Abstract
The diagnosis and therapy of heart failure with preserved ejection fraction (HFpEF) are complicated by the disease’s varied etiology. In the field of heart failure (HF), N-terminal pro-B-type natriuretic peptide (NT-proBNP) has become an important biomarker, providing information on diagnosis, prognosis, and treatment monitoring. This in-depth investigation looks at the connection involving NT-proBNP levels and HFpEF, offering information on possible diagnostic applications as well as consequences for patient care. The study reviews the literature on the connection concerning NT-proBNP and HFpEF and emphasizes how NT-proBNP aids in the distinction of HFpEF from various cardiac and non-cardiac conditions. HF is known to be associated with elevated NT-proBNP levels, but the complex link with HFpEF necessitates careful examination. The abstract explores the difficulties in using NT-proBNP as a diagnostic tool for HFpEF, taking into account the potential influence of renal function, age, and comorbidities on NT-proBNP levels. Additionally, the predictive importance of NT-proBNP in HFpEF is explored, emphasizing its capacity to forecast death and severe cardiovascular events. The abstract emphasizes that diagnosing HFpEF requires a multimodal approach that includes clinical assessment, imaging modalities, and NT-proBNP measurement. In order to accurately manage patients with HF, particularly HFpEF, the abstract highlights the significance of integrating clinical judgment and other diagnostic measures, even though NT-proBNP is still a useful marker in this regard. Lastly, the study offers suggestions for future research trajectories that should further our knowledge of NT-proBNP’s function in HFpEF and improve its clinical applicability in this intricate and varied illness.
Introduction
Heart failure with preserved ejection fraction (HFpEF) is a clinical condition in which individuals have normal or nearly normal left ventricular ejection fraction (LVEF; ≥50%) but exhibit signs and symptoms of heart failure (HF) due to excessive left ventricular (LV) filling pressure. 1 In addition to having normal LV volumes, majority of HFpEF patients also exhibit aberrant diastolic filling patterns, or diastolic dysfunction. 2 A 2017 study of the global population estimated to be affected by HF was 34% higher than in 1990 and 16% higher than in 2007. HF is a serious health issue. 3 Between 2012 and 2030, there is a projected 46% increase in the prevalence of HF. 4 In the Indian Cardiology Society of India-Kerala Acute Heart Failure patients with HFrEF had greater 90-day hospital death rates (7.7% and 12.3%), in comparison to Heart Failure with mid-range ejection fraction patients (5.2% and 9.9%) and HFpEF patients (5.6% and 10.6%), respectively. 5 As per the National Heart Failure Registry from India, the rates of 90-day death for HFrEF, HFmrEF, and HFpEF were 15.7%, 11.0%, and 12.5%, respectively, and the rates of in-hospital mortality were 7.5%, 5.1%, and 5.5%. 6 The majority of patients with HFpEF at this time are elderly women who have multiple coexisting conditions, such as renal disease (51%–58%), obesity (32%–46%), diabetes mellitus (20%–45%), atrial fibrillation (AF) (15%–65%), ischemic heart disease (35%–70%), and obstructive lung disease (24%–30%). Furthermore, women have concentric LV myocardial remodeling and HFpEF as a result of increased systemic vascular and LV stiffness compared to men.7, 8 In patients with HFpEF, the co-existence of diabetes mellitus, chronic renal illness, and hypertension increases systemic arterial and ventricular stiffness. The pressure wave expelled from the LV is reflected back to the heart as arterial stiffness increases. This results in an increase in LV afterload, a decrease in diastolic ventricular function, and an increase in the heart’s hydraulic effort and oxygen requirements for the myocardium. Reduced coronary flow reserve, elevated ventricular diastolic filling pressures, reduced ventricular diastolic function, and pulmonary vascular congestion are the results of these hemodynamic consequences. Breathlessness is a consequence for the patients. 8 HF prognosis assessment, diagnosis, and prediction have all made extensive use of natriuretic peptides (NP).9, 10 A higher likelihood of subsequent death or morbidity has been linked to an increase in N-terminal brain natriuretic pro-peptide (NT-proBNP) concentration over time. However, real-world data regarding the use and dissemination of NT-proBNP concentrations in patients with HF, particularly in those following a deteriorating heart failure event (WHFE), is scarce. 11 The American guidelines on HF for 2022 state that, in cases when a patient has acute symptoms, NT-proBNP may be assessed before discharge in order to better monitor the patient’s course and develop a post-discharge strategy. It could also be carried out as a part of the workup for differential diagnosis before or during a real hospital stay. 12
Biomarkers in HF
The National Institutes of Health Biomarkers Definitions Working Group defines a biomarker as “a characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention.” 12 Treatment guidance has been greatly aided by the diagnosis of HF using many diagnostic techniques, including cardiac magnetic resonance (CMR), echocardiography (ECG), and chest X-ray.13–16 The European Society of Cardiology (ESC) and the American Heart Association (AHA) have added NP, such as brain-type natriuretic peptide (BNP) and N-terminal prohormone of BNP, and cardiac troponin readings in their guidelines for HF diagnosis and treatment.16, 17 In order to better guide HF treatment, additional diagnostic biomarkers like oxidative stress, inflammation, and cardiac remodeling, such as galectin-3, soluble ST2 receptor, and growth differentiation factor-15, may be added. 17 Various biomarkers with their physiological role, indicative conditions, and their role in heart failure management is summarized in Table 1.
