Abstract
Right bundle branch block remains an intriguing cardiac conundrum, presenting challenges in diagnosis, prognostication, and management. Despite being a relatively common electrocardiographic finding, its clinical significance and underlying pathophysiology continue to perplex clinicians and researchers. This case series aims to delve into the enigmatic nature of right bundle branch block, exploring its epidemiology, etiology, diagnostic modalities, clinical implications, and therapeutic considerations. Through a comprehensive review of current literature and emerging insights, we endeavor to shed light on this intriguing cardiac phenomenon and pave the way for enhanced understanding and management strategies.
Keywords
Introduction
Right bundle branch block (RBBB) represents an electrocardiographic anomaly characterized by a delay or a block of conduction within the right bundle branch of the heart’s conduction system. Right bundle branch block is classified according to the Minnesota Code criteria into complete RBBB (prevalence 0.9%-1.5%) 1 when QRS duration >120 ms, and incomplete RBBB (prevalence 3%-4.7%) 2 when QRS duration 100-120 ms. Right bundle branch block is characterized by an rsR pattern in lead V1 with R > r, a slurred S wave in lead I and V6, and a QRS duration >120 ms. Right bundle branch block is often considered benign, but it may signify underlying cardiac pathology and pose challenges in clinical decision-making. Despite its prevalence, controversies persist regarding its clinical significance, optimal diagnostic approaches, and management strategies. This article aims to provide a comprehensive overview of RBBB, addressing its various pathophysiologies, clinical presentation, diagnostic techniques, and therapeutic considerations, thereby unraveling the enigma surrounding this intriguing cardiac phenomenon.
Right bundle branch block is often considered benign, but it can serve as a marker of underlying cardiac pathology and confer an increased risk of adverse cardiovascular events, including heart failure, arrhythmias, and sudden cardiac death. Furthermore, RBBB can complicate the interpretation of electrocardiograms (ECGs) in patients presenting with chest pain, syncope, or other cardiac symptoms, necessitating a thorough clinical evaluation. Right bundle branch block has been associated with certain cardiovascular risk factors and comorbidities, including hypertension (HTN), coronary artery disease, and structural heart disease.
Case 1
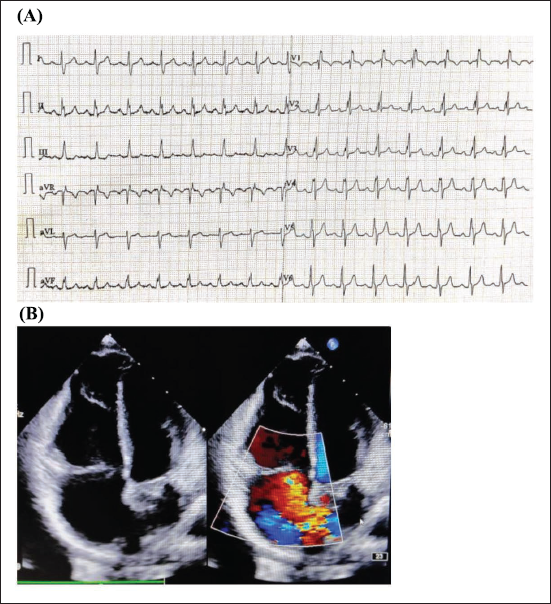
A 33-year-old male presented with complaints of palpitations and effort intolerance. Electrocardiogram showed incomplete RBBB with right-axis deviation and notching of R waves in inferior leads (Figure 1). Echocardiography (ECHO) showed underlying ostium secundum—atrial septal defect (ASD).
(A) Electrocardiogram Shows Right Axis Deviation, Incomplete Right Bundle Branch Block (RBBB), and Characteristic Notching of R Wave in Inferior Leads (Crochetage Sign), Which has High Specificity for Secundum Atrial Septal Defect (ASD). (B) Echocardiography with Color Doppler Shows Ostium Secundum-ASD (OS-ASD) with Left to Right Shunt.
