Abstract
We report a case of dilated cardiomyopathy because of chronic mercury poisoning. A 12-year-old girl was repeatedly admitted with severe congestive heart failure. She received indigenous medication for vitiligo for several months. A transthoracic echocardiogram showed severe ventricular dysfunction (left ventricle fractional shortening: 14%). She had a stormy course in the hospital, complicated by ventricular arrhythmias and multiorgan dysfunction. Careful interrogation and analysis of local medication and patient serum showed high levels of mercury. She was treated with British anti-Lewisite. She improved over the period of time, and her left ventricle fractional shortening improved to 22%.
Introduction
Dilated cardiomyopathy (DCM) is defined as the dilatation and impaired contraction of 1 or both ventricles. 1 The disease is considered idiopathic if primary and secondary causes are excluded by meticulous evaluation, including clinical examination with thorough history taking, laboratory testing, electrocardiogram, echocardiography, coronary angiography, endomyocardial biopsy, and others. 2 Treatable causes of DCM include calcium deficiency, tachyarrhythmias, thyroid disturbances, hypertensive heart disease, and infective diseases, such as diphtheria. 3 Mercury poisoning is rare and may cause injury to the central nervous system and kidneys. DCM because of chronic mercury poisoning is extremely rare. 4 We report a case of refractory heart failure because of chronic mercury poisoning in a child.
Clinical Summary
We present the case of a 12-year-old girl who had repeated hospital admissions over the past 2 months because of acute decompensated left ventricular failure and multiorgan dysfunction. There was hepatic and renal derangement because of the cardiogenic shock. Renal derangement needed peritoneal dialysis. She had repeated episodes of ventricular tachycardia, needing direct current cardioversion. She was evaluated for all correctable causes of cardiomyopathy, which were negative. She was diagnosed with idiopathic DCM. After the third hospitalization, a repeat detailed history revealed the chronic consumption of indigenous medication for vitiligo. Urine analysis for toxicology was done, which revealed elevated mercury levels, and then was confirmed on blood levels done using the inductively coupled plasma optical emission spectroscopy method (mercury blood level, 96.67 µg/L [reference range, 0.46–7.5 µg/L]). She was immediately started with British anti-Lewisite (BAL) chelation and charcoal hemoperfusion therapy for mercury toxicity, and then she showed gradual clinical improvement. Her liver and renal function tests became normal within 2 weeks. Blood mercury levels decreased to 25 µg/L after 6 months of treatment. Indigenous medicine, which the child had taken, was also sent for analysis, which revealed high mercury content (1 g of powder has 14.826 parts per billion of mercury).
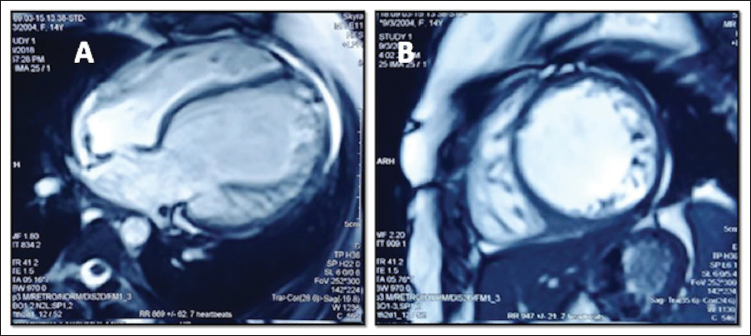
Cardiac magnetic resonance imaging done after 6 months of treatment was suggestive of global hypokinesia of a dilated and thinned-out left ventricle without any early or late gadolinium enhancement on T1-weighted images (Figure 3). There are no regional or global signal intensities on T2-weighted images. Her left ventricle function improved gradually over 12 months (left ventricle fractional shortening is 22%, Figures 1 and 2).
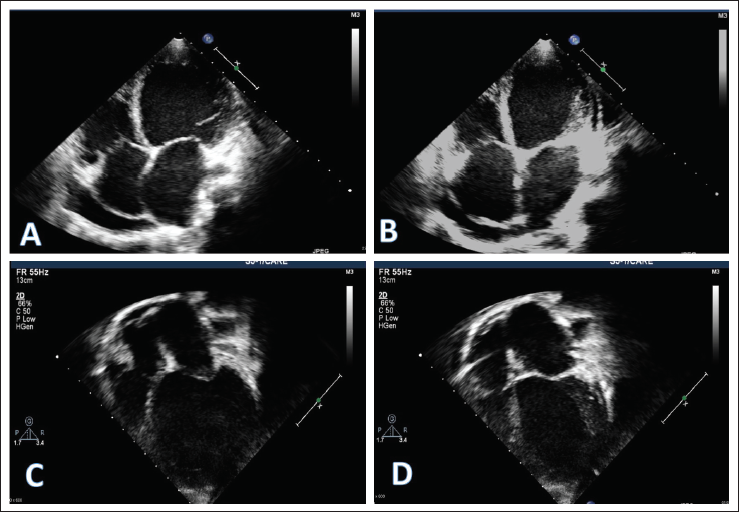
Apical 4 Chamber View Showing Improvement in Left Ventricular Dimensions Before (A, B) and After Treatment (C, D). End Diastole (A) and End Systole (B) Before Treatment Showing Minimal Change in Left Ventricular Dimensions Suggestive of Severe Left Ventricular Dysfunction Along with Pericardial Effusion. After Treatment, There Is Resolution in Pericardial Effusion, with Improvement in End Diastole (C) and End Systole (D) Left Ventricular Dimensions and Function.
