Abstract
Advanced heart failure is an entity where irreversible structural heart disease is associated with persistent, refractory symptoms and quantitative decrease in cardiopulmonary capacity. Despite advanced and comprehensive medical therapy, patients are at high risk of death due to cardiogenic shock or cardiac arrest. Heart transplantation was developed as a surgical intervention to replace the failing recipient heart with a healthy heart from a recently deceased donor. In this article, we discuss the current state of cardiac transplantation, more than 5 decades after the first human cardiac transplantation was performed. Apart from an historical overview of the development of surgical techniques, we focus on appropriate patient selection, pretransplant evaluation, and recognition and treatment of post-transplant complications.
Introduction
Heart failure is a major public health burden with an estimated global prevalence of over 26 million, accounting for substantial economic loss.1, 2 Severe advanced heart failure or ACC/AHA stage D heart failure is estimated to be present in 1 to 2 million people worldwide, and these patients are at high risk of death (up to 75% in 1 year) in the absence of advanced therapies (ventricular assist device or cardiac transplantation).3-5
Patients with advanced heart failure need systematic evaluation and timely intervention. Cardiac transplant remains the gold standard treatment for these individuals, but cardiac transplantation needs careful patient selection to ensure good outcomes. With the development of dedicated advanced heart failure centers across the world, the number of patients deemed to be transplant candidates now outstrip the number of available deceased heart donors globally (approximately 5,000 every year). 6 For patients who are not transplant candidates, and for those on a transplant waitlist, durable ventricular assist devices have proved to be viable alternatives. Therefore, advanced heart failure centers utilize exhaustive criteria to identify optimal transplant candidates, to whom available donor hearts can be allocated.
In this review, we discuss the evolution of the heart transplant procedure and the art of transplant medicine in the 21st century (section I). Additionally, we discuss the indications, pretransplant evaluation, and post-transplant care in the Indian context (Section II).
Section I: 50 Years of Heart Transplantation
Heart Transplantation: Historical Perspective
After several animal experiments over the course of the early 20th century, the first successful human orthotopic heart transplant was performed by the eminent Dr Christiaan Barnard in Cape Town, South Africa on December 3, 1967.7, 8 The recipient was a middle-aged male with advanced ischemic cardiomyopathy and the donor was a young woman who was the victim of a road traffic accident. This initial patient survived 18 days and ultimately died of pneumonia. The first heart transplant in the United States was performed at Stanford University just about a month later. Remarkably, over the next 12 months, over 100 heart transplants were performed globally. 9 However, initial outcomes were plagued by cardiac graft rejection and graft failure which was inadequately understood in the early days. Subsequently, under the leadership of the pioneering Dr Shumway at Stanford University, the cardiac transplant team determined clinical, electrocardiographic, and echocardiographic criteria to diagnose acute cardiac rejection. With institution of appropriate immunosuppressants for cases of rejection, improved survival was achieved in the early 1970s (49% survival at 6 months, 30% at 2 years). 9
In 1973, the Stanford Group instituted the use of routine and frequent endomyocardial biopsy in the post-transplant phase for histological confirmation of rejection and appropriate use of immunosuppression. Along with early detection of rejection, one of the most important advancements in heart transplant care was the in vitro development of cyclosporin A. The effective immunosuppressive action of Cyclosporin was confirmed in vivo in porcine cardiac allografts, 10 and subsequently incorporated into practice at Stanford Hospital. By 1985, 1-year survival rates of 83% were being achieved as a result of standardized post-transplant care and improved immunosuppression to prevent rejection. 9 These refinements in post-transplant care allowed cardiac transplantation programs to grow across the world, until the mid-1990s after which the annual global transplants have levelled off due to limited supply of donor hearts. 9
Orthotopic Versus Heterotopic Heart Transplantation
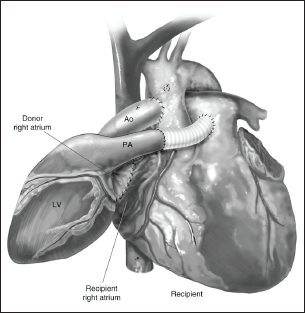
