Abstract
Sudden cardiac arrest is a major public health problem as it accounts for nearly 1,000 deaths per day worldwide. An estimated 80% of these occur outside of hospitals, with less than 20% survival for out-of-hospital victims and around 30% for in-hospital victims. Delays in recognizing sudden cardiac arrest and initiating high-quality cardiopulmonary resuscitation result in significant neurological problems like post-anoxic coma and vegetative states. Human expertise integrated with artificial intelligence will contribute to a dramatic improvement in sudden cardiac arrest outcomes by aiding emergency physicians in making critical decisions in the management and prognostication of patient outcomes.
Introduction
Sudden cardiac arrest (SCA) is defined as sudden and unexpected death due to a cardiac etiology occurring within 1 h from the onset of symptoms or in a person who was symptom-free 24 h before the event. It is associated with significant mortality and disability worldwide. Delays in the recognition of cardiac arrest and initiation of cardiopulmonary resuscitation (CPR) result in neurological problems, post-anoxic coma, and vegetative states.
Integrating artificial intelligence (AI) and CPR technology could signal a turning point in emergency cardiac care. AI-augmented CPR and emergency care in cardiology are poised to become a reality. AI will significantly improve outcomes, enrich lives, and save countless lives through enhanced human expertise and technology.
A majority of automated external defibrillator (AED)-delivered DC shocks do not result in spontaneous circulation (ROSC) due to various factors like demographic, clinical, environmental, and genetic influences. The occurrence of within-hospital cardiac arrest (IHCA) is 8.27 per 1,000 hospitalizations, with 25% survival at hospital discharge and only 22% of the survivors being discharged without neurological impairment.
Novel predictors of SCA in individuals, employing high-end technology to prevent, identify, and treat the event, are the need of the hour. AI technologies like machine learning (ML) and deep learning (DL) can assist clinicians in making critical decisions and improving patient outcomes in SCA and also in prognostication.
An Overview of Artificial Intelligence
Artificial intelligence is a computer system that can perform tasks as good as or better than humans. AI programs can display all relevant medical information in quick time to facilitate the deployment of emergency medical services (EMS) and improve patient outcomes through cognition-driven decision-making.
Artificial intelligence is reforming health care through the early identification of risk factors, timely detection of cardiac events, ensuring effective CPR, and prediction of outcomes in patients post-SCA.
Machine learning develops algorithms and models derived from large datasets and makes accurate forecasts or recommended decisions. Causal ML investigates causal inference to assess heterogeneity in treatment outcomes. Reinforcement learning learns to select actions that lead to the optimal treatment strategy with the lowest mortality rates. Large language models (LLMs) can comprehend and generate natural language, enabling prehospital record generation without the need for manual inputs. This enables the medical community to afford better patient care without distractions.
Natural language processing (NLP) enables machines to understand, interpret, and generate human language in a meaningful and useful manner. Text and research data, as well as social media posts, have made NLP an essential tool for gathering valuable insights and updating tasks automatically.
Machine learning techniques are broadly categorized as supervised and unsupervised learning. The former can create training models based on correctly labeled data inputs to predict outcomes based on dysrhythmia profile. Common supervised ML algorithms include DL, random forest, K-nearest neighbor (k-NN), and support vector machines (SVMs). Additional ML and DL algorithms, programs, models, and tree learning techniques are logistic regression (LR), decision tree (DT), light gradient boosted machine (light GBM), extreme gradient boosting (XGboost), multilayer perceptron, Autoscore, deep neural networks, embedded fully convolutional networks, gradient boosting, artificial neural networks, regression tree, Lagrangian support vector machine (LSVM), Naïve Bayes classifiers, reduced support vector machine (RSVM), regularized LR like Lasso, Ridge, and Elastic Net and predictive voting classifier (Figure 1).
Spectrum of Artificial Intelligence (AI).
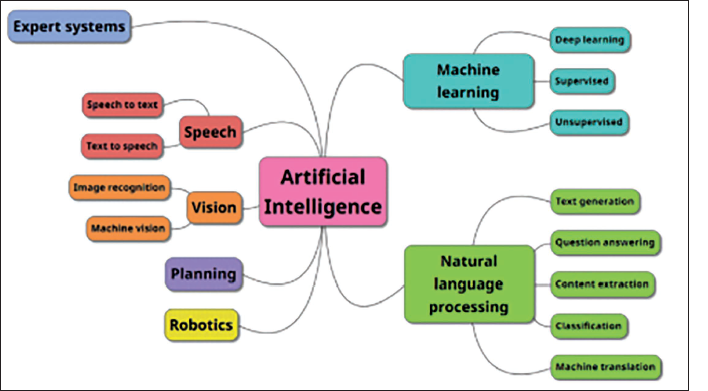
Unsupervised ML modalities like clustering can identify subgroups with similar clinical characteristics, biomarkers, and genomic data to unravel hidden patterns to facilitate comprehension and visualization of data.
