Abstract
Purpose
To determine the prevalence of sarcopenia and sarcopenic obesity (SO) in patients with type-2 diabetes mellitus (T2DM) and established cardiovascular disease (CVD).
Methods
This cross-sectional study included 60 T2DM patients (30 with CVD and 30 without, aged 40-70 years, 80% males). Data collection included body composition analysis using bio-impedance, hand grip strength measurement and gait-speed assessment. Sarcopenia was defined using the Asian Working Group for Sarcopenia (AWGS-19) guidelines, while SO was defined using a body-mass index, waist circumference and percentage of body fat. SPSS software version 23.0 was used. P < .05 was significant.
Results
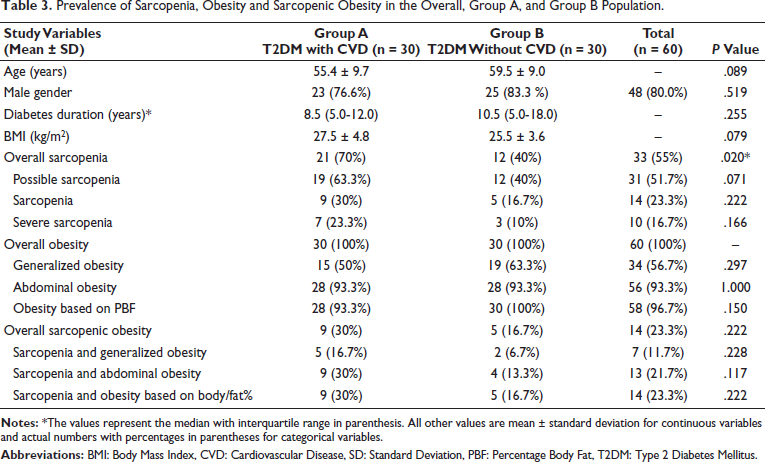
Overall sarcopenia (including possible sarcopenia or sarcopenia) was found to be significantly higher in patients with CVD as compared to those without CVD (70% vs. 40%; P = .020). Although numerically greater (in patients with CVD), no statistically significant difference was found in the prevalence of SO (30% vs. 16.7%; P = .222).
Conclusion
Our study found that the prevalence of overall sarcopenia was significantly higher in patients with T2DM and CVD compared to those without CVD. Sarcopenia appears to be a potential independent risk factor in predicting risk of CVD in patients with T2DM.
Introduction
“Sarcopenia” was first described in the geriatric population as a progressive and generalized skeletal muscle disorder marked by a reduction in muscle mass and quality, decreased muscle strength and/or impaired physical performance.1, 2 Sarcopenia is a relatively young disease which has recently (2016) been assigned a disease-specific code (ICD-10-CM) and also gained popular interest worldwide because of the vast amount of clinical and research potential it carries in being identified as an independent metabolic entity. 3 Sarcopenia, together with obesity, is now well-recognized as, “Sarcopenic obesity” (SO). 4
The recent recognition of sarcopenia and SO as indicators of clinical outcomes has fueled extensive research, revealing a rising prevalence of these conditions in the community. This increase is attributed to their multifactorial pathophysiology, which includes risk factors akin to those associated with obesity.5–7 The relationship of sarcopenia and SO with type-2 diabetes mellitus (T2DM) 8 and cardiovascular disease (CVD)9, 10 is intriguing, as this includes not only a higher prevalence and predictability of sarcopenia and SO in patients with T2DM and CVD, but also the possibility of sarcopenia and SO contributing independently to an increased risk of CVD in T2DM patients. Also, there is a significant association of sarcopenia and SO with mortality (all-cause and/or CVD-related) as an outcome measure,8–13 thereby implicating the magnitude of these health risk factors. Hence it is imperative to investigate such associations beyond the traditional risk factors.
However, research in this domain has indicated a lot of heterogeneity because of the lack of a universally accepted definition and diagnostic criteria, and the socio-demographic factors of the population studied. Moreover, there are no studies having truly representative data investigating the association of sarcopenia and SO in T2DM patients with CVD in the Asian-Indian population.
Therefore, with the current unabated epidemic of non-communicable diseases, there is an ever-growing need for increased awareness toward greater research in sarcopenia and SO. This in turn may just prove to be the missing link for formulating a working definition and defining cut-off values for correctly estimating the prevalence of sarcopenia and SO in Asian Indians. Positive efforts in this direction will effortlessly reflect in optimally defining future CVD risk and mortality eventually in patients with T2DM, and ultimately also drive the formulation of primary preventive strategies reflecting in the betterment of patient health outcomes.
