Abstract
Introduction
Torsades de pointes (TdP), a form of polymorphic ventricular tachycardia that manifests in the setting of an acquired or congenital QT interval prolongation. Polymorphic VT can occur in hyper acute phase or healing phase of an acute myocardial infarction. The presence of necrotic area caused by the infarction explains the existence of a possible circuit as well as the appearance of a re-entry arrhythmia.
Aims and Objectives
Our aim is to study the incidence of long Qt induced TdP in acute MI patients and its prognostic implications. We also want to analyze the impact of primary percutaneous transluminal coronary angioplasty in the management of these subset of patients.
Methods
In this cross-sectional observational study done from May 2021 to May 2022, We have studied 320 cases of acute MI, of which 6 were found to have TdP (1.9%). Four patients had inferior wall myocardial infarction and 2 patients had Non-ST elevation MI. Thrombolysis was done in 2 cases. Two cases underwent primary angioplasty. One case was recommended for coronary artery bypass grafting. All cases underwent appropriate electrolyte correction, besides cardioversion, as needed.
Results
All patients with TdP had prolonged QT interval. 50% of patients had mild hypokalemia. Two patients had complete heart block. Three patients had severe left ventricular dysfunction. Patients who underwent primary angioplasty survived. Thrombolysis had less impact on survival in Acute Mi with TdP.
Conclusion
Early percutaneous coronary angioplasty, combined with electrolyte correction, improves the prognosis in TdP complicating acute MI.
Keywords
Introduction
Torsades de pointes (TdP) is a type of polymorphic ventricular tachycardia (PVT) that occurs when there is a prolongation of the QT interval, whether acquired or congenital. TdP is characterized by certain key features, including a preceding prolonged QT interval, an irregular ventricular rate ranging from 160 to 250 beats per minute with varying RR intervals, and a cyclic twisting pattern of the QRS axis every 5 to 20 beats.1, 2 The QRS complexes in TdP appear to twist around the isoelectric line, giving rise to the name “torsades de pointes” or “twisting of the points.” This type of ventricular flutter commonly occurs in the presence of advanced atrioventricular block and is often accompanied by syncopal episodes. TdP episodes are typically brief and tend to resolve on their own. However, many patients experience multiple episodes of this arrhythmia, which can recur rapidly and potentially progress to ventricular fibrillation, resulting in sudden cardiac death.
Materials and Methods
In our institute, we have studied 320 cases of acute myocardial infarction (MI) in one-year period (May 2021-May 2022). All cases of TdP (polymorphic VT) were analyzed in the study population. Acute MI (AMI) was diagnosed clinically and confirmed by ECG and biomarkers. QT interval is a measure of the interval between onset of QRS to the end of T wave. Corrected QTc was calculated using Bazett’s formula as well as Fridericia formula to increase the sensitivity. Torsades was diagnosed on ECG with its typical twisting pattern around the baseline. Echocardiography was done in all cases according to standard protocol. All cases were treated (thrombolysis/ angioplasty) according to their presentations. We have further analyzed these TdP cases with respect to the management issues and prognosis. Proper consent was obtained and ethics committee approval was taken.