Machine learning techniques are more frequently used to train the AI model as available data are primarily structured (allowing comparison between normal baseline and abnormal vital signs in disease). DL works best with unstructured data. 1
Artificial Intelligence in Critical Cardiac Events
Electrocardiogram and Dysrhythmias
Artificial intelligence can be applied even before acquiring an electrocardiogram (ECG). AI-based models can effectively predict the need for an ECG at triage in the ED. Implementing an ECG decision support system may enhance decision-making, reducing delays in the chaotic ED environment. 2
Clinicians can employ AI for the interpretation and detection of dysrhythmias, ST- and QT-segment abnormalities, and various other ECG abnormalities. AI can accurately identify 27-30 ECG abnormalities and outperform interpretation by cardiology residents.2–8
New-onset atrial fibrillation (AF) and sudden cardiac death can be diagnosed by DL learning models in high-risk patients. 9
Artificial intelligence can alert clinicians and patients to dysrhythmias like AF or bradycardia, track ECG changes during exercise, sleep, or stress tests, accurate calculation of QT duration, and classification of premature ventricular contractions. 10
Artificial intelligence can also process signals to filter noise, artifacts, and interferences, thereby improving ECG quality and accuracy. Detection of heart rate variability (HRV) and beat-to-beat intervals before it becomes apparent in ECG traces is also a major advantage. 11
Artificial intelligence-based ECG analysis can detect and track noncardiovascular disorders such as dyskalemias, hyperthyroidism, cirrhosis of the liver, and anemia using raw ECG data. AI ECG can improve hemodynamic monitoring and therapeutic drug response. AI-based ECG analyses can also detect SARS-CoV-2 infection with 99.2% certainty.11–16
Artificial intelligence can assist in optimizing treatment schedules, reducing symptom-to-treatment duration and costs. It can detect ST-segment deviations and analyze patient response to antidysrhythmic drugs to avert cardiac toxicity. 17
Artificial intelligence can facilitate precision medicine by integrating ECG data with imaging, genomic, proteomic, and biomarker inputs.
Artificial intelligence-based ECG can serve as a screening tool and interpret single-lead ECG data almost as well as 12-lead data. 18
As more sophisticated algorithms are developed, AI will aid ECG diagnosis and case management. AI reduces data processing time and makes available real-time information for better clinical management. 19
With the availability of wearable consumer devices like watches and rings utilizing photoplethysmographic and electrocardiographic sensors, better risk stratification for high-risk cardiac patients has been facilitated. The KardiaBand (KB) records a 6-lead ECG in Apple watches. Paired with an app that uses a convolution neural network (CNN), AF can be detected with 93% sensitivity and 84% specificity against a 99% sensitivity and specificity by cardiology residents. Human oversight is, therefore, necessary for the best results. Wearable technology is not capable of autonomous functioning. It serves as a useful accessory for high-risk cardiac patient monitoring.20, 21
Cardiac Arrest Risk Prediction and Management
Cardiac arrest prediction is based on three inputs:
Analysis of variables and parameters. Development of an early warning system or prediction model. Stratification of patients at a high risk of cardiac arrest.
Various characteristics from patient ECG readings, especially the variation in time between each heartbeat or HRV, genetic data, personal history (smoking habits, medications, stressors, and so forth), and nursing documentation are all analyzed to predict the cardiac arrest risk level. Abnormalities in physiological parameters often precede clinical deterioration by several hours. The 2001 Modified Early Warning Score (MEWS) is a bedside tool for identifying patients at risk for catastrophic events. Artificial intelligence can predict cardiac arrest or life-threatening arrhythmia by analyzing ventricular fibrillation waveform amplitude and frequency and QRS waveform size and amplitude inputs from manual defibrillators, AEDs, or Holter monitors. AI can also sense thoracic impedance data received from manual defibrillators. AI can display end-tidal carbon monoxide values during out-of-hospital cardiac arrest (OHCA) CPR. It can also forecast outcomes based on age and sex. The deep early warning systems (DEWS) outperform MEWS by a 50% reduction in mean alarm count in both the internal and external validation cohorts. A well-calibrated DEWS can predict IHCA events 24 to 0.5 h earlier through positive and timely alerts and improve outcomes through timely interventions. 21
Decision tree can predict the risk of cardiac death with inputs of clinical data together with single-photon emission computed tomography (SPECT) and is superior to Lasso and XGBoost. 21
Exercise testing results in movement artifacts and inferior signal quality that can result in erroneous HRV measurement. The ultra-short-term heart rate fluctuations (1-min ECG samples) with detrended fluctuation analysis (DFA) at rest is a reliable predictor of SCD risk in those with angiographically verified coronary artery disease. This parameter can be integrated into a wearable device like a smartwatch or a smart ring. 22
More than 50% of OHCA victims develop refractory ventricular fibrillation, defined as the absence of ROSC after 30 min of CPR. Extracorporeal membrane oxygenation (ECMO)-facilitated resuscitation programs demonstrate 100% successful cannulation, 43% functionally favorable, and 23% neurologically intact survival rates at hospital discharge and at 3 months post-arrest. An ECMO predictive algorithm (ECMO PAL) demonstrates better performance and superior accuracy across ECMO methods and outperforms the SAVE score, Modified SAVE score, and ECMO ACCEPTS scores.23, 24
Role of Artificial Intelligence in Prehospital Sudden Cardiac Arrest Settings
Artificial intelligence facilitates early detection and dispatch of OHCA and reliably predicts ROSC and survival outcomes from inputs like initial rhythm, age, time to CPR initiation, EMS response, scene, and transport times.
