Abstract
A saphenous vein graft (SVG) is a bypass conduit that is frequently employed in surgical revascularization operations. One of the primary issues with SVGs is that they can encounter blockage rates of up to 25% within the first year. Saphenous venous graft aneurysms (SVGAs) are extremely uncommon, yet they can develop at any time. While early SVG degeneration may result in SVGAs, the classic indications of myocardial ischemia are often associated with late SVG degeneration. Other methods of imaging, including coronary computed tomography and graft angiography, can detect SVGA that would otherwise go undetected. This issue may cause mortality to be underestimated, resulting in fatalities that go unnoticed and uninvestigated.
The major goal of this case report was to offer a detailed account of an early SVGA episode in a patient who had a documented non-ST-segment myocardial infarction and significant myocardial ischemia, as confirmed by coronary artery angiotomography.
Introduction
A significant proportion of patients who have cardiac revascularization surgery typically receive a saphenous venous graft (SVG) as a bypass conduit. One of the primary challenges associated with the utilization of SVGs is the occurrence of early problems, which encompass an occlusion rate that can reach as high as 25% during the initial year. The development of saphenous venous graft aneurysm (SVGA) is rare, however, the time of their manifestation can differ. 1
Although late degeneration of SVG leading to the development of the SVGAs is a long-standing phenomenon associated with classic signs of myocardial ischemia, it is important to remember that this condition can also manifest in the early stages. SVGA sometimes do not exhibit symptoms and are sometimes discovered inadvertently during imaging procedures such as computed tomography conducted for unrelated reasons. In a comprehensive analysis of 5,579 venous grafts, Dieter et al. determined an incidence of SVGA at 0.07%. This complication may cause deaths that go unrecognized and uninvestigated, leading to an underestimation of the actual incidence. Severe reporting bias in the literature may impede the development of reliable estimates of SVGA epidemiology and management approach. 2
As a result, the primary goal of this case report was to describe the early manifestation of an SVGA in a patient who presented with non-ST-segment myocardial infarction and significant myocardial ischemia, which was confirmed by coronary computed tomography angiography.
Case Report
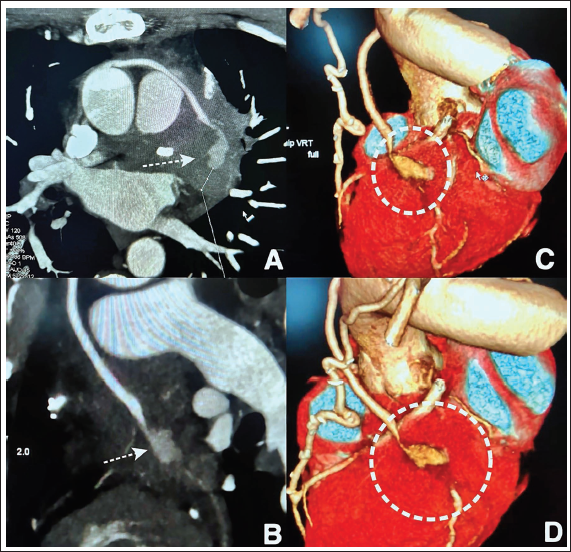
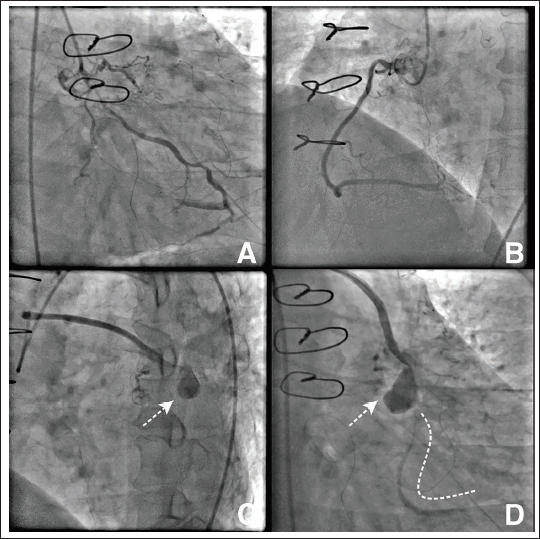
A 56-year-old male admitted complaining of severe chest pain on exertion for the last 15 days. He underwent coronary artery bypass surgery (CABG) a year ago. On arrival in the emergency room, his vitals were stable, but an electrocardiogram demonstrated ST-segment depressions in inferolateral leads. His laboratory parameters were normal except for mildly elevated troponin I levels (0.48 ng/dL; Normal value < 0.12 ng/dL). He was subjected to computed tomography-guided coronary angiography, which demonstrated native coronary artery disease (CAD) with a patent left internal mammary artery graft to the left anterior descending artery (LAD), but there was a focal fusiform aneurysm of the SVG to the obtuse marginal (OM) artery (Figure 1A-D). For further delineation and to plan interventional management of the lesion, conventional coronary angiography (CAG) was done, which revealed an ostio-proximal total occlusion of the LAD and critical in-stent restenosis of the ostio-proximal segment of the left circumflex (LCX) artery with bridging collaterals across the stenotic segment with thrombolysis in myocardial infarction (TIMI) 2 flow. The angiography of the SVG to OM artery revealed a fusiform pseudoaneurysm followed by critical stenosis at the distal edge of the SVGA with TIMI two flow (Figure 2A-D).
