Abstract
Assessment and support of hypoxic patients is common in intensive care medicine. Simple oxygen therapy systems are typically used at the beginning of the patient journey, but their performance is influenced by the device used and the patient’s respiratory pattern. A solution to overcome this issue is presented, which eliminates device factors and the patient’s respiratory pattern from early simple medical gas administration; whereby an oro-nasal mask uses a novel interface combined to visual feedback. The resultant accuracy of medical gas administration achieved, should aid early assessment, support, and rescue of patients in respiratory distress.
Keywords
Introduction
Hypoxic patients being referred to intensive care is a common clinical scenario. By the time of intensive care clinical contact, the patient is invariably on simple oxygen therapy, often by high concentration reservoir (non-rebreathing) mask.
Simple oxygen therapy devices are classically divided into variable (medium or high concentration masks, tent masks, low flow nasal cannula) and fixed performance (Venturi mask) devices. For variable performance devices both device factors (device volume, oxygen flow, vent resistance, leak area) and patient factors (inspiratory flow, length of expiratory pause) are pertinent to performance. 1 Venturi masks aim to overcome these issues, but they are subject to them nonetheless, 2 with the total input mask flow decreasing as the oxygen percentage of the Venturi increases. 3 The high concentration reservoir mask though, is most definitely a variable performance device, delivering between 60% and 90% oxygen at 15 L/min oxygen flow. 3 Historically, it has also been the device of choice for heliox administration, but its’ variable performance can lead to significant dilution of the inhaled heliox mixture. 4
The ideal situation is to remove device factors and the patient’s respiratory pattern from simple medical gas administration, to give known accurate delivery. 2 With this capability, accurate and repeatable measures of the patient’s pathophysiological respiratory function with regards to oxygen, will be within the grasp of the attendant team within minutes of patient encounter.
Controlling simple medical gas administration means controlling the gaseous environment that the patient breathes from. For a mask solution, this means controlling the gaseous environment immediately in front of the nose and mouth. Given that the desire is to give gases other than atmospheric air, a means to separate atmospheric air from the gas to be breathed is necessary. Thus, the administration system must be closed or else the flow of gas to be breathed is so high as to completely displace atmospheric air whilst the patient breathes in. If using the former closed system, the clinician needs to know that the flow and storage of gas within the device is adequate for the patient’s respiratory pattern, and for safety, anti-asphyxia protection is needed. If using the latter high flow technique with a semi-open system, whilst the asphyxia concern is lifted, this approach requires high gas flows, potentially placing high demands on medical gas supplies, especially if the clinician wishes to give higher oxygen concentrations or specialist therapeutic gases. Even then, the clinician lacks real time knowledge that the flow into the device is adequate to control the breathing environment, for all patients, all the time.
This introduces the concept that feedback from the device to the clinician could be helpful for delivering efficient accurate gas administration. The other issue is that no two patients are the same; their faces are different, their respiratory systems have different volumes and capacities. Thus, the administration device and its practical utility need to take this into account.
Closing the gaseous environment that the patient breathes from, to control the inhaled gas, can be achieved using a mouthpiece with a nose clip and an appropriate breathing system, or else a hood. Mouthpieces, however, bypass the nose and invariably require the patient to use fatigable skeletal facial muscles, to ensure correct continued use. Hoods have high dead space, reducing feedback, and require high gas flows, reducing efficiency.
A different approach is to take a non-rigid, inflatable, soft mask, which is designed to mould to the bony skeleton of the face and upper neck tissues to create a bulk seal, around the nose and mouth. Next, all the gas to be breathed is pushed up into and against that bulk seal, to ensure that it is closed, using a discrete easily visualised inflatable conduit, before that gas does anything else. Then, given that that gas has not escaped, it is free to flow into a storage reservoir bag and be breathed by the patient, via an inner breathing chamber bounded by one-way valves and the seal just so tested.
Methods
An oro-nasal mask has been designed incorporating these features (Figure 1).5,6 The mask is non-rigid, using a shaped, soft, deformable foam material (memory foam in current prototypes), compression moulded against the facial bony skeleton and upper neck tissues to create a bulk seal. This seal is continuously tested by the total inflow of gas entering the mask, via a discrete inflatable circumferential conduit in front of the compressed foam. This conduit vents under the foam where it bears against the face, such that when the mask is fitted correctly the vents are largely closed and a microscopic space with high outflow resistance is created, just beyond the vents, which is flooded by the gas to be administered (Figure 1, left panel). If this dynamic molecular seal is good, then atmospheric air is excluded and additionally the gas to be administered does not leak out, allowing it to proceed around the conduit and through a connector into the reservoir bag. An anti-asphyxia valve kept shut by the gas flow through the connector ensures safety. Inspiration is from an inner breathing chamber, fed via a one-way valved pathway from the reservoir bag. Expiration is via a simple low-pressure valve to atmosphere. The key point of use is to deliver a flow of gas, such that the reservoir bag fills completely and promptly during expiration (Figure 1, right panel), as this is when the reservoir bag’s filling best reflects the mask’s seal. This is the feedback to the clinician that the “moulded gas interface” is sound (device closed to atmospheric air) and the gas flow adequate for the patient’s respiratory pattern. The reservoir bag should be of adequate volume for the patient (at least two litres for an adult).
