Abstract
Advanced heart failure (HF) with persistent and progressive clinical decline despite maximal medical therapy portends a high mortality in the absence of advanced therapies, such as ventricular assist devices or heart transplantation. A subset of these advanced HF patients deteriorates into refractory cardiogenic shock, that is challenging to manage with vasoactive agents alone. Temporary mechanical circulatory support (MCS) device options have evolved over the years and provide a viable option to rescue and rest the myocardium of patients in cardiogenic shock. The goal of such therapy is to serve as bridge to recovery, or more often, a bridge to durable advanced therapies. For those patients with progressive advanced HF despite extensive medical therapy, durable MCS devices (ventricular assist devices) are available when heart transplantation is not feasible. In this article, we review currently available temporary and durable MCS devices, with a focus on their hemodynamic profiles, to inform optimal device selection for patients with advanced HF.
Keywords
Introduction
Heart failure (HF) is a progressive clinical syndrome with an estimated global prevalence of over 26 million. 1 Over the past 2 decades, remarkable progress has been made in terms of pharmaceutical therapies for HF, particularly for those with HF reduced ejection fraction (HFrEF). Yet, in patients with advanced refractory HF (classified as stage-D HF by the American College of Cardiology [ACC]), medical therapy alone is often inadequate and associated with high mortality. It is estimated that approximately 5% to 10% of the HF population has stage D HF. 2 This translates to several hundred thousand Americans and Europeans.3, 4 Limited data are available for the Indian population, and have demonstrated high mortality (59% at 5 years) for patients hospitalized with HF. 5
Stage D HF patients warrant multidisciplinary evaluation and in addition to maximally tolerated guideline directed HF-specific medical therapy, they also benefit from concomitant percutaneous or surgical interventions to address underlying coronary stenoses, atrial, or ventricular arrhythmias, severe valvular stenosis or regurgitation and cardiac dyssynchrony. 6 Universally, advanced HF patients pose a heavy clinical and economic burden on global health-care systems. In a 2012 study of 197 countries, it was estimated that HF care results in an economic loss of $108 billion annually. A significant portion of this estimate is indirect costs and productivity loss from premature death, prolonged hospitalization, and disability. 7
While heart transplantation is the treatment of choice for stage D HF patients, the number of global transplants per year (∼5,000) is several folds lower than those eligible for heart transplantation. 8 With medical therapy alone, 1-year mortality stands at 75% for stage D HF patients. 9 The alternative option in such patients is to consider durable mechanical circulatory support (MCS)/left ventricular assist devices (LVAD) either as destination therapy (DT) or as a bridge to transplantation. 9 In a subgroup of patients, who present with cardiogenic shock, these patients may need temporary MCS in order to survive until more durable therapy (LVAD or transplant) can be offered. In this review, we discuss the percutaneous and surgical MCS options currently available for advanced HF.
Spectrum of Advanced Heart Failure
INTERMACS (Interagency Registry for Mechanically Assisted Circulatory Support) Profiles.

Outpatient Management of Advanced Heart Failure
ACC/AHA Stages of Heart Failure.

Patients in stage D HF often need referral to specialized HF/transplant programs where the diagnosis can be reviewed and verified, medications can be optimized, and detailed evaluation for advanced therapies can be pursued. Progressive clinical deterioration in functional capacity, multiple HF admissions, requirement for chronic inotropes, decreased tolerance of HF medications due to hypotension, worsening renal function, recurrent arrhythmias, and defibrillator shocks are some clinical triggers for referral to an advanced HF program or institute. In addition, stage D HF patients are at high risk for deterioration into cardiogenic shock at any time which portends a high risk of death and requires timely evaluation and intervention.
