Abstract
Atrial fibrillation (AF) secondary to organophosphate poisoning is rare but can be life-threatening. A 53-year-old female with no prior cardiac history presented to the emergency department with altered mental status, excessive salivation, vomiting, and respiratory distress following ingestion of an unknown quantity of 35% emulsified endosulfan pesticide. Electrocardiography initially revealed sinus bradycardia, but within hours, the patient developed rapid, irregular tachycardia with no discernible P waves. Cholinergic toxicity was confirmed based on clinical presentation and reduced serum cholinesterase levels. The patient was treated with atropine for cholinergic symptoms and pralidoxime to reactivate acetylcholinesterase. Beta-blockers and calcium channel blockers were avoided due to concerns about exacerbating hypotension. Instead, rate control was achieved with cautious administration of amiodarone. Supportive measures, including intravenous fluids and oxygen therapy, were provided. Sinus rhythm was restored within 36 h of initiating treatment. The patient showed progressive clinical improvement, with resolution of atrial fibrillation and normalization of serum cholinesterase levels. She was discharged on day 8 with no residual cardiac dysfunction. At the 1-month follow-up, the electrocardiography remained normal, and she had no recurrent arrhythmias. Acute atrial fibrillation can be a rare but severe complication of organophosphate poisoning, likely due to autonomic imbalance, electrolyte disturbances, and direct toxic effects on the myocardium.
Introduction
Organophosphate (OP) poisoning, primarily resulting from pesticide exposure, is a well-documented cause of autonomic dysfunction, neuromuscular impairment, and cardiovascular complications. 1 However, acute atrial fibrillation (AF) as a direct consequence of OP poisoning is a rare clinical manifestation. AF is an unusual cardiac manifestation of OP poisoning, often triggered by autonomic dysfunction and excessive vagal stimulation. 2 OPs exert their toxic effects by irreversibly inhibiting acetylcholinesterase, leading to an excessive accumulation of acetylcholine at synapses. 3 This cholinergic overactivity can precipitate a spectrum of cardiovascular abnormalities, including bradyarrhythmias, QT prolongation, ventricular arrhythmias, and, in rare cases, AF.
The underlying mechanisms may involve autonomic nervous system imbalance, direct myocardial toxicity, electrolyte disturbances, and oxidative stress. 4 While AF is often associated with structural heart disease, metabolic disturbances, and systemic inflammation, toxicological causes are less frequently reported. Acute cardiac arrhythmias, ranging from bradycardia to AF, are common in OP poisoning due to excessive cholinergic stimulation. 2 Diagnosis relies on clinical history, electrocardiography (ECG) findings, and tests like serum cholinesterase levels. 5 Treatment involves reversing poisoning with atropine and pralidoxime (2-PAM), while managing arrhythmias with rate control or electrical cardioversion as needed. 6 This case report describes a rare presentation of acute AF triggered by OP poisoning. The report highlights the clinical features, diagnostic approach, and management strategies employed, emphasizing the need for early recognition and intervention to prevent potential complications.
Case Presentation
The patient was a 53-year-old female stay-at-home mom who was brought to the emergency department by neighbors who found her unconscious. She was last seen in stable condition the previous evening. She had ingested an unknown quantity of a 35% emulsified concentration of endosulfan pesticide approximately 4.5 h prior to arrival. Upon arrival, she exhibited profound cholinergic toxicity, including miosis, hypersalivation, lacrimation, muscle fasciculations, bradycardia followed by tachyarrhythmia, hypertension transitioning into hypotension, and respiratory distress. Her symptoms worsened rapidly, and an irregularly irregular tachycardia was detected. She had no history of hypertension, diabetes, coronary artery disease, or thyroid dysfunction. She had never been diagnosed with arrhythmias or structural heart disease. She had no previous episodes of poisoning or psychiatric illnesses, was not taking any regular medications, and had no known drug allergies. There was no history of cardiovascular diseases, arrhythmias, or sudden cardiac death in her family. No familial predisposition to pesticide toxicity was reported. The patient had been living alone after divorcing her husband. She had no known history of smoking, alcohol use, or illicit drug use. However, she had been experiencing significant financial difficulties and psychological distress, raising concerns about possible self-harm. She had no occupational exposure to pesticides.
