Abstract
The standard duration dual antiplatelet therapy (DAPT) is considered as gold standard for post percutaneous coronary intervention (PCI) medical therapy, as mentioned in American College of Cardiology/American Heart Association 2016 and European Society of Cardiology 2017 guidelines. Recently it has been challenged, in terms of duration and composition of this therapy. Many newer regimens and therapeutic drugs are being tried in large randomized clinical trial studies and found to be as effective as DAPT if not superior. There is general trend to introduce better antiplatelets like P2Y12 inhibitor (prasugrel and ticagrelor) as monotherapy for longer duration and restricting use of aspirin beyond 3 months. This review article helps us in understanding the evolution of DAPT therapy, formation of guidelines, and what are the new and evolving concepts in post-PCI medical therapy.
Keywords
Introduction
Dual antiplatelet therapy (DAPT) has become cornerstone of the management of the coronary artery disease (CAD). The 2 major guidelines by American College of Cardiology (ACC)/American Heart Association (AHA-2016) and European Society of Cardiology (ESC-2017) have stated Class-IA indication to initiate DAPT for a minimum duration of 12 months, post acute coronary syndrome (ACS).1, 2 The guidelines also addressed some conditions where DAPT duration can be modified to get maximum benefit and by reducing major adverse cardiac events (MACE). The rationale of modifying DAPT duration hinges on the concept of ischemic risk and bleeding risk of each individual patient being treated for CAD. This article tries to review the evolution of DAPT and current concepts of DAPT therapy.
Ischemic vs Bleeding Risk
The CAD patients can be divided into high bleeding risk (HBR) group and high total ischemic burden (TIB) group (Figure 1). Bleeding risk can be estimated by DAPT-score, PRECISE-DAPT score, Paris registry or BARC-score, all of these scores are of moderate predictive value. Bleeding risk category is said to be high if they satisfy 1 major and 2 minor criteria or if there is stepwise increase in risk of BARC-3 to BARC-5 bleeding.3, 4 The major/minor criteria for bleeding includes parameters such as age (≥75 yr), comorbidities (renal disease, liver disease, cancer), blood labs (anemia, thrombocytopenia), nervous system (stroke, intracranial hemorrhage, arteriovenous malformation), bleeding history (bleeding diathesis, prior transfusion) and iatrogenic (oral anticoagulants, NSAIDs, elective surgery on DAPT, trauma). 