Methods
Study Design
A cross-sectional study in design was carried out in a tertiary care hospital in Northern India. The study participants were recruited from the Endocrinology & Diabetes, and Cardiology outpatient departments (July to October 2022). The sample size was calculated after statistical consideration of the “pilot” study design to be a total of 60 consecutive T2DM subjects which were included in the study. Two groups have been described in our study: Group A: 30 T2DM subjects with CVD, and Group B: 30 T2DM subjects without CVD.
Selection Criteria
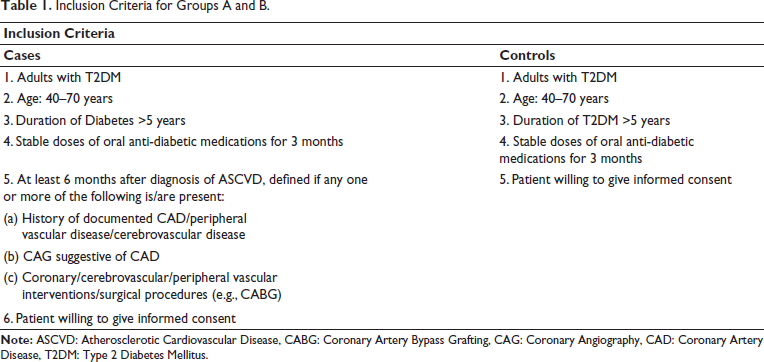
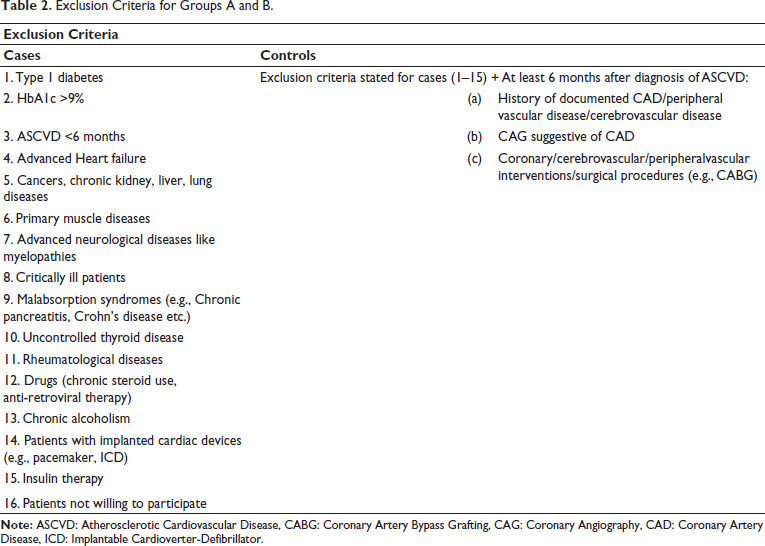
Inclusion (Table 1) and exclusion criteria (Table 2) for cases and controls have been tabulated below.
Inclusion Criteria for Groups A and B.
Exclusion Criteria for Groups A and B.
Data Collection
Data was recorded manually using a pre-designed non-validated performance. Upon voluntary participation (informed consent) by the patients, a participant informant sheet was provided to the patients describing the research study and their role as a participant. Institutional Ethical Committee (IEC) and Institutional Review Board (IRB) approvals were obtained.
Vitals (pulse and blood pressure) were recorded using the American Heart Association (AHA) guidelines. 14 Height was measured by stretch stature (SS) using a fixed wall-mounted stadiometer (HOLTAIN limited-Crymych, Dyfed) with a sliding headboard as per the standardized protocol. 15 Anthropometric parameters included measurement of hip circumference and waist circumference in accordance with the World Health Organization (WHO) guidelines (three trials of data measurement were performed, and their average and mean were calculated thereafter to obtain the final value). 16 Comprehensive body composition analysis, muscle-fat analysis, obesity analysis and segmental lean muscle mass analysis were done using InBody-570 multifrequency body impedance analyzer (BIA) machine, as a standard instrument with the cutoffs in accordance with the Asian Working Group for Sarcopenia (AWGS-19) criteria. 17 Skeletal muscle strength (handgrip strength) was measured using a hydraulic type (Jamar) dynamometer. 17 Gait speed (6 meters) was used to assess physical performance (normal pace from a moving start, without deceleration) as per the AWGS-19 criteria. 17
Definitions
Sarcopenia: defined using the AWGS-2019 criteria.
