field has been the sub-classification of secondary TR into distinct phenotypes based on the primary driver of this distortion. 1
Ventricular Secondary Tricuspid Regurgitation (V-STR)
This form is driven by right ventricular (RV) dilation and dysfunction. It is typically a consequence of chronic pressure or volume overload from conditions like left-sided heart disease (e.g., mitral regurgitation, aortic stenosis) or pulmonary hypertension (PH). The enlarging RV displaces the papillary muscles, leading to apical tethering of the leaflets and preventing their effective coaptation.
Atrial Secondary Tricuspid Regurgitation (A-STR)
This more recently defined entity is characterized by severe annular dilation that is out of proportion to any RV changes. It is primarily driven by dilation of the right atrium (RA) and tricuspid annulus, most commonly in the setting of long-standing, persistent atrial fibrillation. In A-STR, leaflet tethering is often minimal, and the regurgitation is mainly due to the enlarged annular area preventing the leaflets from meeting. This distinction is prognostically important, as patients with A-STR may have a more favorable course than those with V-STR.
Cardiac Implantable Electronic Device-related Tricuspid Regurgitation
With the widespread use of pacemakers and defibrillators, CIED-related TR has emerged as a significant and distinct etiology. The transvenous lead can directly interfere with valve function through several mechanisms: impingement on a leaflet preventing closure, adhesion to a leaflet or chordae, or direct perforation of a leaflet. This direct mechanical interaction is a key consideration when planning an intervention, as T-TEER may be challenging, and transcatheter tricuspid valve replacement (TTVR) requires a strategy for managing the jailed lead. 9
Understanding Tricuspid Valve Anatomy: The Foundation for Intervention
A thorough appreciation of the complex and variable anatomy of the TV apparatus is fundamental to the planning and execution of any transcatheter intervention. Unlike the more robust mitral valve, the TV is a delicate and intricate structure whose components must be carefully considered to ensure procedural success and avoid complications. The TV apparatus consists of the annulus, leaflets, chordae tendineae, and papillary muscles, all situated within the complex geometry of the right heart.
Annulus
The tricuspid annulus is the largest of the four cardiac valve orifices. It is a dynamic, elliptical, and distinctly non-planar structure, possessing a saddle shape that changes throughout the cardiac cycle. This three-dimensional geometry is a critical consideration for the sizing and seating of both annuloplasty and replacement devices. Furthermore, the annulus has a close and critical relationship with the surrounding structures. The septal portion of the annulus is in direct proximity to the atrioventricular (AV) node and the bundle of His, making this region a “danger zone” during device anchoring, particularly for TTVR, where radial force can lead to compression and permanent conduction disturbances. 12 Additionally, the right coronary artery (RCA) often runs in close proximity to the posterolateral annulus, creating a risk of compression or perforation during the implantation of annuloplasty devices that utilize anchoring screws. 13
Leaflets
The term “tricuspid” is often a misnomer. While the valve is classically described as having three leaflets—anterior, posterior, and septal—this configuration is present in only about 60% of individuals. A significant portion of the population has two, four, or even more leaflets, separated by a series of commissures and smaller indentations.14, 15 The anterior leaflet is the largest and most mobile, followed by the septal and then the smaller, often scalloped, posterior leaflet. This anatomical variability has profound implications for T-TEER, where the strategy for leaflet grasping depends entirely on the number and orientation of the leaflets available to create a durable tissue bridge. The presence of multiple leaflets can complicate the procedure and may be associated with less effective TR reduction. 15
Subvalvular Apparatus
The leaflets are tethered to the right ventricle (RV) by the chordae tendineae, which originate from a group of three or more papillary muscles. The subvalvular anatomy is complex and can impede the delivery and positioning of large TTVR systems. The distance from the annulus to the papillary muscles and the moderator band must be assessed by imaging to ensure sufficient space for a replacement device to be implanted without impinging on these structures.12, 16 For T-TEER, the density and location of chordal structures can interfere with the grasp of the leaflets by the clip arms. A comprehensive understanding of this entire apparatus is, therefore, a prerequisite for safe and effective intervention.
