Abstract
Acute obstruction of a modified Blalock-Taussig shunt may result in life-threatening hypoxia in patients with shunt-dependent pulmonary circulation. We here report successful relief of shunt blockage due to thrombosis in immediate postoperative period with transcatheter thrombolysis by Urokinase in an infant. Our case highlights the need of early diagnosis as well as the important role of emergent catheter-directed thrombolysis as an effective rescue strategy to restore the patency of acutely thrombosed Blalock-Taussig shunt.
Keywords
Introduction
Acute occlusion of a modified Blalock-Taussig shunt (MBTS) is not uncommon and is a life-threatening situation. Emergent transcatheter intervention is a safe and effective method to restore the patency of occluded shunt and avoid redo surgery. We here report successful relief of acute obstruction of the Blalock-Taussig (BT) shunt after 3 days of surgery with catheter-directed thrombolysis by Urokinase in an infant.
Case Description
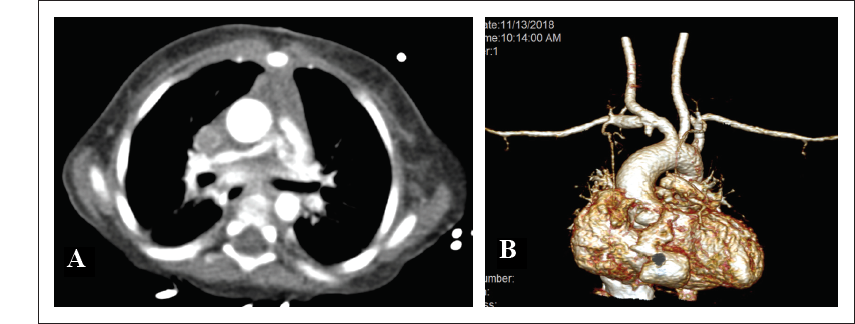
A 3-month-old girl infant was incidentally detected to have central cyanosis and murmur on routine check-up. On echocardiographic evaluation, she was detected to have cyanotic congenital heart disease, pulmonary atresia with intact interventricular septum, confluent small branch pulmonary arteries, and tiny left patent ductus arteriosus (PDA) supplying pulmonary blood flow for which she underwent right MBTS (4 mm Gore-Tex graft) at 5 months of age. The ductus arteriosus was not ligated. Postoperative echocardiography revealed well-patent shunt. Initial anticoagulation was given in the form of Heparin infusion (0-2nd postoperative day). Ecosprin was started on 0 postoperative day for continued anticoagulation effect. Her activated clotting times were kept in the higher range throughout her initial intensive care unit (ICU) stay. But from early 4th postoperative day, she started deteriorating clinically with gradually worsening hypoxemia with saturations dipping to 30 s. There was narrow pulse pressure and shunt murmur was not audible. Echocardiography revealed nonvisualization of the BT shunt flow with tiny PDA providing pulmonary blood flow for survival. This was initially managed with 100 IU/kg heparin bolus followed by high-dose heparin infusion. Despite this, her shunt did not open. Computed tomography angiography was done urgently which revealed blocked BT shunt with a small stump patent at the right subclavian artery end and presence of partial thrombus in left pulmonary artery (LPA) (Figure 1).