Natriuretic peptide
NT-proBNP, mid-regional atrial natriuretic pro-peptide (MR-proANP), and BNP are among the molecules collectively referred to as NPs, or moving cardiovascular biomarkers of myocardial stretch and biomechanical stress. 9 Research findings indicate that patients with HF and elevated NP levels, as opposed to those with low levels, experienced a greater risk of death or HF rehospitalization. 18
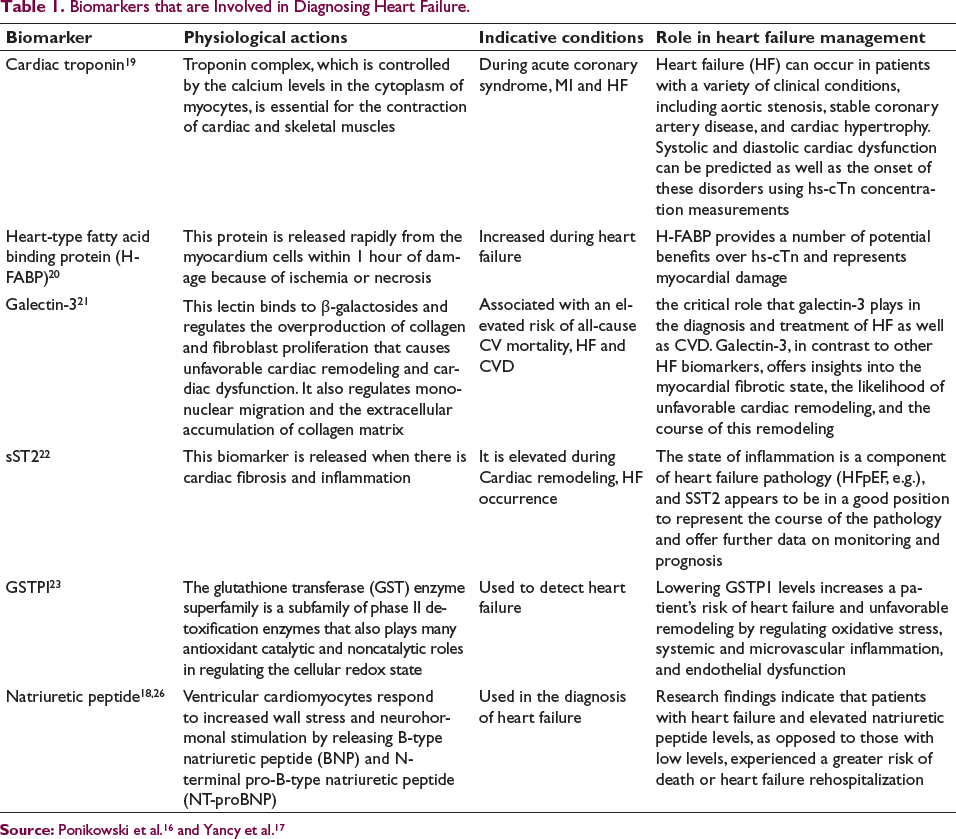
Biomarkers that are Involved in Diagnosing Heart Failure.
