Abstract
Optical coherence tomography (OCT) is an optical analog of intravascular ultrasound (IVUS) used to examine the coronary arteries with a higher resolution. Based on polarization properties, OCT can differentiate tissue characteristics (fibrous, calcified, or lipid-rich plaque) and identify vulnerable plaque. Because of the strong attenuation of light by blood, OCT requires a field of view that is free of blood. OCT can provide detailed information on coronary intervention such as dissection, tissue prolapse, thrombus stent malapposition, and under expansion. Despite the diagnostic utility of OCT, facilitated by its high resolution, the impact of intracoronary OCT on clinical practice has thus far been limited for various reasons. Herein, we provide a comprehensive up-to-date overview on the utility of OCT in coronary artery disease, specifically acute coronary syndromes along with a summary of the evidence, work in progress, and future directions.
Introduction
Angiography is a two-dimensional lumenogram with a set of limitations. Intravascular imaging―intravascular ultrasound (IVUS) and optical coherence tomography (OCT)―together help in obtaining a cross-sectional tomographic image of the coronary artery and thus providing useful additional information that complements the information obtained by angiography. Intravascular imaging is also helpful in the selection of the appropriate treatment strategies to be employed for percutaneous coronary intervention (PCI). Registries,1–3 randomized controlled trials,4, 5 and meta-analyses6, 7 have consistently shown the procedural benefits of intravascular imaging-guided PCI in the long term.
Role of OCT in PCI
Intravascular OCT: Basic Technology, Advantages, Limitations, and Various Available Imaging Platforms
OCT works on the principle of near-infrared light directed at the vessel wall through a rotating single optical fiber coupled with an imaging lens within a short-monorail imaging sheath. OCT provides us with high-resolution images of the vessel microstructure in cross-sectional and three-dimensional format by measuring the amplitude and time delay of the backscattered light. It is required to flush with contrast to displace the blood during OCT acquisition, as blood strongly scatters light and attenuates the OCT signal.
The infrared light in OCT has a shorter wavelength (1.3 µm) as compared with the ultrasound waves in IVUS (~40 µm at 40 MHz) which yield greater axial resolution (10–20 µm versus 50–150 µm) but have poor penetration depth (1–2 mm versus 5–6 mm), which limits the OCT imaging, especially when there are highly attenuating structures present, like a red thrombus or lipid/necrotic core.
Of the various OCT systems that have been developed, the OPTIS™ system (Abbott Vascular, Santa Clara, CA, USA) that provides combined OCT and angiographic visualization (co-registration) and the Lunawaveۚ system (Terumo, Tokyo, Japan) are currently the most popularly employed systems. Novasight Hybrid™ (Conavi Medical Inc., Toronto, Canada) and Dual Sensor (Terumo) are currently available hybrid OCT/IVUS catheter systems that are used for research, which combine the strengths of OCT and IVUS in a single catheter in clinical practice. 8
Practical Steps for OCT Image Acquisition
The basic components of the commercially available OCT imaging system consist of an OCT imaging catheter, a drive motor operating control unit, and an imaging software. To set up the OCT system, the imaging catheter must be attached to the purge syringe which should be flushed with the same material that has been chosen for flushing the coronaries. This enables us to ensure consistent results in the index of refraction. It is then attached to the drive motor operating control. Before OCT imaging, intracoronary nitroglycerine is given to eliminate the spasm caused by catheter irritation. The guiding catheter is then engaged at the coronary ostium in order to achieve clearance during catheter pullback. The subsequent imaging steps include advancing the OCT catheter over the coronary guidewire and placing it distal to the target lesion (~10 mm distally), after which the catheter is purged and a small volume of flush is “puffed” through the guide catheter (in case of marginal clarity, check whether the guide catheter is properly engaged to the target vessel, and if not, it should be adjusted accordingly), and then the pullback is activated. In cases of severely stenotic lesions, where it is difficult to advance the OCT catheter beyond the lesion, predilation with a small balloon can be performed. Flushing can be done either manually or by a power injector. The pressure limit is set at 300 psi for an automated injector to a flow rate of 4 mL/sec for a total volume of 14–16 mL for the left coronary artery and a total volume of 12–14 mL at a rate of 3 mL/sec for the right coronary artery. Contrast injection should be stopped immediately as soon as the guide catheter is seen on the pullback in order to minimize contrast waste. Cine angiography is performed during pullback to utilize contrast injection for angiography and OCT co-registration if the feature is available. OCT acquisition in the coronary arteries is not completely free of risk, but significant complications due to OCT acquisition are rare and can be avoided mostly by using meticulous techniques described in this review.