17
Possible sarcopenia: Low handgrip muscle strength <28 kg in males (M) and <18 kg in females (F) Sarcopenia: Low appendicular skeletal muscle mass (ASM): <7.0 kg/m2 (M) and <5.7 kg/m2 (F) (BIA) + low handgrip muscle strength or low physical performance: 6m walk gait speed <1.0 m/s Severe sarcopenia: Low ASM + low handgrip muscle strength and low physical performance Overall sarcopenia: Any patient having possible sarcopenia or sarcopenia
Obesity: defined using different diagnostic criteria described below:
Generalized obesity: Body mass index (BMI) ≥25 kg/m2 as defined by the WHO guidelines.
18
Abdominal obesity: Waist circumference (WC) >90 cm (M) and >80 cm (F), as stated by the WHO guidelines.
18
Obesity defined using body fat%: Percentage body fat (PBF) >20%, assessed using InBody-570 multifrequency BIA machine. Overall obesity: Any patient having obesity according to any criteria.
Sarcopenic obesity: defined using the following combinations:
Sarcopenia + Generalized obesity: Sarcopenia + Obesity (BMI) Sarcopenia + Abdominal obesity: Sarcopenia + Obesity (WC) Sarcopenia + Obesity using body/fat%: Sarcopenia + Obesity (PBF) Overall sarcopenic obesity: Same as D (sarcopenia + obesity using body/fat%)
Coronary artery disease (CAD): defined if there is any one or more of the following present:
Documented history of myocardial infarction in past, History of myocardial revascularization, and Coronary angiography demonstrating at least 50% stenosis in one of the major epicardial coronary arteries.
Outcomes
The primary outcome of our study was the prevalence of sarcopenia, obesity and SO in the overall population, and in groups A and B using each respective definition as given above. The secondary outcomes included a comparison between defining study parameters (dominant handgrip muscle strength, gait speed and ASM mass) and different patient characteristics between sarcopenic, obese and sarcopenic-obese populations, and also between groups A and B, respectively.
Statistical Analysis
Categorical variables were expressed as absolute numbers and percentages. Descriptive analysis of quantitative parameters was expressed as mean ± standard deviation (SD) for continuous variables and actual numbers with percentages for categorical variables. The significance of difference between the groups was analysed and the association was studied using the Chi-square test and independent samples t-test for categorical data and continuous data respectively. Mann-Whitney U test was also used in sub-group analysis for comparing different groups. P < .05 was considered significant. All analysis was done using SPSS software, version 23.0.
Results
Primary Outcome-prevalence of Sarcopenia and SO
Overall in T2DM patients (n = 60), sarcopenia, obesity and SO were present in 55% (n = 33), 100% (n = 60), and 23.3% (n = 14) subjects, respectively. The prevalence of sarcopenia, obesity and SO among cases versus controls was 70% (n = 21) versus 40% (n = 12) (P = .020); 100% (n = 60) versus 100%; and 30% (n = 9) versus 16.7% (n = 5) (P = .222), respectively (Table 3).
Prevalence of Sarcopenia, Obesity and Sarcopenic Obesity in the Overall, Group A, and Group B Population.
Secondary Outcomes-study Parameters
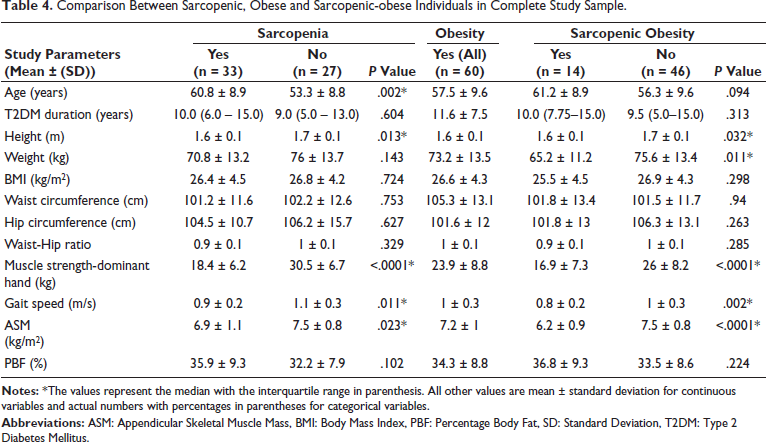
Mean ± SD values of dominant handgrip muscle strength (kg), gait speed (m/s) and ASM mass (kg/m2) in patients having sarcopenia and SO were 18.4 ± 6.2 kg and 16.9 ± 7.3 kg, 0.9 ± 0.2 m/s and 0.8 ± 0.2 m/s, and 6.9 ± 1.1 kg/m2 and 6.2 ± 0.9 kg/m2, respectively (Table 4).