3 TIB refers to all the ischemic events (symptomatic/asymptomatic) suffered by an individual. As shown in many studies, the majority of stent thrombosis is observed in first 30 days post percutaneous coronary intervention (PCI). 5 The ischemia could be either primary angina (reduced blood supply) or secondary angina (increased workload) or mixed angina. 6 The TIB can be estimated by various factors such as history of plaque rupture/ACS, patient comorbidities (DM, CKD, smoking, prior PCI, or CABG), complex CAD and at-risk myocardial territory. It becomes extremely important for cardiologist to balance the ischemic and bleeding risk and optimize the dose and duration of antiplatelet therapy.
An illustration showing fine balance between Ischemic burden and Bleeding risk seen among patients with coronary artery disease, requiring careful management
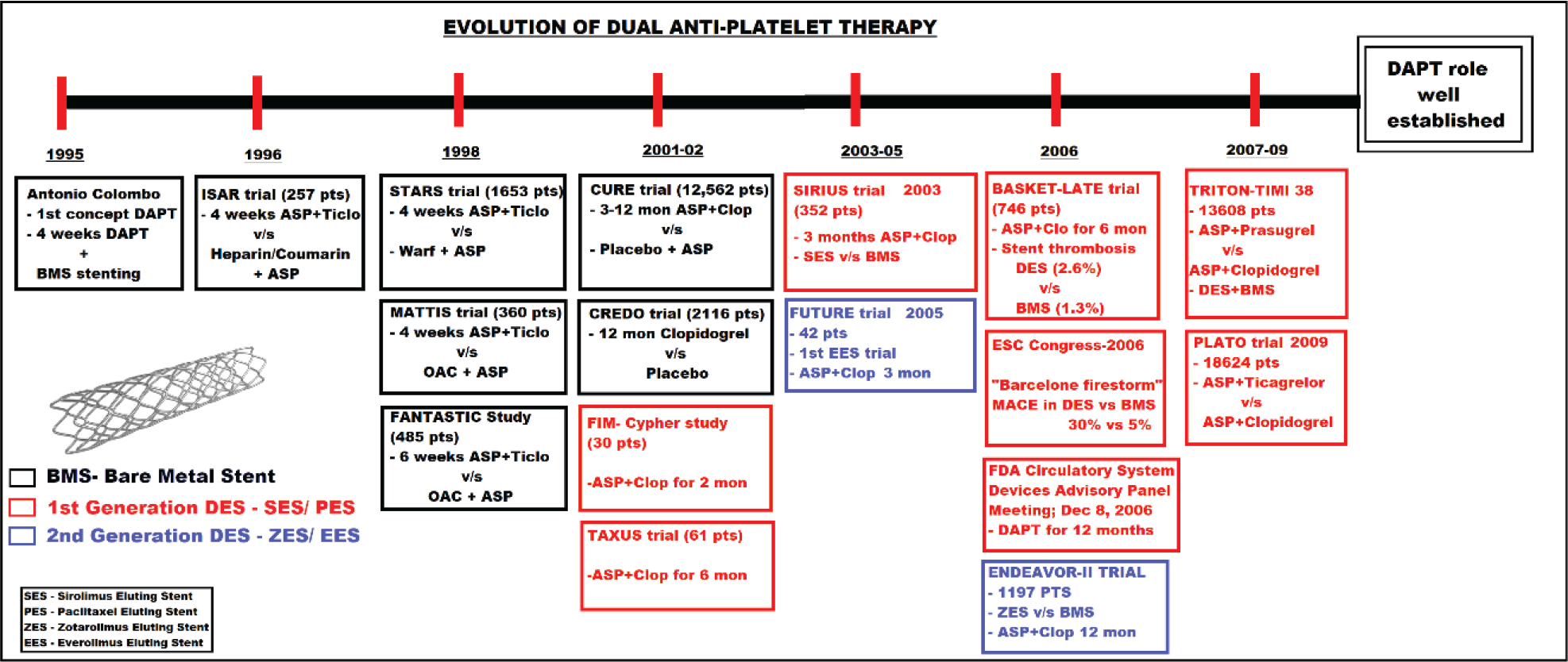
Evolution of DAPT
After performing the first successful coronary angioplasty by Dr Andreas Gruntzig in 1977, he used aspirin as preprocedure medication and followed by oral anticoagulant during postprocedure period. 7 Thus the concept of use of single antiplatelets therapy (SAPT) with single-anticoagulant was born, which was later reinforced by further studies into a well-established medical therapy to prevent thrombosis and restenosis postprocedure. 8 It is to the credit of Antonio Colombo, who proposed the concept of use of DAPT, post-PCI with bare-metal stent (BMS) in 1995. The study involved use of short-duration DAPT therapy with ticlopidine 250 mg bid for at least 4 to 8 weeks along with Aspirin 325 mg for 3 to 5 days. 9
Subsequently very important randomized clinical trial (RCT) trials like STARS trial, which included 1653 patients, revealed superiority of DAPT (aspirin + ticlopidine) when compared to aspirin (ASP) alone and aspirin with warfarin combination in reducing post-PCI stent thrombosis for a period of 4 weeks. 10 Similarly MATTIS trial too showed superiority of DAPT over ASP + OAC (oral anticoagulant). 11 Other trials like FANTASTIC and ISAR trials from Europe show similar results.12, 13
In 2001, PCI-CURE trial revealed not only superiority of aspirin + clopidogrel (20% TIB reduction) over aspirin alone therapy, 14 but also use of DAPT for 3 to 12 months had better outcomes when compared to 4 weeks post-PCI therapy using BMS. Similar outcomes were observed in studies like CREDO trial. 15 Till 2001-02, majority of the studies used BMS, such as ISAR, STARS, MATTIS, FANTASTIC, CURE, and CREDO trials (Figure 2). From 2002, with improvement in technology, the 1st generation drug eluting stenting (DES) were being used in majority of the large studies such as CYPHER, TAXUS, SIRIUS, TRITON-TIMI-38, and PLATO trials.
