Abstract
Transcatheter aortic valve implantation has taken the world by storm. The past decade saw expanding indications of Transcatheter aortic valve implantation to include patients at low surgical risk for surgical aortic valve replacement, patients with degenerated aortic bioprosthesis, and repurposing existing transcatheter aortic valves to treat pathology involving other valves. Also around the same time, various iterations of existing transcatheter heart valves and newly launched valves were made available for human use in India. This article highlights salient features of all the commercially available transcatheter heart valves in India. The article also provides a narrative to help a cardiologist choose a particular valve that would best suit the patient taking into account the valve design characteristics and features.
Keywords
Since its first human use in 2002, transcatheter aortic valve implantation (TAVI) has become the standard of care for treating severe symptomatic aortic stenosis in patients at high risk for surgical aortic valve replacement (SAVR) and an attractive minimally invasive alternative to low or intermediate surgical risk patients. As the patient pool for TAVI continues to expand, different types of transcatheter heart valves (THVs) have been introduced for commercial use, each with its unique characteristics. This review article provides an overview of the various THVs approved for commercial use in India.
The Basic Design of a Transcatheter Valve
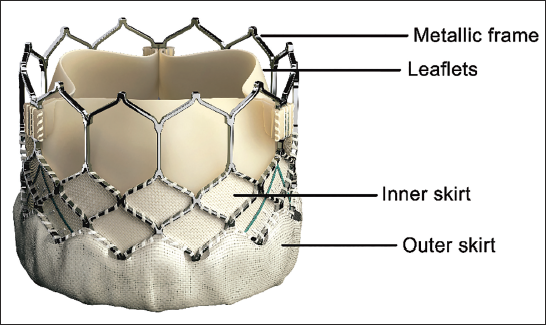
Key design elements of a THV include (a) a metallic frame, (b) leaflets, (c) an inner skirt, and (d) an outer skirt (Figure 1). A few valves have additional structural elements, adding to their uniqueness, which will be highlighted in the respective sections.
Components of a Transcatheter Heart Valve. Shown in the image is Sapien 3 (Edwards Lifesciences).
The Metallic Frame1–3
The frame defines the basic three-dimensional shape of a THV, is the main support structure that anchors to the aortic annulus, and provides a platform on which bioprosthetic leaflets are sutured. The frame is made up of either an MP35N alloy or a nitinol alloy. Caution is advised for its use in patients with documented allergy to metals such as cobalt, nickel, titanium, chromium, molybdenum, manganese, silicon, and/or other polymers. At 300–600 microns thickness, conventional THV frames are much thinner than surgical valve struts, allowing for a larger internal diameter compared to a similarly labeled surgical bioprosthesis.
MP35N is a multiphase cobalt-based alloy containing a high percentage of nickel, chromium, and molybdenum. The alloy has a high tensile strength and corrosion resistance. MP35N is used in balloon-expandable valves such as Sapien 3 (Edwards Lifesciences) and Myval/Octacore (Meril Lifesciences).
Nitinol is a nickel–titanium alloy. It has super-elasticity and shape memory. Nitinol can be deformed and will stay in the deformed state at lower temperatures. As the ambient temperature increases, nitinol assumes its original shape. A valve frame made up of nitinol is kept in an ice-cold bath during the valve loading process in vitro so that it can be crimped inside the capsule with ease. Once the valve is unsheathed inside the human body, the core body temperature facilitates the nitinol to self-expand to its original shape. This unique feature of nitinol is used in all self-expanding THVs such as the Evolut series (Medtronic Inc.), the Acurate Neo series (Boston Scientific), Portico and Navitor (Abbott), and Hydra (Vascular Innovations).
The leaflets
THV leaflets are derived from bovine or porcine pericardial tissue. Evolut (Medtronic Inc.) and Acurate Neo (Boston Scientific) valve series have porcine pericardial leaflets, while others use bovine pericardium. The carefully selected pericardium is laser-cut into a preformed leaflet shape and then sutured onto the metallic frame. Leaflets can be mounted within the intra-annular portion of the metallic frame or higher up, making a THV intra-annular or supra-annular, respectively. All the commercially available valves have a tri-leaflet design. The biological leaflet tissue is treated with proprietary anti-calcific treatment that varies between different manufacturers.
The Inner and the Outer Skirts
The proximal portion of the metallic frame, below the leaflet insertion, has a skirt sewn at the adluminal surface of all the transcatheter valves (the inner skirt). Few THVs also have an outer skirt attached to the abluminal surface of the valve frame that interacts with the aortic annulus. The inner skirt provides a seal, thus preventing the backflow of blood from the aortic sinus into the left ventricular outflow tract through the metal struts. The outer skirt increases the apposition area of the THV as it interacts with the aortic annulus, minimizing paravalvular leak (PVL). Skirts can be made up of either polyethene terephthalate (PET) fabric or pericardial tissue.
