Abstract
For more than six decades, ischemic heart disease has sustained as the world’s leading cause of death. Growing evidence suggests chronic inflammation as the underlying pathophysiology of this illness. Due to this, inflammatory markers have become a mainstay of diagnosing, prognosticating, and following up the effectiveness of therapy. Colchicine remains a contentious treatment option for coronary heart disease, despite mounting evidence to the contrary from recent studies. This article discusses the basics of inflammation, the role of colchicine, various clinical studies, and the latest guidelines on colchicine. Colchicine has the potential to develop into a cheap, novel, and effective therapy for coronary artery disease.
Overview of Inflammation as the Precursor of Atheroma
For more than a century, atheroma was thought to be a simple, passive accumulation of lipids in the intima of an artery. 1 High-dose statins are responsible for reducing coronary artery disease (CAD) risk by 30%. The residual untreated risk (70%) for CAD includes residual inflammatory risk (RIR), residual cholesterol risk (RCR), residual thrombotic risk (RTR), and residual triglyceride risk (RTgR). A further RCR of 13% can be reduced using a newer drug such as PCSK-9 inhibitor to 9% only. 2 Even though the concept of inflammation as one of the major causes of atherosclerosis appears to be a new concept, but in the mid-nineteenth century, Rudolf Virchow was the first to recognize atherosclerosis as a chronic inflammatory process. 3 However, the concept gained popularity recently after the CANTOS study, which is a significant “proof of concept trial” confirming the definitive role of anti-interleukin-1 antibody (antibody directed against inflammation) in reducing major cardiovascular events. This was followed by the inclusion of inflammation as a cause of CAD in various European and American guidelines. Blood markers for this inflammation are interleukin-6 (IL-6), interleukin-1 (IL-1), interleukin-7 (IL-7), highly sensitive C-reactive protein (hs-CRP), matrix metalloproteinase (MMP), tumor necrosis factor-alpha (TNF-α), and tissue growth factor-beta (TGF-β). Among all the markers, hs-CRP is a commonly used marker. The hs-CRP marker prognosticates cardiovascular disease (CVD) outcomes. It increases the recruitment of inflammatory cells like phagocytes/monocytes into atheroma plaques leading to inflammation. The hs-CRP has opsonizing properties that help to attract inflammatory cells to atheroma. The third most important function of hs-CRP inhibits endothelial nitric oxide production leading to endothelial dysfunction. 4
Large Scale Evidence of Inflammation in Atheroma
The subintimal tissue has smooth muscle cells (SMC). SMC’s function is to secrete an extracellular matrix that traps lipids from the bloodstream. This trapped lipid gets transformed into oxidized LDL-C and subsequently into reactive oxygen species (ROS). ROS is known to activate endothelium leading to the expression of integrins and selectins. This receptor expression leads to the transmigration of inflammatory cells which consume these ROS and are also getting activated leading to the release of IL-2, Il-1β, and IL-6 factors. These inflammasomes stimulate the liver leading to an increase in hs-CRP. This in turn increases the transmigration of inflammatory cells. These inflammatory cells consume the lipids stuck in the subintimal matrix and form foam cells. These foam cells and accumulation of lipid form plaque and atheroma subsequently 5 (Figure 1).
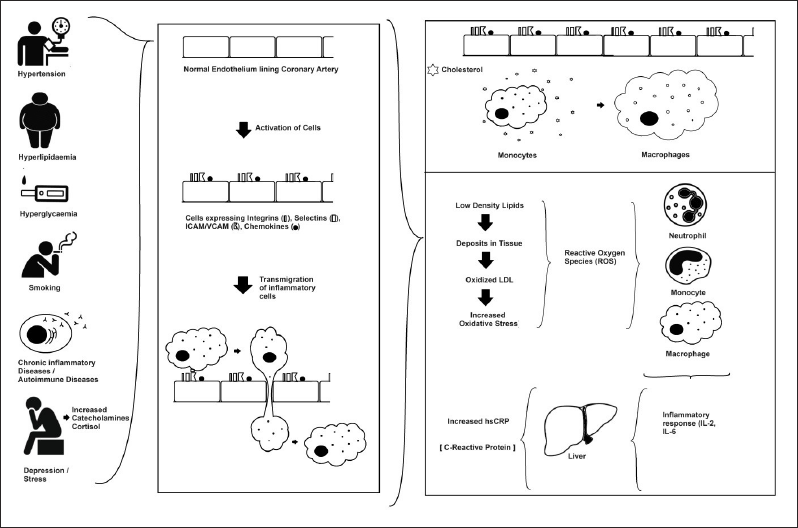
Multiple large trials (like the JUPITER trial) revealed that statins reduced both serum hs-CRP and low-density lipoprotein cholesterol (LDL-C). These trials also showed an overall reduction in CVD risk. The reason for this risk reduction is hard to determine whether there was a reduction in inflammation and LDL-C or LDL-C alone which led to better outcomes, 6 whereas another trial, the CANTOS trial, provided a definitive proof of concept study where inflammation (selective inhibition of interleukin-1β) reduction led to a reduction in hs-CRP and subsequent decrease in cardiovascular risk. 7 This proved the role of inflammation in atheroma formation.
