Abstract
An agonal rhythm is a variant of idioventricular rhythm often preceding asystole. It reflects significant myocardial or metabolic derangement and has a grave prognosis. Ebstein’s anomaly accounts for 0.3–0.7% of all cases of congenital heart diseases. The electrocardiogram in Ebstein’s is seldom normal across the spectrum and plays a major role in establishing diagnosis. This case report describes an electrocardiogram with extreme low voltage and bizarre complexes mimicking agonal rhythm in a patient having Ebstein’s anomaly, presenting with severe biventricular failure.
Introduction
An agonal rhythm is a very slow ventricular rhythm often recognized as the terminal rhythm preceding asystole. It is a variant of idioventricular rhythm, indicating very poor prognosis. 1
Ebstein’s anomaly is a rare congenital heart disease occurring in ≈1 per 2,00,000 live births. 2 It is characterized by downward (apical) displacement of either the septal, posterior, or rarely anterior leaflet of the tricuspid valve and resultant dilatation of the atrialized portion of the right ventricle. Electrocardiogram (ECG) plays a pivotal role in establishing diagnosis, and various electrophysiologic abnormalities have been associated with the disorder.
Case Report
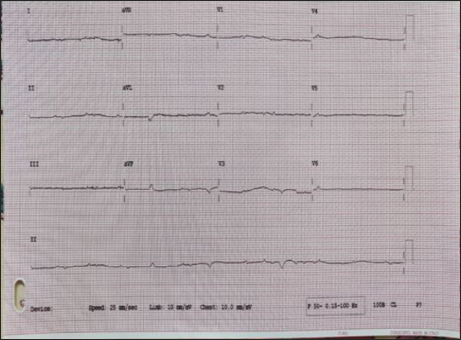
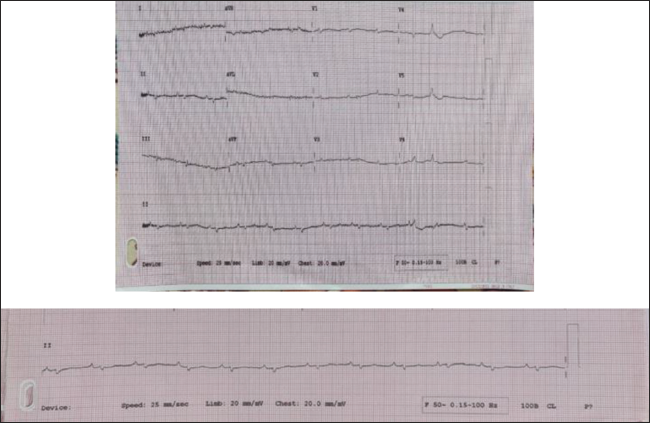
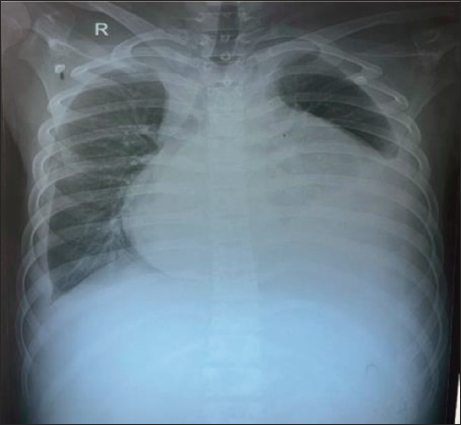
A 16-year-old adolescent boy born out of a non-consanguineous marriage presented to the emergency with progressive dyspnea of 10 months’ duration. The birth, perinatal, and developmental history were normal. He was conscious, oriented, with low volume pulse of 90 bpm, blood pressure recording of 100/60 mmHg, and saturation at room air of 90%. A 12-lead ECG with normal standardization revealed low voltage and bizarre complexes with ventricular premature complexes mimicking agonal rhythm (Figure 1).ECG with double standardization (20 mm/mV) along with right sided and posterior leads were obtained, which revealed regular rhythm with intrinsic complexes, P wave followed by QRS with P wave amplitude more than QRS due to atrialized ventricle (Figure 2). Biochemical profile was done to rule out metabolic and electrolyte disturbances. Chest x-ray revealed massive cardiomegaly (box-shaped heart)—typical of Ebstein’s anomaly (Figure 3).
12-Lead ECG of Normal Standardization Showing Bizarre Complexes Mimicking Agonal Rhythm.
ECG of Double Standardization Showing Regular Intrinsic Complex.