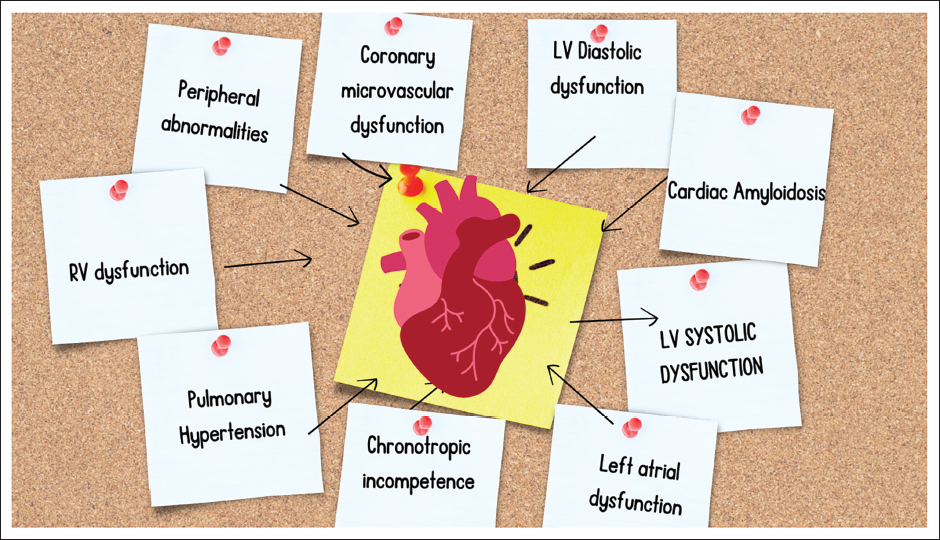
Pathophysiology of HFpEF
HFpEF is a clinical condition in which individuals have normal or nearly normal LV ejection fraction (LVEF; ≥50%) but exhibit signs and symptoms of HF due to excessive LV filling pressure. 1 Most HFpEF patients also exhibit normal LV sizes and aberrant diastolic filling patterns, or diastolic dysfunction. 24 Different cardiac, vascular, and non-cardiac problems contribute to the pathogenesis of HFpEF, even though diastolic dysfunction is the main and essential factor. impairments in the diastolic and systolic function of the LV, the structure and function of the LA (i.e., LA myopathy), abnormalities in gas exchange and pulmonary hypertension, dysfunction of the right heart, autonomic deregulation, vascular stiffening, myocardial ischemia, endothelial dysfunction, kidney disease, and peripheral abnormalities in skeletal muscle in fat. 25 Pathophysiology of HFpEF is summarized in Figure 1.
Diagnosis of HFpEF
In the context of diastolic dysfunction and evidence of preserved LVEF, the diagnosis of HFpEF necessitates clinical symptoms and/or HF indicators. 26 The most frequent causes are CAD, valvular disease, and hypertension. Finding other underlying problems could result in treatment that improves results.27, 28 The identification of potential HF and subsequent referral for two-dimensional transthoracic echocardiography (TTE) to confirm the diagnosis constitute the initial stage in the diagnosis of HFpEF. 29
Diagnostic Criteria
There are a number of accepted standards for diagnosing HF based on clinical symptoms.
1. Framingham criteria 30 :
Diagnosis of HF requires the simultaneous presence of at least two major criteria or one major criterion in conjunction with two minor criteria.
Minor criteria are acceptable only if they cannot be attributed to another medical condition (such as pulmonary hypertension, chronic lung disease, cirrhosis, ascites, or nephrotic syndrome).
2. Diagnostic algorithm based on guidelines framed by Paulus et al. 31
This recommendation requires patients to have three things: no impaired LV systolic functioning (LV ejection fraction >50% and indexed LV end-diastolic volume <97 mL/m2), evidence of LV diastolic dysfunction, and signs and/or symptoms of HF. The recommendation from Paulus et al. for evidence of diastolic dysfunction is that either intrusive hemodynamic measurements (pulmonary capillary wedge pressure, PCWP, >15 mmHg, or LV end-diastolic pressure, >12 mmHg at rest) or tissue Doppler measurements (E/e’ >15) offer sufficient stand-alone evidence of diastolic dysfunction Any E/e’ value between 8 and 15 or elevated levels of NT-proBNP (>220 pg/mL; or BNP > 200 pg/mL) need to be accompanied by at least one other indicator of diastolic dysfunction, such as a low E/A ratio and a high deceleration time, pulmonary venous flow patterns suggestive of diastolic dysfunction, high indexed left atrial volume, AF, and/or LV hypertrophy.