The BASKET-LATE study showed that stent thrombosis was common with DES (2.6%) compared to BMS (1.3%), this is because of discontinuation of clopidogrel after 6 months therapy. 16 This was followed by misleading discussion in ESC congress 2006 at Barcelona that “DES may kill!” The initial enthusiasm for DES was dampened, due to wrongful contemplation of the results of DES studies. 17 This was followed by a consensus developed after careful study of trials by FDA circulatory system devices advisory panel in 2006, which advised to continue DAPT for a period of 12 months post-PCI procedure. It was then that standard 12 months course of DAPT was recommended. 18 With introduction of newer antiplatelets and second-generation DES the incidence of stent thrombosis and stent restenosis reduced dramatically. These outcomes were shown conclusively in studies such as Endeavor-II trials for second-generation DES, TRITON-TIMI-38 trial for prasugrel and PLATO for ticagrelor.19-21
The figure showing evolution of the concept of usage of 'dual antiplatelet therapy' along with development of stent technology over the years. [DAPT: dual antiplatelet therapy, BMS: bare metal stent, ASP: aspirin, Ticlo: ticlopidine, Clop: clopidogrel, OAC: oral anti-coagulant, PTS: patients, mon: months, FDA: food and drug administration, Warf: warfarin, DES: drug eluting stent, SES: sirolimus eluting stent, PES: paclitaxel eluting stent, ZES: zotarolimus eluting stent, EES: everolimus eluting stent]
Different Trials/Studies Conducted to Know the Ideal Duration of the DAPT Therapy
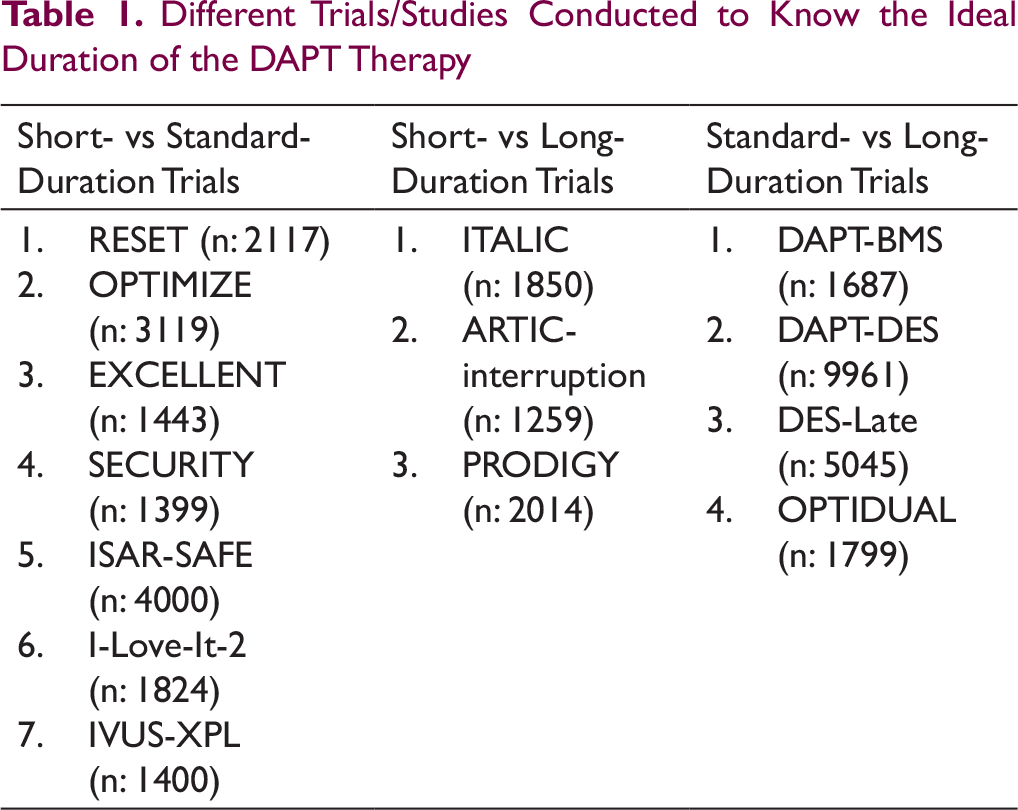
In the later part of 2010, multiple studies were conducted to determine the best duration for continuing DAPT. This was triggered due to concerns of long-term bleeding risk raised by multiple sources like bivalirudin trials, CRF group and Sunil Rao and Duke group. Multiple simultaneous studies comparing different duration of DAPT were conducted (Table 1).