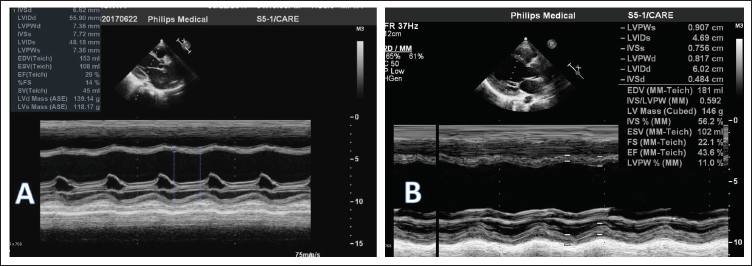
M-Mode Echocardiography Done Before (A) and After (B) Treatment. Left Ventricular Fractional Shortening Improved From 14% to 22% with Treatment.
Cardiac Magnetic Resonance Imaging Done 6 Months After Treatment. Axial (A) 4 Chamber Cut and Short-Axis Mid-Biventricular Cut (B) Showing Thinned-Out Left Ventricular Walls with Dilated Left Ventricle. No Evidence of Late Gadolinium Enhancement.
Discussion
Mercury (hydrargyrum, Hg) is a heavy metal (atomic number 80), has 3 valence states (elemental, inorganic, and organic), and can exist in several forms, which are toxic. 5 Mercury has no physiological role in humans and cannot be excreted from the body in any form. 4 Its toxicity depends on the form, type, and duration of the exposure. Acute inhalation of high levels of elemental mercury vapor predominantly causes interstitial pneumonitis, whereas chronic exposure to a lower concentration of the elemental mercury vapors results in predominant nervous system involvement that may be severe and irreversible. Acute ingestion of inorganic mercury in the form of mercury salts may cause severe abdominal pain accompanied by hemorrhagic gastroenteritis and severe volume depletion within several hours of ingestion. Chronic exposure to a lower concentration of inorganic mercury salts causes neurological symptoms and, occasionally, nephrotic syndrome. Organic mercury has higher lipophilicity than other forms, leading to early absorption in the gastrointestinal tract. It can cross the blood–brain barrier, causing various neurological manifestations. It can also affect the kidneys. Organic mercury, such as methylmercury, can cause cardiac manifestations, such as early atherosclerosis, myocardial infarction, hypertension, and ventricular dysfunction, and also affect its rhythm by the involvement of the parasympathetic nervous system. 6
Our patient presented with a unique set of symptoms, such as DCM and severe biventricular dysfunction and recurrent tachycardia and acute kidney injury with elevated liver enzymes. This suggests that there is multiorgan dysfunction because of both cardiogenic shock and the effects of mercury on the liver and kidneys. The mechanism by which mercury produces toxic effects on the cardiovascular system is not fully elucidated. A possible hypothesis can be mercury ions (Hg++) act as a calcium (Ca++) antagonist at the actin–myosin complex, inhibiting the sarcomere contraction and ultimately reducing the myocyte function (Figure 4). Another possibility is that Hg++ causes disruption of the microtubule network, a major cytoskeleton component in most of the cardiomyocytes, causing changes in the biomechanical integrity of sarcomere and affecting excitation–contraction coupling. 7 Glutathione is the most potent antioxidant, protecting against oxidative stress and reducing cardiovascular diseases. Mercury causes depletion of glutathione, binds to metallothioneins by replacing zinc and copper, and competes for selenium, reducing the effectiveness of metalloenzymes. 4 This increases the production of reactive oxygen species and mitochondrial dysfunction, leading to sarcomere disruption.8, 9 Organic mercury exposure also causes heart rate variability by the parasympathetic change in children.
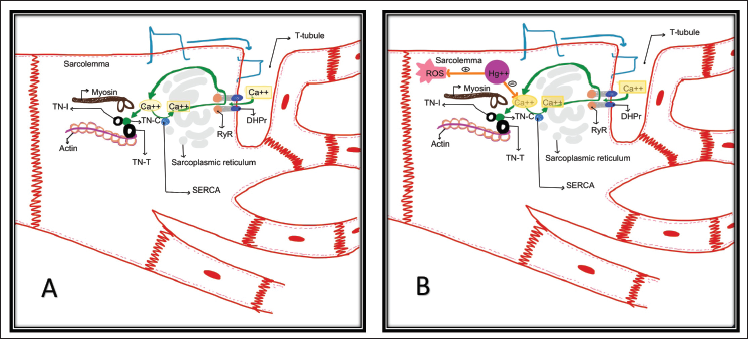
Global involvement with severe biventricular dysfunction along with recurrent ventricular arrhythmia was noted in the index patient. The presence of thinned-out myocardium without late gadolinium enhancement on cardiac magnetic resonance imaging is suggestive of interference with the excitation–contraction coupling and disuse atrophy being predominant mechanisms rather than necrosis of myocytes with consequent scar formation. Involvement of other organs with elevated liver enzymes was also suggestive of heavy metal poisoning.
Chelation is an important therapy in mercury poisoning. BAL (2,3-dimercaptopropanol) contains sulfhydryl groups, which are useful in chelation. BAL can be given orally and through an intravenous route. 10
One cycle of charcoal hemoperfusion along with BAL therapy resulted in clinical improvement along with a reduction of the blood mercury level. Improvement of left ventricular function after chelation therapy is suggestive of reversible pathology like calcium antagonism. Persistence of mild left ventricular dysfunction (left ventricle fractional shortening: 22%) even after 12 months of therapy is suggestive of irreversible injury to the myocardium.
Conclusion
Meticulous history taking and vigilant search for a secondary cause are important in every case before making a decision as idiopathic DCM. DCM because of chronic mercury poisoning is very rare, and a high degree of suspicion is needed based on the social background of the patient. Chelation with BAL is a promising medication for treating such cases.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