Early recognition of OHCA will help initiate telephone CPR and ensure the provision of appropriate EMS resources. Both ML and DL frameworks are capable of analyzing linguistic and phonetic characteristics of speech during the dispatch call to recognize OHCA. However, humans may not comprehend or trust the AI-based interpretations and ignore its alerts. Reliance and user confidence in ML models are needed to incorporate AI and ML into actual clinical practice.
Natural language processing can be applied to text translation, voice recognition, text summarization, and chatbots. Tokenization is the process of dividing text into smaller words or sentences or by stemming and lemmatization, that is, reducing words to their base or root forms and deleting common words that may not carry significant meaning.
Artificial intelligence-based analysis of data and outcome prediction can ensure the best possible OHCA care for victims of SCA. Different AI classification algorithms can predict survival outcomes and/or neurologically intact survival. This will enable the emergency care providers to discontinue resuscitation efforts after informing victims’ families of its futility.
Electrocardiogram-based classification algorithms can detect the victim’s pulse during CPR and provide early warning of rearrest in the immediate post-ROSC period.
The combination of AED and AI can improve outcomes for victims of SCA through improved detection of shockable rhythms from surface ECGs and OHCA data.25–28
Artificial intelligence can identify locations with a higher likelihood of SCA and aid emergency response planning and resource allocation like the placement and distribution of AEDs in public spaces more appropriately.
Out-of-hospital cardiac arrest in remote locations has lower survival rates as the lack of timely access to AEDs is a significant drawback. Drone delivery of AEDs reduces the time to defibrillation significantly and improves outcomes. AI can work out the shortest routes for drone delivery and also alert the nearest medical patrol units. However, adverse weather conditions or flight-path restrictions can affect drone deployment. 29
Artificial Intelligence and Automated External Defibrillator/Implantable Cardioverter Defibrillator
The basis for an AED or Implantable cardioverter defibrillator (ICD)-delivered shock is the recognition of a shockable rhythm such as VF and VT. These devices must have >99% specificity and >90% sensitivity for VF detection. Few AEDs can obtain a >75% sensitivity for VT and >95% specificity for SVT. Hence, an improved VT and SVT discrimination will be a desirable aspect for AI. 29
Present-day algorithms cannot identify shockable and nonshockable rhythms during CPR due to motion artifacts. An interruption of CPR is, therefore, needed for a reliable diagnosis. This implies a 15 s or more hands-off period for rhythm classification. Uninterrupted chest compressions while analyzing cardiac rhythm would be useful as even a 5 s break would decrease survival by 18%. The “Analyze While Compressing” (AWC) is a two-step process wherein a shock is advised after initial rhythm analysis and then confirmed during the “all clear” no-compression period, just prior to shock delivery. In 34.4% of nonshockable rhythms, accurate assessment of the rhythm is not possible by this method, requiring an interruption in CPR to confirm the need for a shock. 30
The decision to implant a subcutaneous implantable cardioverter defibrillator (S-ICD) through individual risk stratification can be facilitated by AI. Inputs like the left ventricular ejection fraction and MRI assessment of myocardial scar size, location, and texture enable AI to evaluate the risk of serious rhythm disturbances with 92.6% accuracy and facilitate S-ICD provision on an individual basis. 31
Outcomes After Sudden Cardiac Arrest: Artificial Intelligence Prophecies
Prognostication of SCA victims by the application of self-fulfilling prophecy and feedback loop carries the possibility of a cryptic false-positive bias that can result in a clinician discontinuing further treatment and missing the opportunity for a successful resuscitation. There is a need, therefore, for a multimodal approach to accurately predict the outcome of cardiac arrest patients to minimize the potential harm of false-positive prophecies. When advanced AI models are developed, clinicians must remain aware of the risk for amplified bias through these self-fulfilling prophecies and feedback loops. 32
The extreme gradient boosting (XGB) DT model outperforms the SVM and LR in predicting the 30-day survival and survival-to-discharge rates post-SCA.33, 34
Artificial Intelligence-based Neurological Outcomes in Sudden Cardiac Arrest
The presence and severity of hypoxic-ischemic brain injury (HIBI) post-ROSC is difficult to assess within the first few days of an event.
