Abstract
We report about a middle-aged male patient with rheumatic heart disease who had undergone an older generation tilting disc prosthetic aortic valve (Björk–Shiley valve) implantation 36 years back. He presented with severe mitral stenosis and a normal functioning prosthetic aortic valve. This case may be one of the longest reported functioning Björk–Shiley valve at aortic position, associated with an uneventful course over four decades. Patients with durable older generation prosthetic heart valves may be encountered in clinical practice and need to be managed in line with current guidelines.
Keywords
Introduction
A 48-year-old male patient presented to the outpatient department with symptoms of exertional dyspnea for a few months. He had been previously diagnosed with rheumatic heart disease (RHD) during childhood and had undergone aortic valve replacement (AVR) with a mechanical prosthetic heart valve (PHV) in October 1988 (at 12 years of age) at another institution. The surgery was performed for possible severe aortic stenosis. Unfortunately, no previous surgical records were available with the patient. He had no other comorbidity and was on oral anticoagulation with warfarin (international normalized ratio (INR) mostly in the range of 2.0-2.5) and tablet enalapril for several years. On clinical examination, the patient was hemodynamically stable and in sinus rhythm. Both opening and closing clicks of the PHV were audible on auscultation, with no abnormal associated murmurs noted. The first heart sound was accentuated, with a mid-diastolic rumbling murmur noted at the apex.
Transthoracic echocardiography (TTE) revealed a metallic PHV at aortic position, possibly of tilting disc morphology (Figure 1). Due to intense acoustic shadowing, further delineation of the nature of PHV was not possible. The peak/mean pressure gradient across the valve was 21/11 mmHg, Vmax of 2.28 m/s, with an early peaking triangular continuous wave Doppler (CW) contour. The acceleration time (AT) of the CW jet was 69 ms and aortic ejection time (ET) was 254 ms. The AT/ET ratio was 0.27. The Doppler velocity integral (DVI) was 0.44, while the calculated effective orifice area by the continuity equation was 1.25 cm2. Mild aortic regurgitation (AR) was noted. No paravalvular AR, thrombus/vegetation was noted.
Transthoracic Echocardiography Showing Aortic Prosthetic Heart Valve (PHV) and Rheumatic Mitral Stenosis (a), Closed PHV with Mild Valvular Aortic Regurgitation (AR) (b), Severe Mitral Stenosis by Planimetry (c), Pressure Gradient Across PHV (d).
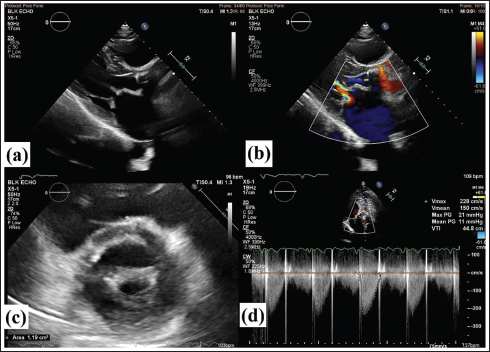
The mitral valve (MV) was thickened with anterior mitral leaflet (AML) doming in diastole and posterior mitral leaflet (PML) restricted. No significant calcification was noted in the valve leaflets or commissures. The peak/mean pressure gradient across the MV was 37/20 mmHg. The planimetered MV area (MVA) was 1.19 cm2, suggestive of severe mitral stenosis (MS). Mild mitral regurgitation (MR) was noted. The calculated Wilkins’ score of MV was 7. The left atrium was dilated (5.4 cm). Concentric left ventricular hypertrophy was present, with other cardiac chamber dimensions within the normal range. The left ventricular systolic function was normal with an ejection fraction of 55%. There was mild tricuspid regurgitation (TR), with a resting calculated pulmonary artery systolic pressure (PASP) of 33 mmHg.
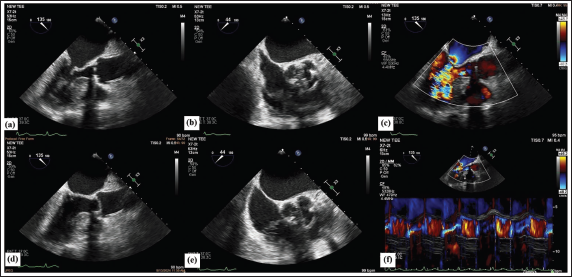
Transesophageal echocardiography (TEE) confirmed the findings of RHD, severe MS, and mild MR, with a valve appearing suitable for balloon mitral valvotomy (BMV). There was no left atrial appendage (LAA) thrombus. The aortic tilting disc PHV was better appreciated on TEE; however, acoustic shadowing was still noted (Figure 2, Supplemental Video Files 1-4). A valve size of 27-29 mm was estimated. No obvious thrombus, vegetation, or significant pannus ingrowth was noted. A mild valvular AR was seen.
Transesophageal Echocardiography Demonstrates Closed (a, b) and Open (d, e) Aortic Prosthetic Heart Valve (PHV) on Long and Short Axis Views, Mild Valvular Aortic Regurgitation (AR) (c), Color M-mode Image Showing AR (f).
