Abstract
Tachycardia is one of the important reversible causes of ventricular dysfunction. More recently, it has been recognized that premature ventricular complexes and atrial fibrillation can result in or contribute to ventricular dysfunction in the absence of a rapid ventricular rate. The term arrhythmia-induced cardiomyopathy was introduced to include all these potential causes. In this review, we will discuss our understanding of AiCMP and current best practices for its diagnosis and management.
Keywords
Introduction
Cardiomyopathies are a group of diseases that affect heart muscle and, in the phenotype of dilated cardiomyopathy, result in ventricular enlargement and reduced ejection fraction. Reversible causes are important because idiopathic dilated cardiomyopathy has a bad long-term prognosis. Arrhythmia-induced cardiomyopathy (AiCMP) is one of the important reversible causes where a cardiac arrhythmia results in or contributes to the ventricular dysfunction so that control of the arrhythmia can completely or partially reverse the cardiomyopathy. In this review, we will discuss our understanding of AiCMP as it has evolved over time and also the current best practices for diagnosis and management of these patients.
History and Terminology
Tachycardia-induced cardiomyopathy (T-CMP) was first described by Gossage et al. 1 in 1913 in a patient with atrial fibrillation (AF) with rapid ventricular response. Subsequently, the development of reversible heart failure (HF) with rapid pacing was demonstrated in an animal model. 2 The term T-CMP was then used to denote the development of ventricular dysfunction due to rapid ventricular rate irrespective of type of tachycardia.
Over the last few decades, it was recognized that frequent premature ventricular complexes (PVCs) could cause ventricular dysfunction.3,4 Although these patients were initially labeled as T-CMP, they did not have sustained tachycardia and different mechanisms were likely responsible for the ventricular dysfunction. So, the term PVC-induced cardiomyopathy was used for this condition.
Similarly, it was recognized that although AF can result in T-CMP due to a rapid ventricular rate, ventricular dysfunction could also be seen in patients with AF who had a controlled ventricular rate. Thus, the term AF-induced cardiomyopathy was used to describe these patients. 5
Since various arrhythmias can result in the development of reversible ventricular dysfunction with the mechanism not limited to tachycardia alone, the broader term “AiCMP” was introduced and is favored now.5–7 This term includes tachycardia, frequent PVCs, and AF as a cause for CMP. The defining characteristic of AiCMP is presence of ventricular dysfunction in a patient with an arrhythmia and its partial or complete recovery once the arrhythmia is effectively suppressed. AiCMP does not include conduction abnormalities induced ventricular dysfunction such as left bundle branch block, preexcitation, and chronic right ventricular pacing. 5
Of note, arrhythmias not only can induce ventricular dysfunction and HF in a normal heart but can also worsen ventricular function in a person with structural heart disease. Effective suppression of arrhythmia in such cases leads to improvement, but not normalization of ventricular function.8,9
Epidemiology
Incidence and prevalence of arrhythmia-induced CMP is not clear and, on the whole, it appears to be an underrecognized condition. Many different forms of tachyarrhythmias can lead to ventricular dysfunction and HF (Table 1).10–13 Atrial tachycardia (AT) is a classic arrhythmia that can result in AiCMP when it is incessant. In studies of focal AT in adults, T-CMP was reported between 8.3% and 10% of patients.14,15 In a study of focal AT in children, T-CMP was observed in 28% patients. 16 Permanent form of junctional reciprocating tachycardia, although a rare condition, is usually incessant and is typically associated with AiCMP. However, other arrhythmias which are not typically associated with AiCMP, like atypical atrioventricular nodal reentrant tachycardia 17 or fascicular ventricular tachycardia 13 , can also be present in the occasional patient with reversible ventricular dysfunction when they are slow and incessant.
Tachyarrhythmias That Can Lead in Cardiomyopathy.