Long Duration vs Standard Duration Studies
The largest trial with regards to long-duration DAPT therapy is “DAPT trial.” In this trial 9961 post-PCI patients were included and started on DAPT therapy (ASA + thienopyridine) for a period of 12 months and continued to 30 months only if they had no bleeding events. This study showed moderate benefits (reduction in MACE 4.3% vs 5.9%; HR: 0.7; P < .001; NNT: 63) of continuing DAPT therapy in patients who tolerated 12 months of therapy. It is important to know that 51.2% of stents used were older generation and rest 46.7% were everolimus eluting stent (EES). A substudy involving PCI using EES showed no benefit (4.3% vs 4.5%; HR: 0.89) of using long-duration DAPT therapy over standard-duration therapy. 22
In Bittl et al, long-duration DAPT (18-48 months) when compared to standard-duration (12 months) of DAPT therapy showed 1% to 2% absolute decrease in late-stent thrombosis (OR: 0.82; 0.47-1.42) and ischemic complications (OR: 0.87; 0.65-1.18); there is 1% absolute increase in bleeding complications (OR: 1.67; 0.99-2.84). For every thousand patients per year, there were 6 fewer MIs and 3 fewer stent thrombosis along with 5 additional major bleedings in the meta-analysis. The MI and stent thrombosis groups overlap; therefore, the overall benefit will be lesser than summation of both. Therefore, the benefit of long- over standard-duration DAPT appears to be marginal with no major change in mortality. 23
Similarly, in Navarese et al, similar findings were revealed from a meta-analysis of 32 287 patients. Here 48% of patients had stable angina, 45% with NSTE-ACS and 7% STEMI. When compared to standard-duration therapy the long-duration therapy had borderline increase in mortality (OR: 1.3; 1.02-1.66) and increase in major bleeding (OR: 1.62; 1.26-2.09), whereas definite reduction in MI (OR: 0.53; 0.42-0.66) and stent thrombosis (OR: 0.33; 0.21-0.51). So, for per thousand patients treated will cause 4.2 deaths, 7.4 patients would suffer major bleeding, the therapy will prevent 13.3 MI and 6.6 stent thrombosis. The effects of preventing post-PCI complications cancel out mortality and major bleeds. 24
Another important study is PEGASUS, 21 162 patients, with follow-up of 1 to 3 years post MI, where ticagrelor either 90 mg/60 mg twice ([OR: 0.85; 0.75-0.96] and [0.84; 0.74-0.95], respectively) compared with placebo. The major bleeding incidences were similar between 90 mg and 60 mg group (2.6% vs 2.3%), but much higher than placebo group with 1.1%. For 1 patient to benefit (prevent ischemic event) the number needed to treat (NNT) per ten thousand were 40 to 42, similarly 1 patient would suffer adverse outcome (major bleeding) when 31 to 41 per ten thousand patients were exposed to treatment, ie, number