Results
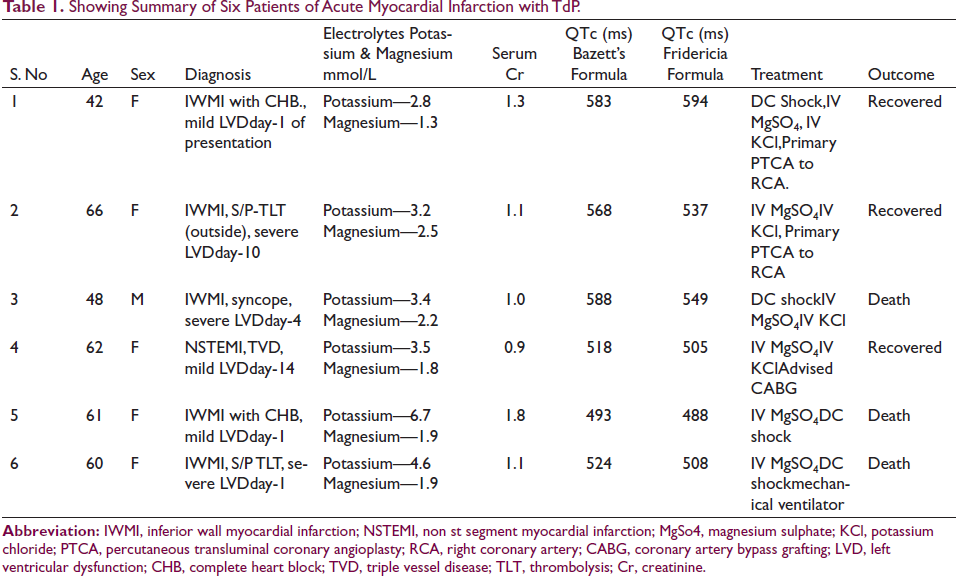
Out of 320 cases of AMI presenting to our hospital during the study period, 6 cases of TdP have been identified which accounts for 1.89% of all AMI cases. Except 2 non-ST Elevation MI (NSTEMI) cases all other cases of TdP were of inferior wall myocardial infarction (IWMI). No cases of anterior wall myocardial infarction were documented in our study population. All the patients of TdP had prolonged QT (100%), of which 5 patients had QTc more than 500 ms. Two cases presented with complete heart block (Figures 1 and 2). Out of 6 TdP cases, 3 cases had PVT (Figures 3 and 4) within 12 to 24 h of MI, whereas the other 3 cases presented during 4 to 14 days. Five among the cases of TdP were female (83.3%). Three cases of TdP had severe left ventricular dysfunction. Three cases had mild hypokalemia (50%) and one case had hyperkalemia (16.6%). One patient had hypomagnesemia (16.6%).
Showing ST-Elevation in Leads II, III, and Avf, Heart Rate of 32/Min and Complete AV Dissociation.
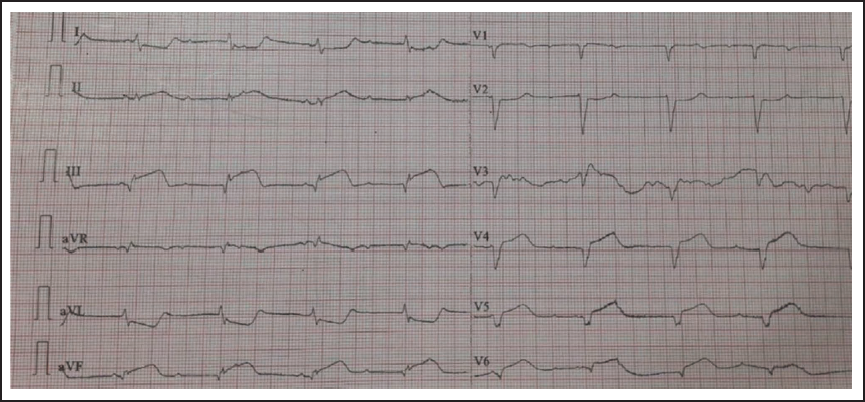
ECG Tracing Showing Prolonged QT with CHB.
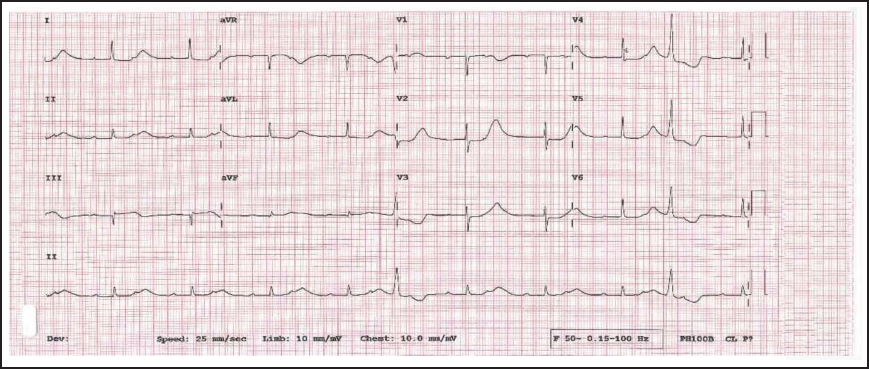
ECG Tracing Showing Prolonged QT with Torsades Preceded by VPC with Short Coupling Interval.