Over the years, 2 techniques of heart transplantation have been developed in animal models and then in humans. The first human transplant was an orthotopic transplant, in which the donor heart is placed in the correct anatomical position by removing the recipient heart. Subsequently, the heterotopic heart transplant was attempted in humans (developed in the early 20th century in animal models), in which the recipient heart is left in situ and the donor heart is “piggy-backed” to the recipient organ and placed to the right side of the recipient heart (Figure 1). 11 The rationale is that the donor heart functions as an “assist heart” for the recipient. Heterotopic heart transplantation was first performed in humans by Dr Barnard in 1974 effectively to function as a “biventricular assist device”—it was primarily to support the native diseased heart. 12 Heterotopic heart transplant was also used for severe pulmonary hypertension, once again to function as a “right ventricular (RV) assist device.” There was also a school of thought to utilize the donor heart in heterotopic fashion wherein despite the occurrence of acute rejection and graft dysfunction, the residual native heart function would keep the patient alive. However, since the advancements in immunosuppressive regimens and decrease in acute cardiac graft rejection, the use of heterotopic heart transplantation has significantly reduced. 13 Additional indications for heterotopic heart transplants include small donor hearts (where the donor heart alone may not meet the recipient’s circulatory requirements) or compromised donor hearts. Today, heterotopic heart transplants are used only by some programs as a biological left ventricular (LV) assist device. Over the years, outcomes with heterotopic heart transplants have not been as good as orthotopic heart transplants mainly because it is a technique used with marginal donor organs (suboptimal donor heart) transplantation. 11
Surgical Techniques of Orthotopic Heart Transplantation
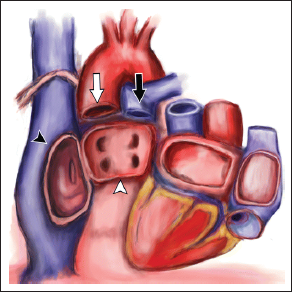
Over the past 5 decades, one major change in the surgical technique of orthotopic heart transplantation is the transition from the standard (biatrial) transplantation to the current standard of bicaval anastomosis. 14 The biatrial method entails excision of the native heart while leaving behind a cuff of right and left atrium as well as the aorta and pulmonary artery (Figure 2). 15 The donor heart (which is harvested from the donor just prior to the transplant surgery and maintained in an ice box to prevent ischemia) is then prepared and anastomosed to the native left atrial cuff. Subsequently, the donor right atrium is anastomosed to the native right atrium such that the native superior vena cava (SVC) and inferior vena cava (IVC) drain blood into the donor heart. The benefit of the biatrial technique is that it is simple; in addition to connections to the native aorta and pulmonary artery, it requires only anastomosis to the 2 atria. However, these very suture lines along the right atrium (running vertically from the SVC to IVC) and those in the posterior left atrium are sites of incisional atrial arrhythmias. Additionally, there is a higher rate of sinoatrial node injury from the right atrial anastomosis and permanent pacemaker implantation (up to 10%). 16
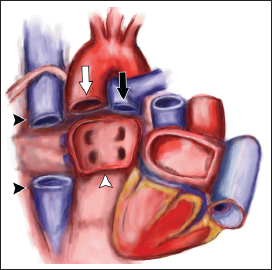
These limitations led to the development of the newer bicaval technique, developed in the early 1990s. 17 In this technique, the native SVC and IVC are divided and sutured to the donor cavoatrial junctions (Figure 3). 15 Therefore, there is no suture line in the donor right atrium per se and the permanent pacemaker implantation rates are lower (around 5%). Additionally, the bicaval technique appears to have slightly improved 30-day survival (odds ratio 0.83, confidence interval 0.75-0.93). Trends in heart transplants suggest that in the modern era, more than two-thirds of orthotopic heart transplants utilize the bicaval method in the 21st century. 16
Ischemic Time, Organ Preservation, and Future Directions
When a donor heart is procured, it is arrested with cold cardioplegia and then transported (in an ice box) to the operating room where the recipient heart has been resected. After completion of cardiac transplantation, the heart-lung machine is discontinued and reperfusion is initiated. The time taken from donor heart arrest to reperfusion in the recipient’s body is called the “ischemic time.” The duration of ischemic time predicts recipient survival and traditionally, the goal is to keep ischemic time <4 h, particularly for older donor hearts. Recent studies have confirmed that in younger donor hearts, prolonged ischemic times can be tolerated and does not affect long-term survival. 18 Therefore, while assessing a donor, both the donor age and anticipated ischemic time are to be considered. This is particularly relevant when out-of-city donor hearts are utilized; our preference is restricting to utilization of younger donors. Additionally, for interinstitutional transfers in India, utilization of “green corridors” has been helpful in decreasing ischemic time. 19
During transport, hypothermia (using an ice box) is the primary mode of organ preservation. Various cardioplegic solutions have also been studied to rapidly achieve noncontractility and minimize tissue ischemia and to protect the organ during transport. Commonly used cardioplegic solutions include University of Wisconsin or Custodial HTK (histidine-tryptophan-ketoglutarate solution). Our transplant program uses the HTK solution which is a low viscosity, low potassium-containing solution with lower cost than the University of Wisconsin solution.