On general examination, the patient appeared disoriented, restless, and in respiratory distress. Her skin was cold and clammy, with increased sweating. Her vital signs revealed a blood pressure of 97/65 mmHg, a heart rate of 129 beats per minute with an irregular rhythm, a respiratory rate of 25 breaths per minute, an oxygen saturation of 87% on room air, and a body temperature of 36.8°C. Her weight and height were 64 kg and 1.71 m, respectively. Neurological examination showed altered mental status, muscle twitching, and hyperreflexia. Cardiovascular examination revealed an irregularly irregular pulse, no murmurs, and mild peripheral edema. Respiratory examination showed bilateral crackles, suggesting pulmonary congestion, along with signs of mild respiratory distress. Gastrointestinal examination revealed abdominal tenderness and hyperactive bowel sounds.
A range of biochemical, hematological, and toxicological tests was performed to evaluate systemic involvement and confirm the diagnosis. The patient’s white blood cell count was elevated at 18,300/mm3/mm³. Hemoglobin (12.9 g/dL) and platelet count (182,600/mm³) were within normal limits. The patient’s sodium level was 137 mmol/L (normal range: 135-145 mEq/L), and magnesium was 2.1 mg/dL (normal range: 1.7-2.2 mg/dL), both within normal limits. Potassium was 2.8 mEq/L (normal range: 3.5-5.0 mEq/L), which is low and considered critical. Serum creatinine (1.0 mg/dL) and blood urea nitrogen (16 mg/dL) were also within normal limits. The patient’s aspartate aminotransferase (AST) was 41 U/L, and alanine aminotransferase (ALT) was 36 U/L. Total bilirubin and alkaline phosphatase levels were normal. The results showed a pH of 7.14, partial carbon dioxide of 59 mmHg, and bicarbonate of 11 mmol/L. The patient’s cholinesterase level was 2,430 U/L (reference range: 5,000-12,000 U/L). The patient’s troponin I was 0.07 ng/mL (reference range <0.04 ng/mL), and creatine kinase-MB was within normal limits. The patient’s thyroid-stimulating hormone was 1.9 mIU/L, and free T4 was 1.6 ng/dL. The toxicological results were positive for OP compounds, specifically endosulfan. The patient’s prothrombin time, activated partial thromboplastin time, and international normalized ratio were all within normal ranges. The patient’s random blood glucose was 89 mg/dL, within the normal range.
Electrocardiography confirmed AF with a rapid ventricular response, with no evidence of ST-segment elevation or ischemic changes. QT prolongation was noted. Chest X-ray showed pulmonary congestion without signs of aspiration pneumonia. Echocardiography demonstrated normal left ventricular function with no structural abnormalities. A brain computed tomography (CT) scan was performed to rule out stroke due to the new-onset AF; however, it showed no acute ischemic changes or evidence of bleeding. Based on the clinical presentation, laboratory findings, and ECG results, the patient was diagnosed with acute AF secondary to OP poisoning due to endosulfan ingestion.