3. Integrated diagnostic approaches
Utilizing the clinical composite score H2FPEF can also help with risk assessment and direct the diagnostic process. 32 This score creates a weighted summation of patient risk factors, including age over 60, AF, and obesity. Patients with a score of more than six have a high probability of having HFpEF, while those with a score of less than two have a low probability. For patients in the middle of the score, more investigation is necessary. This process makes it possible to diagnose HFpEF using a Bayesian method, in which patients with a low or high risk of disease are diagnosed, while those having an intermediate pre-test probability of disease remain eligible for more costly and definitive testing. Crucially, the H2FPEF score may be computed with readily available echocardiographic data and basic clinical features from everyday clinical practice. 32
The ESC/HFA suggested the HFA-PEFF score in 2019 as an additional scoring system to aid in directing the diagnostic assessment of HFpEF, where the domains include functional, morphological, biomarker in sinus rhythm and AF in which major criteria scores 2 points and minor criteria scores 1 point. 33
Echocardiography
Among the diagnostic tools most useful in this situation is ECG, which is essential for HFpEF diagnosis. It allows precise filling pressure calculation with excellent spatial and temporal precision, is conveniently accessible, and provides information on the general anatomy of the heart. It is not surprising that the vast majority of diastolic dysfunction indicators are the subject of ECG in HFpEF, since diastolic dysfunction is the fundamental pathophysiological mechanism in this condition. Considering that the echocardiographic variables related to diastolic dysfunction are influenced by heart rate and loading conditions, the main question revolves around whether ECG, and Doppler measurements in particular, are useful for evaluating diastolic dysfunction. 34 Together with E/e, other echocardiographic markers of diastolic dysfunction include E/A ratio, E’s deceleration time, assessment of pulmonary venous flow, left atrial volumes, and mass of the LV. 35 They are only useful as a diagnostic tool for patients with equivocal e/e’ in cases where HFpEF is suspected (between 8 and 15). 36 An increased E/e’ >15 as indicators of diastolic dysfunction appear adequate to identify increased LV pressures during the preliminary evaluation of individuals who may have HFpEF; however, filling pressures fluctuate over time based on volume status. Penicka et al.’s study, however, revealed that filling pressures might not increase in the early phases of the illness. 37 Researchers found that 30% of stable outpatients with unjustified dyspnea and invasively diagnosed HFpEF had normal E/e’ indices. Furthermore, only 25% of HFpEF patients satisfied the prerequisites for the current diagnosis, while 20%–40% of controls had borderline E/e’ values, suggesting that even when paired with additional echo indications of diastolic dysfunction, E/e’ had low specificity and sensitivity. 37 Therefore, diastolic dysfunction cannot be ruled out even in the absence of an increased E/e’.
Differential Diagnosis
Patients with HFpEF usually exhibit symptoms of exhaustion, exercise intolerance, dyspnea, or evidence of peripheral or pulmonary congestion in addition to acute HF. The symptoms and indicators of HFpEF and HFrEF can sometimes be confused. Patients with diabetes mellitus, systolic hypertension, obesity, age, and African American ancestry are more likely to present with HFpEF than with ischemia. Another more typical indication of HFpEF is AF. Individuals with diastolic function disorders rely on atrial contraction for filling, and pulmonary edema is produced when AF develops as a result of LA dilatation, which lowers ventricular filling and cardiac output. 38 A patient’s presentation determines the differential diagnosis for HFpEF. A high-output condition, extracardiac fluid overload, and cardiac or respiratory etiologies are among the differential diagnoses made for patients who arrive in the emergency room with acute HF. Obesity and physical deconditioning can be included in the differential in individuals who report as outpatients with dyspnea or exercise intolerance. 39 The differential diagnosis of HFpEF includes the following cardiac conditions: intracardiac shunt, severe hypertension, atrial myxoma, pericardial disease, restrictive or hypertrophic cardiomyopathy, valvular disease, and acute coronary syndrome complicated with pulmonary edema.
The overlap between pulmonary hypertension, chronic obstructive pulmonary disease (COPD), and HFpEF may make it challenging to differentiate respiratory disorders from HFpEF. 40 Conditions that cause extracardiac volume overload, such as nephrotic syndrome, anemia, hypothyroidism, arteriovenous fistula, or extracardiac shunting, are among the other diagnosis in the differential. 38 The diagnosis of heart failure with heart failure (HFpEF) is based on the presence of symptoms of HF along with hemodynamic, Doppler echocardiographic, or biomarker evidence of aberrant LV relaxation, filling, distensibility, or stiffness, according to the criteria set forth by the ESC. This disease’s clinical spectrum starts with DD and moves on to HFpEF.
NT-pro BNP in Diagnosing HFpEF
NT-pro BNP is not broken down by the enzyme neprilysin, which breaks down BNP, BNP stays in the bloodstream for a longer period of time than BNP. In reaction to prolonged LV stretch or elevated wall strain, these peptides are released. Patients with HFpEF had lower BNP levels than patients with HFrEF, but for a given BNP level, patients with HFpEF have an at least equal risk of all-cause death and hospitalization for HF. It has been demonstrated that high filling pressures and the degree of diastolic dysfunction are connected with elevated BNP or NT-proBNP levels, which are a reliable indicator of the prognosis. 41 Van Veldhuisen et al. examined the use of BNP in patients with HF both at baseline and after 18 months. They found a relationship between the BNP levels and either mortality alone or a composite of all-cause mortality and HF hospitalization. 42 Additionally, BNP can be utilized to distinguish HFpEF from concomitant illnesses such obesity, anemia, and COPD that might exhibit symptoms similar to those of HF. The BNP may be normal or slightly higher under various circumstances. 42 Treatment response and outcome can be predicted by BNP and NT-proBNP. HFPEF is suggested by a BNP of >200 pg/mL or an NT-proBNP of >300 pg/mL.