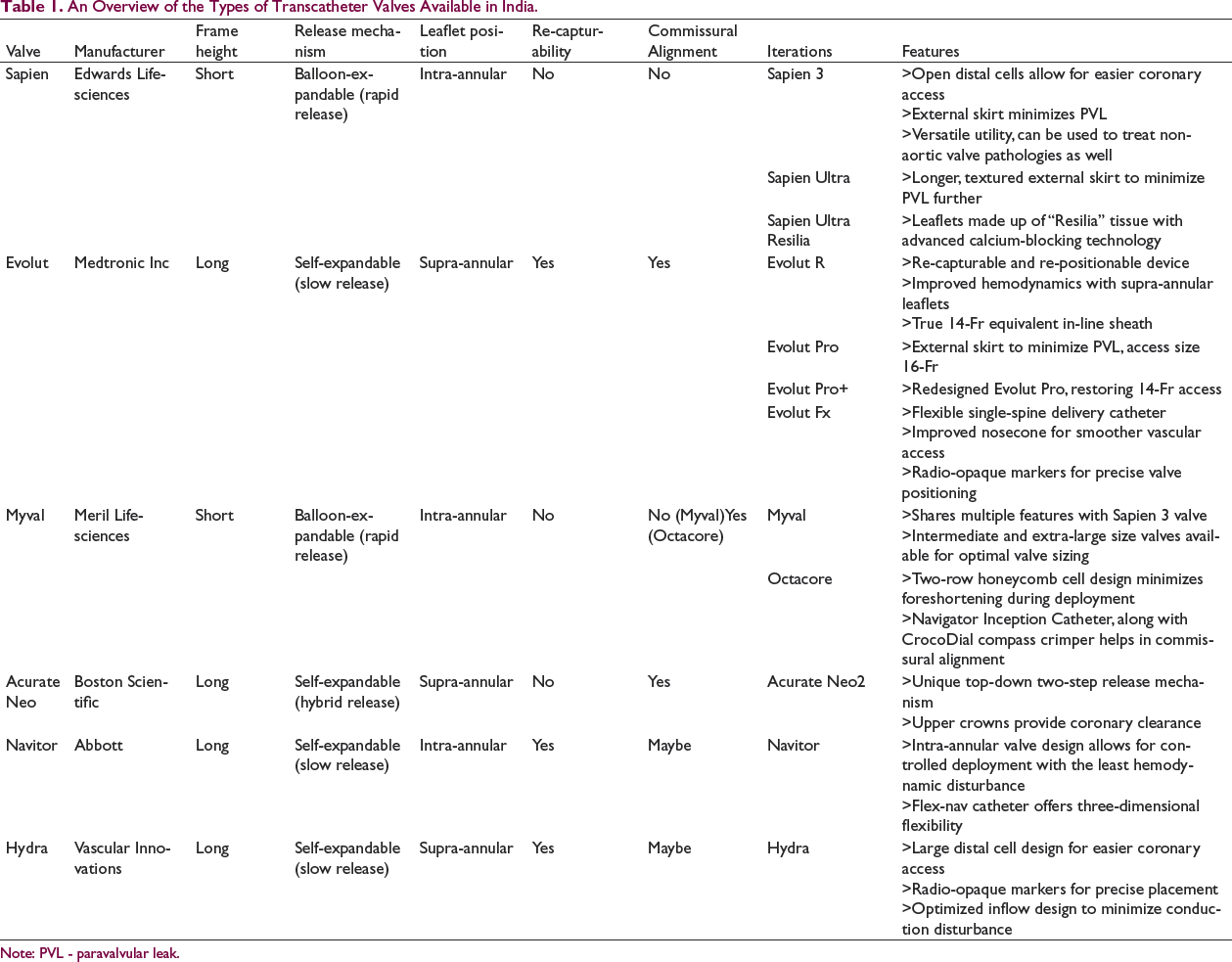
Types of Transcatheter Heart Valves (Table 1)
THV can be classified based on its frame height, leaflet position, release mechanism, or whether it can be recaptured and repositioned.
An Overview of the Types of Transcatheter Valves Available in India.
Frame Height
Commercially available valves can be grouped into short-frame valves (SFVs) and long-frame valves (LFVs). SFVs have frame height ranging from 15.5 to 22.5 mm depending on the valve type and size. SFVs almost always have the leaflets mounted in an intra-annular position. Using standard THV deployment techniques, the distal edge of an SFV may not cross the coronary ostia plane in many patients, allowing for easier access to coronaries post-TAVI. Also, except in a few patients with short sinus height, SFV does not reach the sino-tubular junction. This reduces the risk-plane in case a future valve-in-valve TAVI is needed. LFVs have frame heights ranging from 45 to 55 mm. The tall frame allows for the leaflets to be mounted in the supra-annular position, and the frame universally extends beyond the sino-tubular junction.
