Abstract
Symptoms of heart failure (HF) should be alleviated as quickly as feasible. Apart from standard pharmacological therapy, supplementation of oxygen is lifesaving in hypoxemic (SpO2 <90% or PaO2 <60 mmHg) HF patients. For sick patients with SpO2 <85%, respiratory distress (respiratory rate >25 breaths per min) and pulmonary edema, along with oxygen administration, noninvasive positive pressure ventilation (NIPPV) methods, including BiPAP/CPAP or humidified high-flow nasal oxygen, should be tried to optimize oxygenation, reduce work of breathing and to reduce the venous return. If these noninvasive ventilatory support methods fail to improve hypoxemia, endotracheal intubation and mechanical ventilation should be instituted. Short-term mechanical circulatory support (extracorporeal membrane oxygenation) devices are reserved for cardiogenic shock patients due to advanced HF associated with reversible end-organ failure and hypoxia. These temporary mechanical circulatory support devices are also useful as a bridge to long-term mechanical circulatory support or heart transplant in end-stage HF with refractory cardiogenic shock. Even though the use of additional oxygen is a standard practice for hypoxemic patients, routine use of high concentrations of oxygen via inhalational therapy or NIPPV for normoxemic HF patients may result in hyperoxemia. Hyperoxemia has been shown to have negative hemodynamic effects like increased systemic vascular resistance, reduced cardiac index, and decreased coronary blood flow. Detailed prescription by the treating physician, careful monitoring, and meticulous titration of oxygen is essential for safe oxygen treatment for HF patients.
Keywords
Definitions
SaO2: The percentage of oxygen saturated hemoglobin related to total hemoglobin in the arterial blood measured by blood gas analysis. (Normal values: 95% and 100%)
SpO2: The percentage of oxygen saturated hemoglobin related to total hemoglobin in the arterial blood measured by pulse oximetry.
PaO2: Partial pressure of oxygen in the arterial blood.
FiO2: Fraction of inspired oxygen; the amount of oxygen in the inspired air. 0.21 is the room air FiO2 (21%).
Hypoxia: Low oxygen at the level of the tissues.
Hypoxemia: Low oxygen concentration in the blood. (SaO2 <90% or PaO2 <60 mm Hg)
Hyperoxemia: Arterial oxygen PaO2 >120 mm Hg.
Introduction
Heart failure (HF) is a condition that develops where the heart does not pump enough blood for the body’s needs. Patients in HF usually present with symptoms of breathlessness, fatigue, cough, dizziness, confusion, wheezing, pedal edema, palpitations, and signs like raised jugular venous pressure, pulmonary crepitations, tachypnea (respiratory rate >24/min), tachycardia, or dependent edema. These signs or symptoms may not be evident in the early stages of HF and in patients who have received proper treatment. However, as the disease progresses, there will be a gradual decline in cardiac output and quality of life. Patients may also experience episodes of acute decompensation due to myocardial infarction (MI), lung infections, dysrhythmias, uncontrolled hypertension, or patient’s noncompliance with diet or medication.
Acute heart failure (AHF) is a syndrome defined as the new onset or worsening of symptoms and signs of HF, mostly related to systemic congestion. 1 The pathogenesis of AHF is exceedingly varied, even though the underlying heart disease and triggering events may be comparable. Retention of fluid leads to systemic and pulmonary congestion, resulting in organ failure due to low cardiac output and hypoxemia.
Breathlessness is the common presentation of HF. In addition to the standard therapy for HF, supplemental oxygen is indicated when the oxygen saturation is <90% or partial pressure of oxygen (PaO2) is less than 60 mm Hg.2, 3 Oxygen is only useful when dyspnea is due to hypoxemia. Even though many of these patients may not have hypoxemia, supplemental oxygen is commonly given with the intention that oxygen may be useful and causes no harm. In nonhypoxemic breathless patients, oxygen offers no added benefits and can result in hyperoxemia, which is associated with negative hemodynamic consequences due to increased systemic vascular resistance (SVR) and reductions in cardiac output and coronary blood flow. Many recent HF management guidelines recommend caution for the routine use of oxygen inhalation therapy and NIPPV for normoxemic HF patients because of concerns related to hyperoxemia.4-6
In this review article, we aim to describe the mechanism of hypoxia in HF, the indications for supplemental oxygen, and the appropriate oxygen delivery devices that need to be used, along with the adverse effects of oxygen and guidelines for oxygen supplementation in HF.