Case 2
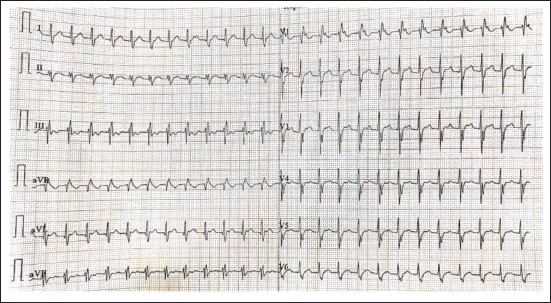
A 24-year-old male with complaints of palpitation and dizziness came to the casualty department. On examination, the patient was conscious and oriented with a heart rate (HR) of 180 bpm; blood pressure (BP): 122/74 mmHg; no murmurs on auscultation. Electrocardiogram showed incomplete RBBB with an HR of 172 bpm (Figure 2). A diagnosis of supraventricular tachycardia (SVT) was considered, and injection adenosine was administered. There was no resolution of symptoms or HR. Echocardiogram showed no structural abnormalities. Beta-blockers were administered, but the symptoms persisted. In view of RBBB morphology, fast ventricular rate, and non-responsiveness to medications given for SVT, the possibility of fascicular ventricular tachycardia (VT) was considered. The patient was given verapamil and responded to it. After initial stabilization, an electrophysiology study was done. The left ventricle was entered anterogradely via the transseptal approach, and activation mapping was performed during VT. The earliest ventricular activation was at the base of the posterior papillary muscle. Pace mapping from the posterior papillary muscle region showed a 12/12 match. Ablation was done with a 4 mm tip irrigated catheter (45°, 40 W) at the earliest site, which terminated the VT within 10 s. Two pulses of around 120 s were delivered. Post radiofrequency ablation (RFA), there was no inducible tachycardia despite isoproterenol infusion and aggressive pacing protocols. The ectopic origin was successfully ablated. On follow-up, there is no recurrence.
Electrocardiogram Shows Narrow QRS Complex Tachycardia with Heart Rate (HR) of 180 bpm and Incomplete Right Bundle Branch Block (RBBB) Pattern.
Case 3
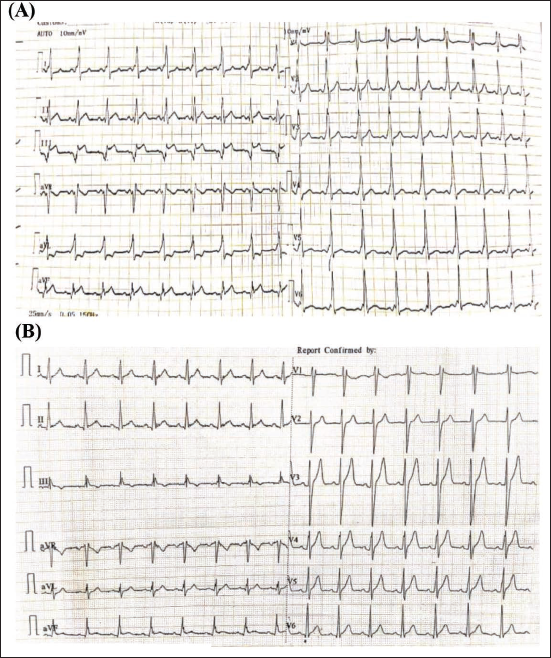
A 30-year-old male presented with complaints of chest discomfort and palpitations for 2 months. The patient had been reviewed in an outside hospital with the above complaints and ECG changes, and he was advised to undergo a coronary angiogram. Coronary angiogram revealed normal epicardial coronaries. In view of the persisting symptoms, the patient came to our hospital. On examination, the patient was hemodynamically stable. Electrocardiogram showed the HR of 102 bpm, RBBB pattern involving V1, ST-segment elevation in III, aVF, and II. On careful analysis, delta waves were noted in all the leads (Figure 3A). A diagnosis of Wolff–Parkinson–White (WPW) syndrome, in the left posterolateral pathway, was considered. Electrophysiology study was done, which was suggestive of an accessory pathway in the posterolateral wall, which was successfully ablated with RFA. Post-procedure ECG showed normal sinus rhythm (Figure 3B).
(A) Electrocardiogram Shows Right Bundle Branch Block (RBBB) Pattern in V1. Q Waves with ST-segment Elevation in III, aVF, and II. Short PR Interval, Delta Waves in All Leads. (B) Post Radiofrequency Ablation (RFA) of Accessory Pathway Shows Resolution of Delta Waves but Persistence of RBBB Pattern.