PCI Guidance
Assessment during pre-PCI includes quantitative and qualitative analysis. OCT helps us identify the culprit lesion, assess the morphology of the lesion to guide lesion preparation strategies, and characterize the precise stent landing zones, thus allowing precise selection of the diameter and length of balloons and stents. MLD MAX mnemonic (morphology, length, diameter, medial dissection, apposition, and expansion) helps in the proper guidance of PCI.
Guidance During Pre-PCI
Morphology
A simplified algorithmic approach for the interpretation of an OCT image in native coronary arteries is depicted in Figure 1. It describes the most frequent pathological abnormalities of the vessel wall which include low-attenuating and signal-rich lesions indicative of fibrous plaques, low-attenuating but sharply delineated and signal-poor regions indicative of calcific plaques, high-attenuating and signal-poor regions that are covered with a fibrous cap indicative of lipid-rich plaques, and those that are inside the lumen such as the low-attenuating white thrombus and the high-attenuating red thrombus.
An Algorithmic Approach to Optical Coherence Tomography Image Interpretation of Common Morphologies.
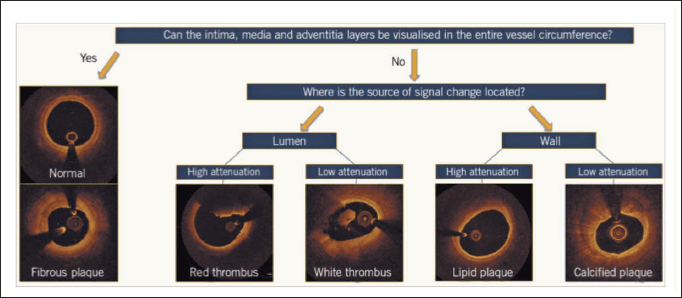
In most cases, both calcification arc and thickness can be determined by an OCT. An OCT-based scoring system has been designed and validated for calcified lesions to determine various morphologies that lead to under-expansion of the stent.9, 10 The factors that are identified may be effortlessly memorized as the “rule of 5s:” 1 point for calcium length >5 mm, 1 point for maximum calcium thickness >0.5 mm, and 1 point for maximum arc of calcium >50% of the circumference of the vessel (i.e., >180° of the circumference). When all factors are present (i.e., the total score is 3), it results in stent under-expansion in target lesions, which requires atherectomy or intravascular lithotripsy for calcium modification, whereas there is an adequate expansion of stent in the lesions with scores of ≤2.
Length
The OCT software by incorporating a multiplanar reconstruction of the three-dimensional data provides a two-dimensional lumenogram of the artery, referred to as the lumen profile. Initially, a region of interest is created by the largest lumen proximal to the lesion and distal to the lesion on the lumen profile. By navigating at these sites through the cross-sectional images of OCT, the segments that appear most “normal” within 5 mm are selected as the proximal and distal reference frames (i.e., these segments have good visibility of the medial [external elastic lamina (EEL)] and adventitial layers of the arterial wall as they have minimal atherosclerotic plaque burden). The OCT software then automatically calculates the length of the lesion. Usually, this number may not correspond to a commercially available drug-eluting stent (DES), so either the proximal or distal frame of reference, wherever there is a disease-free normal segment, is adjusted to an available DES length. This method significantly minimizes the stent edge problems, including geographic miss. The presence of thin cap fibroatheroma (TCFA) at the reference segments may predispose the risk of early stent thrombosis and post-PCI target lesion failure as they may cause significant dissections. 11
Diameter
An EEL-guided sizing strategy is preferred to a lumen-guided strategy as it leads to the selection of a larger device size (≈0.5 mm) which leads to a larger lumen area without increasing post-procedural complications. If two separate diameter measurements are made in the arterial wall at two different locations, which are at least one quadrant apart from each other, then EEL measurements may be utilized. Greater than 180° of EEL could be visualized in ~80% of cases in the ILUMIEN III trial. 12 To determine the device size, EEL-based measurements should be rounded down to the commercially available DES. If EEL is not visualized clearly, then the mean lumen diameter recorded from the automated lumen profile feature is utilized for sizing the device. To determine the device size, lumen-based measurements should be rounded up, between 0.25 and 0.5 mm. 13 The size of the stent and the balloon size post-dilation at the distal segment of the stent are determined by the distal measurements, whereas the proximal measurements help in determining the size of the balloon to be achieved for post-dilation at the proximal segment of the stent.