Comparison Between Sarcopenic, Obese and Sarcopenic-obese Individuals in Complete Study Sample.
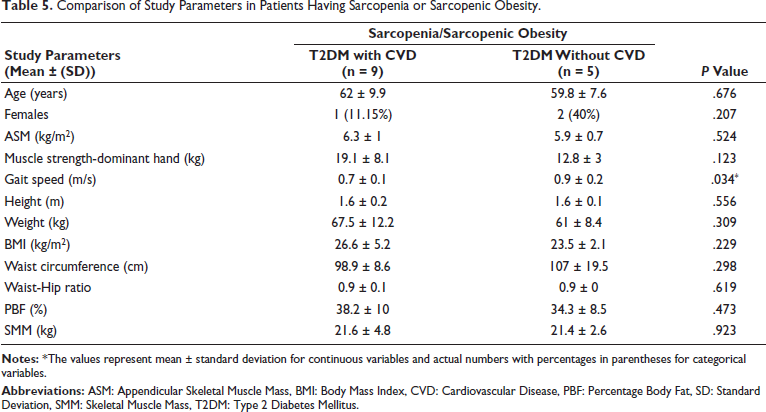
On comparing CVD versus non-CVD patients having sarcopenia/SO, mean ± SD ASM (kg/m2), dominant handgrip muscle strength (kg) and gait speed (m/s) were found to be 6.3 ± 1 versus 6 ± 0.7 (P = .524), 19.1 ± 8.1 versus 12.8 ± 3 (P = .123), and 0.7 ± 0.1 versus 0.9 ± 0.2 (P = .034), respectively (Table 5).
Comparison of Study Parameters in Patients Having Sarcopenia or Sarcopenic Obesity.
Discussion
Sarcopenia and SO with T2DM
Current published evidence indicates a strong association between sarcopenia/SO and T2DM independent of obesity and other metabolic risk factors.13, 19–22 However studies which aim to estimate the prevalence or relevance of sarcopenia/SO have presented diverse and at times dichotomous results, which can be explained by the socio-demographic differences of the population studied, and more importantly, lack of a universally accepted definition.
Verma et al. 23 (n = 65,562 aged ≥45 y; Longitudinal Aging Study of India-LASI (2017-18) wave-1 survey) using the AWGS-19 criteria, found the prevalence of possible sarcopenia, obesity and SO (using possible sarcopenia and obesity) to be 41.9%, 27.1%, and 8.7%, respectively. Whereas, the Sarcopenia-Chandigarh Urban Bone Epidemiological Study (Sarco-CUBES) using the European Working Group on Sarcopenia in Older People (EWGSOP2) criteria in apparently healthy young individuals (n = 804 aged ≥20 y) found the prevalence of probable sarcopenia to be 14.6% (M = 16.8%, F = 13.1%), sarcopenia to be 3.2% (M = 4.7%, F = 2.1%) and severe sarcopenia to be 2.3% (M = 2.6%, F = 1.9%). 24 This dichotomy of prevalence is surely due to different criteria used to define sarcopenia. Hence, necessitating formulation of diagnostic criteria for optimally defining the prevalence of sarcopenia/SO in our population, like was done with obesity.