The patient was immediately admitted to the intensive care unit for close monitoring and supportive management. Airway protection was ensured, and she was placed on oxygen therapy via nasal cannula at 4 L/min. Due to respiratory distress, she was subsequently intubated and mechanically ventilated. She received 20 mL/kg of intravenous fluids (normal saline) to maintain hemodynamic stability. Atropine was given as a 2 mg IV bolus, repeated every 5 min until secretions decreased, followed by a continuous infusion of 0.03 mg/kg/h for 1 h. 2-PAM was administered at an initial dose of 1 g IV over 30 min, followed by a continuous infusion of 0.5 g/h for 24 h. She received potassium chloride intravenously at a rate of 10 mEq/h for 12 h. Once her serum potassium reached ≥3.0 mEq/L, the treatment was transitioned to 10 mEq of oral potassium supplementation once daily. The oral supplementation was discontinued after her hypokalemia was fully corrected. Beta-blockers and calcium channel blockers were avoided due to concerns about exacerbating hypotension. For AF, she received an intravenous amiodarone bolus of 150 mg over 10 min, followed by a continuous infusion of 1 mg/min for the next 6 h for rate control. Amiodarone was administered after her hypokalemia was corrected, with her potassium levels transitioning from 2.8 to 3.0 mEq/L. Because of the severity of the poisoning, rhythm control was initially deferred, and electrical cardioversion was withheld. Anticoagulation is generally not recommended for her, as the risk of stroke is minimal based on a CHA2DS2-VASc score of 1/10 (Table 1). In this patient, a score of 1 is considered low risk for stroke, and anticoagulation is typically not indicated. She had no additional risk factors that would significantly elevate her stroke risk, suggesting that the AF was likely secondary and transient. She received diazepam prophylactically to prevent seizures.
CHA2DS2-VASc Scoring System for Withholding and Recommending Anticoagulation in Atrial Fibrillation.

The patient remained in the ICU for 60 h, during which she showed gradual improvement. Spontaneous conversion to normal sinus rhythm occurred after 36 h without requiring electrical cardioversion. By day 2, she was weaned off ventilatory support and transferred to the inpatient ward. A psychiatric evaluation was conducted, revealing severe emotional distress but no underlying psychiatric disorder. She was discharged on day 8 with appropriate counseling and monthly follow-up with a cardiologist and a psychiatrist.
Table 1 is interpreted as follows: A score of 0 in men or 1 in women indicates low risk, so anticoagulation is usually not needed. A score of 1 in men or 2 in women indicates moderate risk, and anticoagulation may be considered. A score of 2 or more in men or 3 or more in women indicates high risk, and anticoagulation is recommended.
Discussion
Organophosphate poisoning primarily affects the nervous system through excessive cholinergic stimulation. Cardiovascular manifestations, though less common, include arrhythmias, conduction abnormalities, and myocardial dysfunction. 7 Acute AF caused by OP poisoning is a rare but significant complication. 8 OPs like endosulfan, a widely used pesticide, inhibit acetylcholinesterase, leading to the accumulation of acetylcholine at nerve synapses and overstimulation of muscarinic and nicotinic receptors. 9 This disruption in neurotransmission can result in a range of clinical manifestations, including cardiovascular arrhythmias such as AF. The typical clinical manifestations of acute OP poisoning include muscarinic effects such as excessive salivation, sweating, nausea, vomiting, and bradycardia, as well as nicotine-like effects such as muscle twitching, fasciculations, and weakness. Central nervous system (CNS) symptoms may range from confusion to seizures or coma. 10 In this case, the 53-year-old woman, after ingesting the pesticide, likely presents with classical cholinergic and CNS symptoms, along with AF. This arrhythmia may be exacerbated by underlying electrolyte imbalances (e.g., hypokalemia) and acidosis, which are relatively rare in poisoning scenarios.
