Abstract
A 22-year-old male having cardiomegaly with prominent right atrium (RA) enlargement in his chest X-ray underwent echocardiography which revealed an anterior mediastinal cystic lesion. Contrast-enhanced computed tomography of the thorax showed saccular contrast filling outpouching from ostia of the right coronary artery (RCA) of size 5.9 × 6.4 cm in anterior atrioventricular groove compressing RA and right ventricle along the RCA course. The left main coronary artery also appeared to be dilated with a diameter of 1.1 cm proximally. The patient denied any prior angina, palpitation, and dyspnea on exertion. This case is reported for its rarity and the dilemma involving its appropriate medical and surgical management.
Introduction
Coronary artery aneurysm (CAA) is a very rare clinical condition and is defined as an abnormal focal dilatation of the coronary artery that exceeds the 1.5-fold diameter of the adjacent normal artery segment. 1 If there is a similar but diffuse lesion involving >50% of the coronary artery length, it is termed coronary artery ectasia. 1 When there is a very large-sized CAA, that is diameter greater than 20 mm or four times the size of the reference vessel it is considered a giant CAA (GCAA). 2 Etiology and pathophysiological mechanisms are not well understood, but atherosclerosis, connective tissue disorders, and inflammatory reactions with proteolytic imbalance are thought to be involved in aneurysmal formation. The management of CAA is challenging, particularly in younger individuals. It must be individualized depending on clinical presentation, CAA characteristics, and patient profile. Multiple therapeutic approaches including medical treatment, covered stent angioplasty, coil insertion, and surgical resection have been described. 3 We herein report a case of rare GCAA along with its various management-related perspectives.
Case Report
A 22-years old male patient, was normal and asymptomatic when he developed high-grade fever and cough for three days. He went to a pulmonologist who gave him tablets of azithromycin, paracetamol, and levocetirizine. A chest X-ray was done outside which revealed cardiomegaly. Hence, he was referred to the cardiology department. When the patient came to our hospital, the patient had become afebrile. He only had mild cough and dyspnea on exertion functional class II. His vitals were stable with a pulse rate of 84 per minute, oxygen saturation of 99% on room air, and blood pressure of 124/78 mm Hg. Routine lab and sputum investigation reports were unremarkable and shown in (Table 1). Chest examination showed normal bilateral vesicular breath sounds with no added sound. Cardiovascular system examination was clinically normal with no murmur. Chest X-ray done at our hospital revealed Right atrium (RA) enlargement (Figure 1A). Electrocardiogram showed sinus rhythm with normal PR interval (143 ms) and no significant ST-T changes (Figure 1B) and echocardiography (ECHO) showed a mediastinal cystic lesion present in the anterior atrioventricular (AV) groove with normal biventricular function. The size of that lesion was approximately 6.5 cm. The lesion caused significant RA compression with no Color Doppler flow between them (Figure 2). A transesophageal echo was done which revealed similar findings along with spontaneous echogenic contrast inside that lesion (Figure 2). No vegetation was seen in the ECHO. Differential diagnoses at this stage were an aneurysm (coronary or sinus of Valsalva), pericardial mass, and hydatid cyst. Contrast-enhanced computed tomography thorax revealed saccular contrast filling outpouching arising from ostia of the right coronary artery (RCA) measuring 5.9 × 6.4 cm in AV groove compressing RA and right ventricle along the RCA course and faint flow was noted in distal RCA. The left main coronary artery (LMCA) also appeared to be dilated and ectatic with a diameter of 1.1 cm along its length of 1.6 cm. Both the left anterior descending artery and left circumflex artery originated from the distal LMCA ectasia with no flow-limiting lesions (Figures 3 and 4).
Relevant Laboratory Investigations.