History of Colchicine and its Uses
The “colchicine-like derivative” has been known since ancient times, especially in Indian and Egyptian civilizations. Egyptian records like “Ebers Papyrus” written in 1500 BCE describe the properties and uses of “colchicine-like derivative”. It is also mentioned in Ayurveda as “Suranjan” or “Hiranya Tutha”. 8 It is extracted from onion-like tubers of the Colchicum autumnale (Autumn crocus) and Colchicum luteum plants. It is mostly used to treat joint pains and joint swellings. It was derived in purified form in the early 1800s and since then has been used to treat Behcet’s disease, acute gout, rheumatism, familial Mediterranean fever, and inflammatory pericarditis. 9
In the last century, the mechanism of action of colchicine has been unraveled in detail. The main crux of the colchicine mechanism includes the inhibition of polymerization of cytoskeletal structures called microtubules (MTs). This unraveling of the colchicine action has helped us to explore its use in various other medical conditions. The use of colchicine in cardiovascular conditions is gaining traction. Some conditions where colchicine can be used include CAD, postoperative (cardiac surgery) atrial fibrillation, and heart failure with preserved ejection fraction. 10 Low-dose colchicine is commonly used in such conditions where colchicine is consumed in smaller doses like 0.5−1.0 mg/daily, which in turn reduces adverse effects of the drug and still retains the therapeutic effects. Another indication where colchicine has been used successfully is viral myocarditis (like coxsackievirus 3). Here, it has been shown to improve left ventricular function 11 (Figures 2 and 3).
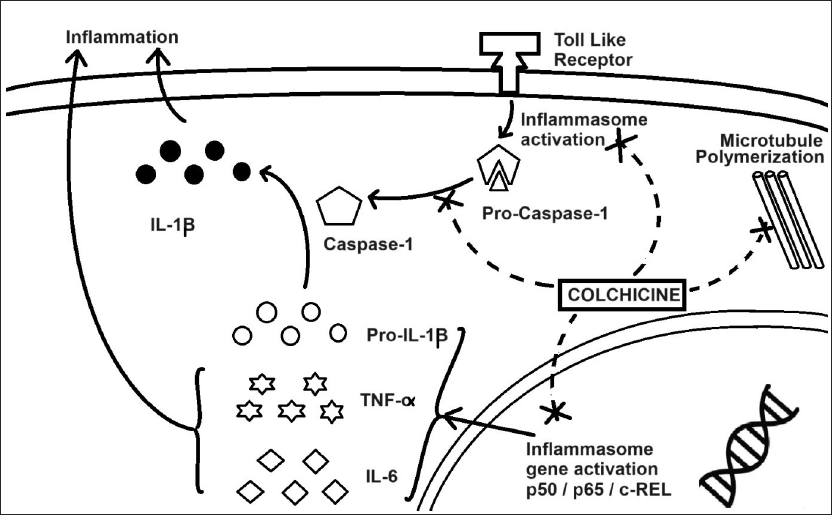
The Pharmacological Mechanism of Colchicine.