Angiographic Co-registration
To reduce manual errors in the co-registration of angiography with OCT frames, an automated software has been developed with real-time point-to-point correspondence between the coronary angiogram and OCT. A more precise stent deployment can be attained by OCT-angiography co-registration, which was shown in one randomized study, thereby eliminating large geographic miss (>5 mm) and thus reducing major stent edge dissection compared with angiography guidance. More recently, to minimize protrusion of stent struts into the main branch, identification and mapping of the side branch ostium while ensuring full coverage of the side branch ostium, referred to as BOOM, that is, bifurcation and ostial OCT mapping, has been described. 14
Post-PCI Guidance
Medial Dissection
High resolution of OCT has been criticized for providing “too much” information, which identifies dissections in up to 40% of PCI, of which ≈80% were not detectable by angiography, with no clinical impact. 15 In the CLI-OPCI registry, a major dissection is defined as a linear rim of tissue with a width >200 µm and it resulted in a 2.5-fold increased risk of major adverse cardiac events such as target lesion revascularization, myocardial infarction (MI), death at the distal edge, but no such risk at the proximal edge. The ILUMIEN IV study (ongoing) defines major dissections as the ones having medial dissection ± intramural hematoma ≥60° in an arc from the center of the vessel and ≥3 mm in length from the stent edge.
Apposition
Stent malapposition is defined as the lack of full contact between stent struts and the vessel wall following PCI. Malapposition can occur immediately after stent placement (acute stent malapposition) or it may occur later (late stent malapposition), which may be termed as late persistent malapposition or late acquired malapposition. Acute stent malapposition usually occurs after DES implantation, which could be identified on an average of 15% of the stents by IVUS 16 and at a higher rate of 50% by OCT 17 due to its higher resolution, flushing of the lumen, and automatic detection of the malapposed struts. There has been a matter of controversy regarding the potential impact of acute stent malapposition on stent failure rates. There has been no consistent correlation between the presence and/or extent of acute stent malapposition and early, late, or very late stent thrombosis, or adverse events after implantation of DES in several larger intravascular imaging studies. 16 The stent failure rates did not increase when there was acute stent malapposition without under-expansion. Proximal stent edge malapposition may result in difficult re-wiring, and immediate stent optimization is required in case of gross malapposition for long segments (>3 mm), or malapposition associated with under-expansion. Low-pressure inflation of semi-compliant balloon is sufficient for opposing stent struts as opposed to high-pressure inflation with non-compliant balloons to expand the stent struts.
Expansion
The stent is considered adequately expanded if the stent expands the lesion to diameters close to or equal to the diameter of the artery. Minimal stent area (MSA) ≥80% of the mean reference lumen area and/or >4.5 mm2 on OCT is considered acceptable according to current European consensus. 20 OPTIS™ Integrated System using the AptiVueۛ imaging software (both Abbott) provides assessment of stent expansion in two modes: by comparing the expansion automatically to the nearest respective proximal or distal reference in each half of the stented segment (dual reference mode) or calculation of the expansion automatically depending on interpolation of the size of vessel, considering OCT-detected side branches (tapered reference mode).
The segments immediately adjoining the stent are considered the reference segments and used to re-measure the size of the vessel, if stent under expansion is identified on post-PCI OCT, usually by EEL guidance, and if not lumen guidance can be considered on failure to visualize by EEL. For inflow/outflow disease, segments within 5 mm from the edges of the stent are considered the proximal and distal segments of reference and are examined following OCT-guided optimization of stent expansion. No further treatment is necessary if the minimal lumen area (MLA) is ≥4.5 mm2 at both the proximal and distal reference segments. If there is an untreated disease in either proximal or distal reference segments, defined as a focal MLA <4.5 mm2, unless contra-indicated anatomically (e.g., tapering of the vessel, distal diffuse disease, or unavailability of a landing zone), additional DES must be implanted.
OCT Image Artifacts and its Limitations
It is essential to assess the quality of the image and identify artifacts for accurate OCT image interpretation. With appropriate modifications, some artifacts can be corrected. OCT artifacts can be categorized as the artifacts that arise from light propagation in the catheter, lumen, or vessel wall and the artifacts that are related to catheter location and movement (Figure 2). OCT light is strongly attenuated by some lumen or vessel constituents, casting shadows that reduce the OCT signal intensity in the arterial wall. Signal-rich areas within the lumen are generated by inadequate blood clearance, thereby causing interference which decreases the OCT signal intensity from the arterial wall (Figure 2A, B). Blood swirling may be mistaken for thrombus or plaque erosion, particularly at the beginning or end of a pullback (Figure 2C, D). OCT light is attenuated by red thrombus, whereas metallic stent struts and guidewires block the path of incident OCT light, thereby producing shadows on the vessel wall.