(A-D) Computed Tomographic Coronary Angiography Revealed a Large Thick-walled Circular Aneurysmal Dilatation (Dashed White Arrows) Across the Mid-segment of the Saphenous Venous Graft (SVG) to Obtuse Marginal (Cross-Sectional and Longitudinal Views, Panels A and B). 3D Volume-rendered Reconstructed Image Revealed a Pseudoaneurysm of SVGA With Significant Stenosis of the Downstream With the Reduced Flow (Dashed White Circles, Panels C and D).
(A-D) Conventional Coronary Angiography of the Left Coronary Artery Demonstrated Chronic Total Occlusion of the Ostio-Proximal Segment of Left Anterior Descending Artery. The Critical In-stent Restenosis of the Ostio-proximal Segment of the Left Circumflex Artery Extends to the Large 1st Obtuse Marginal Artery. The Right Coronary Artery was Normal. The Left Internal Mammary Artery to LAD was Patent (not Shown) While Angiography of the Saphenous Venous Graft to OM1 Revealed a Large Saccular Aneurysm of the Mid-Segment Followed by Critical Stenosis of the Distal Segment With Thrombolysis in Myocardial Infarction Flow II.
The potential choices between surgical/percutaneous intervention and medical management were explored, taking into account the risk of SVGA progression resulting in rupture, in situ thrombosis, and distal embolization, as well as the recurrence of acute coronary syndrome, heart failure due to mitral regurgitation, and loss of myocardial viability. The patient chose conservative treatment after seeing improvement in symptoms with the use of dual antiplatelet therapy (aspirin and clopidogrel), anticoagulant (low molecular weight heparin for 7 days), beta-blockers, and long-acting nitrates and trimetazidine. A patient has completed a follow-up of one year on optimal medical management with no recurrence of the symptoms.
Discussion
Fistula formation, rupture, acute chest discomfort, haemoptysis, and hemodynamic compromise due to compression of surrounding vascular and cardiac structures have all been documented after the accidental discovery of SVGA on imaging. 3 The occurrence of an aneurysm in the SVG usually occurs at a significant time interval following the initial CABG procedure, with an average detection period of approximately 13 years post-surgery. Indeed, the incidence of SVGA detection within the initial year following CABG was about 4%; however, a significantly higher proportion of 68% was observed more than a decade post-surgery. 4
SVGAs can be classified as either true or pseudoaneurysms. Pseudo SVGAs are infrequent and typically occur at the proximal or distal anastomotic site which is rare to involve at the mid segment of the SVG as in our case. The prevalent categories of SVGAs include true SVGAs and are often observed several years following CABG. 5 Early occurrence in the initial months after surgery is frequent, although there are few documented cases of late incidence occurring years later. The development of SVGA can also be classified as either early (within 15 days to a few months after CABG) or late (≥5 years after the CABG). It is thought that infections, innate vascular wall weaknesses, or harvesting errors are the root causes of early SVGA. 6 However, it is believed that atherosclerotic degeneration, which impacts the structure of the arterial walls, causes late SVGA. 5 The diagnostic imaging gold standard, CT or traditional coronary angiography, is a great tool for assessing grafts and excluding obstructive disorders, as it was in our instance.
Mattia Vinciguerra et al. discovered 296 relevant scientific articles that encompassed 342 patients and a cumulative count of 368 SVGAs up till 2021, with 71.7% of these cases being classified as true SVGAs. 2 In total, 48.5% of the complications were found when the existing literature was examined. Compression of adjacent structures was observed in 24.5% of these patients, making it the most common complication. Aneurysm rupture occurred in 9.9% of instances. Fistulous communication occurred in 4.6% of patients and myocardial infarction due to prolonged ischemia injury in 2.9% of patients, among other complications as in our case.