Central Role of Diagnostics and Patient Selection
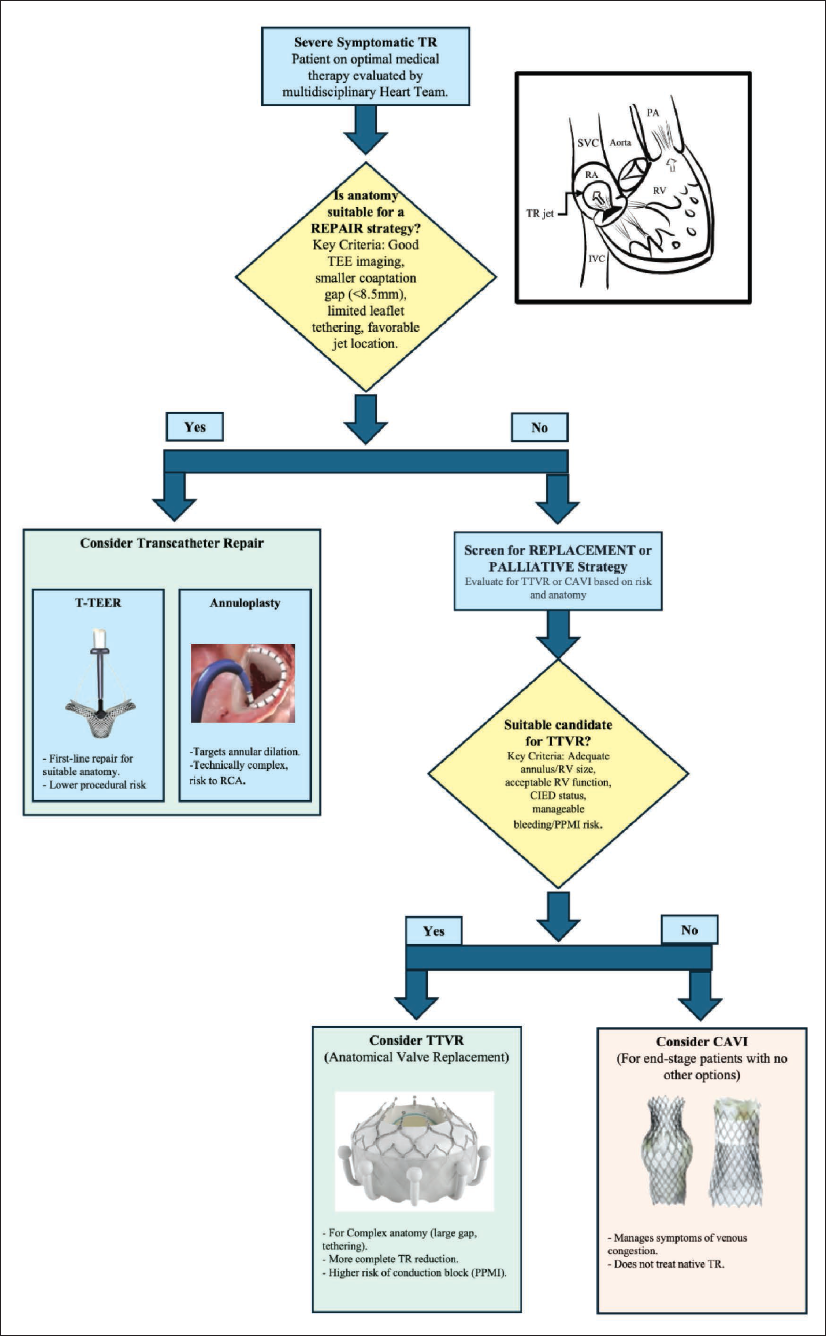
A comprehensive, multimodality approach is the cornerstone of modern TR management, essential for diagnosis, risk stratification, and procedural planning (Figure 1).
