Abstract
A pulmonary artery aneurysm is defined as focal dilation of the pulmonary artery beyond a diameter of 29 mm. The reported incidence is 1 in 14,000 as per the autopsy studies. A diameter of more than 55 mm is defined as a massive pulmonary artery aneurysm which is very rare. These are commonly associated with patent ductus arteriosus. No distinct features for identification and guidelines for their management warrant a detailed evaluation for etiology and close follow-up in this entity fearing fatal rupture. We present a case of a massive pulmonary artery aneurysm associated with the ventricular septal defect.
Keywords
Introduction
A pulmonary artery aneurysm (PAA) is a rare cardiac condition. In a study at Mayo Clinic by Deterling and Clagett, only 8 cases of PAA were identified out of 1,09,571 consecutive postmortem examinations. 1 When compared to aortic aneurysms, PAAs occur in younger age groups with an equal sex incidence. 2 Most of them (almost 90%) involve the main PA and the remaining involve the branches (left most common than the right pulmonary artery (RPA)). 3 There has been no standard cut-off to define PAA. The main pulmonary artery (MPA) mean diameter in normal healthy adults is considered to be 25 ± 3 mm, with 29 mm as an upper limit of normal in males and 27 mm in females. 4 As per the literature available, PAA is defined as dilatation with a diameter >1.5 times the upper limit of normal (43 mm in males and 40 mm in females). 5 Half of them occur in patients with congenital heart disease, most commonly patent ductus arteriosus (PDA). 6 A dilation of more than 50 mm (massive PAA) is exceedingly rare. 7
Case
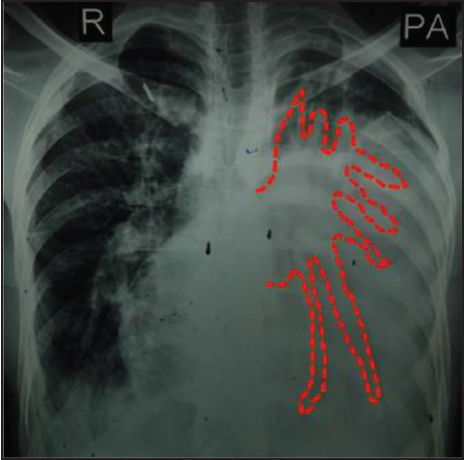
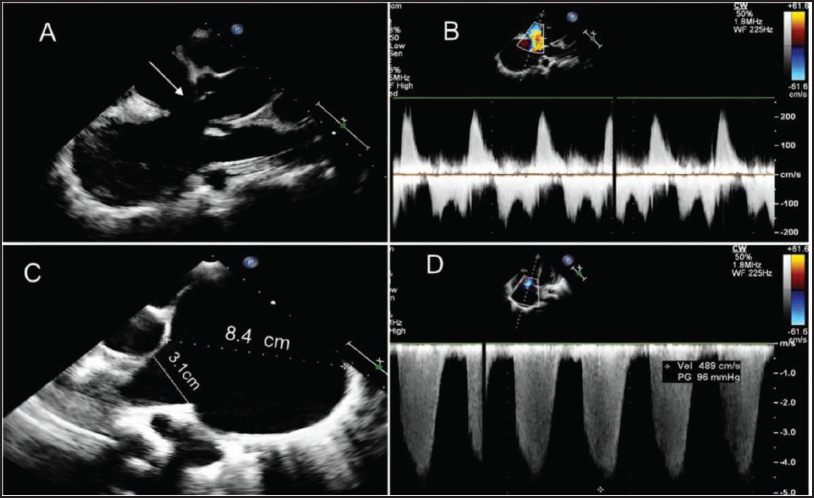
A 25-year male presented to the emergency with progressive dyspnea from NYHA II to IV over two weeks, associated with swelling of legs, abdominal distension, and a decreased appetite. During the illness or prior to the onset of symptoms, there was no history of weight loss, fever, rash, joint pains, cough, hemoptysis, skin, and oral ulcers. There was no history of any surgeries in the past. A chest x-ray was advised and the presence of left massive pleural effusion was noted (Figure 1). A cardiology opinion was taken because of a history of probable congenital heart disease in the past. On physical examination, there was central cyanosis (SpO2 of 78%), grade III pandigital clubbing, bilateral pitting pedal edema, and raised jugular venous pulse. The patient was tachycardic and all peripheral pulses were felt. Their blood pressure was 110/70 mmHg. There was no evidence of marfanoid features, hyperextension of joints, spine abnormalities, or oral and mucosal ulcers. Cardiac auscultation revealed a grade 4/6 pan systolic murmur in the left fifth intercostal space in the parasternal area, a grade 3/6 mid-systolic murmur in the left second intercostal space, and a grade 3/4 early diastolic murmur in left second intercostal space. A provisional diagnosis of ventricular septal defect (VSD) with bidirectional shunt and failure was made. On further evaluation, the electrocardiogram showed a right ventricular strain pattern. The chest X-ray was reexamined which showed broncho vascular markings posterior to cardiac silhouette, favoring cardiomegaly and a negative finding for pleural effusion (dotted line in Figure 1- indicate the bronchovascular markings). The two-dimensional transthoracic echocardiography revealed a large subaortic VSD with a bidirectional shunt. The MPA measured 84 mm in diameter and the right PA was 31 mm on echocardiography along with dilated right atrium, and right ventricle (Figure 2). There was right ventricular dysfunction. There was no pulmonary stenosis or vegetation. The patient was managed symptomatically and improved on medications.