Mechanism of Hypoxia in Heart Failure
Hypoxia is more common in AHF patients than the chronic heart failure (CHF) patients. Two main mechanisms responsible for tissue hypoxia in HF patients include elevated ventricular diastolic filling pressures and decreased cardiac output. The elevated filling pressure of the left ventricle leads to pulmonary congestion, which decreases the diffusion capacity of the lung, causing an increase in intrapulmonary shunting, resulting in arterial hypoxemia. Another mechanism is pump failure, leading to reduced cardiac output and decreased tissue perfusion causing circulatory hypoxia. Patients with low cardiac output without much changes in diffusion of gases have tissue hypoxia without arterial hypoxemia. The presence of anemia may aggravate hypoxia caused by low tissue perfusion. Congestion symptoms (dyspnea or breathlessness) rather than low cardiac output are commonly responsible for the hospitalization of these patients.7-11
Oxygen Therapy in Heart Failure
The rationale behind O2 therapy is to correct the hypoxemia and thus prevent irreversible damage to vital organs resulting from cellular hypoxia. The clinical goals of oxygen therapy include correction of hypoxemia, reduction in the work of breathing, and optimizing the myocardial work. SpO2 and PaO2 are the key clinical parameters that guide us in initiating, monitoring, and adjusting oxygen therapy. These parameters are not useful for assessing hypoxia due to pump failure and other causes of tissue hypoxia. Measurement of lactate levels, mixed venous oxygen saturation, tissue oxygen saturation, gastric intramucosal pH, or venous-arterial carbon dioxide gradient are useful for assessing tissue hypoxia.
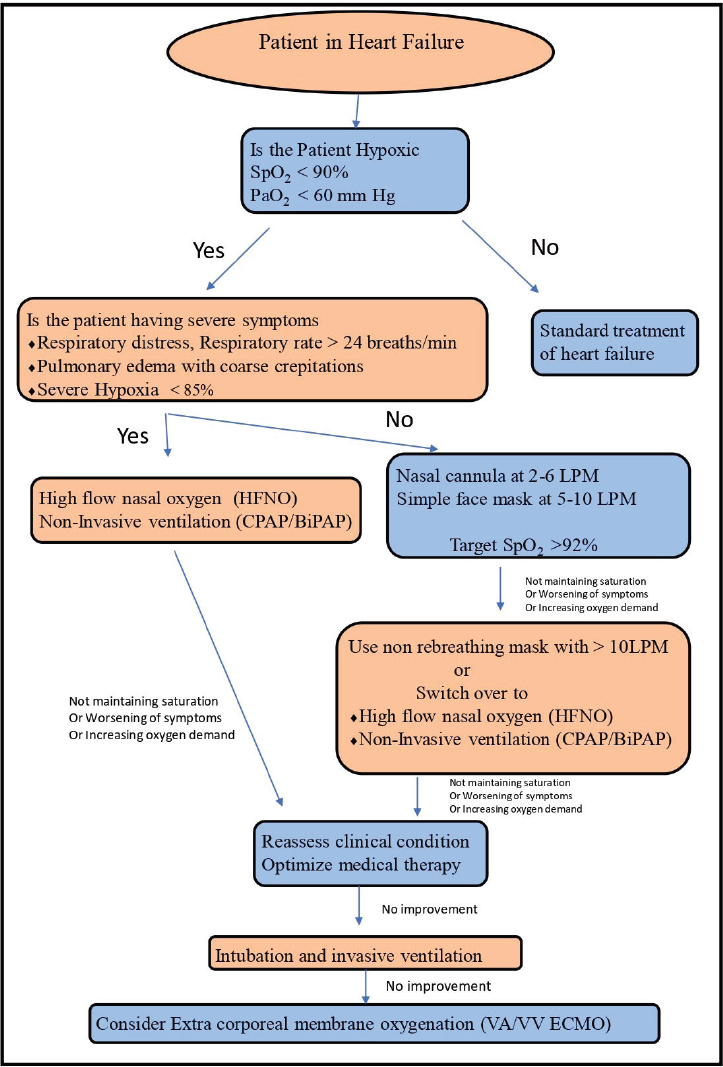
2021 ESC Guidelines for the diagnosis and treatment of AHF and CHF recommended oxygen therapy in patients with AHF and SpO2 <90% or PaO2 <60 mmHg to correct the hypoxemia. 5 The initial management of mild hypoxemic patients must include starting oxygen with simple devices like nasal cannula (1-6 L/min) or face masks (5-10 L/min). 4 If the desired saturation is not achieved, a non-rebreathing mask (NRBM) that can deliver FiO2 of 60 -100% can be used. The oxygen flow rate in a NRBM must always be more than 10 L/min and the reservoir bag must remain inflated during inhalation as well as exhalation to provide adequate inspiratory flow. Tight-fitting NRBMs can achieve 100% oxygen when the oxygen flow rate is >15 L/min, but prolonged use risks oxygen toxicity and reabsorption atelectasis. Humidified high-flow nasal oxygen (HFNO) using specialized equipment can also be considered as an alternative for reservoir masks in these patients.
