Abstract
Although Himalayan P waves are not uncommon in congenital heart diseases involving the tricuspid valve, it is rare in pediatric idiopathic restrictive cardiomyopathy. We are presenting one of the tallest P waves in literature, the Himalayan P in a toddler with idiopathic restrictive cardiomyopathy. Additionally, we reviewed the literature on the value of electrocardiograms in the evaluation of restrictive cardiomyopathy.
Introduction
Himalayan P waves are uncommon in the pediatric age group. Often it is due to congenital heart disease with tricuspid valve involvement. It is classically seen in Ebstein’s anomaly of the tricuspid valve, tricuspid atresia, combined tricuspid and pulmonary stenosis. Restrictive cardiomyopathy (RCM) is a rare cause of Himalayan P in the pediatric age group as many of the adult-associated risk factors are absent in children. Here we are presenting one of the tallest Himalayan P waves mimicking ventricular bigeminy in lead V1 in a toddler with RCM with a brief review emphasizing electrocardiographic abnormalities in pediatric RCM.
Case
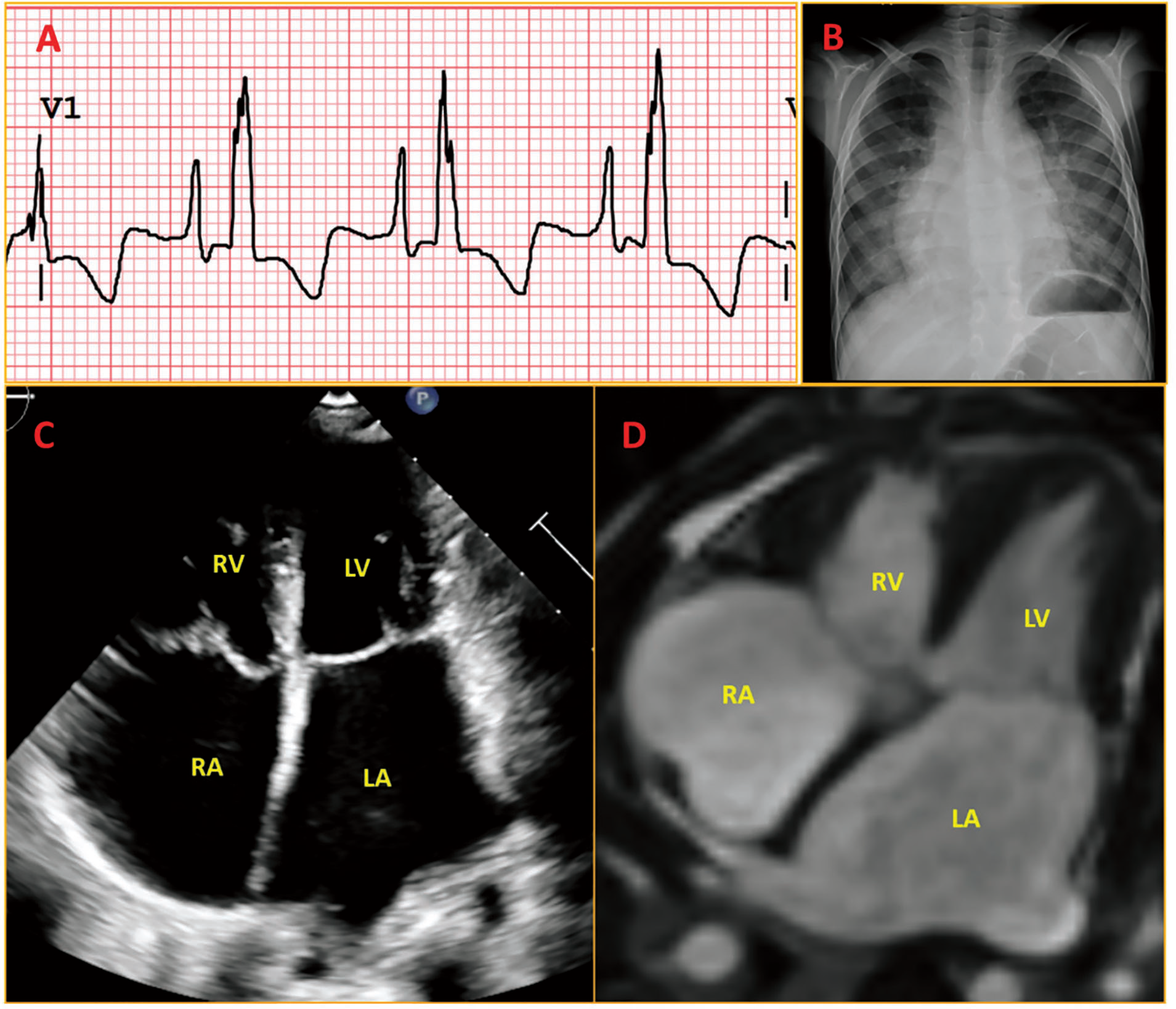
A toddler with an uneventful perinatal history was diagnosed with cardiac disorder while being evaluated for lower respiratory tract infection. On probing, the patient’s mother admitted a three-month functional class 2 dyspnea history. Clinical evaluation showed elevated jugular venous pressure, no pedal edema, no cardiomegaly, loud P2, left ventricular S3, and faint pan-systolic murmur at the mitral and tricuspid area. Electrocardiogram (ECG) showed sinus rhythm with tall peaked P (peak to trough ~9.2mm) wave in V1 and deep and wide terminal negative deflection (Morris’ index: increase in P-wave terminal force in V1 [PtfV1], with terminal negativity of P wave in V1 > 0.1 mV and duration >0.04 s), suggesting biatrial enlargement (Figure 1A, Supplementary Figure 1). There was fragmentation of QRS, suggesting a myocardial disease with intraventricular conduction delay. Chest X-ray showed biatrial enlargement with significant pulmonary venous hypertension (Figure 1B). The echocardiogram revealed a dilated, non-collapsible inferior Caval vein, significant biatrial enlargement, moderate mitral and tricuspid regurgitation, and moderate pulmonary hypertension suggestive of RCM (Figure 1C). Cardiac catheterization showed right/left ventricular end-diastolic, mean pulmonary artery, and right atrial pressure of 16, 34, and 6 mmHg, respectively, confirming the presence of restrictive physiology. Cardiac magnetic resonance imaging confirmed echocardiographic findings with minimal late gadolinium enhancement of the right atrial wall (Figure 1D). Extensive work-up for other causes of cardiomyopathy was negative, like HIV, sarcoidosis, amyloidosis, storage disorders, and hypertrophic cardiomyopathy. Hence he was diagnosed as RCM and listed for a heart transplant.
