Abstract
Atrial fibrillation (AF) and heart failure (HF) are epidemics that deserve greater attention in the current era. With improved medical therapies and robust revascularization strategies for acute coronary syndromes, there has been a substantial increase in the population of people living with stable coronary artery disease, which in turn contributes to the AF and HF burden. AF and HF are unique disease processes, in that each can precipitate the other, and in those with co-existing AF and HF, the burden of morbidity and mortality is amplified. While there has been remarkable progress in the sphere of pharmacological therapy for HF (particularly HF with reduced ejection fraction), medical therapy options for rate and rhythm control for AF have been largely unchanged for several decades. Instead, advances in catheter ablation (CA) technology and strategies for AF ablation have culminated in a robust evidence base, showing improved survival, quality of life, and restoration of ventricular function. The purpose of this clinical review article is to summarize the pathophysiological link between AF and HF and to emphasize the role of CA in the management of such patients.
Keywords
Introduction
The purpose of this clinical review article is to summarize the pathophysiological link between atrial fibrillation (AF) and heart failure (HF) and to emphasize the role of catheter ablation (CA) in the management of such patients. The first section provides an executive summary of the relationship between AF and HF and highlights the role of left atrial remodeling and fibrosis. The second section delves into the pharmacological and non-pharmacological management options at every stage of AF, from stage 3A (paroxysmal AF) to stage 4 (permanent AF). 1
Relationship Between AF and HF
Epidemiology of AF and HF
AF and HF are two major cardiovascular public health problems that increasingly co-exist. Globally, AF prevalence is increasing and was estimated to be 50 million in 2020.2, 3 While prevalence data from India is somewhat limited, it is estimated to be approximately half the prevalence of AF in the United States (~400-500 per 100,000 individuals vs. ~1,000 per 100,000 individuals) as per the Global Burden of Disease (GBD) study.2, 4 The same study pegged the global prevalence of cardiomyopathy and HF at 6.11 million in 2020, which is likely a significant underestimation as the NHANES (National Health and Nutrition Examination Survey) 2017-2020 data estimates the HF burden in the United States alone to be ~6.7 million. 5 Other studies estimate the global burden of HF to be over 23 million. 6
Further, it is estimated that patients with either AF or HF alone have an approximately 40% risk of developing the other condition. 7 In patients with AF, there is an increased risk of the development of both HF with preserved ejection fraction (HFpEF) and HF with reduced ejection fraction (HFrEF), albeit the association with HFrEF may be stronger (incidence rate of 12.8 per 1,000 person-years vs. 4.9 per 1,000 person-years). 8 In patients with established HFpEF, the prevalence of AF is in the 25%-39% range (increasing AF risk with worsening diastolic filling patterns), while with advanced HFrEF, the prevalence of AF can be as high as 49%. 9
While the association between HF and mortality is established and well accepted (~45% survival at five years), 10 AF has traditionally been considered to significantly impact quality of life (QoL) with a modest effect on mortality. 11 However, in patients with HF and AF, multiple studies have demonstrated a higher risk of death. 7
Pathophysiology of AF and HF
While several shared risk factors and co-morbidities contribute to the development of AF and HF individually, often there is an interplay of structural and pathophysiological changes. 12 Shared risk factors such as age, hypertension, diabetes, obesity, sleep apnea, coronary artery disease, and structural heart disease alter the atrial and ventricular myocardium directly and indirectly such that substrate for both AF and HF is created (Figure 1).
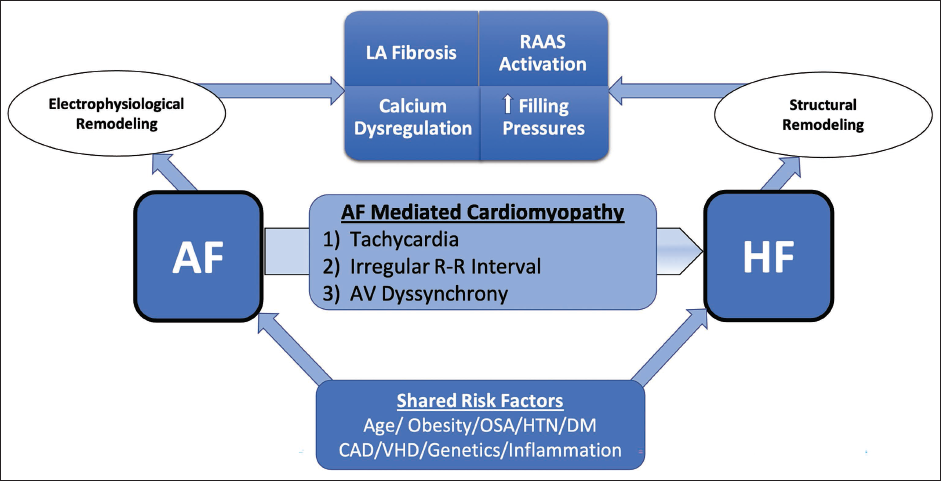
AF, atrial fibrillation; AV, atrioventricular; CAD, coronary artery disease; DM, diabetes mellitus; HTN, hypertension; LA, left atrial; OSA, obstructive sleep apnea; RAAS, renin angiotensin aldosterone system; VHD, valvular heart disease.