Imaging Artifacts of the Optical Coherence Tomography.
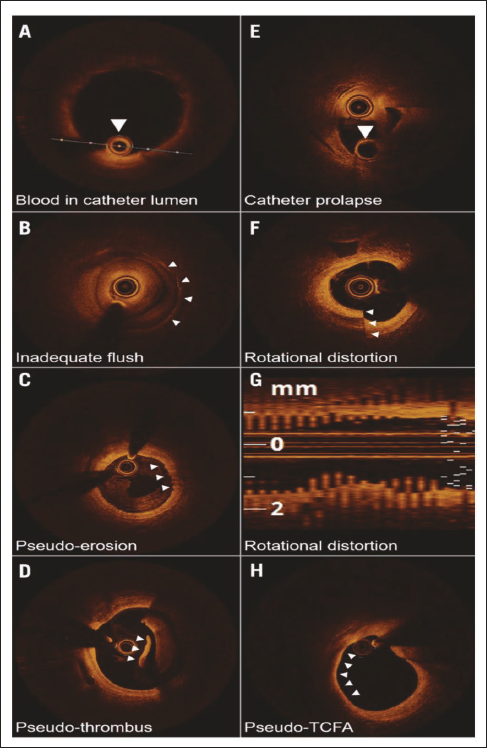
If undue force is used, the catheter may prolapse on itself (Figure 2E). Non-constant angular velocity of the monofiber optical catheter results in non-uniform rotational distortion (NURD) and is visualized as smearing or blurring in the lateral direction. NURD occurs as a result of the rotational resistance inside the catheter due to narrow or tortuous vessels, a crimped catheter sheath, or a tight hemostatic valve (Figure 2F, G). When the imaging catheter is placed close to the arterial wall, such that the light being emitted is parallel to the tissue surface, it would appear as though that part of the arterial wall is attenuating the light, when in fact the optical beam is not even penetrating that region. This artifact is usually confused with TCFA (Figure 2H).
OCT: Acute Coronary Syndromes
The superior resolution of OCT helps in identifying the characteristics of the lesion such as thrombotic debris, dissection, calcified nodules, calcium requiring lesion preparation, plaque erosion, rupture, and TCFA that lead to acute coronary syndromes (ACS).18, 19
Ruptured lipid-rich plaque is the most common cause of ACS in patients with obstructive atherosclerotic coronary disease.20, 21 On OCT, ruptured plaque is visualized as a discontinuity in the fibrous cap with a lipid-rich plaque containing a cavity inside. Overhanging thrombotic material can be seen in acute presentations which is not mandatory for making the diagnosis, as the thrombus may be dissolved by early treatment with antithrombotic and antiplatelet drugs or it could be removed mechanically by aspiration thrombectomy before performing coronary catheterization. Although plaque rupture is the most frequent cause of ACS, plaque erosion may be observed in 20%–30% of patients as shown by various pathological studies. The ability to differentiate between plaque rupture and erosion helps in guiding treatment strategies, perhaps with deferral of stenting in non-critical stenoses with plaque erosion, but before this approach can be broadly recommended, randomized data are warranted.
OCT can identify even subtle changes that commonly occur in plaque erosion such as (a) an intact fibrous cap with a white thrombus on it, (b) an irregular surface of lumen surface present without thrombus, or (c) thrombus attenuating the plaque in the absence of calcification or lipid plaque immediately proximal or distal to the location of the thrombus. Plaque erosion is described as “definite” in the presence of thrombus on an intact fibrous cap or “probable” when thrombus or rupture is absent, but there is luminal irregularity.