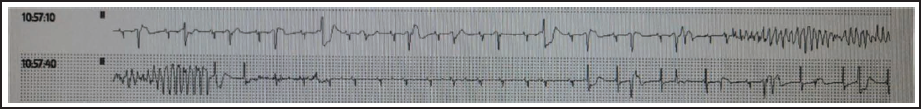
ECG Showing Torsades De Pointes.
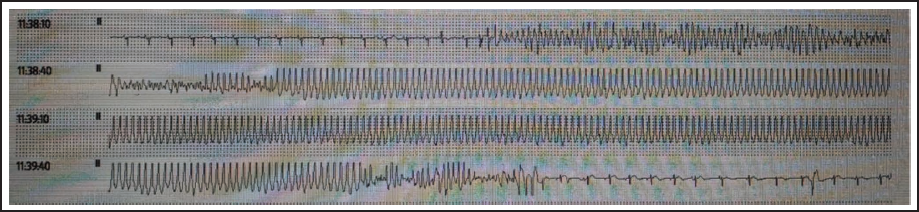
Two cases underwent thrombolysis one with streptokinase and other with tenecteplase. Two out of 6 TdP patients underwent early (primary/rescue) angioplasty. One case was referred for coronary artery bypass grafting. Almost all patients received cardioversion, magnesium, and potassium supplementation as needed. Two cases had temporary pacemaker support. But none of them required overdrive pacing. Of the 6 patients, 3 (50%) survived and got discharged. Two patients who underwent angioplasty survived (100% survival). Thrombolysis had little impact on survival in our series (16.6%). All the details of patients with TdP were mentioned in Table 1.
Showing Summary of Six Patients of Acute Myocardial Infarction with TdP.
Discussion
TdP is a unique type of PVT characterized by a prolonged QT interval. It can be classified into 2 categories based on its etiology: congenital, which is adrenergic-dependent, or acquired, which is pause-dependent.3–5
Symptomatology
Patients with polymorphic VT typically present with one or more of the following: sudden cardiac arrest, syncope or presyncope, “seizure-like” activity, or palpitations. Importantly, polymorphic VT that persists for more than 10 to 15 seconds often degenerates into VF. Idioventricular rhythm is a reperfusion sign within the first 24 hours after thrombolytic therapy which sometimes may be confused with ventricular tachycardia.
Etiopathogenesis of TdP
Post-MI Polymorphic Ventricular Tachycardia and TdP
In the setting of an AMI, polymorphic VT is much less common, occurring in only 0.3% of patients when compared with monomorphic VT and VF. 6 PVT or TdP in AMI can present in 2 phases, either in the hyper acute stage (12-24 h) which is called “Ischemic PVT” or in the healing phase also known as “Infarct related PVT.”
When PVT occurs shortly after the onset of an acute MI, usually within the first 12 hours, it is often accompanied by symptoms or signs indicating recurrent myocardial ischemia. This type of ischemic PVT is typically triggered by ventricular extrasystoles with very short coupling intervals, known as the “R on T phenomenon,” and may not be pause-dependent. 7 Additionally, it may exhibit a normal QT interval, contrary to the typical association of prolonged QT in other forms of PVT. The underlying mechanism of this type of PVT is believed to involve early after depolarizations and triggered activity.8, 9 However, visible QT prolongation is not observed unless a significant area of myocardium is involved in the generation of after depolarizations.10–12
“Infarct related PVT” as described by Halkin et al, 13 occurs in the “healing phase” of MI. A few days after a MI, the maximum difference in the refractory period between adjacent areas in the ventricle is observed, but this discrepancy gradually normalizes over the course of weeks. 14 The mid-myocardial Purkinje cells show more early after depolarizations than the epicardial cells, which results in the heterogeneity of repolarizations between different myocardial layers, which generates multiple waves of re-entry propagating TdP.4, 5 Fluctuations in the length of cardiac cycles can potentially enhance calcium loading, subsequently triggering additional extrasystoles.15, 16 The presence of left ventricular dysfunction can act as a substrate for the occurrence of non-sudden onset PVT in individuals with a lower ejection fraction.