Cardiogenic Shock in Stage D Heart Failure
Cardiogenic shock is defined by hemodynamic parameters (systolic blood pressure <80-90 mmHg or mean arterial pressure 30 mmHg lower than baseline), cardiac index <1.8L/min/m 2 without support or <2.2 L/min/m 2 with support and elevated right or left ventricular filling pressures, with clinical signs or symptoms of hypoperfusion. Patients with advanced HF have lower cardiac reserve, higher biventricular filling pressures, and a greater degree of anaerobic metabolism as compared to cardiogenic shock patients with acute myocardial infarction.16, 17 A recent consensus statement by the Society of Cardiovascular Angiography and Intervention proposes a classification schema to identify the severity of cardiogenic shock, and helps rapidly identify critically ill patients who may benefit from temporary MCS. 18
The universal initial therapy for cardiogenic shock in stage D HF consists of inotropic therapy, often combined with vasodilators (to decrease afterload). However, these agents can be ineffective, increase myocardial oxygen consumption, impair tissue perfusion, and increase the risk of arrhythmias. 19 Accordingly, in many cases medical therapy alone is unlikely to suffice as a bridge to durable treatment for cardiogenic shock in advanced HF. Various temporary MCS options exist to support patients with cardiogenic shock and stage D HF. The goal of temporary MCS is to primarily serve as a bridge to advanced HF therapies (durable LVAD/transplant) and in a small percentage of patients serves as a bridge to recovery from cardiogenic shock. Figure 1 shows the interplay between outpatient and inpatient evaluation for advanced HF therapies such as durable MCS/LVAD.
Patients with progressive advanced heart failure (HF), classified as ACC/AHA stage C and stage D HF may worsen clinically and need evaluation for durable mechanical circulatory support devices (MCS) in the outpatient setting. These patients may also deteriorate and require hospital admission for cardiogenic shock where temporary MCS may be required. The figure shows multiple possible outcomes where temporary MCS may serve as a bridge to durable MCS or allow for recovery and subsequent outpatient evaluation for durable MCS.
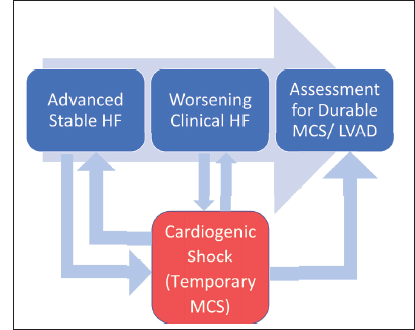
Temporary Mechanical Circulatory Support for Cardiogenic Shock
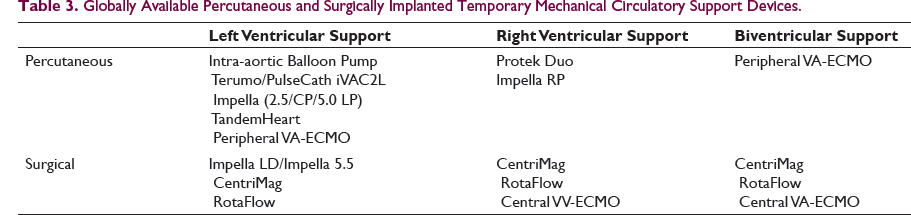
Globally Available Percutaneous and Surgically Implanted Temporary Mechanical Circulatory Support Devices.
Percutaneous Temporary MCS for Left Ventricular Support
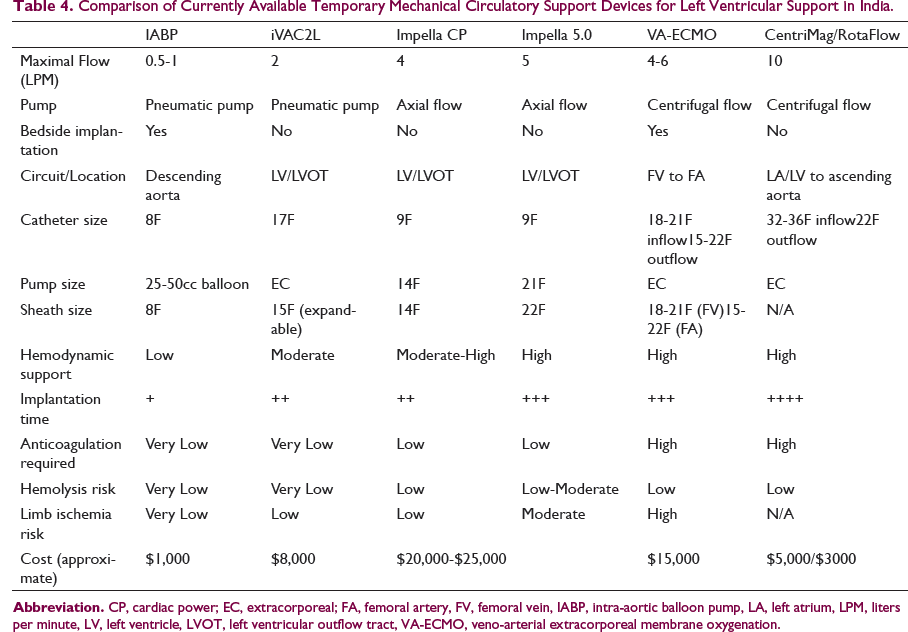
Comparison of Currently Available Temporary Mechanical Circulatory Support Devices for Left Ventricular Support in India.