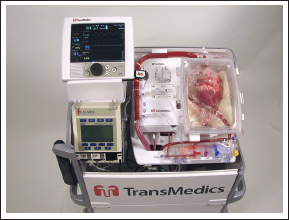
A new development in the area of organ transport is the development of donor hearts in a warm, beating state as opposed to the current practice of hypothermic, arrested state. Utilizing the Organ Care System Heart System (TransMedics, Andover, Massachusetts, USA) ex vivo perfusion system, hearts can be transported for longer durations with less concern about ischemic time (Figure 4). 20 In a randomized trial, 30-day post-transplantation survival with ex vivo perfusion was noninferior to standard techniques. 21 This technology holds great promise in unifying donor pools across geographic areas and maximize donor heart utilization.
Another area of considerable research is the use of donor hearts from patients with circulatory death (as opposed to current practice of neurological death). This so-called practice of “donation after cardiac death (DCD)” is from patients with irreversible brain injury, but who technically do not meet criteria of brain death. Life support is withdrawn and organs are retrieved after circulatory death. While DCD potentially can bring in more donor hearts into the pool for allocation, challenges facing DCD use include ethical dilemmas surrounding withdrawal of care, and ischemic injury to the heart after circulatory death. Currently, this practice has been well studied in Australia and UK,22, 23 but is not legally allowed in India.
Current Status of Heart Transplantation in India
Although the first heart transplant in India was performed in 1968, this was unsuccessful and the first successful heart transplant was subsequently performed by Dr Venugopal at the All India Institute of Medical Sciences in 1994. Transplant volumes remained low, until the past decade where there has been a steady rise in cardiac transplant numbers. This has been possible due to the establishment of dedicated advanced heart failure centers, particularly in South India where organ donation is more readily available. Currently, transplant programs have been established in some capacity in about 17 states and union territories. A major impetus for organ transplantation in India was provided by the setting up of the National Organ and Tissue Transplant Organization (NOTTO) in 2014. The goal of the NOTTO is to integrate data from state-level organ donation programs and create a national donor and recipient registry to achieve transparent allocation. Essentially, the NOTTO functions similarly to the United Network of Organ Sharing (UNOS) system in the United States. Based on NOTTO data, there has been a year-on-year increase in the number of heart transplants in India and was reported at 241 in 2018. While the UNOS system in the United States utilizes 5 categories of heart transplant listings (status 1: sickest patients on mechanical circulatory support, status 5: combined organ transplant) in order to allocate hearts to the sickest patients, NOTTO emphasizes 3 categories (priority 1: emergency, priority 2: semi emergent, and priority 3: elective). Additionally, in India, state-level government organizations (Jeevandan-Telangana, TRANSTAN-Tamil Nadu, Jeevsarthakathe-Karnataka, Mrithasanjeevini-Kerala) work in complement with NOTTO regional authorities to further strengthen transplant allocation.