In patients with HF, direct pressure-related changes can set up AF substrate. This includes increased left ventricular and atrial filling pressures with resultant atrial distension/stretch and atrial fibrosis that enable the electrophysiological milieu to perpetuate AF. The presence of mitral regurgitation as part of the HF complex further is a risk factor for left atrial dilatation and AF development. 13 Similarly, tricuspid regurgitation is associated with right atrial dilatation and increased AF burden.14, 15 Conversely, the presence of AF creates atrioventricular dyssynchrony and eliminates atrial contraction, both of which result in suboptimal biventricular filling and can accentuate diastolic dysfunction. 13 Rapid irregular ventricular rates also interfere with ventricular filling and lead to biventricular pressure rises and adverse ventricular remodeling, including fibrosis. Therefore, AF-induced cardiomyopathy is associated with biventricular dysfunction. 16
In addition to pressure-related structural changes in the atria and ventricles, patients with HF also have calcium homeostatic abnormalities and neurohumoral activation (renin-angiotensin system as well as adrenergic activation) that may promote atrial fibrosis, atrial dispersion, and alteration in the action potential duration and effective refractive periods.17, 18 These maladaptations promote the development of atrial ectopics, atrial tachycardias, and AF. Additionally, systemic inflammation may further be implicated in the pathogenesis, particularly in HFpEF patients. 19 The resulting endothelial dysfunction and inflammation in the atrial and ventricular myocardium can accelerate the development of an electrophysiological substrate for AF genesis and maintenance. This explains the considerable impact of weight loss, exercise, and improvement of the metabolic profile on AF burden in such patients.20, 21
Atrial Fibrillation-mediated/Induced Cardiomyopathy
Since it is mechanistically possible for AF to develop in patients with HF and vice versa, it is important to understand and recognize the clinical entity of AF-mediated cardiomyopathy (AMC) or AF-induced cardiomyopathy (AIC).22, 23 Diagnosing this condition can often be challenging because AMC/AIC may develop even in patients with rate-controlled AF, indicating mechanisms that may implicate myocardial ischemia, inflammation, neurohumoral changes, and energy depletion. 22 AMC/AIC probably accounts for a large subset of tachycardia-induced myopathy (>50%), and as it is a reversible condition, pursuit of aggressive rhythm control is recommended. 24 As such, since several anti-arrhythmic drugs may also propagate the cardiomyopathy in AMC/AIC, American and European guidelines recommend CA as the first line option for rhythm control when AMC/AIC is suspected.1, 25
Left Atrial Fibrosis and AF
As previously discussed, various pathological processes culminate in atrial stretch and subsequent fibrosis. Prior animal and human studies have demonstrated that AF leads to atrial fibrosis.26, 27 In turn, atrial fibrosis perpetuates atrial arrhythmias by establishing an ideal electrophysiological substrate. As the AF burden increases, this leads to a vicious cycle leading to increased atrial fibrosis.17, 28
This interest in understanding the burden of atrial fibrosis in AF patients resulted in the use of delayed enhancement MRI to identify fibrotic changes in the left atrium. These analyses showed that most AF patients have some degree of fibrotic changes in the atria that may vary from minimal to extensive (Figure 2).29, 30 Interestingly, the clinical AF phenotype (paroxysmal vs. persistent) may not correlate to the burden of fibrosis. Therefore, clinically, we encounter several persistent or permanent AF patients with minimal fibrosis and some paroxysmal AF patients with considerable left atrial fibrosis and myopathy.29–31
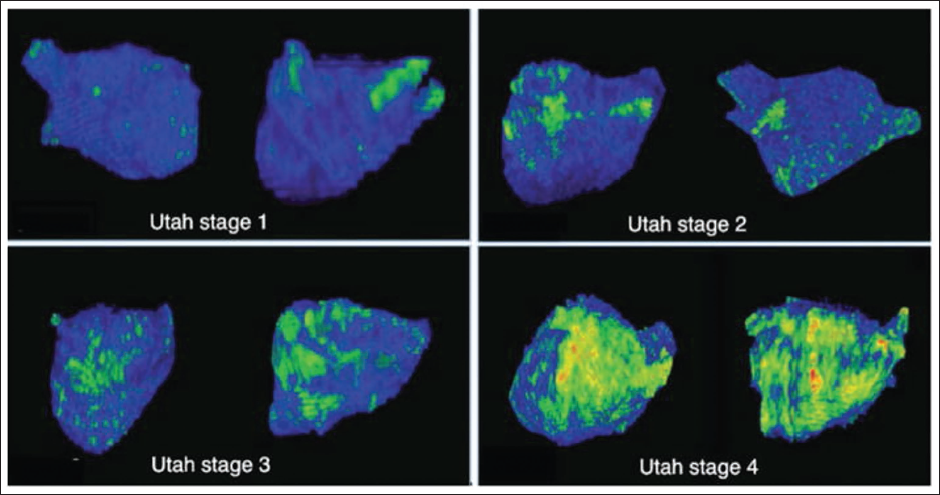
Utah stage 1: <5% fibrosis, Utah stage 2: 5–20% fibrosis, Utah stage 3: 20–25% fibrosis, Utah stage 4: >35% fibrosis. Reprinted with permission from Akoum et al, J Cardiovasc Electrophysiol. 2011 Jan; 22(1): 16–22. With permission from John Wiley and Sons.
Quantification of left atrial fibrosis was developed by Marrouche et al using pre-ablation delayed enhanced cardiac MRI sequences.29–31 In the DECAAF (Delayed-enhancement MRI Determinant of Successful Radiofrequency Catheter Ablation of Atrial Fibrillation) study, the classification schema included four stages of left atrial fibrosis (<10%, 10%-20%, 20%-30%, and >30% fibrosis of the left atrium), and AF recurrence after CA was significantly associated with increased fibrosis burden.
While much of this work is still translational and has not been integrated into routine clinical practice, it provides a framework for the understanding of AF substrate in HF patients and informs the management strategies that are likely to yield greater rhythm control. Ultimately, an individualized approach with greater use of CS in stages 1-2 of left atrial fibrosis and depending on atrioventricular junction (AVJ) ablation and device implants for stages 3-4 may result in better clinical outcomes and reduced procedures.
Management Strategies
In the following section, we will discuss the spectrum of rhythm management options, including pharmacological and catheter-based rhythm control strategies. We also highlight the role of a hybrid rate/rhythm control strategy utilizing AVJ ablation that may be particularly relevant to health care economies like India.
Rate vs. Rhythm Control
Over the years, there has been considerable controversy over the need for restoration of sinus rhythm (rhythm control) as compared to just ventricular rate control using medical therapy or AVJ ablation with ventricular pacing. This ambivalence in therapy options was perpetuated by randomized controlled trials such as AFFIRM (AF Follow-up Investigation of Rhythm Management) and RACE (Rate Control Versus Electrical Cardioversion), which showed no benefit of rhythm control over rate control in the general AF population.32, 33 However, it is important to note that the rhythm control strategies utilized anti-arrhythmic drug (AAD) therapy alone and not CA. Indeed, subsequent analyses showed that even in these studies, those patients maintaining sinus rhythm had better outcomes, and there was definite harm related to AAD therapy. 34 In this review, our focus is on rhythm restoration strategies, and we will highlight pharmacological and interventional techniques to achieve sinus rhythm.