Chest X-Ray PA View Showing Cardiomegaly. Broncho Vascular Markings on Posterior Aspect are Visible (marked by the red color dotted lines over the presumed effusion).
2D Echocardiography. (A) Parasternal Long Axis View Showing Sub Aortic VSD (White Arrow). (B) Doppler Interrogation Across VSD Showing Bidirectional Shunt. (C) Parasternal Short Axis View Showing Dilated MPA (8.4 cm) and RPA (3.1 cm). (D) Tricuspid Jet Velocity of 4.9 m/sec.
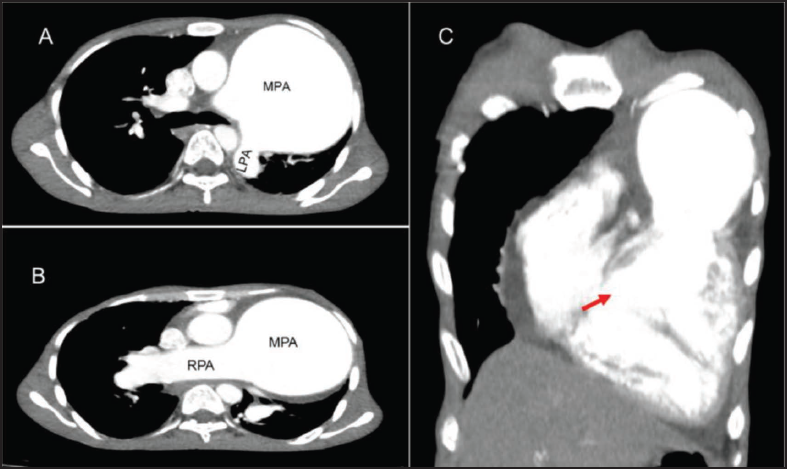
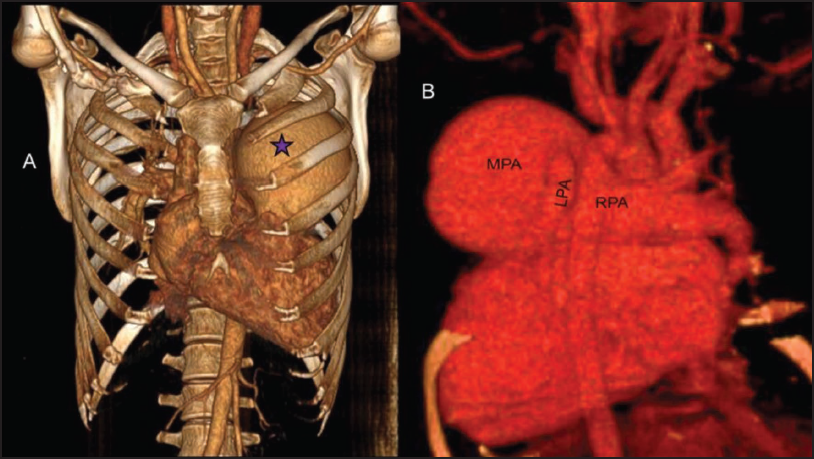
Cardiac computed tomography and pulmonary angiography (CTPA) were done. The echocardiographic findings were confirmed. However, the dimensions were more. The MPA diameter was 92 mm and the RPA diameter was 33 mm (Figures 3 and 4). There was no vegetation. This aneurysm caused posterior compression of the left bronchus. Lung parenchyma was normal.
Cardiac CT. (A) Sagittal Section Showing Dilated MPA and Normal LPA. (B) Sagittal Section Showing Dilated MPA and RPA. (C) Coronal Section Showing Sub Aortic VSD (Red Arrow).
3D Constructed Cardiac CT and CTPA. (A) Dilated MPA (Purple star). (B) Posterior Aspect of Heart Showing Dilated MPA and RPA, Normal Sized LPA.
The biochemical profile revealed deranged renal and liver function. The hemoglobin was 18.5 mg/dl and the other hemogram was normal (Total leucocyte count, erythrocyte sedimentation rate [ESR]). Blood culture, ANA profile, C-reactive protein (CRP), and procalcitonin, were negative. The patient was managed with intravenous furosemide, intravenous dobutamine infusion, oral spironolactone, oral sildenafil, and intravenous unfractionated heparin (as prophylaxis for thromboembolism). His total hospital stay was one week. His lab parameters improved and was hemodynamically stable and discharged.