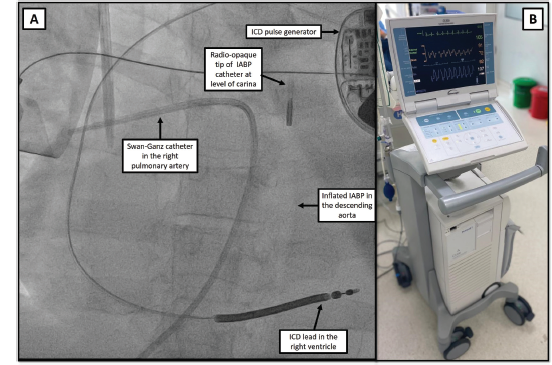
Intra-aortic Balloon Pump (Aortic Pulsatile Pneumatic Pump)
The intra-aortic balloon pump (IABP) has been available for several decades and is manufactured by several companies around the world (Teleflex Corporation, Maquet Holding B.V. & Co. KG. [Getinge Group], Zeon Corporation, Tokai Medical Products Inc, Senko Medical Instrument Mfg Co, Ltd, InterValve Inc, Insightra Medical, Inc). Due to ease of use and low device cost, this device is the most commonly used temporary MCS device. 20 In essence, the device consists of a catheter mounted balloon that is positioned in descending aorta and pneumatically counterpulsed (using helium) in the diastolic period. While the device delivers minimal hemodynamic support (0.5-1 L/min), its usefulness is in the afterload reduction and improved coronary perfusion. Additionally, its hemodynamic effects are helpful in patients with coronary disease and in decreasing afterload for patients already on VA-ECMO. 21 There is also considerable interest in the use of an IABP for longer term use by utilizing an axillary access, which allows for ambulation while awaiting a durable LVAD or heart transplant. 22 Overall, its low-profile catheter size (7-8F) minimizes vascular site complications.
iVAC2L-pVAD (Left Ventricle to Ascending Aorta Pulsatile Flow, Extracorporeal Pneumatic Pump)
The iVAC2L percutaneous ventricular assist device (Terumo Europe, formerly PulseCath) is a single lumen catheter with the catheter tip positioned in the left ventricle and a bidirectional valve positioned in the ascending aorta. It is connected extracorporeally to an IABP console, which powers pulsatile forward flow from the left ventricle to ascending aorta flow. It is able to deliver more flow (2 L/min) than an IABP; however, the catheter size is considerably larger and needs an expandable 15F arterial sheath which increases vascular complications. This device is not sold in the United States and is primarily available in Europe, as well as in India. Its utility is primarily for left ventricular support during high risk percutaneous coronary intervention, but it is can also be used for cardiogenic shock. 23
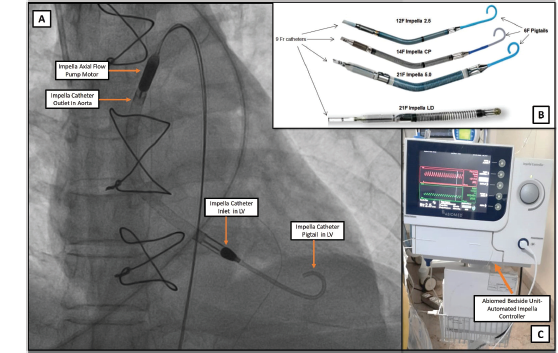
Impella Pumps (Continuous Flow, Microaxial Left Ventricle to Ascending Aorta Pump)
The Impella series of percutaneous devices (Abiomed, Danvers, MA) provide cardiac output augmentation from 2.5 to 5L/min (Impella 2.5, Impella Cardiac Power [CP], Impella 5.0 Left Percutaneous (LP)). The Impella is easy to implant percutaneously (retroaortic implant into the left ventricle) and provides rapid hemodynamic support. Specific to advanced HF and cardiogenic shock, the Impella catheter is useful in unloading the left ventricle and in turn decreasing left and right heart pressures. As with IABPs, the use of axillary Impella implants allow for ambulatory temporary MCS which is attractive for patients with severe HF being bridged to a durable LVAD or heart transplant.24, 25 The 2.5 and CP Impella catheters require 12F and 14F access, respectively, while the Impella 5.0 LP catheter requires a surgical cut-down (21F access). Apart from vascular complications, stroke, hemolysis, and catheter-related ventricular arrhythmias are expected complications. Although this catheter is available for use in India, the major disadvantage is the high cost of the single-use catheter.