Leaflet Position
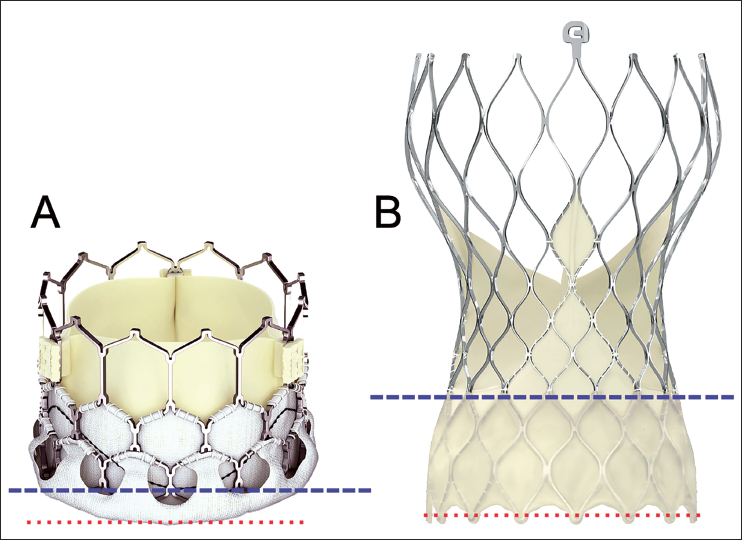
THVs can be classified as intra-annular or supra-annular valves depending on whether the leaflets are positioned within the annular plane or above (Figure 2). The post-implant internal diameter (ID) of a THV at the leaflet level is constrained by the rigid annulus in the case of an intra-annular valve. In contrast, a supra-annular THV can achieve a larger ID at the leaflet level; hence, it is expected to provide better hemodynamic performance. Also, owing to the leaflet design, a supra-annular valve is associated with a lower incidence of leaflet thrombosis due to a smaller neo-sinus. 4
Transcatheter Heart Valves. (A) Myval; (B) Evolut Pro. Note the leaflet attachment plane (broad dotted line—blue) in reference to the annular plane (fine dotted line—red). Myval is an intra-annular valve, whereas Evolut Pro is a supra-annular valve.
Release Mechanism
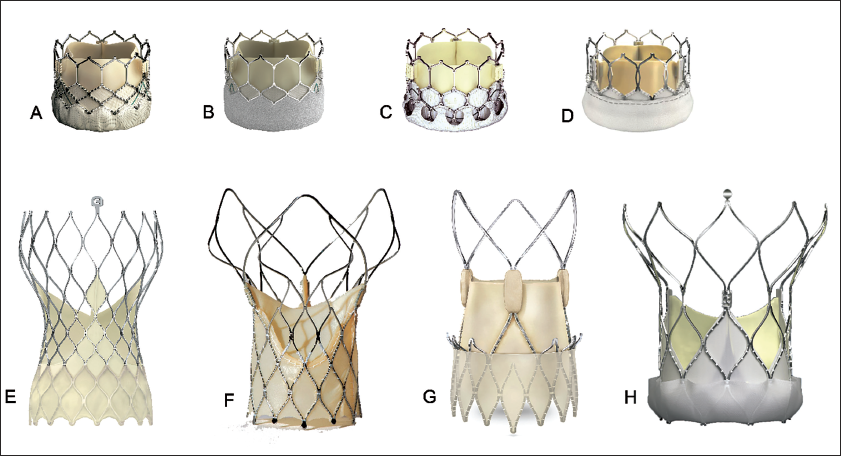
THV has traditionally been classified as balloon-expandable (BE-THV) or self-expandable (SE-THV) (Figure 3). All the commercially available BE-THVs have cobalt alloy frames with an intra-annular leaflet design. SE-THVs are made up of nitinol, an alloy that forms a strong cubic crystal structure referred to as austenite with a high radial strength once exposed to warmer body temperature, allowing it to self-expand once released. SE-THV can have an intra-annular or a supra-annular leaflet design.
Commercial THVs Available in India. (A) Sapien 3 (Edwards Lifesciences), (B) Sapien Ultra (Edwards Lifesciences), (C) Myval (Meril Lifesciences), (D) Octacore (Meril Lifesciences), (E) Evolut Pro+ (Medtronic Inc.), (F) Hydra (Vascular Innovations), (G) Acurate Neo 2 (Boston Scientific), (H) Navitor (Abbott). Valves A–D are balloon-expanding, intra-annular; valves E–H are self-expanding, supra-annular (except Navitor); THV, transcatheter heart valve.
