Abstract
Valve infolding is one of the uncommon side effects of transcatheter aortic valve implantation (TAVI) with self-expandable valves (SEV). Acute hemodynamic collapse brought on by infolding frequently results in complications, such as periprocedural stroke. We present an unusual occurrence of infolding of the Evolut PRO valve. Removal and replacement of the valve should be considered as soon as infolding is discovered.
Introduction
For patients with severe aortic stenosis, transcatheter aortic valve implantation (TAVI) is currently one of the standard procedures. Despite being safe, there are still a few potentially disastrous complications. Valve infolding is a serious complication that is exclusive for TAVI with self-expandable valves (SEV). This case report helps in the identification and subsequent management of valve infolding.
TAVI is recommended for patients with severe aortic stenosis. One of the infrequent complications is valve infolding. Infolding often causes acute hemodynamic collapse and can lead to complications including periprocedural stroke. We present a case report when infolding was recognized before releasing the valve.
Case report
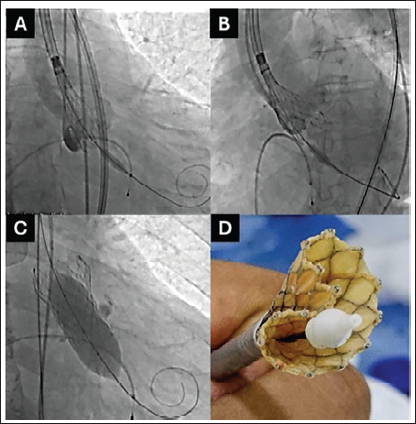
A 70-year-old male with symptomatic severe aortic stenosis was considered for TAVI. CT analysis showed Siever’s type-I LR bicuspid aortic valve 1 with severe calcification and calcified raphe. In view of severe valvular and nodular calcium in the left ventricular outflow tract (LVOT) a 29 mm Evolut PRO+ (EP+) (Medtronic Inc., Minneapolis, MN) transcatheter heart valve (THV) was selected giving an 11% oversize for the annular perimeter of 82.3 mm. The inter-commissural distance at 4 mm was 29.2 mm giving it a flare configuration, 2 hence annular sizing was chosen. A percutaneous transfemoral TAVI under monitored anesthesia care was planned using standard operative recommendations 3 with an 18 French introducer sheath (Cook Incorporated, Bloomington). Pre-dilatation was performed with a 20 mm × 40 mm Z-MED balloon (B. Braun Medical Inc., Allentown, PA) using a handheld inflation device. After verifying a satisfactory valve loading on fluoroscopy, a slow deployment of EP+ THV was begun in a standard fashion aiming for a 3-5 mm implant depth. At 80% deployment level, the depth of the THV was judged to be 0 to 1 mm below the annulus. The THV was recaptured under pacing at 120 beats per minute. The second attempt was deemed satisfactory with a depth of about 2-3 mm in cuspal overlap view and about 4-5 mm in co-planar view below the annulus at 80% deployment level (Figure 1). In the same co-planar view, two radio-opaque lines were noted within the valve frame which were highly suspicious for valve infolding (INF). It should be noted that the radio-opaque lines were not seen in the cuspal overlap view. Since the THV was not completely released at this point, a decision was made to recapture and discard this THV, and TAVI to be re-attempted with a new THV and delivery system. As the valve could not be re-sheathed completely, the valve and the delivery catheter were removed en bloc. Once out of the body, the THV was inspected, and valve infolding was confirmed. The introducer sheath was examined for structural integrity. Another EP+ of the same size was deployed with a final depth of 4 mm below the annulus without any subsequent issues. Post-dilatation with a 25 mm Z-MED balloon resulted in satisfactory hemodynamic parameters (Figure 2).
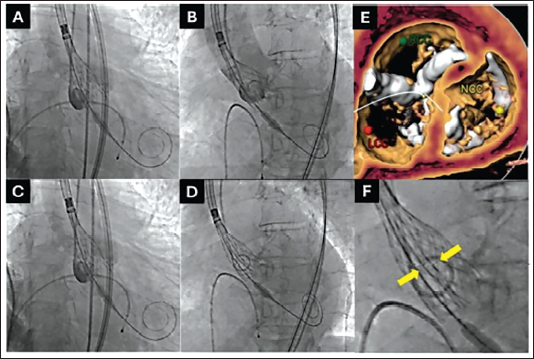
(A, B) Cuspal overlap and coplanar views, respectively, with a new THV showing optimal frame expansion. (C) Post dilatation with 25 mm Z-Med balloon. (D) Infolded valve, as seen ex-vivo, Note the frame invagination at 10 o’clock.