TandemHeart (Left Atrial to Aorta Continuous Flow, Extracorporeal Centrifugal Pump)
The TandemHeart device (LivaNova PLC, UK, formerly CardiacAssist, Inc, Pittsburgh, Pennsylvania) is an extracorporeal centrifugal continuous flow pump that diverts blood from the left atrium, bypasses the left ventricle, and diverts blood flow into the femoral artery. The device essentially decreases left-sided cardiac pressures by venting the left ventricle; however, this is partially offset by increased afterload from retrograde aortic blood flow. 26 The TandemHeart cardiac output augmentation is similar to the Impella CP device (up to 4 L/min). However, due to the requirement for transseptal puncture and placement of a large bore cannula in the left atrium, this procedure requires more expertise and time than an Impella catheter implantation. On the other hand, stable inflow cannula placement in the left atrium may allow for more prolonged use as compared with an Impella, where the catheter tip often interacts with structural components of a dynamic left ventricle (papillary muscles, chordae, contracting ventricular wall) and may require more trouble shooting. Additionally, in the event of increasing oxygenation requirements, an oxygenator can be added to the TandemHeart circuit and it simulates a VA-ECMO circuit. Vascular complications due to large bore access (15F arterial sheath, 21F venous sheath), catheter migration, and high device cost are some disadvantages. The TandemHeart is currently not available in India.
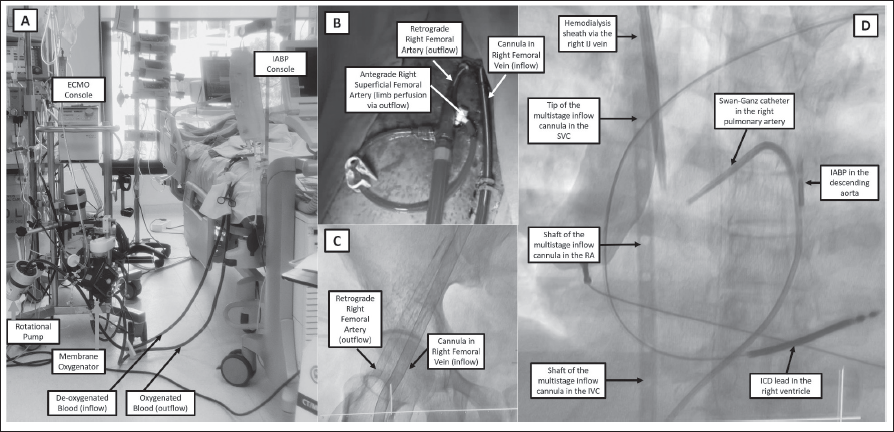
Peripheral VA-ECMO (Right Atrial to Aorta Continuous Flow, Extracorporeal Centrifugal Pump)
The VA-ECMO circuit provides cardiac and respiratory support by oxygenating blood diverted from the femoral vein and subsequently delivering this oxygenated blood into the femoral artery (percutaneous cannulation). Although this extracorporeal centrifugal pump circuit provides up to 4 to 6 L/min continuous flow, it increases afterload and left ventricular workload. 26 Therefore, in cardiogenic shock patients being bridged to durable LVAD or heart transplant therapy, the left ventricle may need to be unloaded by using either an IABP or an Impella catheter (ECPELLA). 26 Although the ECMO device can be placed bedside for an extremely ill, arresting patient, careful attention should be paid to vascular access. Often such bedside cannula placement often is performed by noninterventional cardiologists and as such may be associated with serious vascular complications. Whenever possible, ultrasound and fluoroscopy-guided vascular access should be obtained in a cardiac catheterization laboratory (similar to any large bore vascular access), including the routine use of antegrade perfusion cannulation to prevent limb ischemia. 27 Compared with Impella and TandemHeart, the ECMO setup is less expensive and is the most utilized temporary MCS in India outside of an IABP. Numerous companies manufacture commercially available ECMO machines for heart and lung support (Medtronic plc, Terumo Corporation, LivaNova PLC, NIPRO Corporation, Getinge Group [Maquet], ALung Technologies, Inc, and MicroPort Scientific Corporation).