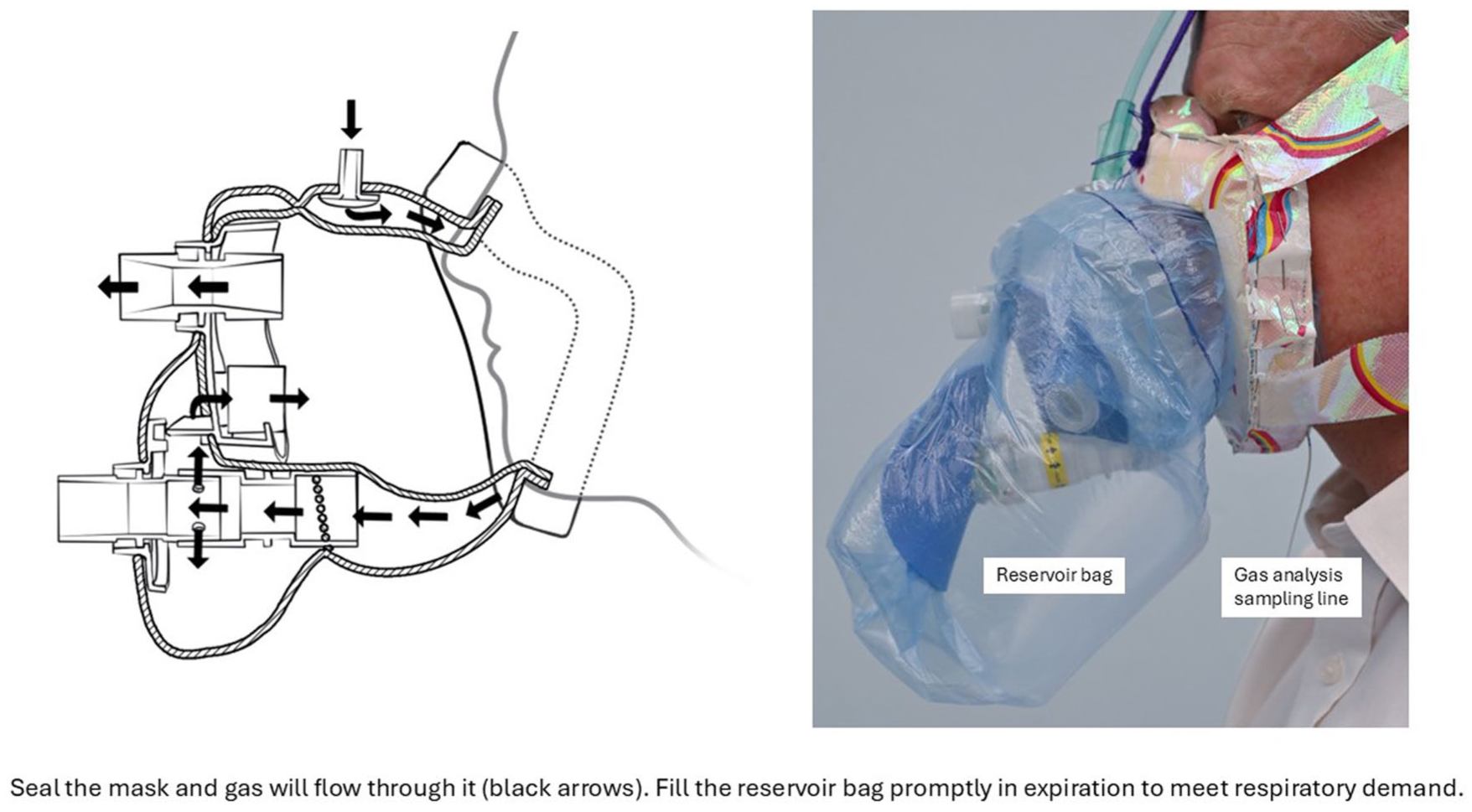
Schematic diagram of proof-of-concept moulded gas interface mask (left panel) with the experiment prototype pictured in expiration (right panel).