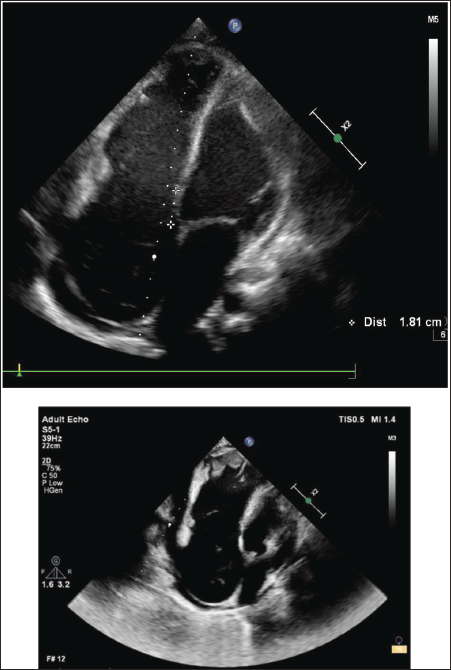
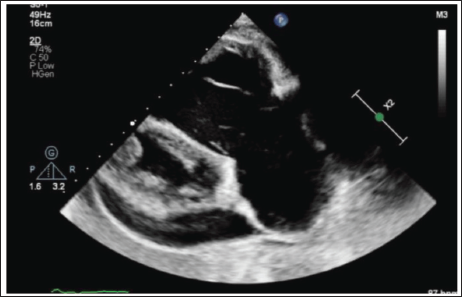
The echocardiography showed an 18 mm apical displacement of the septal leaflet of the tricuspid valve and a tethered dysplastic septal leaflet (Figures 4 and 5). It also showed dysplastic anterior and posterior tricuspid leaflets (Figure 6), severe free tricuspid regurgitation, dilated right atrium and right ventricle, severe biventricular dysfunction (Figure 7) with a small ostium secundum atrial septal defect from the right to left shunt, and mild pericardial effusion. Multiple clots were observed in both ventricles and right atrium.
Chest X-Ray Suggestive of Massive Cardiomegaly and Box-Shaped Heart.
Apical Four-Chamber View of Echocardiogram Showing Tethered Septal Leaflet and Apical Displacement of 18 mm Suggestive of Ebstein’s Anomaly. Note the Dysplastic Septal and Anterior Tricuspid Leaflet.
RV Inflow View Showing Dysplastic Anterior Tricuspid Leaflet and Dysplastic Posterior Tricuspid Leaflet.
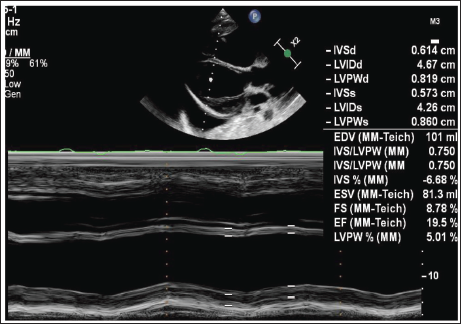
M-Mode Showing Severe Left Ventricular Dysfunction.
Serial ECG monitoring did not show improvement in ECG even after optimal decongestion therapy. The patient’s overall clinical condition improved in the hospital course, and he was discharged in a hemodynamically stable state.
Discussion
Ebstein’s anomaly represents about 40% of congenital malformation of the tricuspid valve. 3 The left heart is involved in around 39% of Ebstein’s anomaly due to altered left ventricular geometry, impairment in systolic and diastolic function, and noncompaction. Myocardial fibrosis in the ventricular septum is one of the proposed causes for left ventricular dysfunction. 4 Low QRS voltage in severe decompensated heart failure is a marker of adverse clinical outcomes. 5
Low-voltage complexes are commonly seen in massive pericardial and pleural effusions, obesity, hypothyroidism, and anasarca. Previous studies show that low-voltage QRS complexes are observed in Ebstein’s anomaly with massive pericardial effusion, 6 and the high amplitude of P wave presages limited survival. 7 Although the patient had signs of congestion, mild pericardial and pleural effusion, it was not large enough to explain the low-voltage complexes. However, in our case extremely low-voltage complexes mimicked agonal rhythm on initial ECG; further double standardization revealed intrinsic complexes.
Electromechanical disturbances are prevalent in this anomaly, and electrocardiogram is a cornerstone for diagnosis. Downward displacement of the septal tricuspid leaflet is accompanied by discontinuity between the central fibrous body and the septal atrioventricular ring, which creates a substrate for accessory pathways. Abnormalities include PR interval prolongation, atrioventricular conduction delay, right bundle branch block, infrahisian conduction disturbances, bizarre QRS complexes, type B Wolff–Parkinson–White preexcitation, supraventricular arrhythmia, and atrial fibrillation.
Patients with acute decompensation due to heart failure who cannot undergo surgery are managed medically, including diuretics and digoxin. Radiofrequency ablation for accessory pathways have low success rates. Approximately 3–4% of the patients need permanent pacing for atrioventricular blocks.
Conclusion
Ebstein’s anomaly can cause bizarre complexes that cannot be picked up by regular ECG, as seen in this case mimicking agonal rhythm. Magnification aspects and technical aspects should be kept in mind in dealing with such cases.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