The least common cause of ACS is a calcified nodule. Studies have shown three different types of plaques at the site of culprit lesions, which include eruptive calcified nodules, superficial calcific sheet, and calcified protrusion. 22 Histopathologic studies have shown that, of all the subtypes, eruptive calcified nodules result in ACS, whereas calcified protrusions and calcific sheets are a part of the underlying fibroatheroma, and do not cause ACS. When cyclic hinge movement occurs in the mid-right coronary artery which is the usual location for eruptive calcified nodules, the calcified plaques are weakened, resulting in fracture. 22
OCT helps in the evaluation of MI with non-obstructive coronary arteries (MINOCA). 23 For women having MINOCA (i.e., coronary artery disease with stenosis diameter of <50% on angiography), OCT is capable of identifying definite or possible culprit lesions in 46% of the participants. The most common OCT findings were layered plaque, plaque rupture, and intraplaque cavity. Similarly, in patients with non-ST-segment elevation ACS (NSTEACS) and multivessel disease, identifying the culprit lesson by coronary angiography alone may be insufficient. An identifiable culprit lesion may not be found in up to 30% of patients with NSTEACS, and on angiography, >10% of patients may have multiple culprit lesions. Even in ST-segment elevation MI (STEMI), in up to 4%–10% of cases, the culprit lesions may not be identifiable by angiography alone. In such instances, OCT helps in resolving the ambiguity pertaining to the culprit plaques.
With or without angiographically discrete lesions, spontaneous dissections and intramural hematoma are easily identified on OCT and are sometimes in patients presenting with ACS.
Comparison Between OCT, IVUS, and Angiography in Registries and Randomized Trials
OCT relatively being a new modality currently lacks robust evidence compared with IVUS-guided PCI. Ongoing and completed trials of OCT-guided PCI are depicted in Table 1. OCT has significantly impacted the procedural strategy in the earliest studies. The CLI-OPCI trial comprising 670 patients compares angiography to an OCT-guided strategy, and adverse features that require further interventions were identified on OCT in 35% of cases. 24 After adjustment for potential confounders, OCT guidance has been shown to significantly reduce the risk of MI or cardiac death at 1 year.
Summary of Clinical Data From Various Studies on Optical Coherence Tomography Versus Intravascular Ultrasound Versus Angiography for Percutaneous Coronary Intervention Guidance.
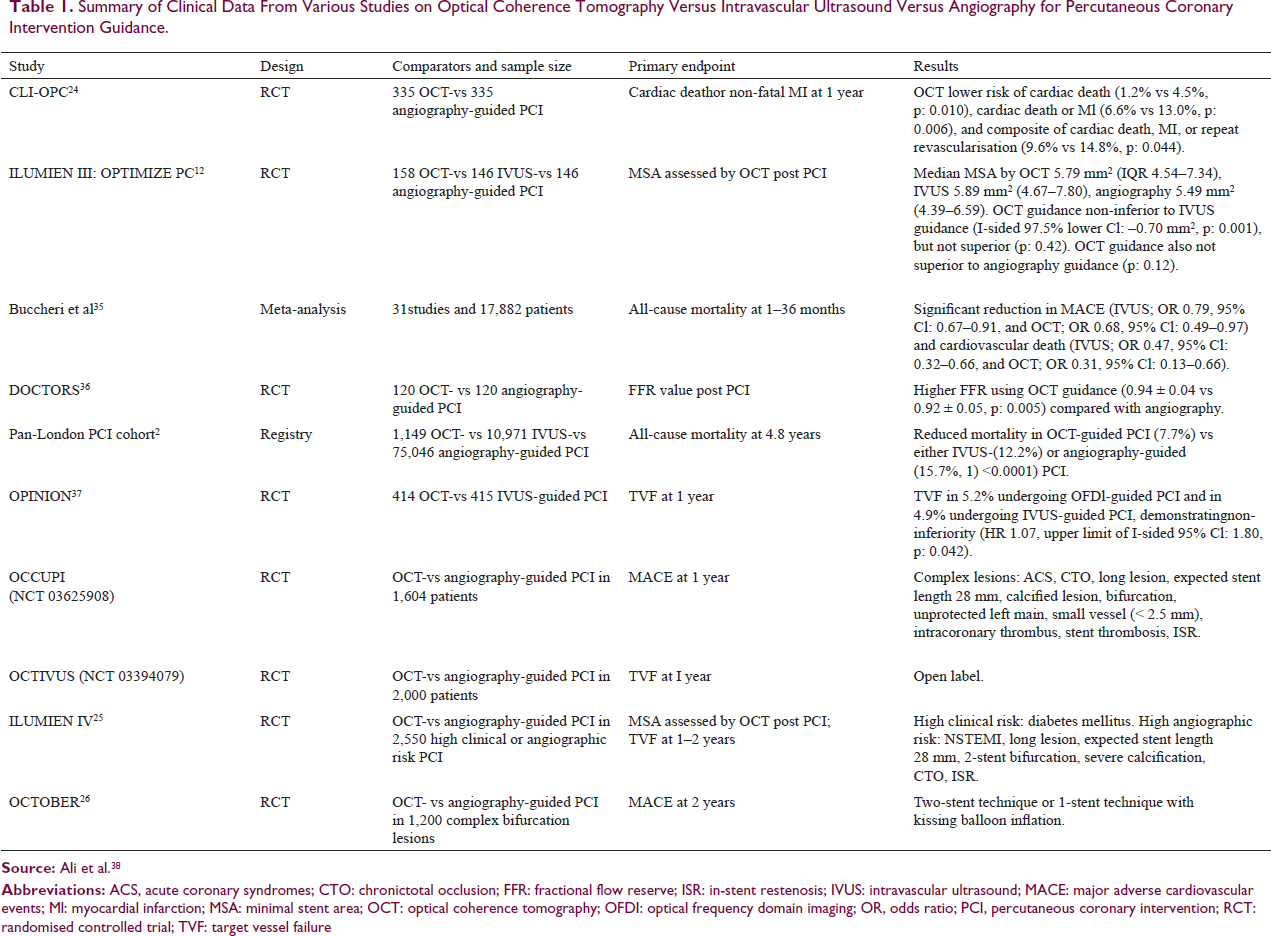
The ILUMIEN IV 25 and OCTOBER trials 26 are the two massive ongoing randomized trials for evaluating the clinical impact of OCT-guided versus angiography-guided PCI. ILUMIEN IV is a single-blinded, global, multicenter, prospective randomized trial comparing OCT-defined EEL-based PCI guidance to angiography-guided PCI in complex coronary lesions. Two-year clinical outcomes of OCT-guided revascularization versus angiography-guided revascularization in patients with complex bifurcation stent implantation will be compared in the OCTOBER trial.