Multiple experiments led to proof-of-concept prototype masks. One such prototype experiment is presented, using a prototype mask (Figure 1, right panel). Whilst, quantitative mask fit testing involves controlled negative pressure manoeuvres or the lack of ingress of aerosols into masks using condensation nuclei counting, 7 for the purpose of testing exclusion of atmospheric air, nitrogen is an abundant and suitable choice to assess mask performance; it has a small molecular size (28 atomic mass units, ~0.3 nm in diameter), a large presence in atmospheric air and is metabolically inactive. Gas analysis (Figure 1, right panel) was undertaken at the author’s mouth, by a multichannel (helium, nitrogen, oxygen, argon, carbon dioxide) mass spectrometry gas analysis system (HPR-20; Hiden Analytical, Warrington, UK) calibrated to atmospheric air, with calibration check post experiment. Prototype mask performance was tested by introducing cylinder medical oxygen (BOC Medical Ltd., Woking, UK) into the device, with gas flow adjusted to the mask’s visual appearance. Cylinder contents were measured before (control) and after experiment (drift).
Results
Calibration and drift error of nitrogen on atmospheric air was low (78.04% (actual 78.08%), falling to 77.55% post experiment). Drift of nitrogen on medical oxygen was low (0.11% falling to 0.08%, drift −0.03%). Test data was corrected for drift for all gases using medical oxygen drift data corrected for time. Prototype test results are given in Figure 2 and Table 1.
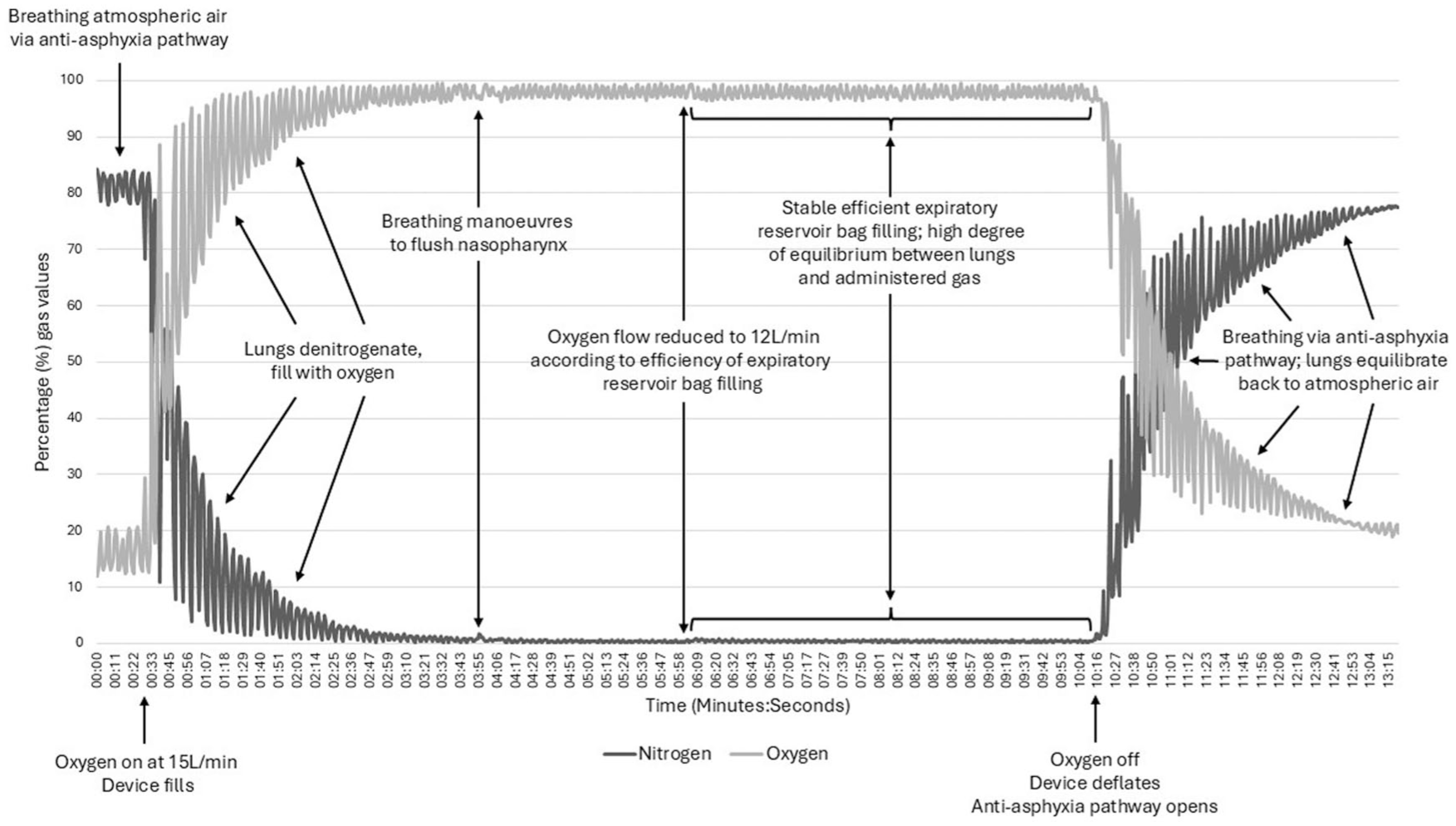
Performance of proof-of-concept moulded gas interface mask. Medical oxygen at 15 L/min from 30 s, reduced to 12 L/min at 6 min according to mask appearance, stopped at 10 min 15 s opening anti-asphyxia valve.