THVs can have a rapid deployment or a slow controlled deployment. All BE-THVs are to be deployed rapidly. All but one SE-THV have a controlled release mechanism. Acurate Neo2 (Boston Scientific) is a SE-THV with a hybrid release mechanism where the initial release is slow but the final deployment is rapid. Controlled release valves can be recaptured and repositioned before the final deployment, whereas valves with a rapid or hybrid release mechanism cannot be recaptured or repositioned.
Transcatheter Heart Valves Available for Commercial Use in India
Sapien 3 (Edwards Lifesciences)
Sapien 3 (S3) is the third-generation valve in the Sapien series of valves. S3 is a balloon-expandable rapid-deployment short-frame THV with an intra-annular leaflet design. The MP35N frame provides high radial strength. The distal cells of S3 have an open cell geometry for easier coronary access. S3 has an outer skirt made up of PET fabric, which is designed to minimize PVL. S3 is available in four sizes (20, 23, 26, and 29 mm) and can treat annuli with area-derived diameters ranging from 18.6 to 29.5 mm.
S3 can be deployed at the aortic position in either retrograde or antegrade fashion. Standard transfemoral TAVI, as well as most of the alternate accesses, utilizes a retrograde approach. Antegrade deployment, although rarely done, can be performed via trans-septal or trans-apical access. S3 is the only US-FDA-approved valve for an aortic and mitral valve-in-valve procedure. S3 has been successfully deployed in pulmonic position as well.
S3 is crimped over the proprietary Commander delivery system (Edwards Lifesciences) and introduced via a 14 Fr expandable eSheath (16 Fr for a 29 mm valve). The recommended minimum mean luminal vascular diameter for the 14 Fr eSheath is 5.5 mm (6.0 mm for 16 Fr). It should be noted though that the outer diameter of the expandable eSheath increases momentarily as the crimped valve and the pusher traverse through it. The Commander delivery system has an advanced “flex” mechanism, which allows smoother navigation at the aortic arch and helps cross the aortic valve in difficult anatomy, for example, horizontal aorta, bicuspid aortic valve, etc.
A dedicated Certitude delivery system is available for trans-apical access. The delivery catheter is introduced via an 18 Fr or a 21 Fr Certitude sheath.
Sapien Ultra is the fourth-generation valve of the Sapien series. It has a textured PET fabric, which is approximately 40% longer than S3 to improve external sealing (Figure 4). The Sapien Ultra Resilia valve features the newer Resilia tissue, which has advanced calcium-blocking technology and is shown to have reduced calcification in juvenile sheep model. Sapien Ultra is expected to be launched in India in the year 2024.
Difference in Height and Texture of the External Skirt (Curly Brackets) of Sapien 3 (Left) vs Sapien Ultra (Right).

Evolut Pro+ (Medtronic Inc.)
Evolut Pro+ (EP+) is the fourth-generation iteration of the CoreValve-Evolut series. EP+ is a self-expandable slow-release long-frame THV with a supra-annular leaflet design. The nitinol alloy enables the frame to self-expand and acquire a high radial strength when exposed to warm body temperatures. EP+ valve can be recaptured and repositioned as required to obtain an optimal implant. The valve frame exerts a differential radial force at each level, conforming to the anatomical requirements. While the high radial force of the inflow segment allows it to conform to the annulus, the low radial force of the outflow segment enables a controlled release. The waist portion, where the leaflets are mounted, has a high hoop strength, which promotes circularity. The cells of the frame allow for up to 10 Fr guide catheter access for future coronary interventions. EP+ has an outer pericardial skirt to minimize PVL. EP+ is available in four sizes (23, 26, 29, and 34 mm) and can accommodate annuli with perimeter-derived diameters ranging from 18 to 30 mm.
The EP+ is always implanted in a retrograde fashion at the aortic position and cannot be used for trans-septal or trans-apical access. Similarly, EP+ is not to be used in mitral, tricuspid, or pulmonic positions.
The EP+ valve is crimped on a proprietary delivery system with an inline sheath. The thin profile of the porcine leaflets allows the valve to be delivered via a 14 Fr equivalent delivery system (18 Fr for 34 size valve) and can be used in patients with a mean vascular luminal diameter of 5 mm or larger. Commissural alignment can be achieved in tricuspid aortic valve cases by introducing the delivery catheter with the flush port oriented at the 3 o’clock position and ensuring that the hat marker is always at the outer curvature.