Previous case reports of OP poisoning rarely emphasize AF as a primary clinical manifestation. Typically, OP poisoning leads to muscarinic and nicotinic symptoms (e.g., bradycardia, miosis, salivation, and muscle weakness). 11 However, cases of arrhythmias, including AF, are documented, though less common, and might result from severe poisoning or concurrent comorbidities. Typically, OP poisoning leads to symptoms like nausea, vomiting, abdominal pain, and, in severe cases, respiratory distress. 5 However, this peculiar case also presents with acute AF, which may be related to electrolyte imbalances, hypoxia, or autonomic dysfunction caused by the poisoning. OP poisoning primarily works by inhibiting acetylcholinesterase, leading to the excessive buildup of acetylcholine. 2 This overstimulation of muscarinic receptors in the heart results in an increase in vagal tone, which typically causes bradycardia. However, direct myocardial toxicity and resultant disturbances in electrolyte homeostasis can predispose the heart to arrhythmias, including AF. Altered action potentials, particularly from disruptions in calcium and potassium channels, contribute to the development of AF. 12
Atrial fibrillation can manifest in three main forms: paroxysmal, persistent, and permanent. 13 Paroxysmal AF occurs in episodes that resolve spontaneously, while persistent AF requires medical intervention for conversion to sinus rhythm. Permanent AF refers to a chronic state where the arrhythmia persists despite attempts at cardioversion. In the case of OP poisoning, the AF is likely paroxysmal, directly linked to the acute toxicity of the pesticide, although it may progress to persistent or permanent AF if left untreated. In this case report, the patient experienced paroxysmal AF after ingesting endosulfan OP. While bradycardia is more commonly observed in OP poisoning due to parasympathetic overdrive, acute AF has been reported in some severe cases. 11 It is generally considered a rare complication but can occur in individuals with severe poisoning or specific risk factors such as age, heart disease, or electrolyte disturbances. The patient developed acute AF, which is a transient arrhythmia that might occur during or after poisoning. The mechanism could involve both direct effects on the heart’s electrical system from acetylcholine and secondary effects from metabolic disturbances.
Organophosphate poisoning typically leads to bradycardia due to the excessive stimulation of the parasympathetic nervous system. 7 However, more severe cases may result in dysrhythmias such as AF, often in individuals with predisposing conditions like electrolyte imbalances or underlying cardiac disease. OPs like endosulfan inhibit acetylcholinesterase, leading to the accumulation of acetylcholine at synaptic clefts (Figure 1). OPs inhibit acetylcholinesterase, leading to excessive acetylcholine, which can trigger parasympathetic overactivity and potentially disrupt cardiac conduction, leading to AF. 4 Oxidative stress and electrolyte imbalances, especially hypokalemia and hypomagnesemia, could further contribute to arrhythmias. 14 This causes overstimulation of both muscarinic and nicotinic receptors, disrupting autonomic control over the heart and possibly precipitating AF. Additionally, the imbalance in electrolytes, hypoxia, and acidosis resulting from severe poisoning can trigger arrhythmias.
Pathophysiology of Acute Atrial Fibrillation (AF) Caused by Organophosphate Poisoning.
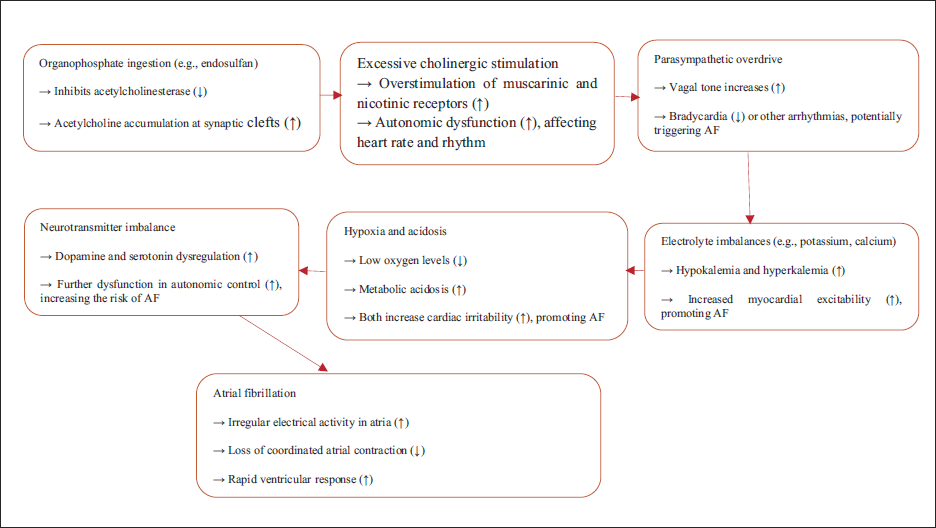
The diagnosis of AF in the setting of OP toxicity requires a high index of suspicion and timely recognition of other poisoning symptoms. An ECG is essential for confirming the presence of AF, which is characterized by an irregularly irregular rhythm and the absence of P waves. 15 The diagnosis of OP poisoning is largely clinical and requires a detailed history of pesticide exposure, such as ingestion of endosulfan. Laboratory tests, such as serum cholinesterase levels, can confirm exposure to OPs, while arterial blood gases can assess acid–base disturbances and electrolyte imbalances.