Approach to Diagnosis of HFpEF
Patients who exhibit symptoms that could indicate HF should be evaluated clinically. A TTE should be recommended for the patient if physical examination findings indicate HF (jugular venous distention, S3 heart sound, or displaced apical impulse) or meet clinical criteria for the MICE or Framingham rules. Patients who do not meet these requirements should have measurements of 3,5 BNP or NT-pro-BNP, and they should be referred for TTE if either the NT-pro-BNP level is 125 pg per mL or higher, the BNP level is 35 pg per mL or higher, or the Netherlands rule score is 13 or higher. The TTE evaluation of diastolic function and LVEF can validate HFpEF. 26
NT-proBNP as a Significant Biomarker
Ventricular cardiomyocytes respond to increased wall stress and neurohormonal stimulation by releasing BN) and NT-proBNP. 43 NPs, also known as circulating cardiac biomarkers of myocardial stretch and biomechanical stress, are a group of molecules that generally include several types, including NT-proBNP, MR-proANP, and BNP, in addition to other subsets of NPs like C-type NP. 9 International HF guidelines advocate measuring NT-proBNP for prognostic stratification, illness severity assessment, diagnosis, and categorization.9, 10 In HF trials, NT-proBNP serves as an eligibility criterion and provides enrichment for cardiovascular versus non-cardiovascular outcomes. 44 Nonetheless, there is a general lack of information regarding the use of NT-proBNP testing in clinical settings. There is no empirical data about the use and distribution of NT-proBNP levels in patients with HF, particularly in those who have just undergone a worsening heart failure event (WHFE) despite the fact that increases in NT-proBNP concentration over time have been linked to an increased risk of eventual mortality/morbidity.11, 43 Age-dependent cut-offs to rule in HF are favored over NT-proBNP concentrations because they are more closely associated with age, sex, and renal clearance than BNP and MR-pro-ANP concentrations. In patients with both acute and chronic HF, MR-proANP was significantly correlated with newly diagnosed acute HF but not with gender or age.45–47 In older adults with several HF risk factors (diabetes mellitus, chronic renal illness, vascular disease, AF, and hypertension), NT-proBNP was found to have a greater diagnostic accuracy for asymptomatic cardiac dysfunction than MR-proANP.48, 49 NPs have been thoroughly evaluated as potential substitute markers of the risk of developing and compensating for HF as well as a predictor of the effectiveness of therapy for HF based on guidelines. 50 All-cause mortality has been found to be adequately predicted by peak concentrations and even a trend of NPs in ambulatory follow-up. 51 According to the diastolic dysfunction and diastolic chronic heart failure (DIAST-CHF) trial, among stable outpatients with cardiovascular risk factors, NT-proBNP was a more accurate predictor of incident AF than HF. 52 Comparing the clinical usage of NPs with other cardiac and non-cardiac biomarkers, it is noteworthy that there is a proven economic benefit. Therefore, it appears that the most economical course of action for Personalized point-of-care (POC) management of HF is based on NP interventions.53, 54 There is compelling evidence that the inclusion of NP in the best guide-based management of HF may aid cardiologists in the following areas: (a) identification of high-risk HF patients; (b) assessment of the patients’ status—determining whether they are stable or have a propensity to worsen; (c) optimization of HF management; (d) enhancement of prognosis assessment; (e) reduction of all-cause and HF-related mortality and re-admission to hospital; and (f) cost savings per year of life. 20
Physiological Factors Affecting NT-proBNP
NT-proBNP is a biomarker used to diagnose and monitor HF, among other cardiovascular conditions. Several physiological factors can affect NT-proBNP levels, influencing their interpretation in clinical practice. Here are key physiological factors and their effects:
Age:
- Effect: NT-proBNP levels generally increase with age. - Mechanism: Age-related changes in cardiac structure and function, such as reduced cardiac output and increased arterial stiffness, may elevate NT-proBNP levels.
55
Sex:
Renal Function:
Effect: Impaired renal function (chronic kidney disease) leads to higher NT-proBNP levels. Mechanism: Reduced renal clearance of NT-proBNP and increased cardiac stress due to fluid overload in renal impairment.
58
Body Mass Index (BMI):
Effect: Lower NT-proBNP levels are observed in individuals with higher BMI. Mechanism: Increased adipose tissue may enhance the clearance of NT-proBNP or alter its production, and obesity-related cardiac changes may influence its levels.
59
Physical Activity:
Effect: Physically active individuals may have lower NT-proBNP levels. Mechanism: Regular physical activity improves cardiovascular health and reduces cardiac stress, thereby lowering NT-proBNP levels.
60
Diabetes Effect: Diabetic patients might have altered NT-proBNP levels, often lower than expected. Mechanism: The exact mechanism is not fully understood, but it may relate to changes in renal function, cardiac metabolism, or autonomic regulation in diabetes.