needed to harm (NNH) per ten thousand. NNT and NNH were similar, which emphasized that careful selection of patients will help in better outcomes. Similarly, other studies like PRODIGY, DES-LATE, and ARCTIC-Interruption did not show reduced ischemic events on use of DAPT longer than 12 months. However longer duration can be considered in patients who had complex PCI, when you use older generation stents or if there was history of stent thrombosis in the first 12 months.25-27
Shorter-Duration vs Standard-Duration Studies
A meta-analysis of 5 RCTs [EXCELLENT, RESET, OPTIMISE, SECURITY, and ISAR-SAFE trials] involving comparison of shorter-duration (3-6 months) vs standard-duration (12 months) DAPT therapy published in ACC/AHA focused update 2016 on “duration of DAPT in patients with CAD” showed no major difference in stent thrombosis, MI risk or mortality between the two. Although, there was borderline increase in risk of bleeding in “standard-duration” when compared to “shorter-duration” therapy (OR: 1.65 [0.97-2.82]). One of the drawbacks of these studies were inclusion of higher percentage of patients with “low-risk” stable-CAD; also, it was underpowered to detect difference in stent thrombosis. 23
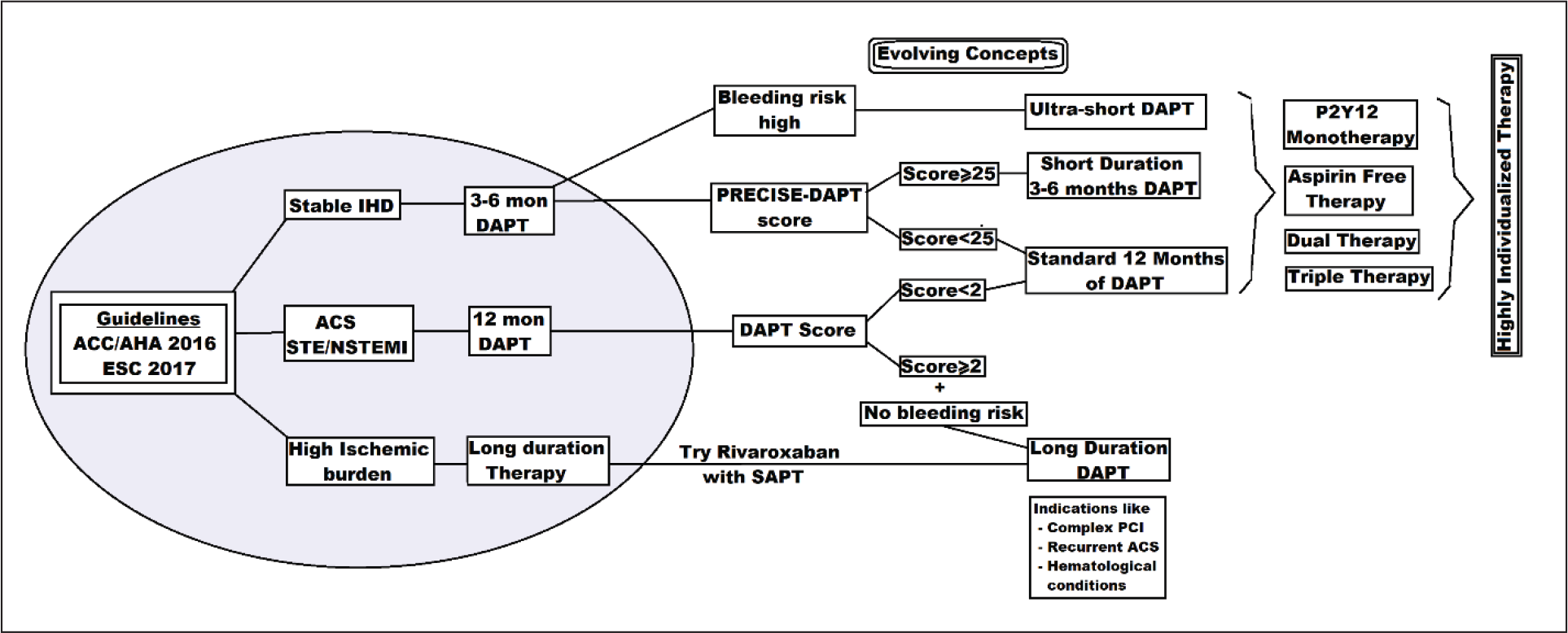
The figure illustrates the duration of the dual antiplatelet therapy according to ACC/AHA 2016 and ESC 2017 guidelines (inside the shaded oval figure). Also it highlights the various evolving concepts for individualization of dual antiplatelet therapy (Outside the shaded oval figure)
Again, similar findings were noticed in Navarese et al, where stable CAD and NSTE-ACS were similar in percentage. There was marginal increase in the risk of major bleeds in standard-duration when compared to short-duration (OR: 0.58; 0.36-0.92), moreover there was no significant difference in the mortality, MI, and stent thrombosis outcomes (OR: 0.91, 1.11, and 1.32).
The Guidelines
These aforementioned studies and meta-analysis helped us to divide the post-PCI patients into 2 groups. The group with HBR (calculated by BARC, TIMI scores, etc) and the other group comprising high TIB. Therefore, the HBR group can be provided with shorter-duration DAPT therapy, thus retaining ischemic benefits (mortality, MI, and stent thrombosis incidence) similar to standard-duration therapy, and at the same time reducing the bleeding incidence. Similarly, the TIB group, can be provided with longer duration DAPT, thus reducing the risk of MI and stent thrombosis.