For patients in severe respiratory distress (respiratory rate >25 per min) with SpO2 <85% and cardiogenic pulmonary edema, early institution of continuous positive airway pressure (CPAP) with a high concentration of oxygen or HFNO will be needed (Figure 1). 4 In CPAP, the machine delivers a constant or continuous pressure of the gas at a set value. This pressure splints the small airways leading to lung recruitment and improved oxygenation. This pressure increases the intraalveolar pressure, thereby decreasing the transudation of fluid into the lungs, thus alleviating breathlessness in congestive HF.
(A) Oxygen Mask with Rebreathing Bag, (B) Humidified High-Flow Nasal Oxygen Device, and (C) Bilevel Positive Airway Pressure (BiPAP) Mask.
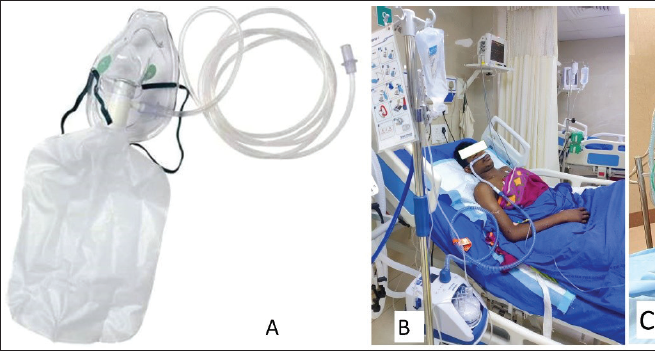
If the patients’ condition does not improve with these devices, noninvasive positive pressure ventilation (NIPPV) with bilevel positive airway pressure (Bi-PAP) with a tight-fitting mask should be considered. The expiratory positive airway pressure or positive end-expiratory pressure (PEEP) acts similar to CPAP and keeps the alveoli open during expiration and improves oxygenation. The inspiratory positive pressure (IPAP) assists the patient’s ventilation by pushing in the air at a preset pressure, that is, IPAP. This improves the volume of air which moves in and out of the lungs, improving the tidal volume, minute ventilation, and thereby improving CO2 clearance. This mode is very useful in patients with type II respiratory failure. NIPPV reduces work of breathing and also reduces preload by decreasing the venous return.