61
Methods to Detect NT-proBNP
As cardiac indicators, BNP and NT-proBNP have been evaluated; these tests rely on non-competitive (sandwich) immunoassay using capture and detection antibodies. In the clinical and diagnostic domain, a range of analytical techniques are available that are tailored to NP. These techniques include radioimmunoassay (RIA), enzyme-linked immunosorbent assay (ELISA), immunofluorescence assay (IFA), microfluidics, the recently introduced electrochemiluminescence immunoassay (ECLIA), and others. Sandwich immunoassays are the foundation of modern techniques used to measure circulating NT-proBNP. 62
Immunofluoroscence Assay
The discovery of immunofluorescence assays allows for the 15–20 minutes detection of NT-proBNP. This quick and easy technique, which is frequently employed in clinical diagnosis, guarantees first treatment for HF. Sandwich immunoassay is the foundation of the IFA concept. In this instance, a monoclonal antibody is coupled with a fluorescent protein and immobilized on a nitrocellulose membrane. Following the addition of a sample (serum, plasma, or whole blood), fluorescently labeled antibodies bound to the analytes, form an antigen-antibody complex. By means of capillary action, this complex travels in the direction of the test line, where it is absorbed by the antibody coating to create a sandwich complex. Analyzer measurements of fluorescence allowed for the calculation of analyte concentration. The intensity of the fluorescence is directly correlated with the analyte concentration. 63
Enzyme-linked Immunosorbent Assay
A quantitative assessment of NT-proBNP is provided by ELISA. Antibodies are paired with an enzyme that is particular to a substrate’s reaction in ELISA. The most often employed enzymes are β-galactosidase, horse radish peroxidase (HRP), and alkaline phosphatase (ALP). When the substrate is applied to the NT-proBNP after these antibodies bind to it, a catalyzed reaction results in a color shift that aids in the quantification of NT-proBNP. The antigen-antibody interaction determines the assay’s quality and quantity. After 30-60 minutes, the enzyme-substrate process is finished, and sulfuric acid is added to halt it. The detection of colored response is done using a microtiter plate reader.64, 65
ELISA has a number of drawbacks, such as the need for a large sample volume, a lengthy detection period (about 4 hours), and high instrument costs. Additionally, in contrast to other assay types, ELISA lacks the sensitivity necessary to identify extremely low NT-proBNP levels. 66
Chemiluminescence Immunoassay
Because of its great stability, ultra-sensitivity, and specificity, the modern and adaptable chemiluminescence immunoassay has drawn a lot of interest and has been used for the quantitative measurement of protein analytes. The definition of chemiluminescence is the release of light as a result of a chemical reaction. Both direct and indirect techniques are possible with the use of luminophore and enzyme markers, respectively. Commonly utilized luminophores are acridinium and ruthenium esters; enzymatic markers employed in the indirect technique include horseradish peroxidase with luminol or its derivatives as substrate and ALP with adamantyl 1,2-dioxetane aryl phosphate (AMPPD) as substrate.
The interaction between biotin and streptavidin binds the immunological complex. The test has a reference range of 5–35,000 ng/L and will only take 18 minutes.67, 68
Latest Test
According to data from the World Health Organization (WHO), cardiovascular diseases (CVDs) remain the primary cause of death globally. 69 Point-of-care testing (POCT) may now accurately and swiftly identify HF because to be economically feasible lateral flow assays (LFAs). For example, the cobas h 232 NT-proBNP from Roche Diagnostics GmbH provides a sensitive platform that can detect NT-proBNP in 150 µL of whole blood, with a detection limit of 60 pg√mL−1 and a range of detection of 60–9000 pg∙mL−1. 70 The fleece fiber and membrane components that make up the LFAs are commonly utilized for sample transportation and as a drying substrate for antibodies specific to the probe. 71 Both require a sizable sample volume, have a relatively large dead volume, and function less well as a result of their intrinsically high analyte and sample adsorption capacities. 72
For POCTs to appropriately meet this requirement, a small finger-prick blood sample must be used instead of a venous blood draw. This adjustment reduces invasiveness, ensures patient comfort, and speeds up sample handling. Therefore, future tests should be as easy to administer as the one-step LFA in order to increase analytical figures of merit while considering patient comfort. The test configuration shown here suggests that reasonably sized supplementary hardware can be used to gain such analytical properties. While doing POC diagnostics without an instrument is no longer possible, the integration of fluorescence detection, active fluid control, and nanotechnology has created new opportunities for very accurate and reliable outcomes. However, enhanced surface-to-volume ratios, improved reaction kinetics, precise fluid management, and enhanced detection have developed an automated method that is easy to use and only requires one-step to complete. 73
Advantages of NT-proBNP
A growing body of research has examined the efficacy of using NT-proBNP and BNP in conjunction with the growing usage of NP testing for HF. Based on research, NT-proBNP has a number of distinct benefits in terms of clinical performance, stability, and compatibility with other techniques.