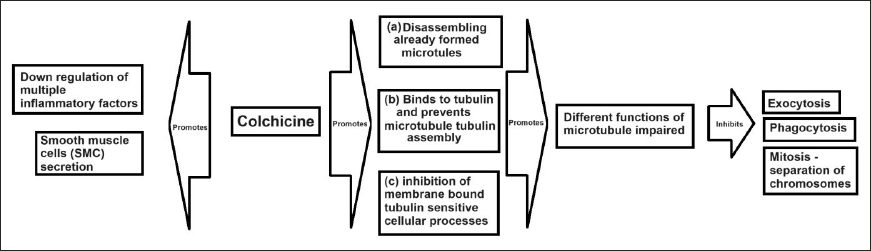
Colchicine Mechanism of Action
Colchicine’s anti-inflammatory mechanism includes impairing cytosolic microtubule function, inhibiting the production of inflammatory factors, and inhibiting SMC secretion of matrix. This impairment of microtubule function involves failure to polymerize microtubule. Furthermore, failure of microtubule polymerization (a) includes structural undoing of polymerized MTs, (b) prevents microtubule assembly formation by inhibiting polymerization between MTs, and (c) also inhibits cellular protrusions which are microtubulin sensitive. This disruption of microtubule polymerization leads to the failure of cellular exocytosis, phagocytosis, and cellular mitosis in monocytes, macrocytes, or SMC. 12 In short, disruption of microtubule polymers leads to cell cycle arrest or even cell death. This restricts inflammatory cell aggregation in atheroma and thus reduces inflammation (Figures 2 and 3).
Colchicine is known to reduce the transmigration of inflammatory cells, inhibits matrix secretion by SMC, and reduces ROS and activation of inflammatory cells.
Pharmacology of Colchicine
Chemically alkaloid, colchicine when taken orally, has a bioavailability of 24%−88% and a half-life of 20−40 h. It is absorbed from the jejunum and ileum with a bioavailability of 45%−50%. It is excreted via the kidney, intestine, and bile. It reaches peak concentration in 1 h and exerts a peak effect in 24−48 h. The intraleukocytic concentrations are more important than plasma concentrations; thus, dose for a desired response might differ between patients. Colchicine once in the cell binds to tubulin and gets activated. It also binds to the other two proteins such as cytochrome P3A4 (CYP3A4) and p-glycoprotein, which helps in metabolizing the colchicine. Intra-cytosolic colchicine concentration peaks at 48 h and chemokine inhibition happens after 6−24 h after the first dose. 12 Colchicine reduces hs-CRP (30%−40%) and IL-6 (16%) by inhibiting chemokines and neutrophil degranulation in chronic CVD. 13
In patients with renal dysfunction or hepatic insufficiency, colchicine in therapeutic doses also causes organ damage in neurons, muscles, and bone marrow. Colchicine is regarded as a medication with a narrow therapeutic index. This is secondary to the blocking of autophagic vacuoles with lysosomes by the colchicine-tubulin assembly. Colchicine is converted to 2- and 3-desmethyl colchicine by the enzyme CYP3A4, which is found in the intestine and hepatocyte cells. An ATPase efflux pump found on intestinal cells, like P-glycoprotein, may interfere with the absorption of colchicine from the intestine. P-glycoprotein is also found in hepatic cells, nephrons, and blood-brain barrier cells thus interfering in the metabolism of other drugs. Therefore, the risk of drug-to-drug interaction depends on the presence of hepatic dysfunction, renal dysfunction, ingestion of CYP3A4-affecting drugs, or colchicine dose consumed by the patient. Colchicine metabolism disorders result from the concomitant use of colchicine with either P-glycoprotein inhibitors (such as amiodarone, carvedilol, cyclosporine, digoxin, diltiazem, dronedarone, felodipine, itraconazole, lidocaine, ranolazine, quinidine, and ticagrelor) or CYP3A4 inhibitors (such as atazanavir, clarithromycin, indinavir, itraconazole, nelfinavir, fluoxetine, grape juice, ketoconazole, saquinavir, nefazodone, and ritonavir). These P-glycoprotein inhibitors and CYP3A4 inhibitors cause reduced metabolism of colchicine thus increasing its blood concentration and ultimately resulting in drug toxicity. Colchicine is also susceptible to drug-drug interactions because of how cytochrome P450 3A4 and p-glycoprotein metabolize it. Monitoring for negative consequences is therefore of utmost significance. Given this, a highly customized strategy is necessary, restricting the use of colchicine to individuals who continue to be at extremely high risk, despite receiving the maximal tolerable GDMT until more data are available.
Evidence of Colchicine in Acute Coronary Syndrome
Some of the initial evidence for the role of colchicine came from multiple retrospective studies on patients who were administered colchicine, suffering from gout. The studies did show a reduction in the incidence of myocardial infarction (MI), stroke, transient ischemic attack (TIA), and all-cause mortality in such patients. Later, two major randomized trials in 2019 and 2020, with 5000 plus subjects with the acute coronary syndrome (ACS), were started on colchicine for 1-year duration and followed up for a period of 24 months from the day of the ACS. The COLCOT trials 13 and COPS trials 14 had a majority (80%) of male patients compared to females (20%). Diabetics were 20% and smokers were 30.6%.