ACC/AHA focus update on DAPT duration in 2016 emphasized on individual care for patients. Categorizing the DAPT therapy into short-, standard-, and longer-duration therapy based on benefit and risk ratio. The patients with stable angina, undergoing PCI with DES implantation, requires a minimum of 6 months of DAPT therapy and patients with ACS will requires at least 12 months of DAPT therapy. If bleeding risk is high, then the therapy duration can be reduced to 3 months (short) or 1 month (ultrashort) in case of SIHD and 6 months in ACS patients. Clopidogrel is preferred in SIHD and in case of ACS, any of the available antiplatelets like clopidogrel, ticagrelor, or prasugrel along with aspirin can be utilized. 1 The ESC-2018 update on DAPT, broadly appears similar to ACC/AHA update with minor changes. The use of ticagrelor (Class I) is preferred over clopidogrel in ESC update, for standard duration as well as extended-duration therapy. The DAPT score was used for ACC/AHA guidelines, whereas DAPT score and DAPT-PRECISE scores were used for calculation of bleeding risk in ESC-2018 guidelines. The ACC/AHA update was focused mainly on duration of DAPT, whereas ESC update was a broad-based document covering multiple clinical scenarios (Figure 3). 28
Post Guideline Data
A critically important trial released soon after guidelines was SMART-DATE trial; it included 2712 patients with ACS (UA, NTE-ACS, STEMI) with second-generation DES with DAPT for 6 months (median: 184 days) vs >12 months (median: 531 days) therapy. During the 18 months of follow-up, it was noticed that a definite divergence of outcomes after performing landmark analysis, MACE in percentage was found higher in 6 months when compared to 12 months therapy (HR: 1.69; 0.97-2.94). This increase in MACE was localized to increased incidence of MI to 1.8% vs 0.8% (HR: 5.06; 1.46-17.5). Therefore, the study concluded that 6 months DAPT is less effective and guidelines should stick to 12 months standard DAPT therapy. 29
Recent Short/Ultrashort DAPT Studies/Trials
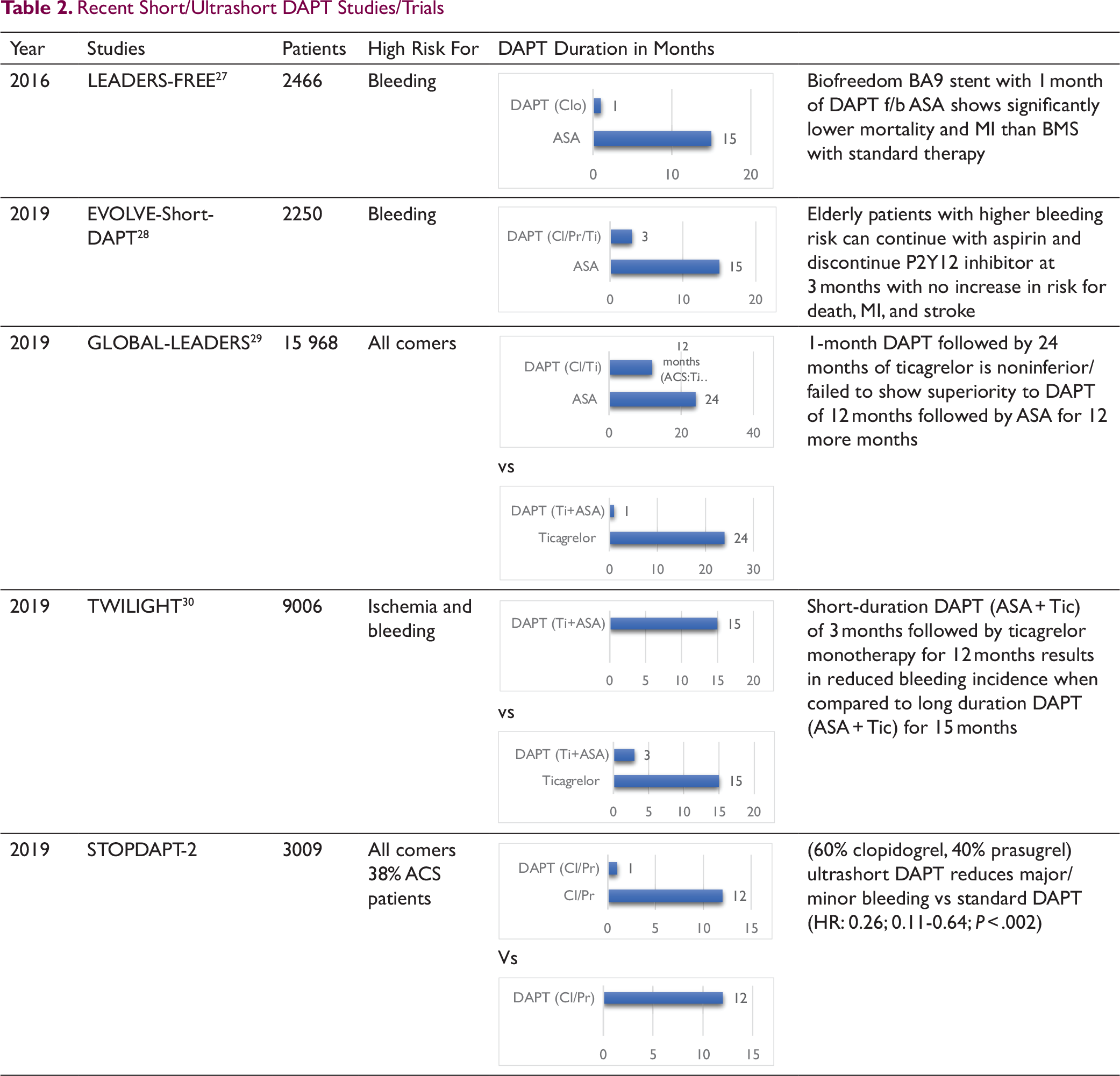
Ultrashort DAPT
The recent trials and studies have concentrated on shorter- and ultrashort-duration DAPT therapy. Some of these studies are LEADERS-FREE, EVOLVE-Short-DAPT, GLOBAL-LEADERS, and TWILIGHT trials.
A general trend in these studies is to adopt DAPT for a period of 1 to 3 months duration and to slowly move toward use of irreversible P2Y12-receptor blocker like ticagrelor instead of continuing aspirin. The details of the studies have been highlighted in Table 2.
With introduction of second-generation everolimus and zotarolimus DES, multiple studies had used short-duration DAPT such as RESET (3 months), OPTIMIZE (3 months), EXCELLENT (6 months), and ISAR-SAFE (6 months). This had an effect on ESC-2015 guidelines for ACS, where P2Y12 inhibitor was advised (Class-IIb) for a shorter duration of 3 to 6 months after second-generation DES deployment in HBR patients. This paved the way for use of short-duration DAPT in future studies.