Utilization of Electrical Cardioversion and Ibutilide-facilitated Cardioversion
Electrical cardioversion (ECV) to restore sinus rhythm may either be the first step in the management of AF and HF patients or may follow a course of oral AADs. This pre-treatment minimizes the chance of ECV failure/ immediate recurrence of AF. 35 Although antero-posterior electrode placement has traditionally been advocated, data suggest that antero-lateral electrode placement is more efficacious.36, 37
An alternate approach to the restoration of sinus rhythm is the use of an ibutilide infusion, which can be used in patients with prior AAD use as well.38–40 QT prolongation and torsades de pointes may occur in 3%-5% of patients, and the risk of this is higher in patients with EF < 30%. Concomitant magnesium administration may mitigate this risk and increase cardioversion success. 41 Finally, in patients who fail initial ECV or initial chemical cardioversion, ibutilide-facilitated ECV (F-ECV) has shown 100% efficacy in a prior randomized controlled trial. 42
Chemical cardioversion/ECV/F-ECV are all important modalities of restoring sinus rhythm at various stages of rhythm management (first occurrence or recurrence). In patients with HF (particularly HFrEF), maintenance of sinus rhythm will invariably need long-term AAD and/or CA.
Pharmacological Rhythm Control of AF for Patients with HFrEF
Pharmacological rhythm control using AADs is challenging for HFrEF patients, mainly because only two AADs can be administered without major contraindications. Amiodarone and dofetilide are both Class III AADs that have similar efficacy; however, adverse effects are not negligible. 43 The primary limitations of dofetilide use are baseline or subsequent dose-related QT interval prolongation and renal dysfunction, both of which can result in proarrhythmia (torsades de pointes). The standard practice is for inpatient admission with QTc monitoring for 48-72 hours during initial dofetilide loading. Traditionally, dofetilide has not been commercially available in India, and familiarity with the drug is low. It is important to note that no study has shown a mortality benefit with AAD use in HF, and the use is primarily for reduction of HF symptoms and hospitalizations.44, 45 This is likely because any mortality benefit conferred by the AAD is counterbalanced by the adverse events associated with long-term use. With studies demonstrating the mortality benefit of CA for AF in HF, increasingly AADs are being used for rhythm control while waiting for ablation or following ablation as an adjunct. It is also imperative to prescribe concomitant guideline-directed medical therapy for HFrEF, as optimal control of HF allows for enhanced action of rhythm-control therapy.
Pharmacological Rhythm Control of AF for Patients with HFpEF
Apart from standard guideline-directed medical therapy, the emphasis of care in HFpEF management is integrated management of all co-morbidities, including AF. Even brief episodes of paroxysmal AF can lead to hemodynamic decompensation. High AF burden can also convert a patient with stable HFpEF to HFrEF due to AMC/AIC. 46 Therefore, early rhythm control is recommended. 47 The findings of the EAST-AFNET4 trial support this strategy. This trial included 798 HF patients, of whom 56% had HFpEF, and patients assigned to early rhythm control did better than usual care patients. 48 In addition to the standard Class III AADs amiodarone and dofetilide, Class Ic AADs flecainide and propafenone may also be utilized in HFpEF patients if they do not have significant coronary artery disease burden or left ventricular hypertrophy. Sotalol and dronedarone (Class III agents) may be used with caution if there are no recent HF exacerbations, but in general, they are less preferable if alternative AADs can be utilized.
Catheter Ablation for AF
Over the past two decades, technology for CA has evolved and technical expertise has expanded, and randomized studies have shown the clear functional and mortality benefits following ablation for AF in HF patients.49–57 Therefore, CA has become the preferred option for restoring sinus rhythm in a durable fashion and minimizing AF burden in the present era.1, 25
Several recent studies have established the primacy of CA for rhythm control in AF patients with HFrEF (Table 1). Randomized controlled studies such as ARC-HF, CAMTAF, and CAMERA-MRI evaluated the CA of AF versus rate control and found improvements in peak VO2 and LVEF change with CA, respectively.52, 53, 56 The PABA-CHF study randomized 81 patients and compared ablation versus AVJ ablation and cardiac resynchronization (CRT) therapy and demonstrated the superiority of CA over AVJ ablation using a composite endpoint of change in LVEF, 6-minute walk distance, and HF symptom questionnaire. 54 The AATAC trial, on the other hand, compared CA with rhythm control using amiodarone and demonstrated the superiority of ablation based on LVEF improvement and greater rhythm control efficacy. 50
Randomized Controlled Trials of Catheter Ablation Versus Medical Therapy in Heart Failure Patients with Atrial Fibrillation.