All cases in our series irrespective of the time of presentation, had prolonged QT indicating that the PVT in our series is TdP. A correlation has been established between the degree of QT prolongation and the development of potentially life-threatening TdP in patients recovering from a MI. 14 According to a study by Yap et al, QTc greater than 500 ms has been linked to a 2 to 3 fold increase in risk of TdP, with each 10 ms increase contributing to a 5% to 7% exponential increase in risk. 17 Five out of 6 patients had QTc more than 500 ms in our series. All cases with TdP were associated with high troponin values indicating ischemia involving large area of myocardium.
In our study, the incidence of TdP among all types of MI was found to be 1.89% (6 out of 320 cases). Similar observations were noted in a previous study. 13 Four out of 6 TdP cases in our study were of inferior wall MI. This is in contrast to other case studies where there is equal or more incidence of TdP in anterior wall MI. No bundle branch blocks were picked up on ECG in our patients.
TdP is a type of ventricular tachycardia that can occur in patients with both inferior and anterior wall MI, but the propensity to cause TdP is more commonly associated with inferior wall MI. There are a few reasons why this may be the case: Significant bradycardia with high degree AV block or complete heart block and significant SA block with pauses and blunted sympathetic activity in IWMI.
18
Delayed conduction in the His-Purkinje system: The His-Purkinje system, which conducts electrical impulses through the heart, has a longer path in the inferior wall of the heart than in the anterior wall. This longer path can cause delayed conduction in the inferior wall, which can result in electrical heterogeneity and a greater risk of TdP.
19
Differences in repolarization reserve: Repolarization reserve refers to the ability of the heart to adjust its electrical activity in response to changes in heart rate or other stimuli. Patients with inferior wall MI may have a lower repolarization reserve compared to those with anterior wall MI, which can increase the risk of TdP.
20
Higher prevalence of electrolyte imbalances may be caused by factors such as stress induced catecholamine release, vomiting, diarrhea, or diuretic use.
Electrolyte Disturbance-Induced Polymorphic Ventricular Tachycardia
Hypokalemia and hypomagnesemia can predispose to TdP. These disorders can occur together since hypomagnesemia can directly cause hypokalemia. Hypocalcemia, alone or induced by hypomagnesemia, is a less common cause. The risk of developing TdP in the presence of hypokalemia or hypomagnesemia is greatest in patients taking anti-arrhythmic drugs. 21 The increase in risk with hypokalemia may be related to enhanced drug blockade of the rapid component of the delayed rectifier potassium current (IKr), and this itself may also be related to decreased IKr activity.22, 23 When potassium levels fall below 3.0 mmol/L, there is a notable prolongation of the Q-T interval, which poses a significant risk for the development of TdP, ventricular fibrillation, and sudden cardiac death.
As per the European Society of Cardiology, hypokalemia is associated with specific ECG changes based on potassium levels: (a) When potassium levels range from 3.0 to 3.5 mmol/L, ECG changes include T-wave flattening or inversion. (b) When potassium levels range from 2.3 to 3.0 mmol/L, ECG changes include Q-T interval prolongation (resulting in a longer duration of the T wave), the presence of a visible U wave, mild ST depression (around 0.5 mm), and ventricular extrasystoles. (c) When potassium levels drop below 2.3 mmol/L, there is a risk of TdP and ventricular fibrillation.
In our case series, only one case had potassium below 3.0 (i.e., 2.8 mmol/L). Other cases had a mild decrease or borderline low potassium levels, indicating that the cause of PVT in these cases is more of an ischemic substrate than electrolyte induced TdP. None of the patients with TdP were on QT prolonging drugs. The average QTc is 530.1 ms among the six patients of TdP.
Table 24, 5, 24, 25 summarizes various risk factors for Torsades in AMI.