In recent times, the use of HFNO has become popular in the intensive care units (ICU) for the management of patients with acute hypoxemia. 12 These electronically powered devices can deliver up to 60 to 100 L per min (LPM) flow of blended air with oxygen through a specially designed wide bored nasal cannula. As there’s less chance of entrainment of room air, we can give a very high concentration of oxygen (FiO2: 95-100%) to the patients. These devices also have an inbuilt heated humidifier with a heated breathing tube with integrated heated wires and a temperature sensor that enables to control the inhaled gas temperature from 31oC to 37oC. Pre-warmed and humidified inspired gases facilitate the mucociliary clearance of the lower airways, prevent dryness of the upper airway, and improve patient compliance. The high flow of gases flushes the upper airway dead space and generates a small amount of PEEP (up to 5-7 cm of H2O) which facilitates oxygenation, airway patency, and reduces the upper airway resistance and work of breathing. 13 Due to these beneficial effects, the use of HFNO has been shown to be as efficacious as NIPPV in terms of reduction of intubation rates in patients with hypoxemic AHF, and also reduces the reintubation rates among patients at high risk of extubation failure.14-17 However, the amount of PEEP developed by the HFNO is significantly lower than the PEEP effect produced by NPPV, in patients with AHF associated with severe pulmonary edema, NPPV (BiPAP) should be considered as a preferable therapy. 18
Invasive positive pressure ventilation with an endotracheal tube can be considered if all these measures fail. Short-term mechanical circulatory support (extracorporeal membrane oxygenation) is indicated in patients with acute pulmonary edema associated with low cardiac output to restore organ perfusion. These temporary mechanical circulatory support devices are also useful as a bridge to long-term mechanical circulatory support or heart transplant in end-stage HF with refractory cardiogenic shock (Figure 2). 19
Simple flowchart for oxygen therapy in heart failure patient.
HF patients who also have concomitant chronic obstructive pulmonary disease (COPD) are at risk of hypercapnic respiratory failure. High concentrations of oxygen in these patients can cause progressive hypercarbia. For these patients, fixed oxygen concentration performance devices like venturi masks which provide an accurate, controlled concentration of oxygen from 24% to 60% are very useful to prevent hyperoxemia-induced hypercapnia.
Home Oxygen Therapy
Domiciliary oxygen therapy or home oxygen therapy (HOT) is often prescribed to alleviate suffering for severely symptomatic CHF patients. Oxygen concentrators or cylinders are commonly used for the administration of oxygen at home. However, unlike patients with COPD and severe hypoxia in whom oxygen prolongs survival, there is no evidence to support the use of HOT in CHF patients. One study was performed to address the question whether HOT had any effect on the quality of life, breathlessness, was terminated early, and found that adherence to HOT was poor and concluded that there is no evidence to support the use of HOT in patients with HF. 20 British Thoracic Society Guidelines for home oxygen use in adults recommends long-term oxygen therapy should be considered for patients with pulmonary hypertension, including idiopathic pulmonary hypertension, when the PaO2 is <60 mm Hg. 21
Another form of oxygen supplementation during physical activities of daily life is ambulatory oxygen therapy (AOT). AOT is recommended for patients in heart failure with preserved ejection fraction (HFpEF) as a part of a rehabilitation program who have shown improved exercise capacity (more than 10% improvement in exercise capacity with oxygen enrichment) and symptomatic relief. during the assessment.4, 22
Monitoring and Target Saturation
Pulse oximeter is widely used to monitor arterial hemoglobin oxygen saturation. British Thoracic Society Guideline for oxygen use in adults in healthcare and emergency settings recommended target oxygen saturation of 94% to 98% for AHF patients. 4 This target should be reduced to 88% to 92% if the patient is at risk of hypercapnic respiratory failure. In the literature, there is no uniform consensus regarding the target oxygen saturation. In contrast to the 94% to 98% target range suggested by the British Thoracic Society, the Thoracic Society of Australia and New Zealand recommended a SpO2 range of 92% to 96% for oxygen therapy for patients who are not at risk of hypercapnic respiratory failure. 23 Beasley et al 24 suggested that a SpO2 target range of 92% to 96% is preferable to a 94% to 98% target range for the supplementation of oxygen to adult patients who are not at risk for hypercapnia, as it is close to the mean minimum SpO2 of healthy aged adults. The HiLo-HF (high vs. low oxygen therapy in patients with AHF) pilot trial explored the effects of supplemental O2 therapy in patients with AHF and concluded that titrating O2 therapy to high (>96%) or low (90-92%) SpO2 targets did not result in changes in biomarkers, symptoms, or clinical outcomes. 25
Arterial blood gas (ABG) gives more information about sick hypoxemic patients with shock or hypotension. PaCO2 and pH values obtained from ABG analysis help the clinician to identify the chronicity of hypoxemia and the type of respiratory failure.