On the other hand, AF, the most common chronic arrhythmia in the general population, may be associated with varying degrees of ventricular dysfunction in many patients and may, therefore, be the most common cause of AiCMP. In patients with left ventricular (LV) dysfunction and AF, significant improvement in ejection fraction is seen in 58% to 68% after ablation suggesting that many of these patients have reversible ventricular dysfunction related to AF.8,18 In different studies of radiofrequency ablation (RFA) of PVCs, PVC-induced ventricular dysfunction varies between 7% and 30%.19–21
Pathophysiology
Pathophysiologic Mechanisms for AiCMP.
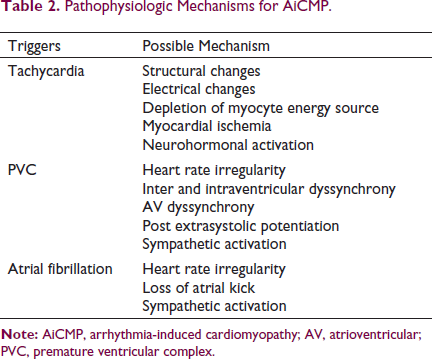
A rapid heart rate is well known to result in ventricular dysfunction and HF but the exact mechanism behind this is not clear. Various animal models have consistently shown the development of ventricular dilatation and dysfunction in response to chronic rapid pacing. All these models have shown a time-dependent ventricular remodeling with development of HF. Hemodynamic changes such as fall in systemic arterial pressure, elevated intracardiac pressure, and elevated pulmonary artery pressure are seen as early as 24 hours of rapid pacing. 22 This leads to increased systemic vascular resistance and elevated ventricular wall stress and activation of the neurohormonal response. With continued pacing, these hemodynamic changes typically plateau at 1 week, whereas cardiac output, ejection fraction, and cardiac volumes may continually deteriorate for up to 3 to 5 weeks. 22
Chronic rapid pacing induces changes at myocyte and myocardium level. Depletion of myocyte energy stores, 23 mitochondrial dysfunction with increased activity of oxidative enzymes, 24 myocyte elongation, decreased myocyte attachment with basement membrane, 25 activation of proapoptotic cascades, and loss of myocytes 26 are the major changes seen. Significant loss of extracellular matrix is also seen. 27 This weakens myocyte support and causes myocyte misalignment. This may contribute to biventricular dilatation with no change or thinning of ventricular wall. 28
Increase in contraction frequency is known to cause a progressive increase in myocardial contractility. But this force frequency relationship is blunted in AiCMP. 29 T tubules depletion with loss of L-type calcium channels, 30 decreased Ca transient,31–33 slow Ca uptake, 34 and reduced Ca stores 35 are seen to contribute to abnormal excitation contraction coupling.
Myocardial ischemia may lead to myocyte injury, ventricular dilatation, and dysfunction. Alteration in myocardial capillary structure and distribution with reduced subendocardial coronary flow reserve is seen in animal models of rapid heart rate-induced ventricular dysfunction.36,37
AF appears to cause AiCMP by mechanisms not limited to a rapid rate alone.8,18,22 Animal models have shown calcium mishandling due to heart rate irregularity in AF and this altered excitation contraction coupling may contribute to ventricular dysfunction. 38 Heart rate irregularity is also seen to produce adverse hemodynamic effects 39 and increased sympathetic activity. 40 In addition to heart rate irregularity, loss of atrial kick in patients with AF may lead in ventricular underfilling, elevation of filling pressure, and sympathetic dysregulation. 41 Further studies are needed to better understand the mechanisms behind AiCMP in AF.
Similarly, development of AiCMP in the presence of frequent PVCs is multifactorial. Major proposed mechanisms are heart rate irregularity, inter- and intraventricular dyssynchrony, AV dyssynchrony, postextrasystolic potentiation, and sympathetic activation. 42 Animal models of PVC-induced cardiomyopathy failed to show inflammation, mitochondrial dysfunction, apoptosis, or fibrosis seen in other animal models of AiCMP. 43 These findings are supported by CMR studies in patients with PVC-induced cardiomyopathy. 44 Electrophysiological remodeling with altered Dyad function appears as a primary reason for PVC-induced cardiomyopathy. 5 These findings suggest primary functional abnormalities behind this condition.