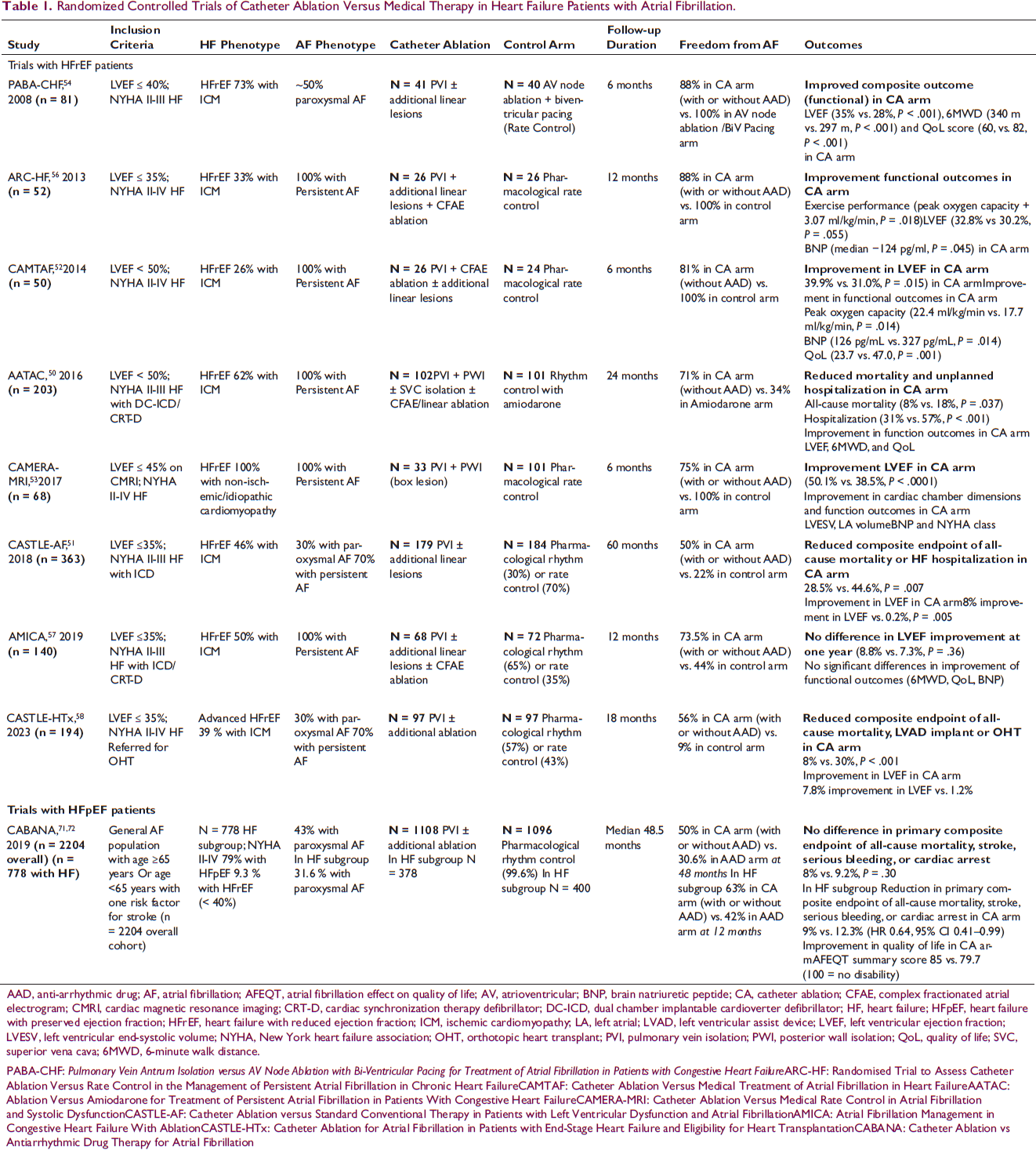
AAD, anti-arrhythmic drug; AF, atrial fibrillation; AFEQT, atrial fibrillation effect on quality of life; AV, atrioventricular; BNP, brain natriuretic peptide; CA, catheter ablation; CFAE, complex fractionated atrial electrogram; CMRI, cardiac magnetic resonance imaging; CRT-D, cardiac synchronization therapy defibrillator; DC-ICD, dual chamber implantable cardioverter defibrillator; HF, heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; ICM, ischemic cardiomyopathy; LA, left atrial; LVAD, left ventricular assist device; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; NYHA, New York heart failure association; OHT, orthotopic heart transplant; PVI, pulmonary vein isolation; PWI, posterior wall isolation; QoL, quality of life; SVC, superior vena cava; 6MWD, 6-minute walk distance.
PABA-CHF: Pulmonary Vein Antrum Isolation versus AV Node Ablation with Bi-Ventricular Pacing for Treatment of Atrial Fibrillation in Patients with Congestive Heart Failure ARC-HF: Randomised Trial to Assess Catheter Ablation Versus Rate Control in the Management of Persistent Atrial Fibrillation in Chronic Heart FailureCAMTAF: Catheter Ablation Versus Medical Treatment of Atrial Fibrillation in Heart FailureAATAC: Ablation Versus Amiodarone for Treatment of Persistent Atrial Fibrillation in Patients With Congestive Heart FailureCAMERA-MRI: Catheter Ablation Versus Medical Rate Control in Atrial Fibrillation and Systolic DysfunctionCASTLE-AF: Catheter Ablation versus Standard Conventional Therapy in Patients with Left Ventricular Dysfunction and Atrial FibrillationAMICA: Atrial Fibrillation Management in Congestive Heart Failure With AblationCASTLE-HTx: Catheter Ablation for Atrial Fibrillation in Patients with End-Stage Heart Failure and Eligibility for Heart TransplantationCABANA: Catheter Ablation vs Antiarrhythmic Drug Therapy for Atrial Fibrillation
The study that provided the most robust evidence for CA was undoubtedly the CASTLE-AF study, which compared CA versus medical therapy (either rate or rhythm control). 51 Three hundred and ninety-eight patients with NYHA Class II-III HFrEF (EF < 35% with implantable cardioverter defibrillator) on guideline-directed HF therapy were included and randomized in a 1:1 fashion. This was the first study powered to assess mortality as an outcome (13.4% vs. 25%, P = .01 with an ARR of 11.6% for CA versus medical therapy). There was a significant reduction in the composite outcome (death or HF hospitalization) in the ablation arm (28.5% vs. 44.6%, P = .006) over a three-year period. While analyzing this study, it is important to note that nearly 10% of patients in the medical therapy group crossed over to the ablation arm, and 15% crossed over from the ablation arm to the medical therapy arm. Therefore, the difference between the two groups may be higher than the result demonstrated. It should also be noted that 25% of the patients in the CA group required repeat ablations. 51
As a follow-up to the CASTLE-AF study, investigators evaluated the efficacy of CA compared with medical therapy in advanced HF patients referred for left ventricular assist device (LVAD) or heart transplantation evaluation. 58 A total of 194 patients were randomized in 1:1 fashion in the CASTLE-HTx trial; the overall profile of patients included NYHA Class II, III, and IV functional classes with mean LVEF <30%, and long duration of AF (>3 years, and >60% were persistent AF). The mean left atrial diameter was <50 mm. In the group assigned to CA, 84% underwent ablation after randomization, and 9% underwent multiple catheter procedures. After 18 months of follow-up, in the intention to treat analysis, the primary endpoint (death, LVAD implant, or urgent heart transplant) occurred in 8% of the ablation group versus 30% in the medical therapy group. The secondary endpoint of mortality was also clinically and statistically significant (6% vs. 20%). This trial was terminated after one year due to efficacy, and indeed, the findings highlight the greater benefit of CA in the higher-risk population, to the extent that it may even delay the need for advanced therapies such as heart transplantation in this selected referral population. 59 The caveat, however, is that such ablation should only be performed at centers of excellence with high-volume, skilled operators.