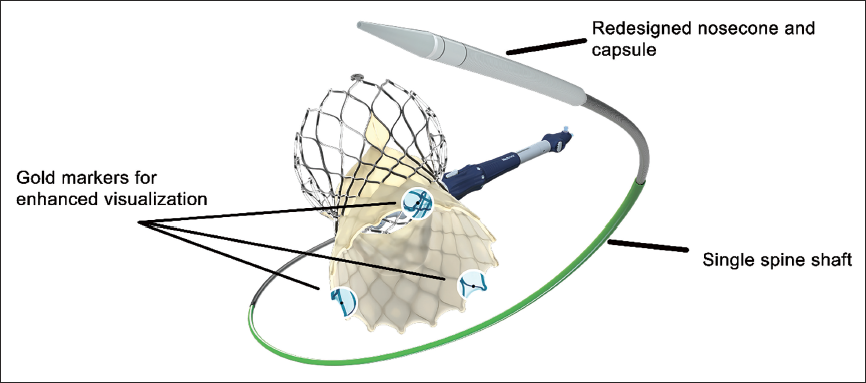
Evolut Fx is the fifth-generation valve with numerous structural iterations. The delivery catheter has a single spine design, as opposed to two spines in earlier generations, adding more flexibility. The nosecone is redesigned to allow for smoother vascular entry and navigation. The valve frame itself has three golden markers 3 mm above the proximal edge of the frame (Figure 5). The markers are placed equidistant to each other, and the position of the valve commissures is marked. The markers are radio-opaque and aid in precise valve positioning as well as commissural alignment. The Evolut FX was made available for commercial use in India in December 2023.
Evolut Fx: The Newest Generation Valve in the Evolut Series. Salient features include single spine shaft, redesigned nosecone and capsule, and gold markers over the valve for enhanced visualization.
Myval (Meril Lifesciences)
Myval is a balloon-expandable rapid-deployment short-frame THV with an intra-annular leaflet design. The MP35N frame provides high radial strength and has open distal cells for easy coronary access. It has an outer fabric skirt to minimize PVL. Myval is available in four conventional sizes (20, 23, 26, and 29 mm), three intermediate sizes (21.5, 24.5, and 27.5 mm), and two extra-large sizes (30.5 and 32 mm). Myval can treat patients with area-derived annular diameters ranging from 18.5 to 32.7 mm. While all the valve sizes are compatible with the 14 Fr expandable Python sheath, larger size THV requires at least 5.5 or 6 mm mean luminal diameter for vascular access.
Myval THV is directly crimped on the balloon of the Navigator delivery system, unlike the S3, making the implant procedure simpler. The Navigator balloon has proximal and distal stoppers to prevent untoward valve migration. The catheter has a “flex” system designed for smoother navigation through the aortic arch and the annulus. In addition to the standard retrograde approach, Myval can also be used for antegrade implantation through trans-septal or trans-apical access. In addition to TAVI and ViV-TAVI, Myval has also been successfully used in mitral and pulmonic positions.
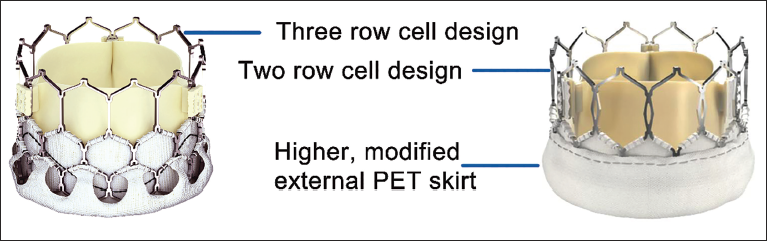
Myval Octacore is the second-generation valve in the Myval series with numerous design iterations. The three-row hybrid honeycomb cell design of Myval has been modified to a novel two-row interlacing geometrically identical octagonal cell design (Figure 6). Each of the two rows forms the inflow and the outflow zone, respectively. The inflow portion has an inner and an outer fabric skirt to minimize PVL. The symmetric cell design minimizes valve foreshortening during deployment. Octacore THV is deployed using a redesigned Navigator Inception Catheter (NIC). The NIC has four radio-opaque balloon markers, one each at the proximal, distal, geometrical mean, and landing zones. The Octacore valve is crimped using the new CrocoDial Compass crimper, which, in combination with the “aligners” on the catheter shaft, assists in commissural alignment.
Comparing Design Characteristics of Myval (Left) vs Octacore (Right) Valves. Octacore has two rows of interlaced octagons stacked one over the other. The design feature minimizes frame foreshortening during deployment.