Recent Advances in Optical Coherence Tomography
In patients with renal disease, the usage of contrast for OCT image acquisition may result in the worsening of renal disease, which is undesirable. Various alternative flushing agents have been investigated. 27 The image quality generated by low-molecular-weight dextran is comparable with radiocontrast28, 29; however, as the mode of excretion of dextran is from the kidneys, it can result in renal injury similar to contrast. 29 Carbon dioxide routinely used for peripheral angiography 30 is not appropriate for coronary imaging as it can precipitate myocardial ischemia. Previously in occlusive OCT acquisition techniques, clear crystalloid solutions such as Ringer’s solution were used, but the recent use of normal saline flush has produced better quality images on par with contrast OCT acquisitions, though problems with blood mixing and risk of arrhythmias with non-contrast flushes exist. There is a need for alternative non-contrast flush media with similar biocompatibility, viscosity, blood clearance capability, and optical transparency which are being investigated.
New OCT systems are being developed in order to acquire OCT images more rapidly (e.g., full sampling both cross-sectionally and longitudinally, within one cardiac cycle), 31 enhanced spatial resolution (e.g., “micro-OCT” with ~2 µm isotropic resolution, helps in the identification of cellular [endothelial cells, macrophages] and even sub-cellular [extracellular cholesterol crystals] structures 32 ), and depth of penetration. Further studies are required to validate these new advances and their impact on clinical outcomes.
A recent trial 33 compared fractional flow reserve guidance versus OCT guidance in patients having intermediate coronary lesions on angiography in a single-center, prospective, 1:1 randomized trial. In 350 patients, the primary endpoint of MACE or significant angina occurred in 14.8% of patients in the physiology arm and 8.0% in the OCT arm (p = 0.048) at 13 months. 33 Thus, the combined benefit of both modalities can be obtained by OCT-based flow ratios. To determine the functional significance of coronary stenosis, the optical flow ratio was developed and validated against fractional flow reserve. 34 FUSION trial (NCT04356027) is a single-arm, prospective, multi-center study designed to evaluate the accuracy of offline, OCT-based virtual flow reserve against fractional flow reserve using ILUMIENۛ OPTISۛ platform (Abbott Vascular).
Conclusion
For treating complex coronary lesions such as bifurcation lesions, OCT is a critical tool in the interventional cardiologist’s arsenal. Compared with angiography and IVUS, OCT provides valuable additional information that helps the operator at each step of the PCI―from planning the most appropriate revascularization strategy to assessing final results. Ongoing randomized trials on the impact of OCT guidance will provide robust evidence in improving clinical outcomes of PCI, and this could increase the use of OCT as the imaging modality of choice in clinical practice.
Footnotes
Declaration of Conflict of Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