One aspect of CA that is not well addressed by the randomized trials is the ablation strategy that is most beneficial for HF patients. In general, there is consensus that paroxysmal AF patients with a lower burden of AF often only need pulmonary vein isolation (PVI). There are centers that advocate for high-dose isoproterenol infusion following PVI to identify non-PV triggers for additional ablation in HF patients. 60 However, this practice is not universally implemented. In selected patients with a clinical occurrence of typical atrial flutter, cavotricuspid isthmus (CTI) ablation should be performed following PVI. Prior studies have not demonstrated the benefit of routine CTI ablation in patients without demonstrating typical atrial flutter. 61 On the other hand, there is considerable ambivalence about the approach for persistent and long-standing persistent AF. One school of thought is to just perform PVI ablation, and this is largely informed by the STAR-AF II trial, where PVI alone outperformed other ablation strategies (additional complex fractionated atrial electrogram ablation or additional linear ablation). 62 Meta-analytic comparison of PVI alone with additional focal impulse and rotor modulation (FIRM) mapping guided ablation also demonstrated better outcomes with PVI alone. 63 As a result, empiric ablation beyond the isolation of pulmonary veins has mostly been relegated by electrophysiologists. 64 Instead, a more nuanced approach is recommended. As discussed previously, the phenotype of AF (paroxysmal vs. persistent) does not necessarily correlate with the atrial arrhythmogenic substrate. Cardiac MRI data and baseline electroanatomical voltage maps can be useful in identifying extensive atrial substrates and tailoring ablation strategies. Since the posterior left atrium is key for arrhythmogenesis, often additional ablation is targeted at achieving electrical isolation or quiescence in this area. 65 It remains unclear if this needs to be performed with the first ablation or is reserved for redo procedures. 66 This lack of clarity in the initial ablation strategy is reflected in the clinical trial designs of the CASTLE-AF and CASTLE-HTx, where additional ablation was not mandated in the study protocol and left to the discretion of the individual operators. Indeed, additional ablation beyond PVI was performed in 51.7% of CASTLE-AF but only in 9% of CASTLE-HTx studies.51, 58
Another addition to the ablation armamentarium to be considered in AF and HF patients is ethanol ablation of the Vein of Marshall (VOM) to prevent VOM-related atrial tachycardias and mitral isthmus-dependent flutters. Both the VENUS trial and the Marshall-PLAN study have demonstrated better outcomes with VOM + PVI as compared to PVI alone.67, 68 It should be noted that both of these studies adopted a strictly anatomical approach to ablation, included all comers with AF, and did not restrict VOM ablation to mitral isthmus-dependent flutters or VOM ridge-triggered AF. Neither of these specifically analyzed outcomes in HF patients.
Our approach is to perform PVI alone for paroxysmal and early persistent AF patients. In long-standing persistent AF, if there is evidence of extensive left atrial substrate (as evidenced by bipolar voltage areas on the electroanatomical map < 0.10 mV), then further ablation on the posterior wall is performed to address the arrhythmogenic substrate. Various case studies are presented from the authors’ clinical practice, where different lesion sets were utilized for CA based on clinical and electroanatomical data.
Case 1: PVI alone (Cryoballoon Ablation)
An 83-year-old male with a history of long-standing premature ventricular contractions presented with AF with a rapid rate (unknown duration) and acute decompensated heart failure. Echocardiography showed LVEF of 30% with regional wall motion abnormalities. Coronary angiography revealed non-obstructive stenoses. Transesophageal echocardiography and direct cardioversion were performed to restore sinus rhythm, and he was started on dofetilide for maintenance of sinus rhythm. He subsequently underwent cryoballoon ablation for early persistent AF with possible AMC/AIC, and he was taken off dofetilide following ablation (Figure 3). His LVEF improved to 55% three months after the ablation.
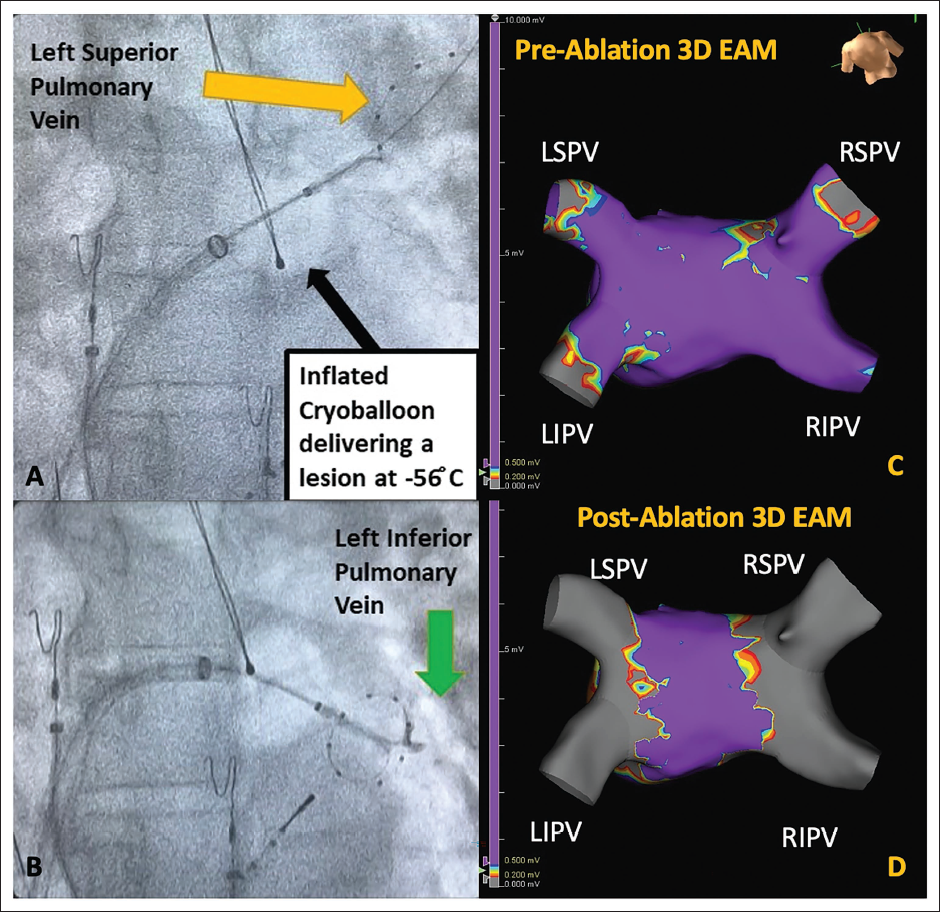
3D EAM, three-dimensional electroanatomical map; LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein.