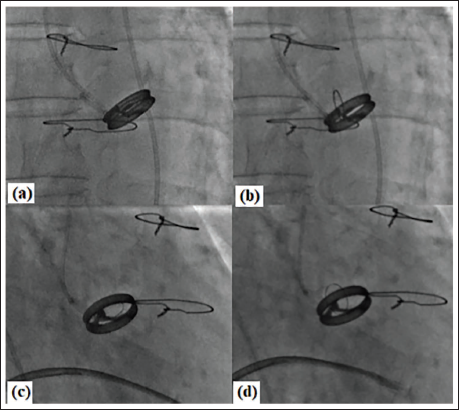
Fluoroscopy confirmed the PHV as Björk–Shiley valve (BSV) at aortic position, with a stable prosthesis (Figure 3, Supplemental Video Files 5 and 6), possibly of concavo-convex disc morphology. Normal excursion of the tilting disc was appreciated, with an opening angle of 70° (Figure 4). Subsequently, the patient underwent a successful, uncomplicated BMV procedure, with >50% increase in MVA from baseline and no increase in severity of MR.
Fluoroscopic Still Images Showing Aortic Prosthetic Heart Valve (PHV) at Closed and Open Positions in Anteroposterior Projection (a, b) and Right Anterior Oblique Projection (c, d).
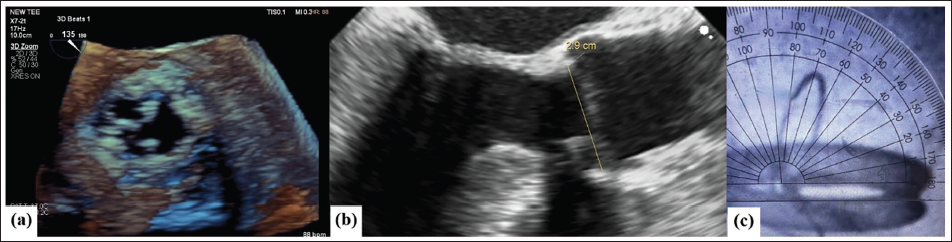
Three-dimensional En-face View of Aortic Prosthetic Heart Valve (PHV) on Transesophageal Echocardiography (TEE) (a), Valve Dimension on TEE (b) and Opening Angle 70° (c).
Our patient has had a normal functioning BSV at aortic position for 36 years, despite having a subtherapeutic INR (a higher target INR of 3.0-4.0 being recommended by current guidelines for older generation PHV).1, 2 In one of the largest studies involving Doppler parameters of normal functioning aortic BSV in 141 patients, the normal range of peak velocity of 2.5 ± 0.6 m/s, mean gradient of 13.9 ± 7.0 mmHg, and DVI of 0.40 ± 0.10 were noted. 3 Our patient’s PHV Doppler echocardiographic findings were within this range, suggestive of a normal functioning prosthesis.
Previous case reports have described normal functioning older generation tilting disc aortic PHV 4-5 decades after implantation, with the longest reported case of 48 years post-AVR.4–7 There have been scarce case reports of normal functioning older generation PHVs from India, despite cardiologists encountering such patients during clinical practice. The longest reported tilting disc PHV in an Indian patient had been implanted at the mitral position 33 years before redo surgery for PHV dysfunction. 8
However, our patient is unique in that he underwent AVR in the pediatric age group. Despite the limitations of achieving therapeutic INR levels and adequacy of testing, the patient never had any thromboembolic complications or structural valve deterioration, which is a rare finding in itself. The valve has demonstrated successful performance during 1.5 billion cardiac cycles over 36 years (assuming an average heart rate of 80 beats/min), far exceeding the fatigue test durability limit of 140 million cardiac cycles. 7
This case highlights the need for cardiologists and imaging specialists to be alert toward the possibility of encountering patients with older generation PHV during clinical practice. BSV was withdrawn from the market from 1986 onwards. There have been reports of late structural deterioration and outlet strut fractures (OSFs) of the BSV at a rate of 0.02%-0.04% per year. 9 Risk factors predisposing to OSF include younger age, larger valve, opening angle of 70°, and implantation at the mitral position. 9 Consequently, prophylactic replacement of these valves with current generation valves has been suggested. An individualized approach is required, balancing between the risk of OSF and loss of life expectancy versus the risk of reoperation and gain in life expectancy. 10
Current guidelines recommend a higher target INR in these patients owing to enhanced thrombogenicity as compared to the newer generation valves.1, 2 Such patients must be counseled about the need for optimum anticoagulation and to report immediately on development of new symptoms. Echocardiographic evaluation is indicated on emergence of new symptoms or clinical suspicion of prosthetic valve dysfunction. A multi-imaging approach using TTE, TEE, and cinefluoroscopy/cardiac computed tomography must be pursued for diagnosing prosthetic valve dysfunction.
Footnotes
Acknowledgment
The authors would like to express their heartfelt gratitude to Dr. T. S. Kler (Chairman and Head of Department Cardiology), Dr. Subhash Chandra, Dr. Ramji Mehrotra, Mr. V. K. Sharma, Mr. Vijay Singh Chauhan, and all the support staff in Non-invasive Cardiology Lab, BLK-Max Super Speciality Hospital, New Delhi, for their assistance and support in preparing this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given his/her consent for his/her images and other clinical information to be reported in the journal. The patient understands that his/her name and initials will not be published, and due efforts will be made to conceal his/her identity, but anonymity cannot be guaranteed.
Supplemental Material
Supplemental material for this article is available online.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