Enumerating the Risk Factors and Mechanisms of Developing TdP in Acute Myocardial Infarction. S No

Management of TdP (Polymorphic Ventricular Tachycardia)
Patients with hemodynamically unstable TdP require prompt defibrillation. In conscious individuals experiencing recurrent TdP episodes, intravenous magnesium sulfate (IV MgSO4) should be administered, and its benefits are observed even in patients with normal baseline serum magnesium levels. Temporary transvenous overdrive pacing, either atrial or ventricular, at a rate of approximately 100 beats per minute, is typically reserved for patients who do not respond to intravenous magnesium. In cases of congenital long QT syndrome, beta-blockers may be utilized to reduce the frequency of premature ventricular contractions and shorten the QT interval. Isoproterenol can be employed as a temporary measure to achieve a heart rate of 100 beats per minute before pacing in patients with PVT triggered by pauses or bradycardia.
For patients with ischemic PVT, treatment may include prompt defibrillation in unstable patients. Metoprolol 5 mg intravenously every 5 minutes (maximum 15 mg). Intravenous amiodarone may prevent a recurrent episode. Urgent coronary angiography and possible revascularization should be done. Short-term mechanical and circulatory support should be given if needed. If the polymorphic VT is due to catecholaminergic PVT (CPVT), beta-blockers should be used.
In our study, we successfully treated the first 2 patients with primary percutaneous transluminal coronary angioplasty (PTCA) to the culprit vessel and corrected electrolyte imbalances. Serum magnesium levels were obtained and IV MgSo4 (magnesium sulphate) 2g slow IV over 10 to 15 minutes of loading dose followed by 2g IV TID (3 times a day) was administered as the first line treatment till ECG changes subsided. Similar dose of MgSO4 was given for the fifth patient. Potassium chloride (KCl) IV 20 to 40 mEq slow infusion was given to the first 4 of our TdP patients. DC shock was given using a bipolar defibrillator when required. However, despite prolonged resuscitation efforts, the third, fifth, and sixth patients could not be revived. The fourth patient was advised coronary artery bypass grafting but she chose to be on conservative treatment (Table 1). This suggests that early revascularization of the culprit artery in the first 2 cases greatly improved their outcomes.
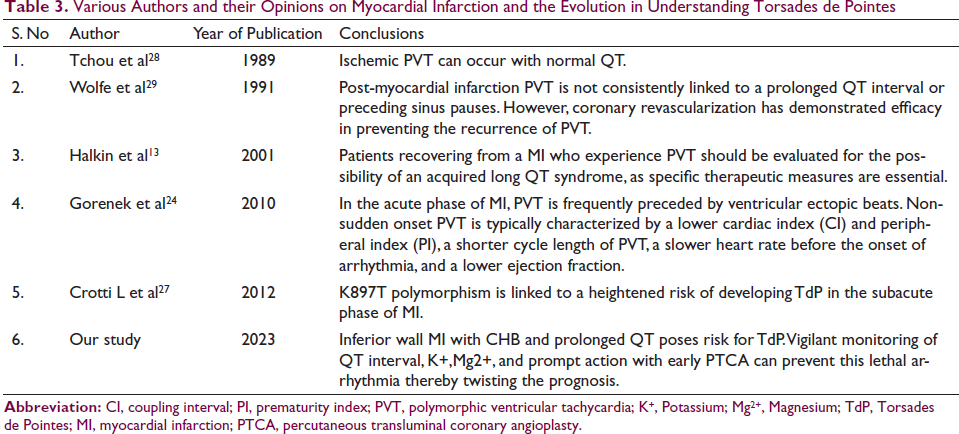
Table 313, 24, 27–29 summarizes various authors’ studies and their conclusions on TdP and MI.
Various Authors and their Opinions on Myocardial Infarction and the Evolution in Understanding Torsades de Pointes
Limitations
It is possible that we have missed some cases TdP as this rhythm abnormality is transient and short lived. This might lead to underestimation of TdP incidence in our study. This might also be the reason for the unreported cases of anterior wall MI with TdP. However genetic polymorphisms unmasked by MI can also have prolonged QT presenting as acquired long QT syndrome.
Conclusions
TdP caused by ischemia or infarction rarely occurs in AMI and should be considered in all cases of prolonged QT and bradycardia irrespective of electrolyte abnormalities. This arrhythmia is associated with ischemia, QT prolongation, and associated with poor prognosis. Early PTCA can prevent this lethal arrhythmia, thereby twisting the prognosis.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Informed consent was taken from all patients. Ethical committee approval was taken.