Acurate Neo2 (Boston Scientific)
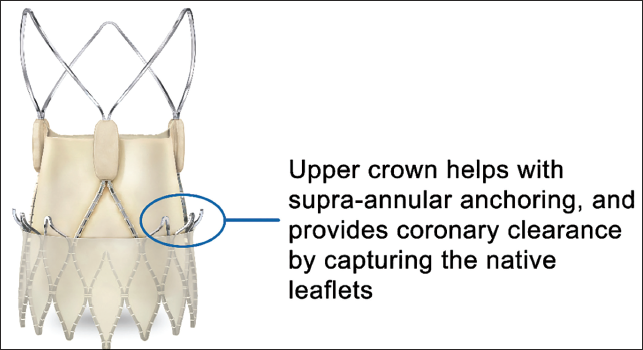
Acurate Neo2 (AN2) is the second-generation device in the Acurate Neo series. AN2 is a self-expandable hybrid-release long-frame THV with a supra-annular leaflet design. There are several unique design elements of AN2 that are worth mentioning. AN2 has an open-frame design, which allows for easier coronary access, and a longer outer porcine pericardial skirt to minimize PVL. The frame has upper crowns, which provide coronary clearance by capturing the native aortic valve leaflets during deployment (Figure 7). This may reduce the risk of coronary occlusion. 5 The lower crown protrudes minimally into the LVOT and has lesser radial force, features that may reduce conduction disturbances. The valve has a two-step top-down release mechanism, in contrast to most of the self-expanding valves. The first step involves the slow release of the distal stabilization arches. At this stage, micro-adjustments can be made for depth correction. The lower portion of the valve is released next in a rapid fashion. Once released, partially or completely, AN2 cannot be recaptured. Radio-opaque posts on the stabilizing arches, along with the free cells located on the lower crown, assist with commissural alignment. AN2 is available in 3 sizes (small: 23 mm, medium: 25 mm, and large: 27 mm) and fits annuli diameters ranging from 21 to 27 mm. A 14 Fr expandable iSleeve introducer sheath is used for valve delivery.
Acurate Neo 2 valve. The upper crown (circled) engages with the native coronaries during deployment, allowing for additional coronary clearance.
Navitor (Abbott)
Navitor (Nav) is the second-generation valve in the Portico series, with optimized radial force and an outer sealing skirt called Naviseal. Nav is a self-expandable slow-release long-frame THV with an intra-annular leaflet design. Nav THV, just like other nitinol frame valves, can be recaptured and repositioned as required to obtain an optimal implant. The Nav frame has larger cell geometry offering easier coronary access. The intra-annular valve design allows for a controlled deployment with the least hemodynamic disturbance. Nav is available in 4 sizes (23, 25, 27, 29 mm), accommodates annuli diameters ranging from 19 to 27 mm, and can be delivered through a 14 Fr integrated sheath (15 Fr for 27 and 29 mm valve sizes). The FlexNav delivery catheter offers a three-dimensional flexibility, which adds to the hydrophilic coating, allowing for smoother vascular navigation.
Hydra (Vascular Innovations)
Hydra is a self-expandable slow-release long-frame THV with a supra-annular leaflet design. The valve can be recaptured, repositioned, and retrieved as required. The valve frame has a large cell design with “tentacles” at the distal end for easier coronary access. The non-flared inflow design is optimized to minimize interaction with conduction tissue while maintaining high radial strength. Two sets of radio-opaque markers are placed over the nitinol frame, at node one and node three, respectively. The proximal markers provide a visual cue for optimal implant depth, while the distal markers indicate leaflet deployment. The valve has an extended inner skirt to mitigate aortic regurgitation but lacks an outer skirt. Hydra is available in three sizes (22, 26, and 30 mm) and can treat annuli diameters ranging from 17 to 27 mm. The newer Aortic Valve Delivery Catheter has a 14 Fr equivalent inline sheath (20 Fr introducer sheath for a 30 mm valve) with an active release mechanism for the final valve release.
Preferred Patient Subset
While all the commercially available THVs are designed to meet the anatomic and clinical requirements of most patients with severe aortic stenosis, certain design characteristics may favor one THV type over the other. Given the paucity of randomized data comparing one THV with another, any preferences mentioned below should be considered an expert opinion at best.
Small Annulus
SAVR or TAVI in patients with small aortic annulus, arbitrarily defined at a mean annular diameter <23 mm or area <430 mm2, is associated with a higher incidence of patient-prosthesis mismatch (PPM).6, 7 Similarly, patients with degenerated surgical bioprosthesis are also at a higher risk for PPM post-TAVI due to the Russian-doll effect. THVs with a supra-annular leaflet position may be preferred in this patient subset since leaflets in the supra-annular position are not restrained within the confines of rigid aortic annulus or surgical valve frame and thus may provide improved hemodynamic outcome 6 For valve-in-valve (ViV-TAVI) cases where TAVI is implanted inside a degenerated surgical bioprosthesis, bioprosthetic valve fracture can be performed for selected surgical bioprosthesis for improved hemodynamics.