Stability in Biochemistry
NT-proBNP has a longer half-life (about 120 minutes) than BNP (approximately 22 minutes), which makes it possible to assess steady-state ventricular performance with less sensitivity to transient physiologic changes. 74 Because NT-proBNP degrades less quickly both in vivo and in vitro, testing blood specimens that are more stable can be used. 75 However, BNP has been shown to have poor in vitro stability in a number of studies, which is definitely caused for concern if testing is delayed after specimen collection.76, 77
Standardization of Assays
Because all licensed NT-proBNP tests are calibrated according to the Roche method, they yield findings that are similar between different testing systems and laboratories. 74
In contrast, BNP tests use a variety of antibodies and targets and are not standardized to a single approach. Consequently, results from various BNP testing platforms may differ. 74
Reliable Clinical Results
Research has demonstrated that NT-proBNP is a more accurate predictor of hospitalization, death, and morbidity in patients with HF than BNP. 78
In a study comparing the diagnostic performance of BNP and NT-proBNP for a variety of HF patients with varying severity, as well as a healthy control group, it was discovered that NT-proBNP was superior to BNP in identifying mild HF. 79 Applying NT-proBNP for determining pharmacological therapy for individuals with chronic HF is associated with decreased all-cause mortality and HF-related hospitalization, however using BNP-guided medication is not significantly associated with lower mortality and morbidity. 80
Difference Between BNP and NT-proBNP
Biomarkers for HF diagnosis and treatment include BNP and NT-proBNP. Although they share the same proBNP precursor molecule, they are not the same in terms of their structure, half-life, or therapeutic applicability. The difference between BNP and NTproBNP are summarized in Table 2. The main variations are as follows:
Difference between BNP and NTproBNP.

NT-proBNP and its Association with HFpEF
In individuals with suspected HFpEF, NP is a key component of the diagnostic work up advised by guidelines. 87 According to a study by Verbrugge et al. shows that patients with HFpEF and normal NP levels, as demonstrated in earlier research, represent a population with distinct, unambiguous cardiac and vascular anomalies that meet predetermined criteria of cardiac failure. As far as we know, this study is the first to demonstrate that patients with HFpEF and normal NP had a higher chance of a negative outcome when compared to controls who do not have cardiac dyspnea. 88 According to a meta-analysis of 51 articles, NPs can detect HFpEF in a chronic context with reasonable diagnostic performance (area under the receiver operating characteristics curve [AUC]: 0.80; 95% CI: 0.73–0.87). 89 Given the increased frequency of non-cardiac diseases in HFpEF, comorbidities have a significant clinical impact on NP circulating levels in both HFpEF and HFrEF. COPD, AF, kidney illness, and diabetic ketosis are among the disorders linked to higher NP levels; on the other hand, obese persons may have dramatically lower NP levels.90, 91 Additionally, older age correlates with higher amounts of NP.90, 92 One of the first significant studies looking into the use of NP in the emergency room to diagnose HF was the Breathing Not Properly Study. 10 In this instance, the BNP levels of 1586 patients who arrived at the emergency room complaining of severe dyspnea were assessed by the authors. BNP values were greater in patients with clinically diagnosed HF than in non-HF patients (mean 675 ± 450 pg/mL vs. 110 ± 225 pg/mL; P = .001). A positive correlation was shown between rising BNP concentrations and an increasing severity of HF, as determined by the New York Heart Association’s (NYHA) functional class (P < .001). When considering all relevant factors such as individual history, physical examination, chest X-ray, and laboratory results, BNP was the most reliable predictor of a final diagnosis of HF. The sensitivity and specificity of a cut-off BNP value of 100 pg/mL were 90% and 76%, respectively. 93 In a research involving 205 HFpEF patients admitted for acute HF, discharge NT-proBNP ≥1500 ng/L (HR: 5.23, 95% CI: 2.87-17.8, P < .001) and ≥50% NT-proBNP reduction between admission and discharge (HR: 0.62, 95% CI: 0.25-0.79, P = .019) were found to be independent predictors of HF-related death and rehospitalization. Additionally, when compared to each variable examined separately, the combination of E/e’ and NT-proBNP levels at discharge greatly enhanced the predictive ability (AUC, NT-proBNP at discharge: 0.80; E/e’ at discharge: 0.77; E/e’ + NT-proBNP: 0.88; P < .01). 94 According to a study conducted by Chen et al. showed that hospitalized individuals with HFpEF frequently have low NT-proBNP levels. A better prognosis was observed in patients with HFpEF and low NT-proBNP levels than in those with increased levels, especially in younger, male, and non- AF individuals. A poorer prognosis in HFpEF is linked to higher levels of NT-proBNP, which suggests a higher chance of fatalities and severe cardiovascular events. Age, renal function, and other comorbidities may have an impact on NT-proBNP levels, which could have an impact on the test’s predictive and diagnostic accuracy which contributes as a major limitation. A combination of clinical assessment, imaging investigations, and biomarkers are used to diagnose HFpEF, with NT-proBNP being one piece of the diagnostic puzzle. Thus, by identifying patients who are more likely to experience negative outcomes, NT-proBNP levels can assist healthcare providers in risk stratification.