In the meta-analysis 15 by Nogic et al., the colchicine cohort has shown significant improvement in the incidence of major adverse cardiovascular events (MACE), cerebrovascular accidents, and urgent revascularizations, with no significant difference in cardiovascular death, resuscitated cardiac arrest, or MIs. Another interesting aspect of this meta-analysis is the increase in all-cause mortality among the colchicine cohort, solely contributed by COPS trial data which showed a significant increase in all-cause mortality. This was found to be secondary to Type-I error in data. Similarly, the meta-analysis showed an overall reduction in the occurrence of MACE mainly contributed by cerebrovascular events and urgent revascularization events. Thus, colchicine does not affect the overall mortality, MI rates, cardiac arrest rates, or cardiovascular death rates (Figure 4).
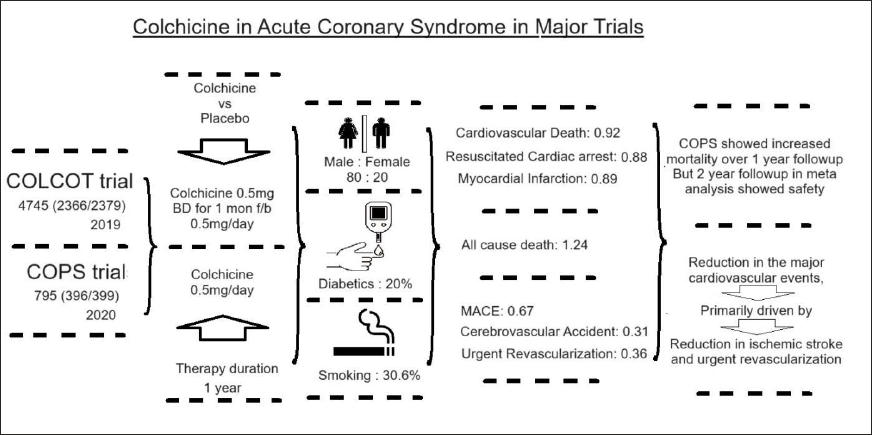
Coming to the adverse reactions, there were no major adverse effects with regard to low-dose colchicine therapy. Even gastric effects (nausea and diarrhea) were like that of the placebo group with no significant rise. Numerically, there was a mild increase in pneumonia cases in the colchicine group (0.9% vs 0.4%).
Recently, a pilot study published in JACC, called “MACT Pilot study.” This proof-of-concept study included all 200 ACS (STEMI/NSTEMI) patients requiring percutaneous intervention (PCI). Here, a drug-eluting stents (DES) were deployed to all patients during PCI. These post-PCI patients were started on low-dose colchicine (0.6 mg daily) with single anti-platelet therapy (ticagrelor/prasugrel). Only a single loading dose of Aspirin was given on the day of PCI. At 3 months, only 1% of the patients had stent thrombosis, and only 1 had significant platelet reactivity. Furthermore, at 1 month, both high-sensitivity C-reactive protein (hs-CRP) and platelet reactivity decreased, indicating that inflammation had diminished. 16
Evidence of Colchicine in Chronic Atherosclerosis of Coronaries
Chronic atherosclerosis leads to chronic coronary syndrome (CCS). CCS is a dynamic process of atherosclerotic plaque accumulation causing reversible inadequate blood flow. Guideline-directed treatment for such patients includes statins and antiplatelet with antianginal drugs. In spite of optimum dosing of these medicines, there is always residual inflammation risk (RIR) which persists. Many studies done earlier were retrospective in nature, and in one such study with 1002 patients suffering from gout, out of them, 501 were on colchicine. Patients were followed up for a median duration of 16.5 months. The results showed higher cardiovascular events (16.4%) among the non-colchicine group when compared to the colchicine group (5.6%) [hazard ratio was 0.51, p = .016]. Similarly, there was a 73% drop in all-cause mortality [HR 0.55, p: .007]. 17 Another retrospective study with 1288 patients showed lower MI rates with the colchicine (1.2%) vs non-colchicine group (2.6%) with a relative risk of 0.46 and p: .03. 18
Colchicine in CCS RCT data came from LoDoCo and LoDoCo-2 RCT trials. The LoCodo-1 trial revealed a reduction in plaque instability and in turn improved clinical outcomes. Primary outcomes (cardiovascular death, spontaneous MI, ischemic stroke, or ischemia-driven coronary revascularization) were seen in 15 patients out of 282 (5.3%) in the colchicine group and 40 patients out of 501 (16%) among the non-colchicine group with HR of 0.33 and p: .001. Similarly, primary outcomes in the LoDoCo2 study happened in 187 patients (5.3%) in the colchicine group and 264 (9.6%) in the non-colchicine group with HR of 0.69, p < .001. Evidently, the colchicine group suffered from a higher risk of non-cardiovascular death when compared to the placebo group in both major trials. Both the cohorts in the LoDoCo2 study were already on aggressive statin therapy. Still the colchicine group showed 25% to 30% relative risk reduction in MACE events. With both above trials, colchicine with aggressive statin therapy has proven itself to be an important add-on therapy in secondary prevention of CCS, and both LoDoCo and LoDoCo2 contain a sizable number of patients. Oral colchicine thus reduces inflammatory factors and stabilizes plaque.19⌓21