Blood pressure should be monitored regularly during NIPPV. The increase in intrathoracic pressures may reduce the venous return to the right heart which can further reduce the cardiac output. NIPPV may also increase pulmonary vascular resistance and right ventricle (RV) afterload which might be harmful to individuals who already have RV dysfunction. Therefore, in individuals with hypovolemia and hypotension, NIPPV should be administered with care.
Humidification of Inspired Oxygen
Humidification of inhalational gases is required to prevent mucociliary damage and thickening of respiratory secretions. Active humidification is not required for the delivery of low-flow oxygen (<5 LPM). However, humidification of high-flow oxygen is recommended to prevent dryness of the upper airway when the planned therapy lasts more than 24 h. 4 Routine humidified oxygen is advised for all patients who require invasive ventilation with a tracheostomy or an endotracheal airway.
Weaning of Oxygen Therapy
Weaning from oxygen therapy can be considered once the patient is clinically stable and the SpO2 is persistently above or in the upper zone of the target range. Before stopping the oxygen therapy, the oxygen flows can be gradually reduced to 2 L/min via nasal cannula or 5 L/min on a face mask. Oxygen should be stopped when SpO2 is >90% on room air. The patient should be carefully monitored for 1 h after cessation of oxygen therapy. A sharp SpO2 drop of >3% indicates acute deterioration of clinical state, which warrants thorough evaluation of the patient and optimal medical therapy.
Adverse Effects of Hyperoxemia
Hyperoxia that develops due to inappropriate administration of a high concentration of oxygen has the potential to cause negative hemodynamic effects, such as increased SVR and blood pressure, reduced cardiac index, and decreased coronary blood flow. 26 Long-term administration of high oxygen concentration (>60%) also attributed to pulmonary oxygen toxicity by causing reactive oxygen species-mediated damage to capillary endothelium and alveolar epithelium. Administration of a high concentration of oxygen may cause absorption atelectasis and inhibition of reflex hypoxic pulmonary vasoconstriction which can further deteriorate ventilation-perfusion mismatch.
The 2 major randomized control trials, AVOID (Air Versus Oxygen in myocardial infarction) and DETO2X-AMI (Determination of the Role of Oxygen in Acute myocardial infarction) trials evaluated the role of supplemental oxygen in normoxemic acute MI patients.27, 28 These studies showed that administration of oxygen in these patients didn’t show outcome benefit but was rather associated with increased infarct size. Another recent multicentric propensity score-matched retrospective study compared the efficiency of routine oxygen therapy with ambient air, in patients with normoxemic (SpO2 >90%) AHF. 29 The authors concluded that routine use of supplemental oxygen in AHF patients without hypoxemia was not found to reduce all-cause in-hospital mortality or ICU mortality.
Another major problem of supplementation of inappropriately high-concentration oxygen to correct the hypoxemia is many clinicians progressively increase FiO2 to treat the worsening hypoxia. Even though this approach transiently improves the arterial saturation, it creates a false assurance about the patient’s clinical condition to the treating physician. This can lead to a delay in recognizing the progressive deterioration of heart function, resulting in a short window of opportunity to intervene.
Future Research
Even though the administration of oxygen is beneficial in hypoxic HF patients, the specific target is not yet clearly defined. There is variable and insufficient evidence available in the literature regarding the oxygen therapy targets, duration of therapy, and methods of oxygen administration. Large and multicenter randomized control studies are required to fill the gaps in the evidence.
Conclusion
Medical oxygen is a drug and must be used meticulously and diligently. Although early and proper use of oxygen is suggested to treat hypoxemia, we believe that in normoxic cardiac patients, inappropriate use of supplemental oxygen may result in poorer outcomes. Hyperoxia has the potential to cause damage to the heart, lungs, and brain. Indications for treatment with oxygen as well as the appropriate modes of delivery must be borne in mind while using oxygen. Oxygen administration like any other drug needs a prescription, with a mention on the flow, the device to be used, monitoring required, SpO2 target, and should be signed by the physician in the drug chart. Careful monitoring and titration of oxygen based on the patient’s response can reduce the risks associated with oxygen therapy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