Standard treatment includes atropine to counteract parasympathetic effects. If arrhythmias, such as AF, are detected, pharmacologic agents like amiodarone or electrical cardioversion may be used. 16 However, most case reports typically focus on treating the poisoning rather than specifically managing AF, unless it becomes life-threatening. Treatment would involve the usual management of OP poisoning, including atropine to reverse muscarinic effects and 2-PAM to reactivate acetylcholinesterase. Management of AF in some cases would require rate control (e.g., beta-blockers or calcium channel blockers) along with careful monitoring of electrolytes and oxygenation. 17 The prognosis in severe cases of OP poisoning with arrhythmias largely depends on early diagnosis and treatment. Patients with AF due to OP poisoning generally have a more favorable prognosis, though the risk of sudden death or other complications from the underlying toxicity or arrhythmia still exists. The prognosis in this case depends on the severity of the poisoning and the speed of intervention; it was favorable because the patient recovered quickly. If treated promptly with appropriate antidotes and supportive care, recovery from both poisoning and AF is possible. 18 However, the presence of AF may increase the risk of complications, such as stroke or hemodynamic instability.
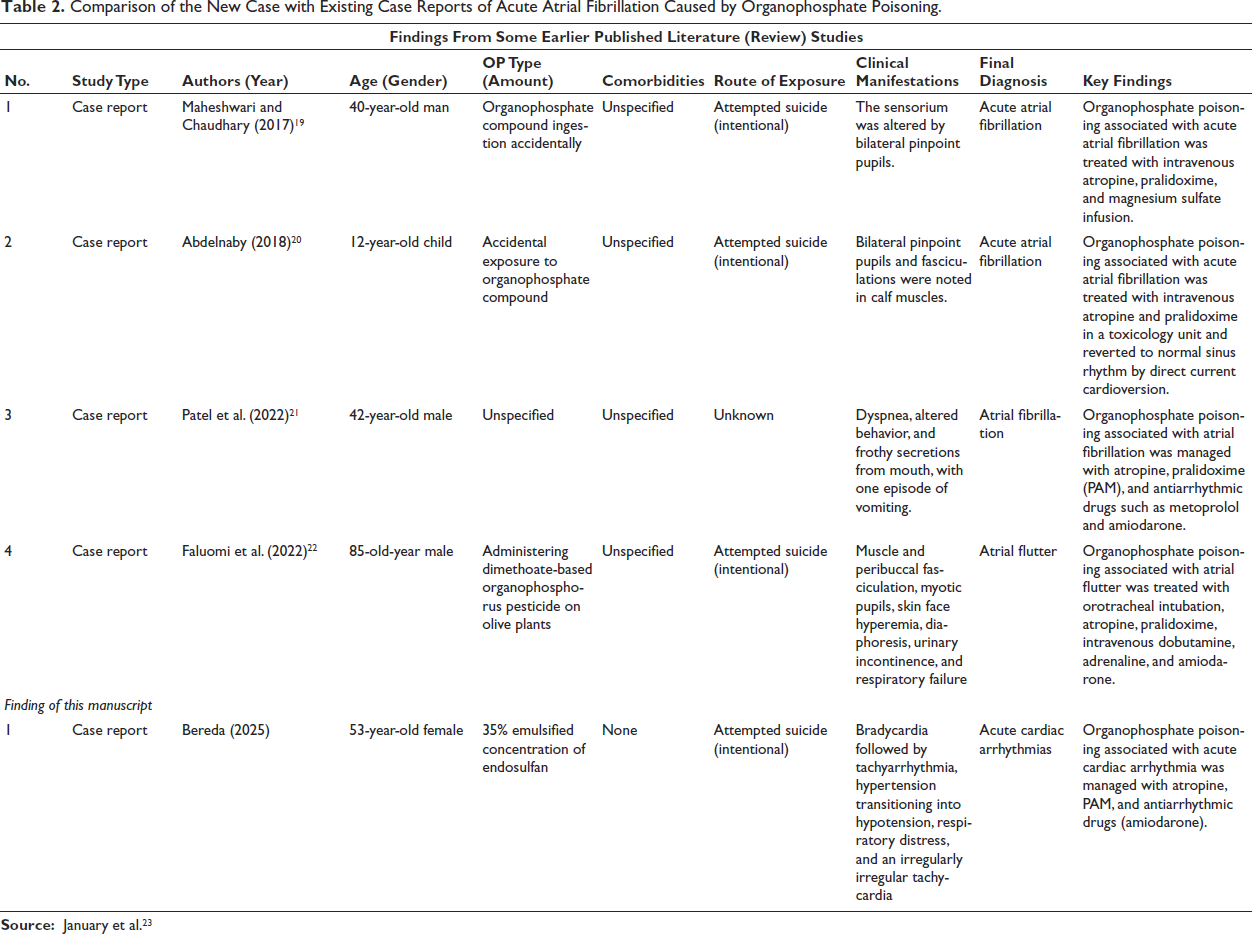
The new case report on OP poisoning compares an unusual presentation with acute AF to many existing reports that typically describe AF alongside clinical manifestations like bradycardia, tachycardia, or QT prolongation. While both cases involve cholinergic symptoms managed with atropine (0.02-0.08 mg/kg/h) and 2-PAM (10-20 mg/kg/h), the AF case required higher atropine doses and additional rate control measures. This case underscores the need for tailored cardiac interventions in OP poisoning and reinforces the importance of continuous cardiac assessment. Existing literature regarding AF typically uses beta-blockers or calcium channel blockers for rate control; however, in this case report, the patient received amiodarone cautiously to avoid exaggerated hypotension (Table 2).
Comparison of the New Case with Existing Case Reports of Acute Atrial Fibrillation Caused by Organophosphate Poisoning.
Strengths and Limitations of the Case Report