Surgically Implanted Temporary MCS for Left/Right and Biventricular Support (Impella LD and Impella 5.5 Pumps)
These pumps are designed for left ventricular support and are surgically implanted via the axillary artery or the anterior aorta. These catheters do not have a distal pigtail catheter design unlike the percutaneous Impella catheters, but the flow circuit is identical. The arterial access size is 21F and the pumps deliver 5 and 6 L/min flow, respectively.
CentriMag and RotaFlow (Continuous Flow, Extracorporeal Centrifugal Pump)
The CentriMag left ventricular assist system (Abbott Laboratories, Abbott Park, IL) and RotaFlow (Getinge Group, Maquet Cardiopulmonary AG, Hirrlingen, Germany) are magnetically levitated extracorporeal centrifugal flow pump that can provide flows up to 10 L/min. These devices were principally designed for surgical implantation, and typically require sternotomy for inflow cannulation either in the left atrium or left ventricle. Outflow cannulation is performed in the ascending aorta. Although predominantly, these devices are used for patients undergoing cardiac surgery; however, it has now been adapted for minimally invasive surgical approaches as a short-term bridge to recovery, durable LVAD, or transplant.28, 29 By adding an oxygenator, these devices can simulate a VA-ECMO circuit. The major advantage compared to an ECMO machine is decreased hemolysis since the rotor has no contact with other surfaces (lack of valves, bearings, seals). Both CentriMag and RotaFlow are versatile devices that can be used as an LVAD (left atrial or left ventricle to aorta), RVAD (right atrium to pulmonary artery), or BiVAD (right atrium to aorta) for up to 30 days. Coupled with lower costs as compared to the Impella/TandemHeart, these are very useful temporary MCS devices.
Central VV and VA-ECMO
This involves using the ECMO device with surgically implanted inflow and outflow cannula (intrathoracic right atrial to aorta cannulation for VA ECMO and right atrial to left atrial cannulation for VV ECMO).
Decision-Making for Temporary MCS Device Selection
As illustrated in the section above, numerous options exist for short-term ventricular assistance of either the left ventricle or the right ventricle. A multidisciplinary approach is required, including a team of specialized HF cardiologists, interventional cardiologists familiar with various percutaneous MCS devices, and cardiac surgeons specializing in MCS and transplant surgery. 30 Timely decision-making, prioritization of resources, and comprehensive determination of future advanced therapies candidacy is key in achieving optimal outcomes. Patient-specific factors including vascular anatomy, comorbidities, acuity of cardiogenic shock, as well as procedural factors such as procedural time, available local expertise, and cost of procedures (especially in low- and middle-income countries) are critical factors that determine the choice between percutaneous and surgical temporary MCS options.
Emerging Temporary MCS Devices
There are several emerging temporary MCS options designed for high-risk percutaneous coronary intervention and/or cardiogenic shock due to advanced HF.
HeartMate Percutaneous Heart Pump
The HeartMate PHP (Abbott Laboratories, Abbott Park, IL) is a catheter-based micro-axial flow device that is an alternative to the Impella catheter system. The device serves to unload the LV and is designed to be rapidly delivered retrograde across the aortic valve. Coupled with a unique design that allows for a 24F transvalvular, axial flow pump to be delivered via a 14Fr sheath, this device presents another temporary MCS option for bridge to durable LVAD/heart transplant. 31 This device can provide 4 to 5 L/min cardiac output augmentation and the PHP is potentially a useful percutaneous temporary MCS device, pending rigorous trial data. 32
Impella ECP (Expandable Cardiac Power)
This is the newest iteration of the Impella family of devices and is similar in design to the Heartmate PHP catheter. The sheath used is even smaller in profile (9F) and the expandable pump is unsheathed in the descending aorta to an 18F profile. The pump is capable of peak flows greater than 3.5 L/min. The device is currently undergoing first-in-human early feasibility study, with the initial indication being hemodynamic support for high-risk PCI. 33
Durable MCS for Chronic Stage D Heart Failure and for Cardiogenic Shock with Temporary MCS Bridge Device
In patients with advanced HF with recurrent HF hospitalizations and progressive lifestyle limitation despite maximal medical therapy, device therapy with cardiac resynchronization and in those who have become inotrope-dependent (such as outpatient milrinone), durable MCS support should be considered. 34 Additionally, in patients with cardiogenic shock, temporary MCS devices previously described may be used as a bridge to implant of a durable MCS/LVAD device. 34
Currently Available Durable Mechanical Circulatory Support Devices.