Case 2: PVI alone (Radiofrequency Ablation)
A patient with stable coronary artery disease and HFpEF had a high burden of paroxysmal AF with resultant HF decompensations. Both cryoballoon and radiofrequency ablation options were discussed. The patient underwent radiofrequency pulmonary vein isolation using an antral radiofrequency lesion set along with carina lesions to achieve isolation (Figure 4).
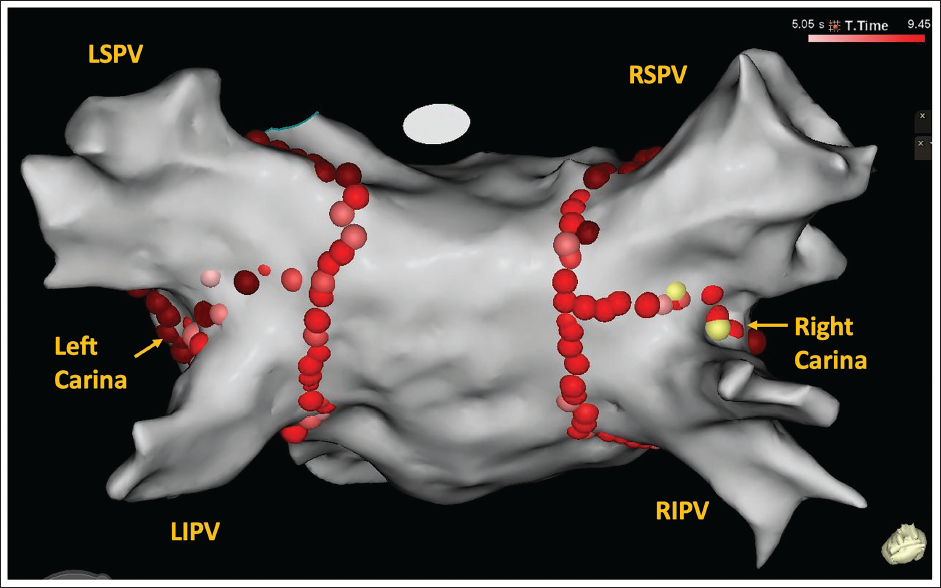
LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein.
Case 3: PVI Plus (Radiofrequency Ablation)
A patient with persistent AF and HFrEF requiring amiodarone presented for rhythm control. He had HFrEF with EF 30% and ICD for primary prevention and alcoholic hepatic cirrhosis. During evaluation for a liver transplant, he was taken off amiodarone, had repeated recurrences, and therefore underwent AF ablation. Left atrial electroanatomical voltage mapping showed a considerable scar in the posterior left atrium. After completion of PVI, roof and floor lines were performed to create a box lesion (Figure 5). This effectively renders the posterior wall of the left atrium electrically silent. This patient remained in sinus rhythm without anti-arrhythmic drugs.
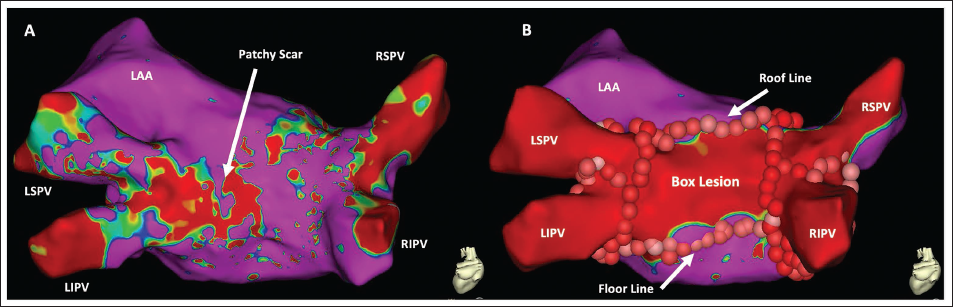
LAA, left atrial appendage; LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein.
Case 4: PVI Plus (Radiofrequency Ablation)
A patient with prior infective endocarditis of the mitral valve and bioprosthetic mitral valve 30 years back underwent a redo of MVR six years prior and developed long-standing persistent AF with AF-mediated cardiomyopathy. She had a prior history of stroke and presented for AF ablation. Following TEE to exclude the left atrial thrombus, she underwent completion of pulmonary vein isolation and posterior box lesion, following which AF converted to atrial flutter. A high-density map of the left atrium showed a counterclockwise mitral isthmus-dependent flutter. The endocardial lateral mitral isthmus line did not terminate the lesion. A Vein of Marshall (VOM) ablation was considered, but a venogram did not show a VOM. Finally, epicardial mitral isthmus ablation was performed in the coronary sinus, overlaying the endocardial lesions, and the flutter terminated with sinus rhythm during ablation (Figure 6). She subsequently maintained sinus rhythm.