The patient was planned for surgical management. He denied surgery, however, is under close follow-up. The diameter of the PAA did not increase during the 12 months of follow-up.
Discussion
PAA is a rarity among the diseases of the pulmonary vasculature. In a study done by Deterling et al., there were only 8 cases of 109,571 autopsies. 1 When translated into the incidence it amounts to 0.073%. 8 In general, PAA can be defined as a diameter ≥40 mm. 6 The usual mechanism for the formation of a true aneurysm is a weak vessel wall and an increased hemodynamic shear stress. 9 The congenital heart diseases with increased flow across the shunt and thereby increased shear stress on the vessel wall will be leading to aneurysm formation. 10 In a study done by Gupta et al. on PAA in Eisenmenger syndrome, the most common cause was PDA and the least was VSD and transposition of great arteries (TGA). 5 Massive PAA is defined as >50 mm, which is exceedingly rare.7, 11 It involves the MPA in 90% of cases, and in remaining the branches (the Left is more common than the right). 3 In our case, the patient had VSD and PAA with a diameter of 92 mm of the MPA. The RPA was involved along with MPA.
The usual presentation is not specific and is secondary to the compression of structures due to an aneurysm.5, 6 Our patient presented with symptoms of right heart failure.
Chest X-ray interpretation is important to differentiate between cardiomegaly and pleural effusion. Both posteroanterior and anteroposterior erect films are insensitive to small amounts of fluid. Features favoring the effusion include blunting of the costophrenic and cardiophrenic angle, fluid within the horizontal or oblique fissures, a lateral meniscus with gentle sloping medially will be seen with large effusion on the PA films, mediastinal shifts away from the effusion (may not occur when there is simultaneous collapse). 11 In our case, no such findings are seen, suggesting it is cardiomegaly.
A focused and complete echocardiogram is required to assess the presence of congenital heart defects, right ventricular outflow, pulmonary artery flow, and diameter. A cardiac computed tomography and pulmonary angiogram or cardiac magnetic resonance imaging is useful in diagnosing and evaluating PAAs.12–14
The natural history of PAA depends on the etiology and associated lesions. Patients with isolated PAA, have better prognosis, compared to those with associated cardiopulmonary disease including pulmonary hypertension (PH) and congenital heart disease. The presence of certain features is considered a red flag in PAA―PAA ≥75 mm, aneurysm diameter growth >2 mm/year, and mean PA pressures ≥50 mmHg; this group of patients has a poor prognosis. 15 The aneurysm can complicate by in situ thrombosis, rupture, dissection, and compression of adjacent structures―left main coronary artery, lung, and adjacent bronchi.7, 16 One-third of all PAAs reported may worsen due to rupture and die of massive hemoptysis, asphyxiation, exsanguination, and sudden death.7, 17
Treatment is surgical management in the presence of a shunt, or else conservative medical management.18, 19 Medical management includes calcium channel blockers, diuretics, anticoagulants, endothelin receptor antagonists, phosphodiesterase type 5 inhibitors, and prostacyclin derivatives. 17 A close monitoring and follow-up is warranted, to look for new or worsening symptoms, increase in size, thrombus in the PA, and for impending rupture or dissection. 15
Surgery is the cornerstone for treatment for PAA. Indications for surgery are described in Table 1. Aneurysmorraphy is a simple procedure, where only the size is reduced and it does not address the abnormal vessel wall. An increase in the shear stress over the wall after reduction may worsen the condition. Aneurysmectomy and repair or replacement of the right ventricular outflow tract is the method of choice. The MPA and the branches can be replaced with a conduit starting in the right ventricular outflow tract with Gore-Tex or Dacron tubes, homografts, or xenografts (porcine aortic grafts or bovine jugular conduits).13, 20
Indications for Surgery. 6

Intervention in PAA is an emerging line of treatment. Coil embolization may be a good treatment option for iatrogenic causes. 21 Complete occlusion of a dissected PAA by a covered stent was also reported. 22
In our patient, right heart catheterization for reversibility followed by surgical closure of VSD and repair of PAA and graft placement would be an ideal line of treatment but here with baseline saturation of 78%, clubbing, RV dysfunction chances of reversibility test to be positive are slim. Even if PAA is repaired with a graft, the persistence of the shunt will lead to failure of the graft and sudden cardiac death if rupture occurs. The patient is in close follow up and fortunately, there was no further increase in size.
Conclusion
PAAs are rare without distinct symptoms favoring their diagnosis. A careful physical examination will enable the etiology. There is a paucity of literature regarding management. A size of ≥55 mm requires a surgical reduction in fear of rupture. Conservative management needs a close follow-up. Percutaneous interventions in the management of this aneurysm are on the line of development.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical permission was not applicable, as this work was a case report, not a research study.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Patient Consent
Patient consent for the publication and sharing of radiologic investigation figures was obtained.