Discussion
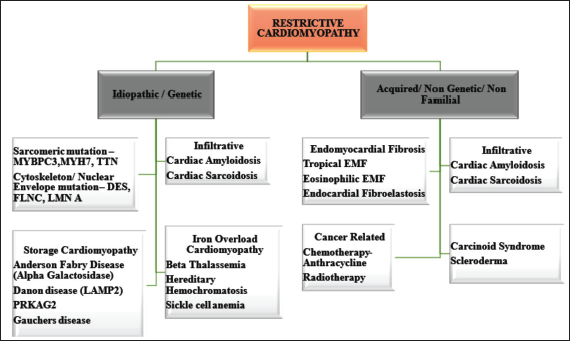
RCM is a rare heart disease to present in the pediatric population accounting for 2.5%–3% of all pediatric cardiomyopathies (Figure 2). 1 Pediatric RCM has a poor prognosis with two years of mean survival of <50%. 1 The mean age of presentation is 6–11 years. 1 It is associated with increased risks of thromboembolic events, arrhythmia, sudden death, and increased incidence of pulmonary hypertension. More importantly, it is less amenable to surgical and medical interventions, with heart transplantation being the only option in most of the cases. One-third of idiopathic RCM in pediatrics is due to sarcomeric gene mutations like—myosin binding protein (MYBPC3), β-myosin heavy chain (MYH7), Titin (TTN), Troponin T (TNNT2), Troponin I (TNNI3), myosin light chain genes, and alpha cardiac actin (ACTC). 1
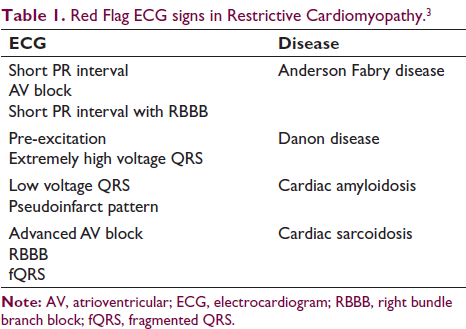
ECG is a very useful screening tool in RCM. It’s abnormal in 90%–99% of patients with idiopathic RCM. ECG features of RCM include (a) Low voltage QRS complexes because of diffuse myocardial infiltration/ fibrosis; (b) non-specific ST segment and T wave changes (80%). Hayashi et al. reported that obliquely elevated ST segment with notched or biphasic late peaking T waves (67%) and ST segment depression with T inversion (25%) were characteristic ECG changes of idiopathic RCM 2 ; (c) biatrial enlargement evidenced by biphasic P wave in precordial leads (91%) and atrial fibrillation (74%); (d) infiltration of conduction system causing bundle branch blocks, atrioventricular block; (e) pathological pseudo-infarction Q waves because of healing granuloma as in sarcoidosis.3, 4 Muraji et al. found that P wave peak to trough >200 µV is sensitive enough to rule in all RCM patients in the pediatric population compared to the age-matched control population. 5
In our case, the total P wave duration in V1 was 110 ms, almost similar to QRS duration (120 ms), giving a false impression of junctional escape echo capture bigeminy (where junctional escape conducts retrogradely, causing retrograde atrial depolarization in turn leading to antegrade AV conduction causing another ventricular depolarization). However, another lead examination and mitral inflow Doppler confirmed it to be a P wave rather than a junctional escape.