Despite the remarkable development in heart transplant programs and increase in surgical volumes, several challenges plague the advanced heart failure medical fraternity. Low levels of awareness are responsible for small organ donation pools; intercity and intracity organ transport can be challenging; rigorous protocols for post-transplant rejection surveillance are rarely followed; and finally financial challenges are often an obstacle at every step. Public-private partnership is one solution to overcome some of these barriers and nongovernmental organizations (such as the MOHAN Foundation) have been advocating for improved coordination, awareness, and arranging financial aid.
Section II: Pre- and Post-Heart Transplant Management
Indications and Workup Prior to Transplantation
Indications for Cardiac Transplantation.

There are 2 main pathways leading to cardiac transplantation for patients with advanced heart failure (a) progressive, advanced heart failure failing outpatient therapies, and requiring frequent hospitalization and/or chronic inotropes and (b) acute decompensation with cardiogenic shock of a patient with known advanced heart failure or de novo cardiogenic shock that requires urgent cardiac transplantation, with or without temporary mechanical circulatory support.
INTERMACS (Interagency Registry for Mechanically Assisted Circulatory Support) Profiles.
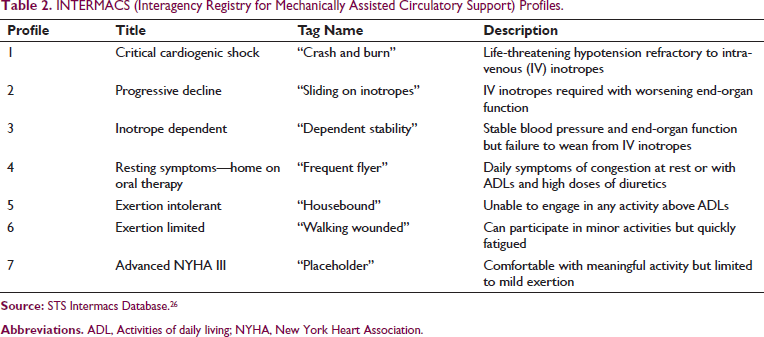
Contraindications to Cardiac Transplantation.

In patients where a transplant is deemed appropriate, further assessment is aimed at the ability to successfully undergo transplantation with good outcomes. 29 Assessment of functioning of noncardiac organs is essential and a panel of investigations is obtained (laboratory testing, urinalysis, 24-h urine creatinine and protein, pulmonary function testing, chest radiography/chest computerized tomography scan, abdominal ultrasonography, carotid dopplers, and ankle-brachial indices as part of a comprehensive assessment). Additionally, infectious serology is routinely obtained (hepatitis B surface antigen and antibody, hepatitis C antibody, HIV, cytomegalovirus, Epstein-Barr virus, varicella zoster, and immunoglobulin G for herpes simplex virus). Vaccinations (influenza, pneumovax, hepatitis B) should be up to date to avoid poor post-transplant outcomes secondary to preventable infections. If clinically indicated, preventive screening for common malignancies should be considered (colonoscopy, mammography, Pap smear test, and prostate specific antigen). Special attention is paid to high-risk behavior (tobacco, alcohol, and recreational drug use), the candidate’s social support and financial assessment to ensure compliance with post-transplant treatments. Mental health evaluation is also a key component of the initial evaluation. 30
Finally, immunocompatibility assessment with ABO blood group typing and human leukocyte antigen (HLA) tissue typing is essential. Other techniques include panel reactive antibody and donor-specific antibody testing. The goal of this profiling is to match a donor against whom the recipient does not have alloantibodies as this would lead to a humoral response against the cardiac allograft (donor heart) after transplantation. 29
Occasionally, combined organ transplantation (heart-lung, heart-kidney, heart-liver, heart-liver-kidney, either simultaneously or sequentially) may need to be considered for patients with end-stage multiorgan dysfunction. Patient selection for combined transplants is even more challenging than for heart transplant alone, and individualized, multidisciplinary decision-making is critical for choosing which patients with multiorgan failure would benefit from a multiorgan transplant. 30
Current Immunosuppression Therapy Following Transplantation
Cardiac transplantation became an acceptable procedure for end-stage heart disease only after the refinements in immunosuppressive therapies in the 1980s, heralded by the discovery of cyclosporine. 14 Innate immunity, T-lymphocytic cells, and B-lymphocytic cells are all important in the immune response and rejection of the cardiac allograft by the recipient’s body. To mitigate this response, 3 categories of immunosuppression are used—(a) induction, (b) maintenance, and (c) rejection management. 14
Induction therapy consists of intensive perioperative immunosuppression to modulate and diminish the acute immune response to the cardiac allograft. Commonly used induction agents are anti-T cell agents (antithymocyte globulin, alemtuzumab) and interleukin-2 receptor antagonists (basiliximab). Not all transplant institutions routinely use induction regimens, and often induction agents are restricted to recipients who are allosensitized to HLA antigens. 31 Our practice is to administer one dose of basiliximab perioperatively.