Evidence of Colchicine in Post-revascularization Patients
In the initial days, the in-stent stenosis in post-PCI patients was the limited cohort where colchicine was considered to reduce in-stent stenosis. But now, the newer group of patients have high RIR, intra-procedural events (like wire-related injuries, high-pressure balloon dilatation-related microdissections, and vascular trauma), and systemic inflammation. These groups seem to aggravate plaque instability and inflammation. The first study on post-PCI colchicine in 1992 had 197 patients who failed to reduce in-stent stenosis rates. 22 In 2013, a prospective RCT with 196 patients who were administered 0.5 mg colchicine showed a reduction in in-stent stenosis of 16% when compared to the non-colchicine group with 33% (OR: 0.38; p 0.007). 23 Another RCT, COLCHICINE-PCI trial with 400 patients revealed no significant reduction in a primary outcome (mortality, non-fatal MI, and target vessel revascularization at 30 days) by 11.7% vs 12.9% in the non-colchicine group. Similarly, PCI-related MI rates also did not reduce significantly. 24
In a study “Colchicine in CABG” by Giannopoulos et al., out of 59 patients, 30 patients who were administered colchicine showed a significant reduction in hs-Troponin-T and creatinine kinase-MB when compared to the placebo group. This could be secondary to the anti-inflammatory action of colchicine. There are many more RCTs in a pipe which can give answers to many of our queries with regard to colchicine. 25
Is Colchicine the New “Aspirin”?
With high-dose statin and DAPT therapy reaching their peak in dealing with cardiovascular risk, now the whole attention is focused on the various residual risks like RIR, RCR, residual thrombotic risk (RTR), and residual triglyceride risk (RTgR). The RCR was to some extent reduced with the help of newer drugs like Bempedoic acid, Ezetimibe and Evolucumab. The RIR was something which was unaddressed. The CANTOS trial using PCSK9 inhibitor gave us “proof of concept” for newer molecules that can control inflammation and in turn reduce cardiovascular risk. Colchicine is used in acute gout, rheumatism, Behcet’s disease, familial Mediterranean fever, and inflammatory pericarditis. Now, it is being explored in the role of reducing cardiovascular risk. As mentioned above, multiple trials have proven the efficacy of the drug in cardiovascular risk reduction. A meta-analysis of LoDoCo1, LoDoCo2, COPS, COLCOT, and Deftereo et al. trials has shown an overall reduction in MACE using low-dose colchicine. 26 Detailed analysis into specific groups showed that low-dose colchicine had consistent results in CCS. Similarly, colchicine in ACS had the majority of the RCTs in favor of use with a significant reduction in risk in the colchicine group. However, colchicine in PCI and CABG is inconclusive and needs further larger trials to comment. The CCS is a comparatively low-risk condition, and the optimization of the therapy can be achieved with great accuracy. However, comparatively, ACS and post-PCI are at higher risk of MACE. The patients going for revascularization depend on multiple confounding factors like the complexity of the coronary artery lesions, the primary operator experience, the technical complexity of the PCI, and the timeliness of the medical therapy given, which play an important role in deciding the outcome. Colchicine in ACS trials has shown some positive outcomes and some insignificant studies. Another aspect of this discrepancy is the usage of inflammatory markers during the RCT trials. No single inflammatory marker (hs-CRP, IL-1b, IL-18, IL-6, and WBC) is considered as an independent, diagnostic marker for MACE. Although hs-CRP is the most widely used marker in the majority of the trials, the accuracy of the sensitivity and specificity of the marker is controversial. Finally, colchicine has been studied in the pathophysiologically similar conditions like cerebrovascular or peripheral vascular disease. There are some studies that have shown benefits in cerebrovascular conditions. 27 Similarly, the role of colchicine in peripheral artery disease is another field of interest, and presently, an RCT trial named LEADER-PAD is undergoing and is expected to be published by 2024.