The report highlights an uncommon complication of OP poisoning, specifically the onset of acute AF, adding new insight into its potential cardiovascular effects. The case report offers a detailed exploration of the mechanisms through which OP toxicity can contribute to arrhythmias, particularly AF, involving autonomic dysfunction, electrolyte imbalances, and other metabolic disturbances. As a single case report, the findings are limited in terms of generalizability to the broader population. The report does not provide long-term follow-up on the patient’s recovery, including the persistence or recurrence of AF after initial treatment.
Key Points
This case presents a unique and clinically significant manifestation of acute OP poisoning with the development of AF, a rarely reported complication. The patient’s AF developed in the absence of underlying cardiovascular disease or structural heart abnormalities, indicating a direct toxic effect of the OP compound on cardiac conduction. Importantly, this case demonstrates that OP-induced AF can be transient and reversible with appropriate antidotal and supportive therapy.
Conclusion
Organophosphate poisoning can trigger acute AF, highlighting the need for careful cardiovascular monitoring in such cases. The development of AF in this context is likely multifactorial—driven by autonomic dysfunction, direct myocardial toxicity, oxidative stress, and associated metabolic derangements such as hypokalemia and acidosis. Early intervention, including the use of atropine and other supportive treatments, is crucial for preventing severe complications like AF and ensuring better patient outcomes. In this case, due to the patient’s hemodynamic instability and risk of exacerbated hypotension, commonly used rate control agents like beta-blockers and calcium channel blockers were avoided. Instead, amiodarone was carefully selected, marking a deviation from standard protocols and highlighting the need for individualized treatment in toxicological emergencies.
Footnotes
Author’s Contributions
GB: Conceptualization, curating the data, investigation, supervision, validation, visualization, writing – original draft, critical review and editing of the manuscript. The author read and approved the final manuscript.
Data Availability Statement
Further details about the report can be made available by request.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from the patient for anonymized patient information to be published in this article.