Before any intervention, patients must undergo medical optimization and a thorough hemodynamic assessment. This involves ensuring they receive optimal medical therapy for underlying conditions, such as heart failure or PH. 17 Right heart catheterization (RHC) is crucial to assess hemodynamics and RV-pulmonary arterial (RV-PA) coupling, as echocardiography can underestimate pulmonary pressures in severe TR. Echocardiography derived metrics, such as the ratio of tricuspid annular plane systolic excursion (TAPSE) to systolic pulmonary artery pressure (sPAP), have emerged as key prognostic indicators, with a ratio <0.41 showing worse outcomes. 18
In addition to medical optimization, multimodality imaging is pivotal in TR management. Imaging should be performed in a euvolemic state. 3D transesophageal echocardiography (TEE) and 3D intracardiac echocardiography (ICE) are crucial for assessing valve morphology and guiding procedures. Intracardiac echocardiography is particularly useful when TEE images are suboptimal. 19 Cardiac computed tomography (CT) is equally essential, especially for TTVR screening, as it offers precise measurements of the TV annulus, right heart chambers, and venous access routes. 16 Cardiac magnetic resonance (CMR) is the gold standard for quantifying RV volumes and function, and can identify fibrosis, which may have prognostic relevance. 20
An Expanding Armamentarium: Transcatheter Treatment Options
The TTVI landscape now includes robust options for both repair and replacement, allowing for a more tailored approach based on patient anatomy and clinical status (Figure 1).
Transcatheter Repair Techniques Including Tricuspid Transcatheter Edge-to-Edge Repair and Transcatheter Annuloplasty
T-TEER is the most established TTVI and remains a frontline therapy, particularly for patients with favorable anatomy (e.g., smaller coaptation gaps, anteroseptal jet location). The fundamental principle of T-TEER is the creation of a tissue bridge between the tricuspid leaflets, effectively forming a double-orifice valve that reduces the regurgitant area. This approach, which surgically originated with the Alfieri stitch, has been successfully translated to a percutaneous technique. Two primary systems have led the field: the TriClip™ system (Abbott) and the PASCAL™ system (Edwards Lifesciences). Both are delivered transfemorally and rely on grasping leaflets under TEE and/or ICE guidance. The TriClip device features an active grasping mechanism, while the PASCAL system utilizes a spacer element designed to fill the coaptation gap and paddles that clasp the leaflets with a passive closure mechanism. Both systems have undergone iterative improvements, resulting in different device sizes and enhanced steerability to accommodate the complex anatomy and challenging delivery angles characteristic of the TV.
The clinical evidence supporting T-TEER is robust, built upon large-scale registries and a landmark randomized controlled trial. The international TriValve registry was among the first to demonstrate the feasibility and safety of T-TEER in a real-world, high-risk population, showing significant TR reduction and symptomatic improvements. 21 This was followed by pivotal single-arm studies for both devices. The TRILUMINATE study of the TriClip system and the CLASP TR study of the PASCAL system both reported high rates of procedural success, with the majority of patients achieving TR reduction to moderate or less at 1 year, coupled with sustained improvements in quality of life and functional capacity.22, 23
The landmark TRILUMINATE Pivotal trial provided the highest level of evidence, randomizing 350 patients with severe TR to either T-TEER with the TriClip system or guideline-directed medical therapy. The trial met its hierarchical primary endpoint, driven overwhelmingly by a profound improvement in quality of life as measured by the Kansas City Cardiomyopathy Questionnaire (KCCQ). At 1 year, patients in the T-TEER arm experienced a mean KCCQ improvement of 12.3 points, compared to just 0.6 points in the control group. The procedure was exceptionally safe, with a 98.3% freedom from major adverse events at 30 days. While the trial did not show a statistically significant reduction in mortality or heart failure hospitalizations at 1 year, it definitively established T-TEER as a safe and effective therapy for improving the health status of patients with severe TR. 24
The success of T-TEER is, however, highly dependent on anatomical patient selection. Key predictors of a successful outcome include a coaptation gap of less than 8.5 mm, a central or anteroseptal jet location, and the absence of severe leaflet tethering or calcification. To standardize this assessment, the GLIDE score was developed, incorporating gap size, jet location, image quality, chordal density, and en-face TR morphology to predict procedural success. 25 Patients with unfavorable anatomy, particularly those with very large coaptation gaps or non-central jets, are less likely to achieve significant TR reduction with T-TEER, often leading to the consideration of replacement therapies. 26
Whereas the transcatheter annuloplasty mimics a foundational surgical technique, it aims to reduce the dilated TV annulus, thereby improving leaflet coaptation. The Cardioband™ system (Edwards Lifesciences) is a transvenously delivered polyester band anchored to the annulus with a series of screws. Once implanted, it is cinched to reduce the annular diameter. The prospective TRI-REPAIR study showed that this approach led to a 16% reduction in the septolateral annular diameter and ≤2+ TR in 72% of patients at 2 years, with corresponding improvements in functional capacity and quality of life. 27 Other devices and suture-based techniques are in earlier stages of development.