Indications and Contraindications for LVAD Therapy.

History of Durable MCS
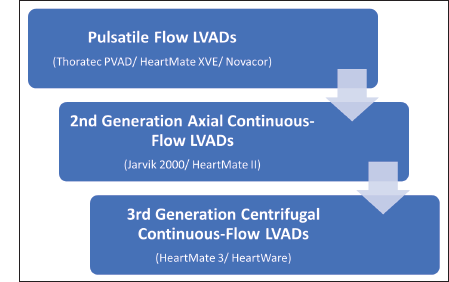
After the first heart transplant in 1967, there was considerable interest in developing MCS devices to serve as a bridge until donor hearts became available. 36 The first US Food and Drug Administration-approved device was the Thoratec Pneumatic Compression paracorporeal ventricular assist device which was developed as a BTT device. This technology then evolved into the first pulsatile flow LVAD controlled by an electronic motor called the Heartmate XVE. The Heartmate XVE was studied in the landmark Randomized Evaluation of Mechanical Assistance for the Treatment of Congestive Heart Failure (REMATCH) trial as a DT LVAD. 37 Considerable progress has been achieved over the past 2 decades in terms of LVAD technology. Pulsatile flow devices have evolved into second and third generation continuous flow LVADs which are currently available for BTT and DT indications. 36 The evolution of durable MCS/LVADs is shown in Figure 6.
Development of Left Ventricular Assist Devices Over the Years.
Durable LVADs (Heartmate II, Heartmate III, HeartWare, and Jarvik 2000)
The currently available continuous flow devices (Table 5) differ significantly from the early pulsatile flow devices. 36 While the second generation LVADs such as Jarvik 2000 (Jarvik Heart Inc, New York, NY) and the Heartmate II (Abbott Laboratories, Abbott Park, IL) are rotor-driven axial continuous flow devices, the third generation LVADs such as Heartmate 3 (Abbott Laboratories, Abbott Park, IL) and HeartWare LVADs (HeartWare Inc, Framingham, MA, USA) have magnetically levitated centrifugal pumps which are built for durability and low risk of hemolysis and thrombosis (Figure 7 shows a third generation continuous flow LVAD). While both generations of LVADs have an inflow cannula that is surgically positioned in the left ventricular apex, an outflow cannula grafted to the ascending aorta, and a percutaneous driveline that connects the pump to the external battery pack, significant differences are present in the pump itself. In second generation, continuous flow devices, blood flow is parallel (axial) to the rotor pump while in third generation devices, the flow in the centrifugal pump is perpendicular to blood flow in the inflow and outflow cannula.
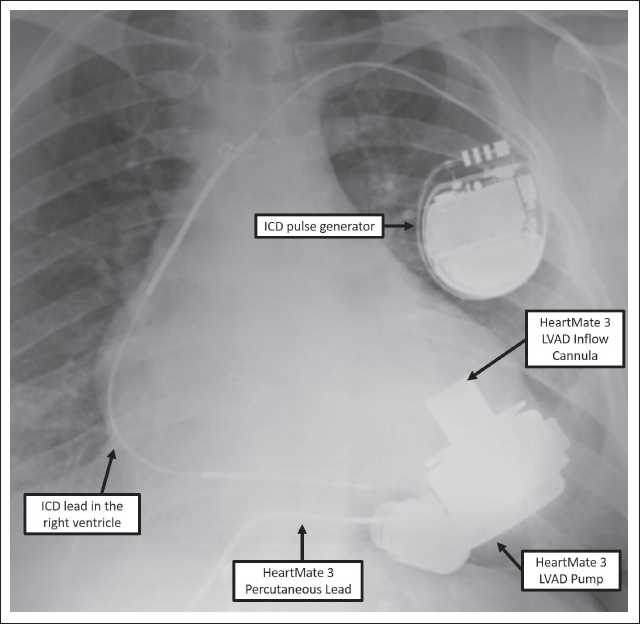
Due to differences in design, survival with the newer centrifugal continuous flow LVADs is higher when compared with second generation axial flow devices (84% vs 74% at 1 year). As such, outcomes with the third generation LVADs are comparable to heart transplant outcomes (79% vs 82% survival at 2 years).38, 39 This represents an exciting evolution in the field of MCS wherein a reliable alternative is available for patients unable to receive a heart transplant. However, a significant limitation in low- and middle-income countries remains the prohibitive cost of durable MCS.