Case 4
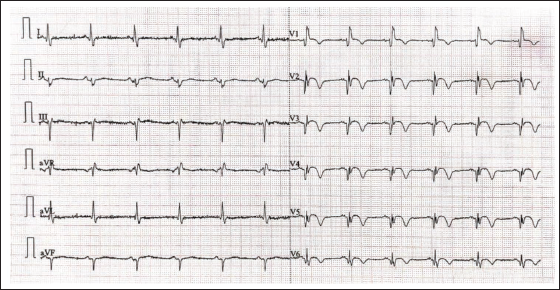
A 60-year-old male K/C/O HTN presented with chest pain. His ECG showed qualitative RBBB (qRBBB) with ST-segment elevation in V1-V6 (Figure 4). Echocardiography was done, which showed hypokinetic mid and apical, anteroseptum, apex, anterior, and anterolateral wall. Ejection fraction (EF): 43%, Grade I left ventricular diastolic dysfunction (LVDD), mild mitral regurgitation (MR), trivial tricuspid regurgitation (TR), pulmonary artery systolic pressure (PASP) 30 mmHg. Patient was taken up for coronary angiography (CAG), which showed multivessel disease. Percutaneous transluminal coronary angioplasty (PTCA) and stenting were done to left anterior descending (LAD), left circumflex (LCX), and right coronary artery (RCA). Post-procedure, there were no new ECG changes. New-onset RBBB is suggestive of underlying multivessel coronary artery disease.
Sinus Rhythm with Heart Rate (HR): 68 bpm, Regular, Left Axis Deviation, Qualitative Right Bundle Branch Block (qRBBB) in V1-V4 with ST-segment Elevation in V1-V6, Left Anterior Fascicular Block.
Case 5
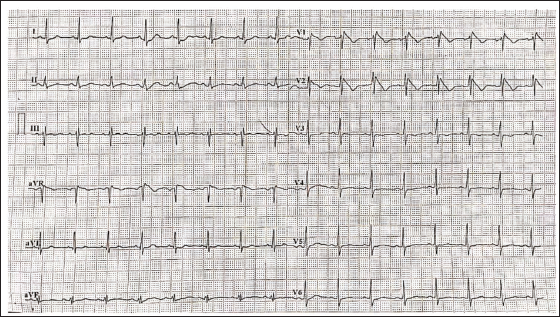
A 22-year-old male presented with two episodes of syncope over the past month. He was referred from the Department of Otorhinolaryngology for further cardiac evaluation. Baseline electrocardiography revealed an RBBB pattern with coved-type ST-segment elevation exceeding 2 mm in leads V1 and V2, suggestive of a type 1 Brugada pattern (Figure 5). Transthoracic ECHO showed a structurally normal heart. The patient reported a family history of sudden cardiac death: his father had died suddenly at 41 years of age while working in the field. Continuous Holter monitoring was performed; however, no sustained arrhythmias were documented during the recording period. Based on the clinical presentation, ECG findings, and family history, a diagnosis of Brugada syndrome was established. The patient was counseled regarding his risk profile and was advised to undergo implantation of an implantable cardioverter-defibrillator (ICD) for primary prevention of sudden cardiac death.
The Electrocardiogram (ECG) Demonstrates Sinus Rhythm with Right Bundle Branch Block (RBBB) Morphology, Characterized by rsR’ Pattern in Lead V1. There is Coved-type ST-segment Elevation Exceeding 2 mm in Leads V1 and V2 with Associated T-wave Inversion, Consistent with a Type 1 Brugada ECG Pattern.