Clinical Presentation
AiCMP may develop within months of onset of the arrhythmia or it may take years. 45 In general, more rapid arrhythmia results in rapid onset of ventricular dysfunction while a slower arrhythmia may take longer to produce ventricular dysfunction. It resembles dilated cardiomyopathy in phenotype. Most common presentations are palpitation, HF, and presyncope or syncope. 46 Patients may also be asymptomatic. Risk of sudden death appears increased in these patients and this may be the first presentation.10,47
Diagnosis and Management
When to Suspect?
In a patient with ventricular dysfunction with no clear cause and persistent or frequent paroxysmal arrhythmia, AiCMP is a potential diagnosis. In patients with paroxysmal arrhythmias or PVCs, ambulatory electrocardiographic (ECG) monitoring may be useful to identify the culprit arrhythmia. 6 Ambulatory ECG monitoring for relatively long duration (2 weeks) may better identify the culprit arrhythmia and provide more accurate measure of PVC burden. In a study, 24-hour ambulatory ECG monitoring identified only 53% patients with PVC burden >10%. Yield of ambulatory ECG monitoring continuously increased with increasing duration of monitoring. 48
Although AiCMP is typically seen without other structural heart disease, some patients with structural heart disease develop ventricular dysfunction out of proportion to the underlying cardiac condition in presence of persistent or frequent paroxysmal arrhythmia. In all such patients, arrhythmia may be additionally responsible for the ventricular dysfunction.
On echocardiogram, AiCMP is characterized by biventricular dilatation with lack of ventricular hypertrophy. When compared with idiopathic dilated cardiomyopathy (DCM), patients with AiCMP are found to have smaller LV-end diastolic dimensions and less LV mass index for similar degree of ventricular dysfunction. 49
B-type natriuretic peptide (BNP) and N terminal pro B-type natriuretic peptide (NT pro BNP) levels are usually elevated in patients with ventricular dysfunction. In patients with ventricular dysfunction, fall of plasma BNP and/or NT pro BNP level after successful suppression of arrhythmia may help in differentiating AiCMP from idiopathic DCM. 50
Predicting Improvement: Role of Cardiac Magnetic Resonance in AiCMP
Cardiac magnetic resonance (CMR) may assist in assessing ventricular dimensions and function and may also help in differentiating patients of AiCMP from idiopathic DCM patients. CMR may also predict response to treatment in patients with suspected AiCMP. In a study of idiopathic ventricular arrhythmias and ventricular dysfunction, late gadolinium enhancement (LGE) was more prevalent in patients who did not respond well after ablation. 44 Another study in patients with frequent PVCs and ventricular dysfunction has shown LGE as independent predictor of response to ablation. 51 In the recent CAMERA-MRI study of AF with ventricular dysfunction, absence of LGE predicted significant improvement in ventricular function after AF ablation. 18 A follow-up CMR after successful treatment of arrhythmia may be done to look for residual fibrosis or scar which may give important prognosis information. 52
Treatment: Drugs Versus Ablation
Initial treatment of AiCMP is optimization of HF therapy with standard drugs (beta blocker, ACE inhibitors/angiotensin receptor blockers, mineralocorticoid receptor antagonists, and Diuretics) to relieve symptoms of HF and attenuate ventricular remodeling. As suppression of arrhythmia can completely or partially reverse ventricular dysfunction in AiCMP, the main component of management is effective suppression of arrhythmia with antiarrhythmic drugs (AADs) or RFA.
Selection of strategy to suppress arrhythmia depends on type of arrhythmia, patient status, and associated comorbidities. For arrhythmias such as atrial flutter, atrioventricular nodal re-entrant tachycardia, atrioventricular reentrant tachycardia, and focal AT, RFA has high success rate and should be considered as first line of treatment.