While infrequent, SVGAs are potentially life-threatening complications that might arise in less than 1% of patients who undergo CABG. The analysis by Ghosh et al. found atherosclerosis and intimal calcification as the main risk factors for SVGA after CABG, with infection being a secondary contributor. Typical risk factors comprise mild right ventricular hypertrophy, left ventricular hypertrophy, mitral regurgitation, aortic valve insufficiency, pulmonary valve stenosis, congestive heart failure, pneumonia, chronic inflammation, hyperlipidaemia, intimal proliferation, and calcification. The prevailing comorbidities observed were dyslipidemia and hypertension, while SVGAs were also detected in several patients who did not have any comorbidities. 7
The treatment guidelines for SVGA are currently ambiguous due to the lack of definitive data that demonstrates the benefits of intervention, compared to conservative management for asymptomatic patients or those who respond to medical treatment. 5 The three most common treatment approaches are surgical intervention, percutaneous therapies, and conservative care. The primary treatment option for SVGA involves occluding or removing the graft, replacing the conduit, and revascularizing the native coronary artery through surgery. 7 The treatment outcomes differ, with mortality rates of 13.9%, 6.1%, and 23.8% for surgical, percutaneous, and conservative therapies, respectively. Our patient showed improvement after receiving optimal medical treatment and has not experienced any symptoms since. The majority of patients achieved stability and were discharged with improved conditions following surgery, with comparable results observed among those who received percutaneous intervention and conservative treatment. 8 Because of the patient’s unwillingness as in our case, the existence of additional medical issues, or the patient’s short life expectancy, appropriate medical therapy and cautious monitoring were utilized in the management of 19% of all SVGAs. 2 Patients at high surgical risk or those in stable condition without symptoms were typically treated with a conservative approach.
The management of SVGAs should be based on their size and rate of growth. Given the patient’s history of co-occurring diseases, which may raise surgical risks and reduce life expectancy, a decision about interventional management should be made only after careful consideration of these factors. Those patients who are at high risk for complications during surgery may benefit from endovascular therapy or intensive observation using optimal medical management.
In order to safeguard viability of the myocardium and avoid rupture, it is necessary to rule out the SVG aneurysm if the afflicted area is large and viable. The surgical options may be considered for patients with a low risk of surgical complications. Surgery was selected as the preferred treatment by 52.6% of the entire group. Out of the mentioned cases, 45.6% underwent aneurysm ligation or resection, whereas 54.4% got a combination of surgical exclusion and repeat revascularization. In total, 11.1% of patients who underwent this revision operation experienced mortality during their hospitalization.
The percutaneous intervention as an option for patients who pose a high-risk during surgery which increasingly a common method with advent of better devices such as coil embolization, vascular plugs, stent-assisted coiling, covered stents, endoluminal grafts, selective thrombin injection with mortality rate of 5%.2, 9, 10 Whether SVG aneurysms that supply blood to non-viable myocardium are surgically excluded or embolized depends on the patient’s risk variables. 1
It is advisable to be cautious when considering conservative treatment for small SVGAs (<40 mm) that were incidentally detected and are asymptomatic but nevertheless require medical treatment. However, it is important to consider that SVGAs are expected to continue increasing in size, therefore any prudent approach should involve periodic reassessment. 2
Conclusion
The condition known as SVGA is rare and is linked with a very high risk of in situ thrombosis and distal embolization. Nevertheless, there remains a therapeutic conundrum in the therapy of this disease because randomized clinical studies are not yet available. Our findings suggest that an early and aggressive interventional approach does not confer a survival advantage, even though the exact treatment for this condition is yet unspecified. The characteristics of the SVGA and the coexisting medical conditions should be taken into consideration while deciding whether to intervene in an SVGA.
Footnotes
Acknowledgment
This manuscript has not been submitted to any journal before for publication, either in part or as a complete version. The authors acknowledge and give full consent and rights to the journal for its publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed in studies involving human participants were conducted in accordance with the ethical standards of the institutional and/or national research committee and the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
Informed consent was obtained from all participants included in the study. Written consent was obtained for participation in the research and the publication of any potentially identifiable information. Patient anonymity was ensured, and all data were handled confidentially.