The LEADERS-FREE trial compared Biolimus-A9 coated, polymer free, drug coated stent (Biofreedom-DCS) with BMS with use of short-duration DAPT therapy. Primary safety end point (Death, MI, STE) was high in BMS compared to BA-9 DCS (P < .005; 12.9% vs 9.4%) with (HR: 0.71; 0.56-0.91). This was an important study as it showed that for high-risk patients for bleeding, DES is preferable over BMS and shorter-duration DAPT is effective. 31
In 2019, many important studies were published and presented at international forums with regards to short DAPT. The 2 most important studies include EVOLVE-Short DAPT and TWILIGHT trials. EVOLVE study was a single arm nonrandomized study mainly involving patients with HBR, whereas TWILIGHT study is a randomized study with 2 arms, 1 arm with short DAPT and other with standard-duration therapy. TWILIGHT included both HBR and ischemic burden patients. Another major difference between the two was use of P2Y12 inhibitor (ticagrelor) instead of Aspirin as SAPT in TWILIGHT. In both studies the researchers focused on single goal of noticing gain from early de-escalation of DAPT. The conclusion from both the studies were applicable to a group of patients whose bleeding risk far outweighs ischemic benefit of longer DAPT use.
EVOLVE-Short-DAPT study used implantation of bioresorbable thin-strut polymer stent (synergy) with 3-month DAPT (aspirin + ticagrelor) followed by aspirin for 12 months. The adjusted outcomes for death/MI were 5.6%, 0.3% for stent thrombosis and 6.26% major bleeding. When this data was compared to historical data as controls, then 3 months of DAPT was noninferior to standard 12 months duration therapy (P: .0016). 32
In TWILIGHT trial, there was a shift in SAPT with ticagrelor being used for a period of 12 months after initial 3 months of DAPT; this was compared to 12 months of DAPT. In this study too, short duration (3 months) of DAPT was found noninferior to standard duration DAPT; all-cause mortality/MI/stroke (3.9% vs 3.9%; P < .001); major bleeding (4.0% vs 7.1%; P < .001); stent thrombosis (0.4% vs 0.6%; P < .05); ischemic stroke (0.5% vs 0.2%; P > .05). In subgroup analysis, patients with NSTE-ACS, diabetes mellitus and those with complex PCI, all of them retained the benefit of short- vs long-duration therapy. 33 SMART-CHOICE and STOPDAPT-2 trials showed similar findings with clopidogrel therapy.
Evolving Concepts
More recent systematic-review article by Yin et al has reaffirmed the aforementioned findings, long-duration DAPT had increased incidence of major bleeding (OR: 1.78; 1.27-2.49) and noncardiac death (OR: 1.63; 1.03-2.59), whereas standard-duration therapy is associated with elevated rates of bleeding (1.39; 1.01-1.92). Thus, short-duration DAPT with newer generation DES implanted showed better outcomes (reduced MI/death/stroke and bleeding incidence) when compared to long- and standard-duration therapy. This study is extremely important as it concluded that “short-term DAPT could be considered for most patients after PCI with DES.” 34
Even though GLOBAL-LEADERS trial showed no additional benefit of 12 months of DAPT (ASA + Tic) over 1-month DAPT (ASA + Tic) followed by 24 months of SAPT (Tic). The ad hoc analysis of GLOBAL-LEADERS trial revealed that aspirin has high incidence of bleeding and did not contribute to the benefit conferred by ticagrelor on ischemia burden. 35 These findings were reaffirmed by GLASSY substudy. 36 Even in TWILIGHT trial once aspirin was removed after 3 months, the cumulative bleeding complications came down drastically (HR: 0.56; 0.45-0.68; P < .001).
Tickled with the idea of aspirin-free SAPT, other studies like SMART-CHOICE and STOP-DART2 have modified the duration of DAPT and SAPT and found noninferior results when compared to standard duration DAPT. In SMART-CHOICE study, 3 months of DAPT (Asp + Tic) followed by SAPT (Tic) vs DAPT of 12 months duration revealed similar all-cause mortality (1.4% vs 1.2%; HR:1.18; P: .61) with significant reduction of bleeding (2.0% vs 3.4%; HR: 0.58; P: .02). 37 In STOP-DART2 trial, ultrashort-duration (1 month) DAPT consisting clopidogrel followed by clopidogrel SAPT showed noninferiority when compared standard-duration DAPT. 38 This is no less than a revelation of sorts, as for the first time, future larger studies/trials can explore aspirin-free antiplatelet therapies after PTCA.