Transcatheter Tricuspid Valve Replacement
For patients with complex anatomy unsuitable for repair, TTVR has emerged as a transformative therapy. TTVR has rapidly evolved from a concept to a clinical reality, offering a more definitive solution for TR by completely replacing the native valve. This approach is particularly advantageous for patients with anatomical features that preclude successful repair, such as very large annular dimensions, severe leaflet tethering, extensive leaflet destruction, or challenging CIED-lead interactions. A variety of TTVR systems are in clinical development, differing in their anchoring mechanisms (e.g., subannular anchors, radial force, leaflet grasping), valve design, and delivery approaches (transfemoral versus transjugular).
The Evoque™ valve (Edwards Lifesciences) is the first TTVR system to receive both CE Mark and Food and Drug Administration (FDA) approval, and has the most extensive clinical data. It is a self-expanding, nitinol-framed valve with a pericardial tissue leaflet design. A key feature is its anchoring mechanism, which utilizes a combination of an intra-annular skirt and nine subannular anchors that grasp the native leaflets and chordae.
The clinical journey of the Evoque valve culminated in the TRISCEND II randomized trial, a landmark study that compared TTVR with optimal medical therapy in 400 patients with severe TR. 28 The trial successfully met its primary hierarchical endpoint, demonstrating the clear superiority of TTVR. At the 1-year mark, key findings highlighted substantial TR reduction, with 95.3% of patients experiencing ≤ mild TR compared to just 2.3% in the medical therapy group. This near-total correction of the valvular lesion is the primary advantage of TTVR over repair techniques. Though there was no statistically significant difference observed in all-cause mortality or heart failure hospitalizations, TTVR notably improved patients’ quality of life, as reflected by the KCCQ score, and enhanced functional class based on the New York Heart Association (NYHA) classifications. Regarding safety, the procedure exhibited a 30-day mortality rate of 3.5%, severe bleeding occurred in 10.4% of cases, and 24.7% of pacemaker-naïve patients required new permanent pacemaker implantation (PPMI).
Recent post-procedural imaging studies have provided further insight into the in vivo behavior of the Evoque valve. Computed tomographic analyses have confirmed its unique design feature: the outer frame conforms to the eccentric shape of the native tricuspid annulus, while a decoupled inner frame maintains an optimal, near-circular geometry to ensure proper leaflet function. These studies, however, also highlighted the issue of hypoattenuated leaflet thickening (HALT), or subclinical leaflet thrombosis, which was observed in approximately 25% of patients, underscoring the critical need for effective lifelong anticoagulation after TTVR. 29
Beyond Evoque, several other orthotopic devices are showing promise in earlier stages of clinical investigation. The LuX-Valve Plus (Jenscare) and Cardiovalve (Venus Medtech) have demonstrated high rates of procedural success and significant TR reduction in first-in-human and compassionate use series, expanding the potential future options for patients.30, 31
Caval Valve Implantation
Caval valve implantation (CAVI) represents a distinct therapeutic strategy, which involves implanting bioprosthetic valves in the superior and inferior vena cava (IVC), thereby bypassing the limitations imposed by the native TV anatomy. It does not treat the native TR directly but instead aims to mitigate its primary hemodynamic consequence: systemic venous congestion that leads to symptoms of right heart failure like edema and ascites. This makes it a valuable palliative option for highly symptomatic patients who are anatomically unsuitable for, or at excessive risk from, orthotopic repair or replacement. The TRICUS EURO study of the TricValve® system (P+F Products + Features) showed significant improvements in quality of life and symptoms at 6 months, and subsequent reports have noted favorable right heart remodeling, underscoring its utility in a select, end-stage patient population.32, 33
Navigating the Challenges of Transcatheter Tricuspid Interventions
While TTVI offers transformative potential, each modality presents a unique profile of challenges and limitations that the Heart Team must carefully weigh in a shared decision-making process.