Discussion
SEV are made up of nitinol which has a shape memory that allows them to regain shape in-vitro. The release mechanism of a SEV involves unsheathing of the THV which should lead to a symmetric valve expansion. Frame mal-expansion is occasionally seen when the inflow portion of the valve frame does not expand properly due to stiff annular anatomy. In contrast, INF is said to occur when there is a persisting wrinkling along the stent frame resulting in an all-over invagination of the inflow and the outflow tract. 4 INF is observed more commonly with larger valve sizes, likely due to lower radial force. 5 INF is not exclusive to THV as it has also been reported in suture-less surgical valves. 4
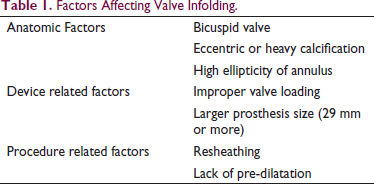
INF has been attributed to occur due to both anatomic as well as device-related factors (Table 1). 6 INF occurs more frequently in severely calcified valves with eccentric openings. Also, re-sheathing seems to be a major contributor for INF. A forward push during re-sheathing, combined with asymmetric THV expansion due to eccentric calcification and/or valve opening, provides a milieu for the valve frame to infold. 5
Factors Affecting Valve Infolding.
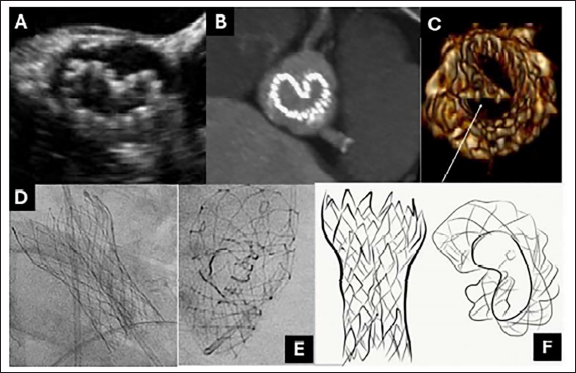
INF can be identified under fluoroscopy as one or two vertical lines on the partially or completely released valve frame, suggesting invagination of the valve frame along with a reduction in the transverse diameter. 4 It must be emphasized that radio-opaque vertical lines may not be seen in every fluoroscopic projection, particularly the cuspal overlap view. A right anterior oblique cranial view can demonstrate the invagination of the valve frame in short axis (Figure 3). In the mid-esophageal short-axis view during transesophageal echocardiography, the Pac-Man sign can be appreciated as well. 7
INF causes incomplete valve apposition leading to significant paravalvular leak with or without a higher transvalvular gradient. 5 The hemodynamic effects of this acute phenomenon can be dramatic, often presenting as resistant hypotension, ventricular tachycardia, or cardiac arrest. The hemodynamic insult, along with the additional procedural steps, leads to higher fluoroscopy time and contrast use, as well as increased risk of periprocedural stroke, acute kidney injury and need for permanent pacemaker implantation. 4
Every attempt should be made to diagnose INF before the final release of the valve. Ensuring optimal valve loading is part of the best practices, with particular attention to ensuring there is no severe crown overlap. Before the final release of the valve, orthogonal images of the valve frame should be obtained to ensure optimal frame expansion, since one view can fail to identify INF as highlighted earlier. 8 If INF is identified prior to the valve release, the THV should be recaptured and TAVI to be re-attempted using a new valve and delivery system. INF identified after the release of the valve needs intervention depending on the hemodynamic stability of the patient. Post-dilatation should be attempted to avoid overzealous balloon oversizing to prevent annular injury. If post-dilatation results in complete valve expansion, or partial valve expansion with mild paravalvular leak and stable hemodynamics, no further intervention is needed. However, if severe INF or leak persists, then valve-in-valve TAVI with a ballon-expandable valve or a surgical conversion should be considered (Figure 4). 4 Although not reported in the literature as a treatment for INF, snaring and pulling the THV out of its annular position followed by TAVI using a new THV may be tried as a last resort.
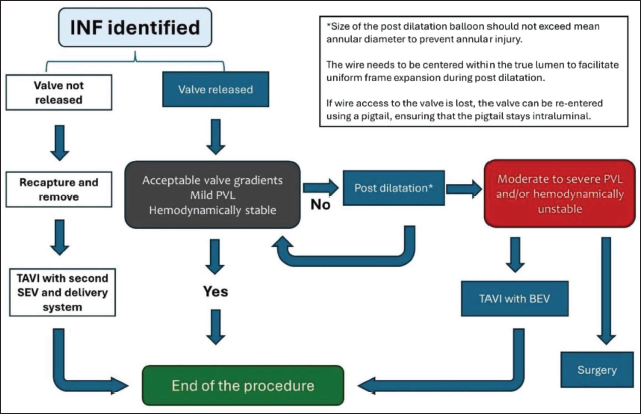
Conclusion
INF is an infrequent complication of TAVI with SEV. Prompt diagnosis is crucial to minimize the impact of acute hemodynamic catastrophe. Remedial steps include post-dilatation and/or implanting a second BEV inside the infolded valve. Be mindful of the higher peri-procedural stroke rate as well as other adverse events.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