The primary outcome of our study found the prevalence of possible sarcopenia, sarcopenia, severe sarcopenia to be 51.7% (n = 31), 23.3% (n = 14), and 16.7% (n = 10) in type-2 diabetic patients. In addition, we found overall obesity and SO to be present in 100% (n = 60) and 23.3% (n = 14) of all T2DM patients (Table 3). The reason for the observed high prevalence in our study is related to the study population characteristics including advancing age; long duration of diabetes and presence of CVD.8–11, 13, 19, 25
Interestingly, the high prevalence of sarcopenia (60.6%; P = .411), obesity (60%) and SO (57.1%; P = .198) in patients who had a T2DM duration between 5 and 10 years, indicates that the loss in muscle mass/strength starts early in T2DM patients of Asian-Indian origin.10, 26–28 This observation is also explained/supported by the ethnic body composition differences in Asian Indians, which include higher total body fat, greater visceral fat and lesser muscle mass when compared to Caucasians. Importantly, as shown by some researchers, the decline in muscle mass may be evident even during the pre-diabetic phase.29–31 Therefore, future studies should also aim at studying the relationship of sarcopenia/SO with T2DM duration to identify sarcopenia/SO at its early stages, which if diagnosed early, might result in a better prognosis.
Association with CVD
Recent studies have found a high prevalence of sarcopenia and SO among CVD patients and have also shown a strong association between sarcopenia/SO with CVD and outcomes, independent of T2DM and other cardio-metabolic risk factors.9, 10, 25, 32
Kim et al. 9 (n = 3320, ≥40 y) using the Framingham risk score to assess the 10-year CVD risk, found that as compared to non-sarcopenic adults (M = 24.1%, F = 11.1%), non-obese adults (M = 42.7%, F = 12.2%), those with SO had a significantly greater 10-year CVD risk (overall ≥20%; M = 43.8% and F = 14.6%). A study conducted in the British population (n = 4,253; 60-79 y) showed that sarcopenia and central adiposity are strongly associated with increased CVD mortality and all-cause mortality, 10 thereby implicating the strong association of sarcopenia/SO with CVD and its outcomes. Sang Ouk Chin et al. 25 (n = 1,578 ≥ 60 y) from the Korea National Health and Nutrition Examination Survey (KNHANES) showed that there was a higher prevalence of CVD in subjects with sarcopenia (13.1% ± 3.1%) and SO (12.3% ± 2.1%) when compared with those only with obesity (10.0% ± 2.2%), thereby indicating that sarcopenia is associated with CVD independent of obesity.9, 10, 25, 32 Finally, to the best of our knowledge at the time of writing this research article, we did not find any large-scale prospective population-based study having truly representative data of the independent association of sarcopenia/SO to T2DM and CVD in the Asian-Indian population.
Also as the primary outcome, our study found the prevalence of overall sarcopenia among group A versus group B to be 70% versus 40%; P = .020. Hence, indicating that presence of sarcopenia may be associated with CVD in patients with T2DM.9, 10, 25, 33 Although SO was seen to be numerically higher in cases than in controls (30% vs. 16.7%; P = .222), a statistically significant relationship could not be established due to a small sample size and obesity being present in all subjects (Table 3). Nevertheless, according to our study, the numerically higher prevalence of sarcopenia/SO among cases may indicate that there is a strong and possibly independent relationship of sarcopenia/SO in T2DM patients with CVD,8–13, 32, 34, 35 however, larger population-based studies are required to correctly estimate the strength of this association.
Study Parameters
As secondary outcomes of our study, all sarcopenia defining parameters (AWGS-19): ASM (kg/m2), dominant handgrip muscle strength (kg) and gait speed (m/s) were found to be numerically lesser among the sarcopenic-obese (SO) subjects in comparison to the sarcopenic subjects (6.2 ± 0.9 vs. 6.9 ± 1.1, 16.9 ± 7.3 vs. 18.4 ± 6.2 and 0.8 ± 0.2 vs. 0.9 ± 0.2, respectively) (Table 3). This indicates that among T2DM patients, muscle mass, strength and physical performance may be more severely affected due to the coexistence of obesity. Whether reduced muscle mass physical performance, and coexisting obesity a better predictors of CVD in Asian-Indian T2DM patients than sarcopenia alone needs to be proven by a larger prospective study.
Interestingly, mean ± SD ASM (kg/m2) and dominant handgrip muscle strength (kg) were higher in group A than in group B (6.3 ± 1 vs. 6 ± 0.7; P = .524 and 19.1 ± 8.1 vs. 12.8 ± 3; P = .123, respectively), while mean ± SD gait speed (m/s) was higher in group B than group A (0.9 ± 0.2 vs. 0.7 ± 0.1; P = .034, respectively) (Table 4). Having CVD affects these parameters, however, this dichotomy of greater muscle mass and poorer muscle performance in cases as compared to controls is mostly unexplained (except for the small sample size being a plausible reason in our study) and needs to be studied by further research for practical understanding.