Case 6
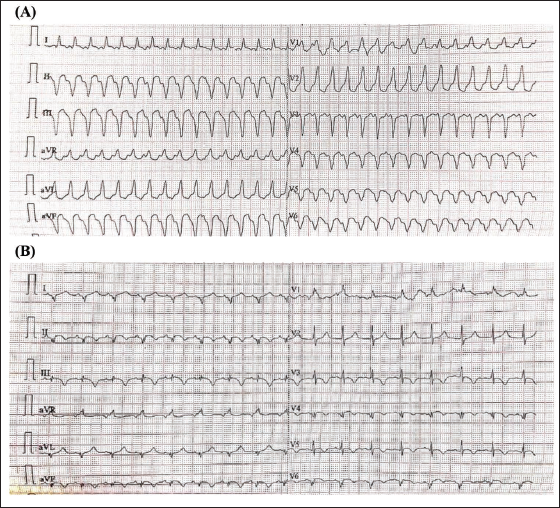
A 36-year-old male with a history of ischemic heart disease and a prior lateral wall myocardial infarction (MI) was previously treated with percutaneous coronary intervention (PCI), involving the drug-eluting stent placement in the LCX artery for single-vessel disease. Two weeks post-procedure, the patient developed intermittent palpitations, dizziness, and a single episode of syncope. He subsequently presented to the emergency department with sustained palpitations and hypotension. Electrocardiography at presentation (Figure 6A) revealed a broad complex tachycardia with RBBB morphology, consistent with monomorphic VT. Immediate electrical cardioversion was performed using synchronized direct current (DC) shock at 200 joules, restoring sinus rhythm. Post-cardioversion ECG demonstrated pathological Q waves in leads I and aVL, indicative of prior lateral wall infarction. A clinical diagnosis of scar-related VT involving the LCX territory was established. The patient underwent electrophysiological study with 3D electroanatomic mapping, which localized the arrhythmic focus to the epicardial region of the left ventricular lateral wall. Successful radiofrequency catheter ablation was performed. At follow-up, the patient remained asymptomatic with no further arrhythmic episodes.
(A) 12-Lead Electrocardiogram (ECG) Recorded During Presentation with Palpitations and Hypotension. The ECG Shows a Regular, Wide QRS Complex Tachycardia at a Rate of Approximately 160-170 Beats Per Minute. The QRS Complexes Exhibit a Right Bundle Branch Block (RBBB) Morphology with a Broad QRS Duration and Predominantly Positive Complexes in Lead V1, Consistent with Monomorphic Ventricular Tachycardia. (B) 12-Lead ECG Following Successful Electrical Cardioversion. The ECG Shows Sinus Rhythm with Persistent RBBB Pattern and Pathological Q Waves in Leads I and aVL, Consistent with Prior Lateral Wall Myocardial Infarction. No Acute ST-segment Changes.
Discussion
Incomplete RBBB is often an incidental finding in healthy individuals, with a prevalence of 3%-4.7%. This form of RBBB is relatively common in the general population, especially among those with high vagal tone or athletic training. In athletes, RBBB is considered a normal adaptation rather than a pathological finding. 3 The ST-segment and T-wave changes are minimal or absent in this benign form. Physiological RBBB does not require specific treatment or intervention. Regular follow-up and assessment are usually sufficient if there are no symptoms or other ECG abnormalities.
In ostium secundum-ASD (OS-ASD), RBBB may appear alongside right axis deviation, right atrial enlargement, and sometimes incomplete RBBB. The RBBB pattern may be subtle, but it is often accompanied by other signs of ASD-related right heart strain. The right ventricular enlargement due to chronic volume overload can disrupt normal conduction pathways, leading to RBBB. This is particularly common in older children and adults with uncorrected ASD. 4 The primary treatment involves closure of the ASD, which can be done surgically or percutaneously.
Fascicular VT originates from the Purkinje fibers or ventricular fascicles, leading to a distinctive RBBB pattern due to the altered activation of the right ventricle. Papillary muscle VT arises from the papillary muscles and can also present with an RBBB pattern with complex morphology that differs from classic RBBB. Differentiating fascicular or papillary muscle VTs from true RBBB is crucial for appropriate treatment. Fascicular VT may require specific antiarrhythmic treatment or catheter ablation. 5 Catheter ablation is often effective for fascicular VTs, while papillary muscle VTs may require treatment of the underlying cause, such as myocardial ischemia. Apart from fascicular VT, broad QRS complex VT presenting with an RBBB morphology may arise in various substrates, including ischemic cardiomyopathy and arrhythmogenic right ventricular cardiomyopathy (ARVC). Differentiating VT from SVT with aberrancy requires careful ECG analysis—looking for capture beats, fusion beats, atrioventricular (AV) dissociation, and extreme axis deviation. Figure 6A demonstrates such a VT with broad RBBB morphology, taken from our case collection.