Horizontal Aorta
Aortic angulation (AA) of >48 degrees defines a horizontal aorta. Early experience suggested an inverse relationship between AA and device success, a phenomenon observed more frequently with SE-THV as opposed to BE-THV. 8 Early randomized trials of SE-THV excluded patients with AA >70 degrees, and as per the instructions for use (IFU), implantation of the Evolut valve should be avoided if aortic angulation is >70 degrees. Historically, short-frame BE-THV with an active flex system was favored in such anatomy, although there are conflicting data regarding the superiority of one valve design over another. 9 However, a recent study of TAVI in patients with horizontal anatomy suggested lower device success with Evolut but not with AN2. 10 While valve design may be a consideration for TAVI for patients with an horizontal aorta, contemporary TAVI performed by an experienced operator using any newer generation device is likely to achieve high device success 11 .
Alternate Access
Long-frame SE-THV has lower tolerance limits for aortic angulation when the right carotid or right subclavian approach is chosen. Caution should be exercised if aortic angulation is more than 30 degrees. In such cases, a short-frame BE-THV may allow for an easier passage and implant. If a trans-apical or trans-septal route is chosen, only short-frame BE-THV can be used (antegrade implant).
Bicuspid Aortic Valve
Patients with bicuspid aortic valves present a heterogeneous anatomic subset where a one-method-fits-all approach is unlikely to work. In patients with eccentric valve orifice or those with asymmetric or raphe calcification, adequate valve expansion can be challenging. A higher incidence of paravalvular leak with SE-THV and a higher incidence of annular rupture with BE-THV were seen with early-generation valves. 12 Bicuspid valve pathology was universally excluded from the majority of the landmark TAVI randomized trials. A systematic conclusion regarding the superiority of one valve design over another cannot be drawn from the available literature. Observational data thus far has shown device success of more than 95% using newer generation valves, and peri-procedural complication rates comparable to that of tricuspid aortic valve pathology13–17 BE-THV, by virtue of higher radial force, has a better ellipticity index and may be associated with a lesser incidence of PVL, but imposes a higher risk for annular injury compared to self-expandable valves. At times, an operator has to balance between the degree of residual PVL acceptable for that patient versus the added risk of annular injury with aggressive balloon dilatation.
Coronary Access
Preserving future coronary access in patients with established, or at risk of developing, coronary artery disease is of paramount importance. Coronary access should not be an issue if the coronary ostia are arising cephalad to the distal edge of the valve frame, as may be the case in many patients where short-frame valves are used. Coronary access may be challenging in patients with low coronary height or narrow-effaced sinuses. THVs with a long-frame design, supra-annular leaflet position, taller sealing skirt, or a higher stent cell density impose additional difficulty. A high THV implant further accentuates unfavorable anatomy for coronary access 18 . Hence, a detailed pre-procedural assessment for future coronary access is a mandatory step in all TAVI cases. Whenever anatomy-valve-related challenges are expected as discussed above, all attempts should be made to achieve commissural alignment (CA). Mal-aligned THV implant is associated with severely compromised, or impossible, future coronary access.19, 20 On the contrary, a high success rate can be expected in engaging the coronaries post-TAVI if CA is achieved. 21 Evolut, Acurate, and Navitor valve series have fluoroscopic markers to facilitate CA. 20 Octacore valve comes with the CrocoDial Compass crimper, which, along with aligners on the catheter shaft, aids in CA. Sapien and Myval valves do not have an established mechanism to achieve CA.
Low Coronary Height
Patients with low coronary heights are at a higher risk for coronary occlusion, as well as future coronary access, particularly if associated with effaced sinuses or long bulky calcified leaflets. Since coronary occlusion occurs due to displaced native leaflets, the choice of valve type may not be relevant since all similarly sized THVs in principle should displace the native leaflets equally. However, in theory, the upper crown of the AN2 valve may engage the native leaflets during deployment, providing additional clearance for coronary flow. Choosing a THV that is “less” oversized and using a controlled release THV to evaluate for coronary flow at the partial deployment stage are strategies that are often used, but not validated, to reduce the risk of coronary occlusion.