Percentage (%) nitrogen values at the mouth during moulded gas interface mask operation compared to percentage nitrogen in the medical oxygen being delivered into the mask.

Discussion
The moulded gas interface mask presented here has been designed as a universal concept, to give the clinician the ability to accurately administer the gas that a self-ventilating patient needs in terms of both oxygen (± carrier gas) content and volume. The inflation of the device is primarily dependent on the quality of the mask’s interfacing seal, assessed by the continuous total flow of gas going into the device. Providing the seal is sound, the flow into the device can be adjusted against the patient’s respiratory demand, according to the dynamic of inflation of the reservoir bag, during expiration.
Thus, the continuous flow of gas into the device gives continuous visual feedback as to the adequacy of both the mask’s seal and the medical gas volume provision; something no other simple oxygen therapy device currently does. If the clinician selects the gas for the patient to breathe and fills the device adequately; then the patient wears the device and breathes. The resultant accuracy, supports gas therapy against clinical indications, be it oxygen in respiratory failure or potentially specialist gases against specific indications, for example, inert gases to protect the brain. 8 Additionally, the ability to control the environment in front of the patient’s airway from atmospheric air, should have infection control benefits.
The work presented in this paper is early proof-of-concept only; further prototype development and testing is required. Key concerns are ease of use and patient comfort and tolerance. Early prototyping is encouraging, with good seals achieved with light compression of soft upholstery foams and easy lifting of the inferior interface margins with quick-release clips on the bottom straps, to allow drinking and mouth care. Compressing infra-orbital tissues can lead to blocking of inferior visual fields, but neck movement is preserved and patient vision is otherwise maintained. Passage of room temperature gases at ambient humidity through the device causes no chamber condensation. The inner chamber offers the possibility for additional passive warming and humidification.
The hypoxic patient on simple oxygen therapy demands clinical decisions surrounding ongoing management. Multiple factors inform these, but early appreciation of disease type and disease severity is key. Assessment of oxygenation is fundamental here and requires knowledge of the fractional inspired oxygen concentration. 9 Being able to simultaneously support the patient’s arterial oxygen saturation appropriately, 3 buys valuable time to put appropriate interventions in place, which can then be assessed for benefit and inform decisions on escalation. The clinician can also avoid the harms of hyperoxia. 10 Overall, such an approach, should improve the system for intensive care teams to safely assess, support, and rescue patients in respiratory distress.
Footnotes
Acknowledgements
The author is grateful to the Division of Anaesthesia, Department of Medicine, University of Cambridge for the loan of the HPR-20 Gas Analysis System, to BOC Medical (Linde plc) for their support of original research (see external funding) and the later use of laboratory space at The Priestley Centre in Guildford, UK. The drawing in ![]() was supplied by i4 Product Design Ltd, Edinburgh and the photograph in Figure 1 was supplied by Mr. Jon Miles. The author is also grateful for the support of the Departments of Research and Innovation, Theatres and Anaesthetics, and Electrical and Biomedical Engineering (EBME) at Worthing Hospital, University Hospitals Sussex NHS Foundation Trust.
was supplied by i4 Product Design Ltd, Edinburgh and the photograph in Figure 1 was supplied by Mr. Jon Miles. The author is also grateful for the support of the Departments of Research and Innovation, Theatres and Anaesthetics, and Electrical and Biomedical Engineering (EBME) at Worthing Hospital, University Hospitals Sussex NHS Foundation Trust.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author’s research leading to the award of an MD degree (University of East Anglia, 2012) was supported by an unrestricted educational grant from BOC Medical and an Inspire Award from The Linde Group (BOC Medical).
Declaration of conflicting interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author is the Director of Sallow Pightle Ltd., the owner of the patent and patent application listed in the references.
Supplemental material
Both the UK patent and the PCT application have been published and are available to view at www.ipo.gov.uk/p-find-publication and ![]() , respectively, using the numbers listed in the manuscript references (patent GB2625567, UK, 2024 and patent application PCT/GB2023/053281, International, 2023).
, respectively, using the numbers listed in the manuscript references (patent GB2625567, UK, 2024 and patent application PCT/GB2023/053281, International, 2023).