Himalayan P/P congenital (>5 mm) waves are indicative of a dilated right atrium and often noticed in Lead 2 where it is even taller than QRS. 6 It is classically seen in anatomical or functional abnormalities of the tricuspid valve like Ebstein’s anomaly, tricuspid atresia, combined tricuspid, and pulmonary stenosis. 7 In adults it has been demonstrated in various conditions like RCM, dilated cardiomyopathy, ischemic cardiomyopathy, hypertrophic cardiomyopathy, and obstructive airway diseases.8, 9 Vlismas et al. Demonstrated a nonspecific interventricular conduction delay, low voltage QRS in the limb leads, and biatrial abnormality with very tall and wide P waves in a 34-year-old man with idiopathic RCM. 7 Gupta et al. from India demonstrated tall P waves (in lead V1 > 22 mm and lead 1 > 9 mm) with right axis deviation and inverted T waves in a 14 years old girl with idiopathic RCM. 10 Canpolat et al. from Turkey reported unexpectedly tall P waves (9 mm in lead V2), left atrial abnormality (negative P wave in lead V1) and right axis deviation in a 22-year-old girl with idiopathic RCM. 11
RCM should be further evaluated with an echocardiogram, cardiac magnetic resonance imaging, genetic study, and cardiac biopsy in selected cases to establish a diagnosis of RCM, especially when red flag signs are present in ECG (Table 1). Poor prognostic markers of RCM include pulmonary congestion, myocardial ischemia, severe left atrial dilation, male gender, reduction of left ventricular fractional shortening, and increased left ventricular wall thickness. Hemodynamic assessment should be done with cardiac catheterization to look for pulmonary vascular disease. 1 If pulmonary vascular resistance (PVR) <6 woods unit, transpulmonary gradient (TPG) <15 mmHg, and diastolic pressure gradient (DPG) <7 mmHg, heart transplantation is considered. 1 If PVR > 6 woods unit, TPG > 15 mmHg, DPG > 7 mmHg, indicators with/without assist devices and pulmonary vasodilators are to be instituted to reassess operability on follow-up in three to six months. Persistently high PVR (>6 woods unit) and high TPG (>15 mmHg) are contraindications for heart transplantation. 1 Hence they are to be offered mechanical support devices (LVAD/RVAD/BiVAD/total artificial heart) with or without pulmonary vasodilators as and when indicated.
Red Flag ECG signs in Restrictive Cardiomyopathy. 3
Conclusion
ECG is a valid screening tool for RCM. Red flag ECG signs should be kept in mind while diagnosing a case of RCM and should be properly evaluated further especially to rule out potentially treatable diseases in the pediatric population where it has a worse prognosis. Many reports of Himalayan P wave in adolescents and adults with idiopathic RCM have been demonstrated. We are presenting a rare finding of Himalayan P in a toddler with idiopathic RCM.
Author Contributions
SS and SM = Conceptualization: equal; formal analysis: lead; writing—original draft: lead; writing—review & editing: lead.
SB = Conceptualization: equal; formal analysis: lead; writing—review & editing: lead.
Ethical Approval
Not applicable.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent
Obtained from the patient in line with COPE guidance.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