Immediately after cardiac transplantation, most programs including ours, incorporate a combination of corticosteroids (prednisone), calcineurin inhibitors (tacrolimus has largely replaced cyclosporine today because of less adverse effects), and antimetabolite agents (mycophenolate mofetil is preferred as it has better outcomes than azathioprine). Since long-term corticosteroid use is associated with many adverse effects, most patients have tapering doses prescribed and steroids are often weaned off by 12 months after transplantation. Main adverse effects related to Tacrolimus is nephrotoxicity, hypertension, and dyslipidemia. In those patients with kidney injury post-transplant or those with coronary artery vasculopathy (CAV), tacrolimus may need to be substituted with mTOR inhibitors (sirolimus or everolimus). 14
Finally, in patients with acute cardiac rejection, determination of cellular rejection versus antibody mediated rejection is key. 32 Cellular rejection is treated with pulse steroids whereas antibody-mediated rejection requires the use of specific drugs such as rituximab, bortezomib, and eculizumab. Given the wide spectrum of medicines used in immunosuppression and their adverse effect profile and drug-drug interactions, transplant cardiologists, surgeons, and pharmacists have to be aware of nuances of drug combinations.
Post Transplantation Follow-up and Management
The immediate post-transplant phase is critical in supporting the donor heart as donor heart dysfunction can occur in 50% of the patients. Hemodynamic monitoring and support is essential and the International Society of Heart and Lung Transplant (ISHLT) recommends intraoperative transesophageal echocardiography to evaluate donor heart function, and continuous monitoring with an intra-arterial line, pulmonary artery catheter, and cardiac telemetry monitoring in the critical care unit. 33 Inotropic and vasoconstrictor drugs are frequently utilized to support the donor heart. Preferred drugs include isoproterenol, dopamine, dobutamine, and milrinone as they augment cardiac contractility and avoid afterload increases seen with norepinephrine and epinephrine. Management in the postoperative period requires prompt detection and treatment of early complications such primary graft dysfunction (either LV/RV/biventricular dysfunction), cardiac tamponade, and RV dysfunction in the setting of high pulmonary vascular resistance. Temporary mechanical circulatory support may be needed for LV or RV dysfunction and allows the donor heart to recover from dysfunctional state and serves to salvage the heart transplantation.33, 34
The goal of long-term post-transplantation follow-up protocols is to ensure cardiac graft function and early detection of complications (graft rejection and CAV) and the adverse effects that stem from prolonged immunosuppressive therapy (infection, hypertension, diabetes, chronic kidney disease, malignancy). To this effect, in the early post-transplant phase, endomyocardial biopsies are performed routinely to detect cellular rejection (weekly in the first month, every 2 weeks in the 2nd month, monthly until 6 months, and then the 9th month and 12th month post-transplant). After 1 year, the risk of cardiac rejection is much lower and the frequency of biopsies is usually patient-tailored and center-specific. With regard to CAV, the gold standard of testing is coronary angiography with intravascular ultrasound. Often, this is performed annually to detect early CAV. 14 Additionally, periodic laboratory evaluation of blood counts, renal, and hepatic function is necessary to detect immunosuppression therapy-related adverse effects.