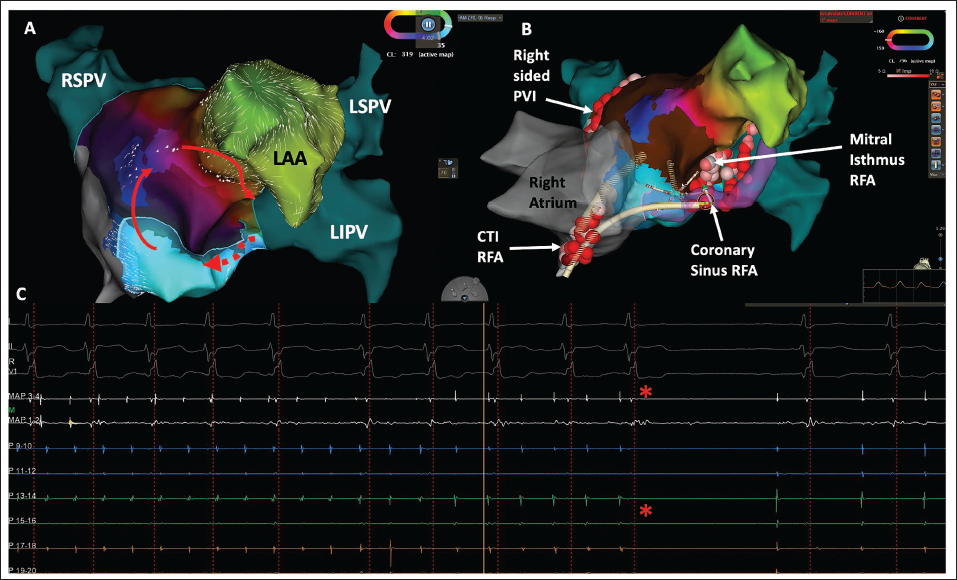
CTI, cavotricuspid isthmus; LAA, left atrial appendage; LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; PVI, pulmonary vein isolation; RIPV, right inferior pulmonary vein; RFA, radiofrequency ablation; RSPV, right superior pulmonary vein.
Case 5: PVI Plus + VOM Alcohol Ablation
A patient with HFrEF and long-standing persistent AF with a duration greater than 10 years presented for AF ablation for durable rhythm control. Initial electroanatomical mapping showed a relatively healthy posterior left atrium (voltage > 0.5 mV shown in pink color), despite long-standing AF. A prominent VOM was identified using a CS venogram, and 8 cc of alcohol was injected, followed by a limited repeat EAM. This showed the development of a low-voltage scar along the mitral isthmus, left-sided carina, and extending into the left superior vein. Subsequently, PVI was performed, and this required limited ablation along the left-sided veins without the need for carina ablation (Figure 7).
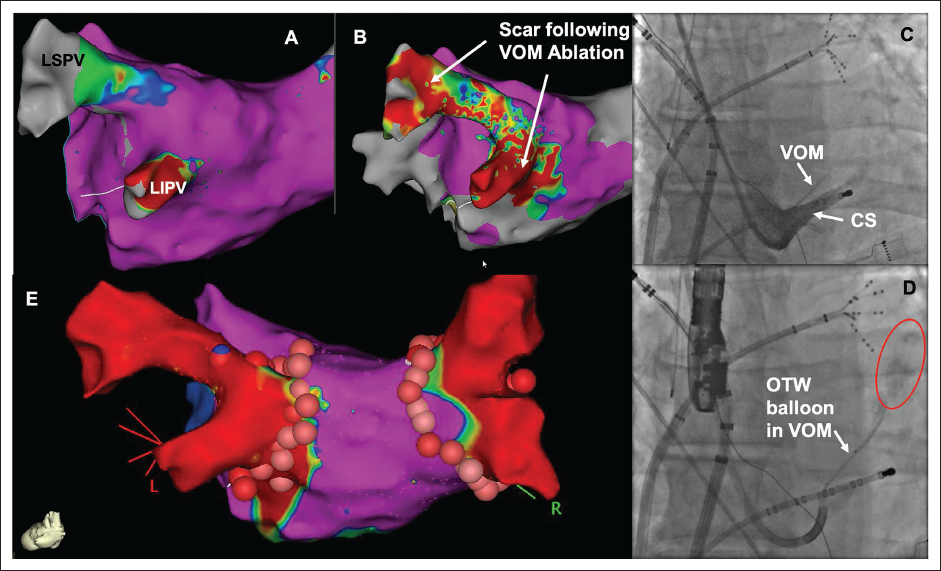
CS, coronary sinus; LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; OTW, over the wire; VOM, Vein of Marshall.