The author considers colchicine as the next big thing in the cardiovascular field. With many ongoing RCTs using colchicine being used during ACS, CCS or peri-procedural PCI/CABG usage may answer many of our queries. One of the most important data from the “MACT pilot study” showed that aspirin can be completely excluded from the therapy after ACS after giving a single loading dose during PCI. This should be followed by the continuation of ticagrelor/prasugrel with colchicine after PCI for ACS. Therefore, after several ongoing trials are finished, colchicine may be added in the recommendations. Colchicine will become one of the important add-on therapies in selected CAD patients. But, as of now, according to the “2023 AHA/ACC/ACCP/ASPC/NLA/PCNA Guideline for the Management of Patients with Chronic Coronary Sisease,” colchicine can be used (class 2b) in secondary prevention in recurrent ASCVD events. 28
Conclusion
The death and morbidity rates for CAD are high. Cholesterol deposition, inflammation, and thrombotic risk are only a few of the pathophysiologic factors that contribute to atheroma development. It is absolutely necessary to develop new strategies to address inflammation in atheromatous lesions. There is recognition of the role played by in the development of atherosclerosis. The two inflammatory markers that are most frequently employed as indications of inflammation are hs-CRP and IL-6. Anti-inflammatory medications, such as low-dosage colchicine, may become the preferred treatment for atherosclerosis inflammation. Colchicine costs little and is generally safe, with rates of gastrointestinal side effects equivalent to those of a placebo. Colchicine when used in CAD reduces the incidence of MACE. Along with aggressive statin therapy, anti-inflammatory agents have a significant role in managing ASCVD risk.
Take Home Message
Anti-inflammatory medications will be added to the existing dual anti-platelet therapy and statins (optimized medical therapy for CAD).
That means a patient who is on guideline-directed optimum medical therapy would still do much better when added to anti-inflammatory therapy.
Anti-inflammatory medication is not a replacement or change to established medical therapy with diet control, exercise, and smoking cessation.
Inflammation forms a large untapped/untreated portion of atherosclerosis.
Colchicine is one of the few anti-inflammatory drugs which are successful in managing atherosclerosis.
In all patients using statins, RIR predicts the future risk of MACE better than RIC.
LoDoCo2 study showed 25%−30% relative risk reduction in MACE events in the colchicine group when compared to the non-colchicine group on top of aggressive statin therapy.
No significant rise in adverse effects was noted, like gastric intestinal side effects and pneumonia.
Colchicine has a narrow therapeutic index and might have detrimental or toxic consequences.
Colchicine is also susceptible to drug-drug interactions because of how cytochrome P450 3A4 and p-glycoprotein metabolize it. Monitoring for negative consequences is therefore of utmost significance.
Given this, a highly customized strategy is necessary, restricting the use of colchicine to individuals who continue to be at extremely high risk, despite receiving the maximal tolerable GDMT until more data are available.
COLCOT (Colchicine Cardiovascular Outcomes Trial) resulted in an increase in QALYs from 1.30 to 1.34 and a 47% decrease in the mean total per-patient expenditures.
2023 AHA/ACC/ACCP/ASPC/NLA/PCNA Guideline for the Management of Patients with Chronic Coronary Disease gives class IIb evidence for the use of colchicine in chronic coronary disease.
Scope of Further Research
Colchicine in atherosclerosis (ACS/CCS) needs the long-term follow-up. The effect of colchicine on the overall immunity, the role of colchicine in patients undergoing CABG, and the effect of colchicine on other co-morbidities like diabetes, hypertension, and dyslipidemia should be further evaluated.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not necessary because this article discusses several previous studies on the subject. The author alone is responsible for the analyses and viewpoints presented in the piece.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Patient Consent
Not necessary because this is not a study and does not need for patient’s permission.