In a patient with ventricular dysfunction and persistent or paroxysmal AF, first target is adequate rate control. If the patient continues to worsen even after appropriate rate control, establishing sinus rhythm should be the goal. AADs have shown lower success rate (35%-70%)53,54 in achieving sinus rhythm with frequent adverse effects in AF. Although studies of AF ablation have shown better success rate (70%-90%)8,18,55,56 and fewer adverse effects, repeat ablation was often required. Till now only 1 randomized controlled trial has directly compared AF ablation and amiodarone for rhythm control in patients with AF and HF. At the end of 2 years, ablation was found superior in establishing sinus rhythm as compared to amiodarone (70% vs 34%) with lower unplanned hospitalization and mortality. 57
Studies of rhythm control strategy with AADs have failed to show benefit over adequate rate control strategy.58,59 But recent randomized controlled trials of rhythm control strategy with AF ablation versus adequate rate control strategy have shown significant benefit in functional class and ejection fraction with AF ablation.8,18 Therefore, early rhythm control strategy with ablation may be considered as first-line treatment in suitable patients with AF.
More than 80% fall in PVC burden is considered as successful suppression of PVCs. 60 Both AADs and RFA have shown long-term success rate of 70% to 80% in different studies. Beta blockers are the most frequently advised AAD for frequent PVCs because of the lack of significant side effects. Other AADs such as amiodarone, dofetilide, sotalol, mexiletine, or flecainide may be more effective than beta blockers but are associated with significant side effects and pro-arrhythmic risk. Current guidelines do not recommend use of Class IC drugs because of increased mortality observed with Class IC drugs in CAST trial. 61 A recent retrospective study has shown effectiveness of flecainide and propafenone in PVC-induced cardiomyopathy without increasing death or ventricular arrhythmia. 62 There is no RCT comparing RFA and AADs for PVCs suppression but a retrospective study has shown that RFA is more effective than AADs. 63 RFA may have limited success or not be possible when PVCs are originating from epicardium, papillary muscles, or near coronaries or conduction system.
Long-Term Outcomes
Initially, AiCMP was thought as a completely reversible entity but evidence suggests it may not be so. In tachycardia-induced ventricular dysfunction, ventricular function improves within a month after elimination of tachycardia, but may take up to 2 to 3 months.10,19,47,64 Even after normalization of ventricular systolic function, some dilatation of ventricle with diastolic dysfunction may persist. Patient may also show hypertrophic response after suppression of tachycardia. This denotes underlying histopathological abnormalities.22,28,65
Studies have reported rapid development of ventricular dysfunction and HF after recurrence of arrhythmia.10,47 Persistent histopathological abnormalities may be responsible for this. Sudden deaths are also reported even after successful suppression of arrhythmia.10,47 Thus, a treatment option with high success or cure rate should be considered for management of AiCMP and one should monitor for recurrences.
Conclusion and Future Perspectives
Tachycardia, frequent PVCs, and AF can trigger ventricular dysfunction with or without an underlying cardiac condition. The term AiCMP encompasses all these conditions. AiCMP is a reversible cause of cardiomyopathy and mimics idiopathic DCM. Clinician has to keep a high index of suspicion to identify this condition. Ambulatory ECG monitoring may be required for diagnosis. Successful suppression of arrhythmia can improve or normalize ventricular function with decrease in morbidity and associated health care expenditure. The histopathological changes may persist and may contribute to rapid deterioration with recurrence of arrhythmia. Sudden cardiac death may occur. CMR may help in diagnosis and may predict response to therapy. A follow-up CMR may give important prognostic information.
Despite great progress in understanding and management of AiCMP, there are definitely gaps in the knowledge. We know little about pathophysiology of PVC-induced cardiomyopathy and AF-induced cardiomyopathy. There is less clarity on timing of interventions. We need more studies comparing ablation and AADs in PVC-induced cardiomyopathy. Our understanding regarding future events in these patients is limited. We need to define the role of CMR in these patients in management as well as in follow-up.