Complications with Durable MCS/LVADs
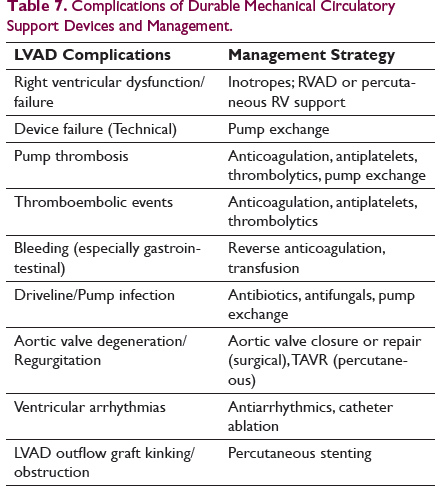
Complications of Durable Mechanical Circulatory Support Devices and Management.
Indications for Urgent Referral to Advanced Heart Failure Centers
As advances are made in the field of temporary and durable MCS devices, advanced HF centers with multidisciplinary decision-making capacity (HF/transplant cardiologist, interventional/electrophysiology cardiologists, transplant cardiac surgeon, critical care physicians, transplant anesthesiologists, perfusionists) are critical to achieving better outcomes. However, establishing advanced HF centers has no meaning without timely and appropriate referrals from cardiologists caring for these severe HF patients and those in acute cardiogenic shock.
Essentially, any patient in cardiogenic shock, who is unable to stabilized with inotropes ± IABP may need advanced MCS such as Impella/VA-ECMO. Since time spent in cardiogenic shock is critical, early discussion should ensue between the referring cardiologist and the advanced HF center. If appropriate, joint decision-making for urgent transfer to the higher center can be facilitated. Among the pool of advanced HF patients failing outpatient medical therapy, those with multiple HF admissions, escalating inotropes, recurrent arrhythmias also warrant prompt referral. In India, the treatment of choice for patients such as this remains heart transplantation, since durable MCS/LVADs are thrice as expensive. Additionally, average wait times for heart transplantation are shorter in India than the West. 40 Appropriate prioritization and decision-making for heart transplantation vs durable MCS/LVAD (as DT) can be carried out only if timely referrals are made and as such education surrounding referrals to advanced HF centers is the need of the hour in India.
Conclusion
We are currently witnessing a revolution in the development of MCS devices and a wide variety of temporary and durable devices are available at our disposal. It is imperative that cardiologists and cardiac surgeons caring for critically ill advanced HF patients are well versed with the nuances of each device, including their specific hemodynamic profiles. Device selection needs to be tailored to patients’ specific hemodynamic needs. High device cost remains a disadvantage for most MCS devices and this adds an additional layer in decision-making for patients in low- and middle-income countries. An important step in providing high-quality care to advanced HF patients is to establish multidisciplinary teams to care for critically ill patients. Ideally, this would include a group of advanced HF cardiologists, interventional cardiologists, and cardiac surgeons with expertise in placement of percutaneous and surgical MCS devices. 30 Such teams would seek input from the referring cardiologists. By necessity, there can only be a few such experienced centers in a given geographic region, since distribution of volume across multiple centers has been found to dilute the outcomes for patients. 41 The establishment of selected, dedicated heart transplant centers with full capacity of services augurs well for the care of critically ill cardiogenic shock/advanced HF patients.
Footnotes
Acknowledgments
The authors would like to thank Dr. Sandeep Attawar (KIMS), Dr. C. Narasimhan (AIG) and Dr. Anuj Kapadiya (AIG) for kindly sharing illustrations used in this manuscript.
Declaration of Conflicting Interests
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