Treatment Approach
According to 2022 AHA guideline for the treatment of HF, the following has been recommended according to class of recommendation (COR), level of evidence where COR is divided into Class 1, 2a, 2b, and 3. 27
Following an official diagnosis of HFpEF, the goals of treatment are to reduce hospitalization, enhance quality of life, and relieve symptoms. In order to maintain a euvolemic condition and get symptomatic relief, a low-sodium diet and loop diuretics must be used. A subgroup analysis of the CardioMEMS Heart Sensor Allows Monitoring of Pressure to Improve Outcomes in NYHA Class III Heart Failure Patients (CHAMPION) trial revealed that patients on diuretic treatment saw a 50% reduction in the frequency of hospitalizations for HF. 95 It has been demonstrated that spironolactone had a negligible effect on LV mass, NT-proBNP levels, E/E′, and the probability of HF hospitalization. 95 Sildenafil, a phosphodiesterase-5 inhibitor, ranolazine, a slow sodium channel inhibitor, and the angiotensin receptor blockers candesartan and irbesartan are some of the well-known drugs that have recently been involved in HFpEF treatment studies. 96
Therapies Targeting NP
The natriuretic and anti-proliferative characteristics of NP, such as BNP and atrial NP ANP, are essential for maintaining fluid homeostasis. 97 It has been demonstrated that HFpEF lowers NP levels; many patients have B-type NP levels less than 100. 98 Inhibiting neprilysin, a protease enzyme that breaks down NPs, may assist patients with HF to experience less fluid overload. 97 Patients with HFpEF have responded favorably to the new medication LCZ696, which combines neprilysin inhibitors with angiotensin receptors (like valsartan). 99 In the phase II PARAMOUNT 31 trial, 308 patients with symptomatic HF, an LVEF of at least 45%, and increased NT-proBNP levels were compared between LCZ 696 and valsartan. NT-proBNP levels were 26% lower in the LCZ696 arm at 12 weeks than in the valsartan group. Furthermore, at 36 weeks, patients in the LCZ696 arm showed higher improvements in left atrial size together with a reduction in symptom severity. 99
Increase in NT-Pro BNP other than HF
BNP levels increase in the following conditions
27
Acute renal failure and chronic renal failure Hypertension (HTN) Pulmonary diseases such as pulmonary hypertension, severe COPD, pneumonia, pulmonary embolism, Adult Respiratory Distress Syndrome (ARDS) Cardiac causes-Myocardial infarction, atrial fibrillation, acute coronary syndrome, cardioversion, valvular heart disease, myocarditis Older age Female sex Liver cirrhosis Hyperthyroidism Sepsis Chemotherapy
NT-proBNP in Dyspnea
NT-proBNP is a valuable indicator in the differential diagnosis of pulmonary and cardiac dyspnea in older adults due to the high sensitivity of the testing method and the strong connection with the severity of HF. 100 It is often very difficult to diagnose HF as the root cause of severe dyspnea in the emergency room. The truth is that signs, symptoms, and systematic testing like electrocardiograms (ECGs), chest X-rays, and blood analysis lack specificity and sensitivity. 101 Acute dyspnea patients are seen at the emergency room and their care is determined by the findings of their physical examination and medical history. Basic laboratory investigations and testing, including ECGs, chest X-rays, echocardiograms, and troponins, are also beneficial. Owing to the limited availability and accuracy of current laboratory and clinical criteria, particularly in acute care hospitals, a sensitive and specific biomarker indicating the changes in hemodynamics caused by dyspnea-related cardiac issues is needed. According to one study, NT-proBNP is important for ruling out acute CHF, and an age-independent cut point of 300 pg/mL should be used for this indication.102, 103
Future Perspective
There is a dearth of research on the prevalence of low NP levels in patients with HFpEF, and it is still unclear how often these levels actually occur. In-depth studies can be done to investigate how low NT-proBNP values can be a prognostic factor in determining all-cause death in patients with HFpEF.
Conclusion
NT-proBNP plays a critical role in the landscape of HFpEF, providing prognostic information, diagnostic clarity, and a useful treatment monitoring tool, as the review concludes. Even if there are still difficulties and subtleties, more research could help to improve its application and clarify the intricate mechanisms that underlie HFpEF. To provide patients with HFpEF with individualized and efficient therapy, NT-proBNP must be integrated into a comprehensive management strategy that includes clinical, imaging, and biomarker data.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Ethical permission was not applicable for this article, as this is a review article drafted from various research articles and not from patients directly.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
Consent was not needed/not applicable, as this is a review article compiled from various research articles and guidelines and not from patients directly.