The assumption of a poor neurological outcome in SCA survivors leads to the withdrawal of life-sustaining therapies (WLST). Those with an unrecognized possibility of substantial neurological recovery are at risk for premature WLST. A multimodal approach and delaying prognostication after ROSC for at least 72 h is recommended to minimize the risk of premature WLST.
Chat Generative Pretrained Transformer 4 (ChatGTP-4) with inputs of baseline characteristics and cardiac arrest-related data has a positive predictive value (PPV) of 85%, a negative predictive value of 76%, and a sensitivity and specificity of 63% and 91%, respectively. 35
The prediction of neurological outcomes with inputs from post-OHCA biomarkers and EEG inputs post-ROSC using ANN models has high reliability (Figure 2). Incorporating daily neurofilament light (NFL) and neuron-specific enolase (NSE) 72 h post-SCA yields minimal false positive and false negative forecast rates.36, 37
Brain Injury Biomarkers for Predicting Neuro Outcomes Post-sudden Cardiac Arrest (SCA).
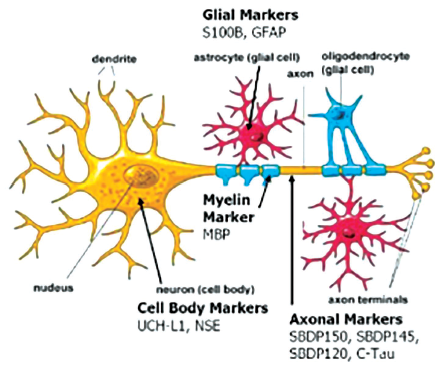
Artificial intelligence can accurately identify the progression of HIBI following cardiac arrest from the early initial CT brain scan. An AI-based prognostic model with CT brain image inputs 3 h after resuscitation is superior to conventional gray-to-white matter ratio (GWR) in terms of area under the receiver operating characteristics curve (AUROC). 38
Limitations of Artificial Intelligence
Artificial intelligence solutions require large, annotated datasets to fine-tune the numerous trainable network parameters. A scarcity of quality-controlled rhythm annotations in OHCA data can cause errors. The rigorous regulatory oversight to ensure the safety and efficacy of these devices can result in delays and is resource-intensive. AI models can be vulnerable to hacking and adversarial attacks that may result in incorrect or harmful decisions. Hence, protection and security from malware is essential.
Data collection and management for developing and applying ML models in clinical settings must respect privacy, comply with the law, be accountable, and consider patient safety. The seven essential requirements are:
Human-based oversight. Superior and safer technical applications. Respecting patient privacy and good data governance. Transparency. Fairness and discrimination-free approach. Consideration of environmental and societal impact. Accountability.
International laws to better protect citizens from the rapidly increasing impacts of AI and ML-driven models need to be implemented. 39
Conclusion
Innovative approaches are needed to improve all aspects of resuscitation and defibrillation for better outcomes. AI’s application in prehospital emergency care can accurately detect shockable rhythms and predict and guide successful outcomes of CPR based on real-time feedback. Better resource allocation can also be ensured if reliable neurological outcomes post-ROSC can be predicted using AI.
Research workers, healthcare providers, and regulatory bodies must collaborate to harness the full potential of AI. Sustained developments in SCA management at all levels are essential to improve survival and decrease adverse outcomes.
Correlation between the clinical data and demographic, laboratory, genetic, and biological data of the patient can improve the accuracy of the prearrest forecast. The percentage of true positives and true negatives in AI prophecy has to be critically evaluated (Figure 3).
Impact of Artificial Intelligence (AI) in Healthcare.
With the increasing adoption of wearable devices with sensors tracking various aspects of health and activity, research opportunities exist to develop techniques to predict and alert patients at risk of OHCAs.
The Utstein format is accepted as the universal data-collecting standard, mainly in prehospital SCA settings. ML models have to be developed based on this to be accurate and reproducible in CPR.
Variations in resuscitation practices exist across global healthcare settings. AI models developed in one context may not apply in another setting due to diverse study populations, different clinical protocols, and resource constraints. AI-driven predictive analysis from large datasets can promote successful resuscitation through developments in CPR protocols, training techniques, and medical interventions.
Artificial intelligence and robotics will have far-reaching impacts and benefits due to their ability to process large datasets swiftly and come up with actionable insights, rapid diagnosis, and precision treatment plans.
Footnotes
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Ethical permission was not applicable for this article, as this is a review article drafted from various research articles and not from patients directly.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Consent was not needed/not applicable, as this is a review article compiled from various research articles and guidelines and not from patients directly.