Future Valve-in-valve Transcatheter Aortic Valve Implantation
Lifetime management of aortic stenosis in low-risk patients must include an action plan for the next procedure when the index THV degenerates. Preliminary data suggests that a ViV-TAVI may be a safer alternative for failed THV than a redo SAVR.22, 23 If a TAVI explant is needed, long-frame THVs are associated with a higher need for ascending aortic replacement. 22
A strategy on how to preserve coronary blood flow during a future ViV-TAVI procedure should be defined during the index TAVI procedure itself. The second THV, when placed inside the first, will splay open the leaflets of the first THV (Figure 8). This, in addition to the inner skirt of the first THV, creates a tubular stent graft-like structure, which extends from the proximal edge of the THV up to the distalmost edge of the splayed leaflets, often referred to as the “risk-plane.” 24 For optimal coronary perfusion post-ViV-TAVI, blood has to traverse outside this “tubular graft,” or the functional neo-skirt. Any impediment in the blood flow due to narrow sinuses or sinus sequestration may compromise coronary perfusion. The aim is to keep the risk plane below the sino-tubular junction and/or have at least a 2 mm gap between the outer valve frame and the aortic wall to allow diastolic blood flow. Intra-annular valves offer a shorter risk plane as opposed to supra-annular valves, hence reducing the likelihood of sinus sequestration. Conversely, a shallow implant during the index TAVI procedure increases the height of functional neo-skirt during a future ViV-TAVI, hence increasing the risk for sinus sequestration.
ViV-TAVI with Evolut R Placed Inside Myval. Note that the Evolut valve splays open the leaflets of Myval, creating a neo-skirt. The distal edge of this neo-skirt denotes the risk plane (dotted ellipse). It is at this plane where at least 2 mm clearance is needed between the external valve frame and the aortic wall for optimal coronary perfusion. ViV-TAVI, valve-in-valve transcatheter aortic valve implantation.
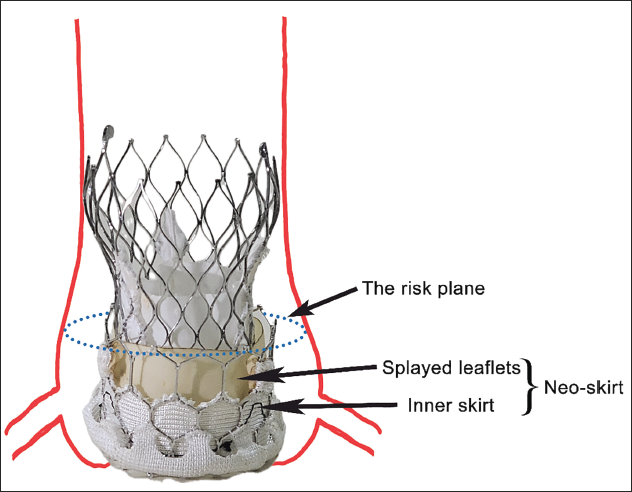
The second issue while choosing a THV is to consider coronary access during future ViV-TAVI procedures. Even if an individual cell size of a THV is large enough to accommodate a 10 Fr guide catheter, unless the cells of the second THV align perfectly over the first THV, the two overlapping frames after a ViV-TAVI will make the effective cell size smaller. SFV, which does not extend beyond the plane of native coronary ostia, may be best suited for index TAVI procedure as it will not only minimize the risk plane but will also avoid metal crowding in front of the coronary ostia, allowing for easier coronary access. If this cannot be achieved, due to either shorter coronary height or a longer valve frame, the index THV should be implanted with optimal commissural alignment and attempts should be made to keep the risk plane below the sino-tubular junction.
Vascular Anatomy
A higher sheath-to-artery ratio is associated with higher vascular complications, particularly in the setting of vascular calcification. In patients with borderline vascular anatomy, using the lowest profile delivery system is prudent. Although the crossing profile of a 14 Fr expandable delivery sheath is approximately 6.0 mm, similar to that of an inline sheath, it expands momentarily to 20 Fr or higher as the crimped valve navigates through it, leading to a transient increase in sheath-to-artery ratio. 25
LVOT Calcium
Annular calcification extending into the LVOT often leads to frame under-expansion, leading to a higher incidence of PVL. Post-dilatation attempts to achieve optimal expansion impose an added risk for tissue injury during balloon inflation. Use of SE-THV may reduce the risk of annular injury as the size of the pre- or post-dilatation balloon, if need be, can be chosen based on the calcium-free dimensions. 26 On the other hand, a higher radial force of BE-THV may provide better sealing, leading to a lesser incidence of PVL, albeit at a slightly higher risk for tissue injury.
Poorly Defined Annular Anatomy
During emergency TAVI or in patients with severely deformed leaflet pathology, defining precise annular anatomy may not be feasible. A generously oversized SE-THV can be chosen in such cases to minimize the risk for valve embolism as well as PVL, without adding much risk for annular injury. A similar approach can be taken for isolated aortic regurgitation cases, where it may be difficult to achieve a stable implant using a nominally oversized THV due to the absence of leaflet calcium.
Conclusion
Today’s cardiologists are equipped with multiple commercially available transcatheter valve systems that can be chosen for a TAVI procedure by matching valve characteristics to that of the patient’s clinical requirements. It is prudent to understand the fundamentals of a valve design as well as the specifics of all the commercially available valves so that an informed physician can choose a valve best suited for a particular patient.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable