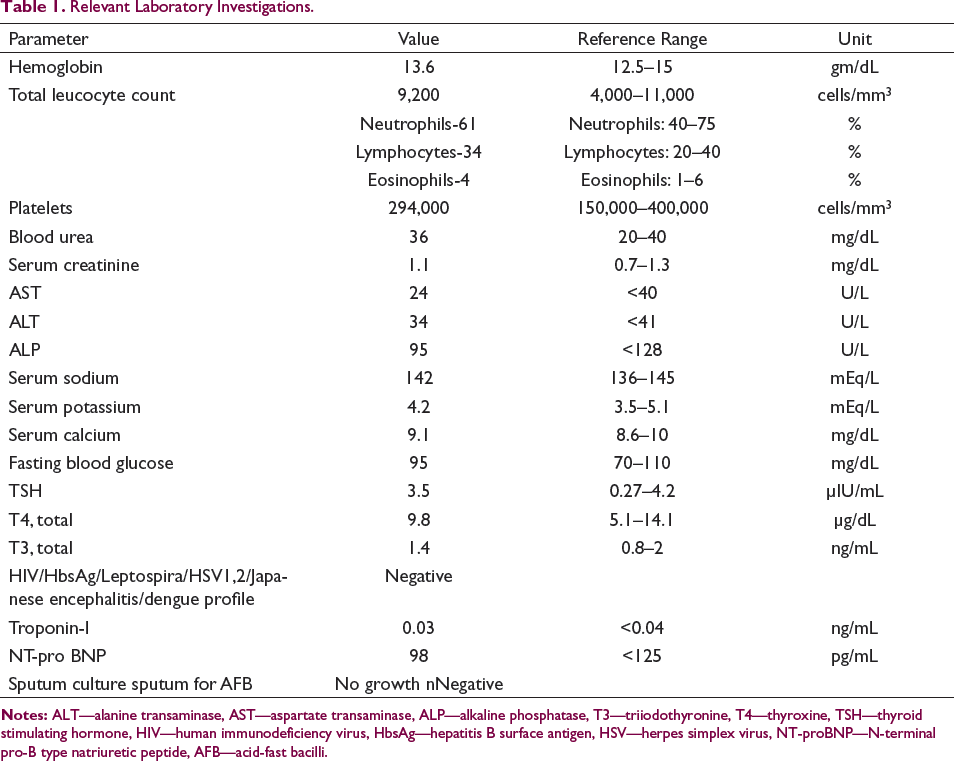
(A) Chest X-ray Posteroanterior View Showing Cardiomegaly with RA Enlargement (Solid Blue Arrow), (B) Electrocardiogram of the Patient Showing Sinus Rhythm, Right Axis Deviation, Normal PR, QRS, and QTc Interval and No Significant ST-T Changes.
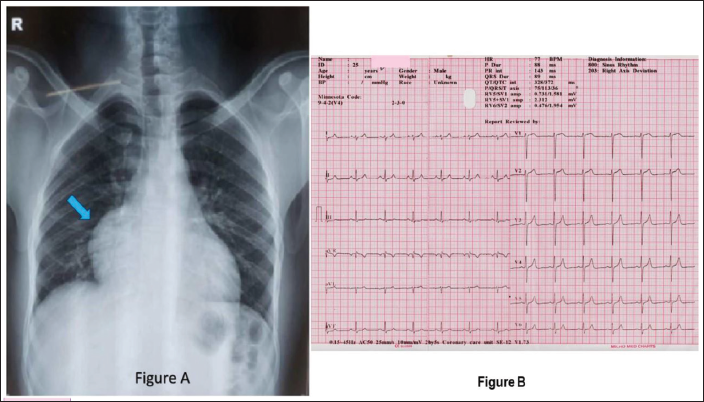
(A) Transthoracic Echocardiography in Parasternal Long Axis View Showing Normal Cardiac Chambers and Aorta. The Cystic Lesion is Not Visualized in This View, (B) Transthoracic Echocardiography in Parasternal Short Axis View Showing Large Cystic Lesion (Solid Blue Arrow) Near Right Coronary Sinus. (C) Transesophageal Echocardiography Showing the Same Large Cystic Lesion (Solid Blue Arrow) with Spontaneous Echogenic Contrast. (D) Transthoracic Echocardiography in Apical Four-chamber View Showing Compressed RA (Solid Blue Arrow) with No Color Doppler Flow Noted in the Cystic Lesion.
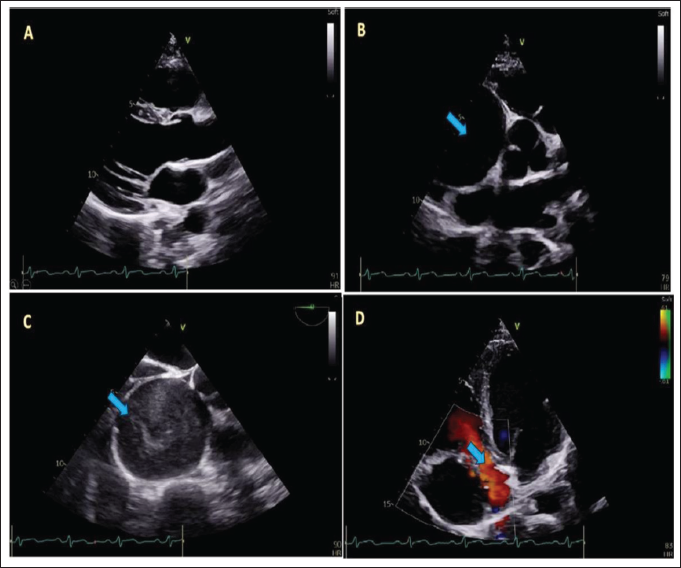
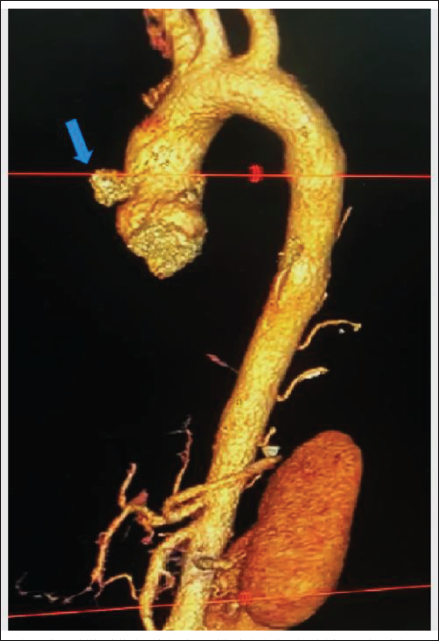
In a further study with cardiac magnetic resonance imaging, fixed perfusion defects were noted in both stress and rest perfusion images in the inferoseptal segments in the mid and apical ventricular region with corresponding areas showing subendocardial late gadolinium enhancement. This was suggestive of ischemia in those mentioned areas. Imaging for left ventricular ejection fraction (LVEF) at rest and after stress was normal. Based on all clinical and investigational details, the final diagnosis was made as saccular aneurysm from RCA ostium, possible Kawasaki disease, with normal LVEF. The patient was treated with single antiplatelet therapy, statin, and warfarin and referred to cardiovascular thoracic surgery for opinion. There he was advised for surgical resection of the aneurysm and coronary artery bypass with graft to distal RCA. He is awaiting the same.
Discussion
A CAA is defined as an abnormal, irreversible, and localized dilatation of the blood vessel lumen that exceeds the diameter of the adjacent normal segment more than 1.5-fold. CAA’s overall incidence ranges from 0.3% to 5.3% and a meta-analysis stated a mean incidence of CAA to be 1.65%. 4 The incidence of GCAA is as low as 0.02%. When compared between males and females, males have a higher incidence of CAA than women, 2.2% as compared to 0.5%, respectively. 4 Although the most commonly involved vessel is the RCA, (40%–70% of CAA), involvement of three coronary vessels or the left main artery may be present in extremely rare occurrences (3.5%) 5 as seen in this present case.
Atherosclerosis is accountable for more than 90% of CAA in adults, whereas in children, Kawasaki disease is responsible for most cases. 6 The pathophysiology of CAA remains unclear but is perceived to be similar to that for large vessel aneurysms. In the study done by Berkoff and Rowe, 7 it was hypothesized that the presence of a thin degenerated media, adjacent to intimal plaque was the major pathological prerequisite leading to plaque rupture. The excavated plaque then leads to aneurysm formation. Matrix metalloproteinases by causing accelerated proteolysis of extracellular matrix proteins are also an important factor in the pathogenesis of CAA formation.
In most cases, CAA is usually asymptomatic. However slow flow of blood on the irregular internal surface of the aneurysm wall predisposes the formation of thrombi with subsequent embolization, resulting in angina pectoris, dyspnea, cough, myocardial infarction, and sudden death. There is no consensus on the optimal management of CAAs. 8 The role of dual antiplatelets or therapeutic anticoagulation in the management of patients with CAA, especially those that are incidentally discovered, is an area of ongoing debate. Vasodilators are usually contraindicated because they may exacerbate myocardial ischemia. Indications for invasive management of a CAA are considered when it is responsible for ischemic symptoms or causes an acute coronary syndrome or when it shows high-risk features of impending rupture. Percutaneous treatment is usually the first choice in these conditions and consists of angioplasty with the deployment of covered stents or coil embolization. However, the lack of purpose-specific devices, high thrombus burden, and challenges of covered stent delivery make percutaneous coronary intervention (PCI) very difficult. 9 Favorable outcomes of PCI in asymptomatic patients with CAA are limited to small case series only. Intravenous immunoglobulin use in patients with Kawasaki disease has been shown to reduce CAA formation and lower incidences of major adverse cardiovascular events. 8 Finally, surgery is the best alternative for patients who cannot be treated percutaneously, and mainly includes, proximal and/or distal ligation of aneurysm, aneurysmal thrombectomy, and aneurysmectomy with or without bypass grafting. 8
Conclusion
CAA is rare and mostly found incidentally during coronary angiography. Precise pathology leading to CAA is not clear. The role of long-term dual antiplatelet therapy or anticoagulation in the primary prevention and intervention of incidentally discovered CAA is not fully answered yet. The learning point here in this rare asymptomatic case of GCAA is about its treatment options. It consists of medical, percutaneous, or surgical interventions, but the optimal treatment for CAA remains debatable. Anticoagulation followed by surgical resection of the aneurysm with bypass surgery was opted for in this present case, but a large-scale study is required for treatment guidelines in the future.
Consent
Written consent has been obtained from the patient for the submission of images and associated text and publication of this case report.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
No ethical approval is required. All identifiable patient information has been removed from this manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