A bifurcation stenting, CTO stenting, stenting of long segments, or deploying more ≥3 stents constitute complex PCI procedure. The duration of DAPT in such complex PCI is mostly left to the discretion of treating cardiologist. A recent trial PRECISE-DAPT concluded that complex PCI had higher risk of ischemic burden and therefore higher ischemic events. These subgroup benefitted from long-duration DAPT therapy in absence of any bleeding risk. 39
Newer oral anticoagulants (NOAC) are gaining popularity in treatment for post-PCI patients. The hypothesis behind this idea is based on the fact that coronary artery thrombus consists of lines of Zahn (alternate red and white bands). These lines of Zahn are alternate layers of platelets and fibrin. 40 Therefore, it makes sense to give a combination of SAPT with NOAC, which acts on both platelet and fibrin together, rather than DAPT that acts on platelet alone. One of the major study mainly done in stable CAD patients was COMPASS trial with 27 395 patients. This study showed that rivaroxaban + aspirin combination fared better in MACE incidence than aspirin alone at 3 years follow-up (HR: 0.76; 0.66-0.86; P < .001; NNT: 77). This trial was stopped early as the mortality benefit shown was significant (Riv + ASA vs ASA; 4.1% vs 3.4%; P < .01; NNT: 143); similar benefit was seen in stroke (1.6% vs 0.9%; P < .001). This study also showed more bleeding: major bleeding (Riv + ASA vs ASA; 1.9% vs 3.1%; P < .001) and minor bleeding (5.5% vs 9.2%; P < .001). This trial did give some in-sights but the role of NOACs was not answered in this study. 41
Another multicenter, double-blind, randomized control study GEMINI-ACS1 included 3037 ACS patients and studied the effect of DAPT vs SAPT (Clo/Tic) + rivaroxaban given for a period of 1 year. This concluded that low dose rivaroxaban (2.5 mg BD) does not cause high bleeding when compared to aspirin (5.3% vs 4.9%; P: .58). There was no benefit in terms of MACE incidence (1.4% vs 1.5%; P: .88). This trial along with ATLAS-ACS2-TIMI-51 trial adds to the growing bank of literature promoting the concept of use of low dose rivaroxaban along with P2Y12 inhibitor in post-ACS patients. 42
In patients who have atrial fibrillation with ACS, such patients need additional anticoagulants along with DAPT, therefore triple therapy/double therapy. The patients who are at higher risk of ACS, or patient with complex PCI can be started triple therapy for a period of 4 weeks followed by SAPT with a NOAC for a period of 12 months, this is followed by continuation of NOACs. Augustus trial included study of “apixaban + P2Y12 inhibitor + aspirin” vs “vitamin K antagonist (VKA) + P2Y12 + aspirin” as triple therapy. The study revealed overall bleeding incidence was higher in VKA group when compared to apixaban group (14.7% vs 10.5%; HR: 0.69; P < .001). Another group compared between placebo (NOAC + placebo) and aspirin group (NOAC + ASA + P2Y12). Here bleeding risk was higher in ASA group compared to placebo group (16.1% vs 9.0%; HR: 1.89; P < .001). Similarly, no difference was seen in apixaban group and VKA group (6.7% vs 7.1%). Thus, there are many studies that show the effectiveness of triple and double therapy. 43
Is There a Role Left for Standard Duration (12 Months) DAPT Therapy?
In SMART-DATE trial, in landmark analysis the risk of MACE between 6 and 18 months was found higher in 6 months duration when compared to 12-month DAPT group, mostly contributed by MI events (2.3% vs 0.5%; P: .025). Therefore, standard-duration therapy still has a role when tailored to individual needs of patients. 29
Authors’ Deductions
We predict that future guidelines will include some basic concepts as recommendations like “Individualized” post-PCI therapy will be stressed more. Routine use of risk scores like TIMI, BARC, PRECISE-DAPT, and DAPT score for assessing ischemic/bleeding risk will be encouraged. Gaining acceptance of P2Y12 inhibitor monotherapy in patients who have undergone PCI with current generation DES. Myocardial territory “at risk” and complexity of the coronary artery stenosis will form very important criteria for deciding the duration of DAPT therapy.
With every new study, aspirin is falling out of favor in DAPT regimen. Therefore, there is a conscious move toward shorter-duration DAPT therapy (consisting of aspirin and P2Y12 inhibitor) followed by P2Y12 monotherapy thereafter, in low-risk patients.
“Short-/ultrashort-duration DAPT” with “P2Y12 monotherapy later” is adequate in patient getting second-generation DES to prevent stent thrombosis and to reduce MACE, whereas older generation/BMS stents require standard/longer-duration DAPT for the same function.
“High HBR with low TIB” group will require newer generation DES with ultrashort-duration DAPT followed by P2Y12 monotherapy, thus avoid bleeding events, and retain the benefit of antiplatelet therapy. “Low HBR with high TIB” group will require either standard-duration 12 months DAPT therapy (SMART-DATE trial) or longer-duration DAPT. Double and triple therapy are gaining ground in patients with high thrombotic risk.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