Transcatheter repair (T-TEER and annuloplasty) presents several challenges. A key issue with T-TEER is its dependency on anatomical suitability. Its effectiveness is limited in patients with complex valve morphology, large coaptation gaps (>8.5 mm), and significant leaflet tethering. In these cases, achieving a durable reduction of TR to moderate or less proves difficult, and significant residual TR is strongly linked to worse long-term outcomes.10, 11 Annuloplasty, particularly with devices like Cardioband, is technically complex and carries risks such as potential injury to the RCA due to its proximity to the annulus. This risk has limited the procedure’s adoption to highly experienced medical centers. 13 Additionally, both repair techniques heavily rely on high-quality TEE and/or ICE imaging to clearly visualize the leaflets, annulus, and surrounding structures for precise device placement, presenting a challenge in patients with poor acoustic windows. 19
Similarly, TTVR has its own challenges. First, the most significant complication is new-onset conduction disturbance (NOCD), which can necessitate PPMI in up to 25% of patients, as reported in pivotal trials. 28 The risk is influenced by device design (e.g., radial force, degree of oversizing) and proximity to the AV node. This necessitates continuous post-procedural rhythm monitoring for at least 72 h and a pre-emptive strategy for valve-sparing pacing. 34 Second, about one-third of TTVR candidates have pre-existing CIEDs, necessitating the “jailing” of leads between the prosthesis and the native annulus. This raises long-term concerns about lead integrity, functionality, and the challenges of extracting an infected lead in the future. Consequently, both these concerns necessitate mandatory multidisciplinary discussions with electrophysiology specialists. 35 The third challenge is that these patients are often frail, face high risks of both periprocedural bleeding from large-bore venous access and subsequent valve thrombosis. This creates a delicate balancing act, requiring meticulous access management and a commitment to lifelong, carefully monitored oral anticoagulation. Finally, the afterload mismatch, particularly in patients with severely compromised RV, becomes a critical concern. The sudden removal of the low-resistance TR pathway can acutely increase ventricular afterload, potentially leading to acute RV failure, a rare yet life-threatening condition that necessitates immediate intervention with inotropes and pulmonary vasodilators.
Caval valve implantation also comes with its own set of challenges. Fundamentally, it is a palliative rather than a curative therapy; it does not address TR at its source but alleviates symptoms of systemic venous backflow, treating the consequences (venous congestion) rather than the root cause (valvular incompetence).1, 32 While it can effectively improve symptoms like edema and enhance the quality of life, it does not restore normal right heart hemodynamics or eliminate the chronic volume overload on the RV, the long-term effects of which remain uncertain. Consequently, CAVI is selectively indicated for highly symptomatic, end-stage patients who are ineligible for TV repair or replacement due to prohibitive surgical risks or complex anatomical considerations.
Future Directions and Conclusion
Transcatheter TV intervention has rapidly evolved from a niche concept to a mainstream therapy. T-TEER is a safe and proven option for improving the quality of life in anatomically suitable patients. Annuloplasty offers a surgical-like approach for select anatomies. Transcatheter tricuspid valve replacement provides a powerful tool that offers near-complete TR elimination for patients with more complex disease, with randomized data now supporting its clinical benefits. Finally, CAVI provides a crucial palliative option for those with no other interventional path.
The role of TTVI in the Indian context is shaped by a distinct disease landscape. Unlike in Western nations, severe TR in India is predominantly caused by RHD, which presents unique anatomical challenges, such as leaflet fibrosis and thickening.36, 37 Current TTVI technologies, developed for functional TR, are often unsuitable for this complex rheumatic anatomy. Consequently, open-heart surgery remains the standard of care for severe rheumatic tricuspid disease. Transcatheter tricuspid valve intervention is currently reserved for a highly selective group of inoperable patients with non-rheumatic TR, with its broader adoption limited by high costs. 37
Future research will focus on optimizing patient selection, refining device technology to minimize complications, and conducting head-to-head trials comparing different TTVI modalities. The ultimate goal is to intervene earlier, before the onset of irreversible right heart failure, and to select the right therapy for the right patient at the right time. The rediscovery of the “forgotten valve” holds promise for a brighter future for patients with TR.
Footnotes
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the relevant ethics committee or Institutional Review Board (IRB).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The participant has provided patient consent for the submission of the article to the journal.