Catheter Ablation in HFpEF
There are no randomized controlled trials specifically evaluating AF ablation for HFpEF patients, and the body of evidence is from observational studies and registry data. While the mortality benefit of AF ablation in HFrEF patients has not been replicated in HFpEF patients, ablation allows for greater maintenance of sinus rhythm and improves functional outcomes as compared with pharmacological approaches, with an acceptable safety profile.69, 70 An exploratory, post hoc analysis of the CABANA trial (which included 2,204 AF patients) identified 778 patients with HF, and of these, 79% were HFpEF.71, 72 Sub-group analysis in these HFpEF patients showed that AF burden was lower, quality of life was better, and it was associated with lower all-cause mortality. Clearly, dedicated randomized controlled studies are needed for this group, but current evidence supports considering earlier ablation for patients with HFpEF as well. 70
Options for Patients Failing Medical Therapy and Not Candidates for Catheter Ablation
While there is increased enthusiasm for CA of AF based on recent trial data, it is not always feasible to achieve rhythm control in this fashion, particularly in patients with severely dilated left atrium (>60 mm), long-standing persistent AF, persistent left atrial appendage thrombus, and those with several left atrial fibrosis. In such patients who develop HF, the alternative “hybrid rate/rhythm control” option is to perform AV junction ablation with simultaneous implantation of a cardiac resynchronization therapy (CRT) device or conduction system pacing (CSP) device, regardless of the underlying QRS duration. The APAF-CRT trial evaluated mortality benefit in HFpEF and HFrEF patients with AF > six months and at least one HF hospitalization. 73 In the 63 patients randomized to AVJ ablation + CRT (43% with EF < 35%), the all-cause mortality was 11% at 29 months, as compared to 29% in the medical therapy group (n = 70). Of note, in this study, an atrial lead was not implanted in the APAF-CRT trials, and the AVJ + CRT was utilized as a superior form of rate control where the benefit was secondary to strict ventricular rate control and regularization of QRS complexes.73, 74
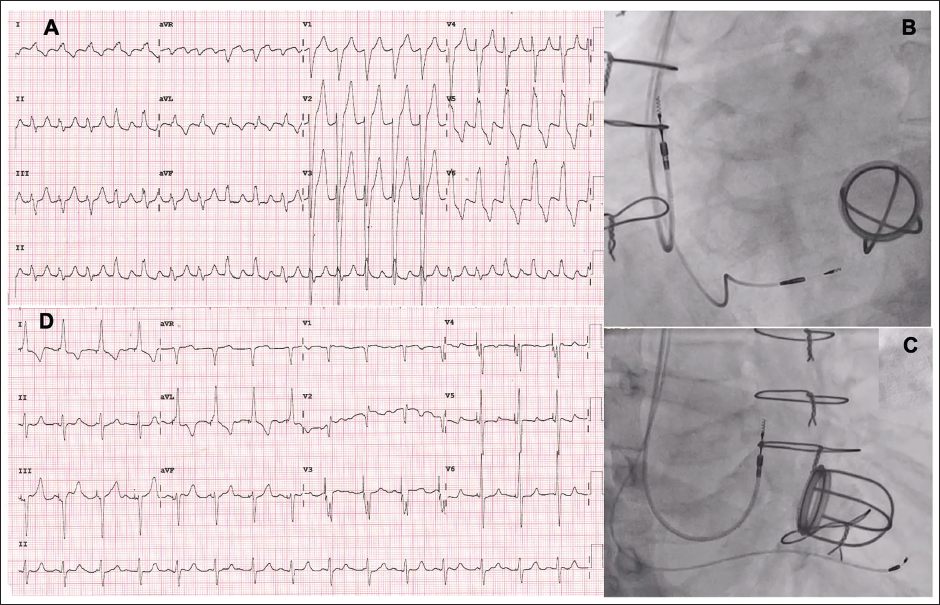
In our practice, we additionally implant an atrial lead in most patients as believe that ventricular rate control, regularization of QRS complexes, and narrow QRS complexes achieved via CRT or CSP will allow for ventricular remodeling and improvement in filling pressures, which in turn may alter left atrial remodeling. This then facilitates the maintenance of sinus rhythm, with cardioversion and AAD administration. The benefit of this “hybrid” strategy is to additionally achieve AV synchrony with atrial sense-biventricular paced rhythm, which may further benefit patients and help them recover from HF. Future research in this area will be needed to determine if this experience can be extrapolated to other centers. One such case is described below.
(Panel A) An Electrocardiogram with Atrial Flutter and Rapid Ventricular Rate with LBBB. (Panels B and C) Left Anterior Oblique and Right Anterior Oblique Projections of the Right Atrial and Left Bundle Branch Pacing Leads. (Panel D) Electrocardiogram Following AVJ Ablation and Cardioversion, with Sinus Rhythm and Left Bundle Branch Paced Rhythm.
Multidisciplinary Team Management of AF and HF
Managing this cohort of patients with advanced heart disease, associated with hemodynamic and electrophysiological compromise, can be particularly challenging. As with all complex pathophysiological syndromes, a multidisciplinary team approach can be very useful in appropriately profiling patients, addressing risk factors, and enforcing remedial therapies. While treating cardiologists can stabilize patients with immediate rate control and/or urgent cardioversion and initiate decongestive and anticoagulation therapies, a collaborative approach involving electrophysiologists and advanced heart failure specialists is important to delineate the best path forward. Since one treatment plan cannot be used as a template to treat every HF/AF patient, an individualized clinical approach is vital. Early rhythm control, preferably with CA by experienced electrophysiologists, may eradicate the need for ICDs and advanced HF therapies in reversible tachycardiomyopathies. On the other hand, prompt diagnosis of inflammatory cardiomyopathies with appropriate cardiac imaging may obviate the need for ablation simply by treating the underlying inflammation and controlling HF. Yet another scenario is the recognition of long-standing, severe, and irreversible cardiomyopathy, where it may be more helpful to focus resources on advanced therapies such as left ventricular assist devices or heart transplantation rather than ICD/CRT implantation or extensive catheter ablative efforts. In this scenario, rhythm control at best may help with symptom control until definitive cardiac therapies can be delivered. Therefore, it is apparent that a “tumor-board style” interdisciplinary discussion would help deliver patient-oriented treatments. Other vital elements of such an interdisciplinary team should include dieticians, pharmacists, physiotherapists, device clinic nurses, and sleep therapists.
In the Indian scenario, where it has been documented that AF is often undertreated in the general population and there is a penchant to utilize pharmacological and device-based therapies, rather than catheter-based therapies for the management of AF, multidisciplinary teams will allow for evidence-based and patient-specific strategic decision-making for this special population of HF patients with AF. 75
Conclusion
AF and HF are increasing in prevalence and may be the cause or consequence of each other. There is increasing emphasis on establishing rhythm control in AF and HF, utilizing pharmacological and/or non-pharmacological techniques. Whichever modality is used for rhythm control, cardiologists, electrophysiologists, and HF teams caring for these patients must choose early rhythm control after diagnosis, as this has been linked to better outcomes. Of the rhythm control options, the evidence base is strongest for rhythm control using CA, particularly in the HFrEF population, as recent randomized studies have established a mortality benefit in this cohort. In HFpEF patients, CA is still helpful in reducing HF hospitalizations and improving quality of life. As a result, European and American guidelines have upgraded CA for AF in HF to a Class I recommendation. While this represents a paradigm shift in the management of such patients, where traditionally pacing and AV junction ablation were most frequently offered to patients, the uptake of CA for AF in HF in the Indian setting is expected to be gradual. As awareness increases, it is vital for centers of excellence to offer advanced AF ablation and for operators to hone this skillset to deliver safe and efficacious care for AF in HF patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