Common Long-Term Complications
With advances in surgical technique, organ preservation, refinements in donor selection, optimal patient selection and immunosuppressive regimens, and median survival after cardiac transplantation in the 21st century exceeds 12 years. 35 Timely recognition and management of complications expected after cardiac transplantation is exceedingly important to improve the quality of care and survival. The most common long-term complication after cardiac transplantation continues to be rejection (20% at 1 year) and this may manifest as graft failure, cardiac arrhythmia, and decompensated heart failure. It may also be asymptomatic and be detected on a routine endomyocardial biopsy. The cornerstone of therapy is induction therapy and augmentation of maintenance immunosuppression regimen. 30
CAV detected by routine coronary angiogram and/or intravascular ultrasound is the second-most common post-transplant complication (8% at 1 year). Increasingly, coronary computed tomographic angiography is being recognized as an alternative, less expensive, safer option for detection of CAV and may become the first-line screening tool in heart transplant recipients in the future. 36 Patients on tacrolimus may need an alternative immunosuppressive drug and percutaneous coronary intervention or bypass surgery should be pursued as clinically indicated. Infrequently, cardiac retransplantation has to be pursued as the only option for severe CAV with nonrevascularizable lesions.
Infection in immune-suppressed patients remains an area of concern, therefore prevention is key. All age-appropriate vaccinations should be completed prior to transplantation if possible. Use of live vaccines is typically contraindicated following transplant due to ongoing immunosuppressive therapy. In the midst of the coronavirus disease-2019 (COVID-19) pandemic, whenever possible the entire vaccination should be performed prior to transplantation. For COVID-19 vaccines, and in fact any other vaccine, urgent cardiac transplantation should not be delayed. Instead, the vaccination schedule should be restarted at least 1-month after transplantation. 37 Apart from vaccination, post-transplant prophylaxis against cytomegalovirus, Pneumocystis jiroveci, Candida species is also routinely utilized.
Long-term immunosuppression also portends a slightly higher risk of malignancy. Although, any malignancy can occur, skin cancers are the most common malignancies encountered in post-transplant patients. Post-transplantation lymphoproliferative disorder is a specific form of lymphoma that needs to be suspected in patients undergoing surveillance. Early detection of malignancy with age-appropriate screening is key to minimizing mortality secondary to malignancies. 38
Additional complications that warrant careful monitoring and treatment include chronic kidney disease, hypertension, diabetes mellitus, and incisional atrial arrhythmias.
Management of Cardiac Transplant Patients in the COVID Era
A new challenge that has arisen for cardiac transplant programs globally is the advent of the COVID-19 pandemic. Data from New York and Northern Italy have demonstrated high fatality rate (25%-29%) in transplant patients on immunosuppression contracting COVID-19.39, 40 As such, greater vigilance is advised and COVID-19 management is similar to nontransplant patients. For patients with moderate-severe illness, immunosuppression (mycophenolate mofetil, mTOR inhibitors, or azathioprine) may be held in the short term without risk of cardiac rejection. 37 Steroids can be safely used when indicated and for the most part Remdesivir has no significant drug interactions with immunosuppressive medications (may lower tacrolimus or cyclosporine levels).
In our own experience at the KIMS Heart and Lung Transplant Institute, we have treated 3 heart transplant recipients with COVID-19. Two had mild disease and were closely monitored at home and 1 was admitted for Remdesivir and supportive treatment. We have also continued to perform selected heart transplants through the pandemic, with critical decision-making on a case-by-case basis—this practice is consistent with that of international centers and supported by ISHLT guidance.37, 41
Conclusion
Heart transplantation is the gold-standard treatment for patients with end-stage heart failure, and the journey of the transplant process is studded with remarkable advances. In the 21st century, it continues to be preferred over LV assist devices, particularly in resource-sensitive countries like India. For any program to achieve successful outcomes, appropriate evaluation and selection of transplant recipients is a key step. To the extent possible, advanced heart failure therapies (medical, device, interventional, and surgical) must be exhausted prior to considering heart transplantation as the supply-demand mismatch in the transplant process is a bona fide problem. Following transplantation, in order to achieve long-term survival, a comprehensive, team-based approach is needed to detect and treat the various post-transplant complications. Therefore, best outcomes are achieved by high-volume, dedicated transplant centers, with qualified and knowledgeable staff who are invested in patient-centered care.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