Wolff–Parkinson–White syndrome is characterized by an accessory conduction pathway that can lead to pre-excitation of the ventricles. The presence of RBBB can complicate the interpretation of ECG findings. Wolff–Parkinson–White typically shows a delta wave and a short PR interval, but when combined with RBBB, the ECG can display a complex pattern where the RBBB pattern may obscure or be combined with the pre-excitation pattern. Right bundle branch block in WPW can make the diagnosis of arrhythmias more challenging. It is crucial to evaluate potential rapid arrhythmias such as atrial fibrillation.6, 7 Treatment usually involves catheter ablation of the accessory pathway. If arrhythmias persist, or if there is a high risk of rapid atrial fibrillation, more aggressive intervention may be required.
Right bundle branch block may occur in the setting of acute coronary syndrome (ACS) and may be indicative of significant myocardial stress or injury. Right bundle branch block in the context of ACS can complicate the interpretation of ST-segment changes, which are critical for diagnosing MI. The presence of RBBB can obscure or mimic ST-segment elevation or depression. 8 Right bundle branch block can make the diagnosis of ST-elevation myocardial infarction (STEMI) or non-ST-elevation myocardial infarction (NSTEMI) more difficult because it can mask or mimic ST-segment changes. It is essential to use other diagnostic criteria and imaging studies to confirm the diagnosis. 9 The treatment of ACS with RBBB includes urgent coronary intervention, such as thrombolysis or PCI, based on the specific type and severity of ACS.
Brugada syndrome is a primary electrical disease characterized by a coved-type ST-segment elevation in leads V1-V3, often accompanied by an RBBB-like pattern. 10 However, the presence of RBBB morphology should not lead to misclassification, as true RBBB is not required for the diagnosis of Brugada syndrome. This distinction is clinically important, particularly in young individuals with syncope or a positive family history, as Brugada syndrome is associated with a risk of sudden cardiac death due to ventricular arrhythmias. Misinterpreting Brugada pattern as benign RBBB could delay appropriate risk stratification and therapy, including ICD placement. 11
Broad QRS complex tachycardia with RBBB morphology most commonly represents monomorphic VT, especially in patients with structural heart disease such as prior MI. In this case, the patient presented with ischemic scar-related VT involving the LCX territory, evident from both clinical history and electrophysiology mapping. The differential diagnosis for a broad complex tachycardia with RBBB pattern includes: (a) VT arising from the left ventricle, as seen in our case; (b) SVT with aberrant conduction (rate-related RBBB); and (c) antidromic AV re-entrant tachycardia in pre-excitation syndromes. Distinguishing VT from SVT with aberrancy is clinically critical, as mismanagement can have serious consequences. Classical ECG clues favoring VT include AV dissociation, fusion beats, capture beats, extreme axis deviation, and concordance across precordial leads. 12 In ischemic cardiomyopathy, VT circuits are typically re-entry-based and often originate from scarred myocardial regions. 13 This case underscores the importance of early recognition and management of scar VT in post-infarction patients. Radiofrequency catheter ablation guided by 3D electroanatomic mapping is an established therapeutic option, particularly when arrhythmia origin is localized to the epicardial surface.
Conclusion
Understanding the diverse presentations of RBBB is crucial for accurate diagnosis and effective management. Right bundle branch block can appear in various contexts, from benign physiological variants to structural heart disease, such as secundum ASD, accessory pathway such as WPW syndrome, or life-threatening complex arrhythmias such as VT or CAD. Each presentation has unique implications for clinical practice, and appropriate interpretation of ECG findings is essential for optimal patient care.
Footnotes
Authors Contribution
Abhishek Samdesi C N: Conceptualization, methodology, data curation, formal analysis, investigation, validation, writing—original draft, writing—reviewing and editing; Pradeep Kumar K.: Conceptualization, methodology, data curation, formal analysis, validation, writing—reviewing and editing; Navya C N: Formal analysis, validation, writing—original draft; Asha Latha: Validation, writing—reviewing and editing; Prakash S S: Validation, writing—reviewing and editing. All authors approved the final manuscript.
Data Availability
No datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest regarding the research, authorship, and/or publication of this article.
Ethical Approval
Based on the regulations of the review board of the Sapthagiri Institute of Medical Sciences & Research Centre, Bengaluru, Karnataka, India, institutional review board approval is not required for case reports or case series.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
Written informed consent had been obtained from the patient to have the case details and any accompanying images published.