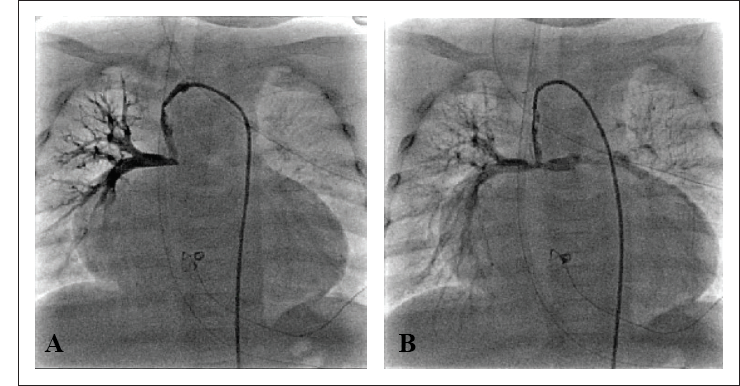
Without waiting any further, the infant was taken up in the Cath lab for shunt thrombolysis on late 4th postoperative day. 4F Judkins right coronary catheter was advanced over 0.035 inch J-tipped Terumo wire into the right subclavian artery and parked at the proximal end of the shunt. Initial shunt angiogram showed irregular filling of the shunt with complete nonopacification of LPA and its branches. Thrombosuction was done followed by 25,000 units of Urokinase (4,400 units per kg intravenous bolus) given as bolus dose into the proximal end of the shunt. Then catheter-directed lysis was continued with slow infusion of 25,000 units of Urokinase (4,400 units per kg per h) over an hour. This proved successful and the shunt opened up with systemic saturation increasing up to 85%. Postprocedure shunt angiogram showed well-patent BT shunt with disappearance of the thrombus and restoration of flow to LPA and its branches (Figure 2). The clinical status of the baby dramatically improved without any hemorrhagic complications. Subsequently, she was put on high-dose anticoagulation in the form of heparin infusion (40 IU/kg/h) along with Ecosprin and Warfarin to prevent reocclusion after thrombolysis. Heparin infusion was stopped once the international normalized ratio (INR) reached 2.5 on 6th postoperative day and she was discharged on low-dose Warfarin and Ecosprin. At 6 months follow-up, she was maintaining saturation of 75% to 80% with well-functioning BT shunt.
Discussion
Acute systemic-to-pulmonary artery shunt occlusion remains a major cause of mortality in early postoperative period in patients with shunt dependent pulmonary circulation which requires emergent surgical or transcatheter interventions to re-establish pulmonary blood flow. Shunt obstruction can occur due to a variety of causes including acute shunt thrombosis, stricture at anastomotic site, neointimal proliferation, and vascular stenosis due to surgery. Few of the risk factors described for acute shunt thrombosis in early postoperative period are dehydration, high hemoglobin, weight less than 3 kg, and smaller shunt size. 1
Balloon angioplasty, use of thrombolytic therapy, and stent implantation are the major nonsurgical treatment modalities described in literature for shunt obstruction.2-10 Systemic thrombolysis was used in noncardiac centers as an emergent treatment2, 9; balloon angioplasty and/or local thrombolysis were mainly described in patients with acute stenotic shunts3-8 and stent placement in cases of chronic obstruction.6, 10
Very limited data are available recently concerning risks and success rate of surgical revision for modified BT shunt obstruction compared to interventional techniques. Redo surgery may expose the patients to risks due to a new sternotomy or thoracotomy hence, more tissue adhesions at the time of future interventions, prolonged ICU stay, infections, arrhythmias, need for blood transfusions, or neurologic complications. Despite some severe procedure-related complications, catheter-based intervention is a less-invasive approach to restore shunt patency in critically ill patients with a good success rate.
The efficacy of thrombolytics is same with either systemic route or local delivery. A relative contraindication is their use within 1 week postcardiac surgery due to high risk of hemorrhagic complications. As bleeding risk is lower in those treated with catheter-directed thrombolysis than systemic thrombolysis, former is the preferred method of administration, if feasible.
A variety of thrombolytic agents have been described in literature. We used Urokinase over recombinant tissue plasminogen activator in view of its slightly greater efficacy and more rapid onset of action. She did not require balloon dilatation or subsequent stent placement as it was a case of acute thrombotic state and the shunt showed complete patency after thrombolysis.
Implications in Clinical Practice
Emergent catheter-directed thrombolysis is an effective and feasible alternative to balloon angioplasty or surgical revision to recanalize the acutely thrombosed BT shunt, especially in life-threatening situation in a severely hypoxic child, with no significant systemic or local complications.
(A). CT angiogram axial view showing filling defects in LPA suggestive of thrombus. (B) Volume-rendered 3D reconstructed CT angiogram showing blocked BT shunt with a small stump patent at right subclavian artery end.
(A). Preprocedure shunt angiogram showing filling defects in right MBTS with complete nonopacification of LPA and its branches. (B) Postprocedure shunt angiogram showing opened MBTS with restoration of flow to LPA and its branches.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