Importantly, our study compares the AWGS-19 17 criteria-based study parameters of muscle mass, strength and physical performance, which to our knowledge, has not been studied so far in Indian patients with T2DM and CVD. Also, we investigated the possibility of defining SO by using three different diagnostic criteria used to measure obesity (BMI/WC/PBF).
According to the findings of this study, which demonstrated a higher prevalence of sarcopenia among subjects with T2DM and CVD, along with its significant associations with other study parameters, and in reference to existing literature, it is reasonable to propose that sarcopenia may function as an independent risk factor in predicting CVD risk in patients with T2DM. On the other hand, even though no statistically significant difference was found in the prevalence of SO, we believe larger studies would enrich our understanding of SO, which may well prove to be an independent risk factor, like obesity, for defining CVD risk in patients with T2DM.
Limitations
First, our study is a cross-sectional study and hence does not prospectively study the incidence of sarcopenia and SO in T2DM patients with/without established CVD. Second, being a pilot study, we described a small select study population which is not truly representative of the population, and hence there is insufficient data to clearly describe different cut-off values or definitions for sarcopenia and SO. Third, even though on average roughly every fourth patient was studied to have SO, a significant difference in the prevalence of SO in T2DM patients with/without CVD could not be clearly appreciated due to the small sample size and all subjects being obese. Fourth, nutrition and fitness were not formally assessed. Fifth, this study was done in T2DM patients with or without CVD, in an outpatient setting, which limits the generalizability of the findings. Future studies aimed at studying these associations in the general population with T2DM might prove to be more relevant. Finally, the roles of other confounding variables like lifestyle, diet and medication use were not discussed.
Though limited by a pilot study design, our study raises the importance of sarcopenia/SO in patients with T2DM and CVD in Asian Indians. Fueling the need to have larger population-based studies studying the independent association of CVD with sarcopenia and SO in T2DM patients, both as a diagnostic and prognostic risk factor. In addition, future studies should also aim at formulating clinically viable nutritional and physical interventions for improving muscle mass/strength and physical performance. This in turn might just prove to be the link in combating sarcopenia and SO, thereby decreasing the risk and prevalence of such metabolic diseases in the community. Positive efforts in this direction will effortlessly reflect in the betterment of patient health with improved outcomes and drive the formulation of primary preventive strategies.
Conclusion
Our study found that the prevalence of overall sarcopenia was significantly higher in patients with T2DM and CVD compared to those without CVD. Sarcopenia appears to be a potential independent risk factor in predicting risk of CVD in patients with T2DM.
Footnotes
Abbreviations
AHA: American Heart Association, ASCVD: Atherosclerotic Cardiovascular Disease, ASM: Appendicular Skeletal Muscle Mass, AWGS: Asian Working Group for Sarcopenia, BIA: Body Impendance Analyzer, BMI: Body Mass Index, CABG: Coronary Artery Bypass Grafting, CAD: Coronary Artery Disease, CAG: Coronary Angiography, CVD: Cardiovascular Disease, PBF: Percentage Body Fat, SO: Sarcopenic Obesity, T2DM: Type 2 Diabetes Mellitus, WC: Waist Circumference, WHO: World Health Organization.
Acknowledgements
The authors gratefully acknowledge the ICMR-STS program for which this research was initially conceptualised and undertaken.
Authors’ Contribution
Amanpreet Singh Wasir: Conceptualization (supporting) Writing – original draft (lead), Visualization (equal), Methodology (equal), Data curation (equal), Investigation (lead). Jasjeet Singh Wasir: Conceptualization (lead), Writing – original draft (equal), Writing – review & editing (lead), Supervision (lead), Resources (lead), Methodology (lead), Project administration (lead). Manish Bansal: Writing – review & editing (supporting), Supervision (supporting), Resources (supporting), Formal analysis (lead), Supervision (equal), Data curation (supporting). Anuradha Rajiv Joshi: Writing – review & editing (supporting), Supervision (supporting), Project administration (supporting).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Institutional Ethical approval and Institutional Review Board (IRB) approval were duly obtained from concerned authorities.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
