Abstract
Ischemic heart disease as a cause of heart failure is common in India, ranging from 48% to 71%. The pathoanatomic basis of LV dysfunction is not only due to infarcted myocardium, but also due to viable but dysfunctional myocardium (hibernating/stunned myocardium) and structural abnormalities (mitral regurgitation/ventricular septal defect aneurysm) consequent to obstructive coronary artery disease. Evaluation of dysfunctional but viable myocardium is a key determinant of recovery of LV function, and magnitude of recovery is proportional to the amount of dysfunctional viable myocardium which if more than 25% LV (4 segments out of 17 segments model) results in improvement in function and size (reverse remodeling). Most of the data regarding therapy come from observational, registry data showing better outcomes with coronary artery bypass graft than percutaneous coronary intervention and medical therapy. The need for more multicentric, randomized controlled trials regarding recovery of LV function by various therapies is more than ever now.
Introduction
The etiology of heart failure varies from region to region in the world and is influenced by the level of epidemiological transition of that country, the type of population sampled (in community or hospitalized patients), and the age distribution of patients presenting with heart failure. Among the various studies of heart failure in India—Trivandrum Heart Failure Registry (THFR), 1 Indian college of cardiology (ICC), 2 National registry heart failure, 3 and International Congestive Heart Failure (INTER-CHF), the most common etiology of heart failure was ischemic heart disease ranging from 71% (in THRF) to 48% (inter-CHF). Thus, heart failure due to ischemic heart disease seems to be common in clinical practice in India, and hence the need for evaluation and appropriate therapy cannot be overemphasized.
Heart failure in coronary artery disease (CAD) patients is not only due to chronic ischemic LV dysfunction but can also occur due to structural disorders consequent to CAD—like severe ischemic mitral regurgitation (MR), LV aneurysm, and acquired ventricular septal defect which needs to be addressed for effective therapy of ischemic heart failure.
The pathoanatomic basis of ischemic LV dysfunction at rest is not only due to infarcted myocardium but also due to reversible LV dysfunction, consequent to significant obstructive CAD. This concept of reversible LV dysfunction was established based on studies of patients undergoing coronary artery bypass graft (CABG) which 4 resulted in improvement and in some cases normalization of LV function. Not only is there an improvement in LV function—regionally and globally, but even the adverse remodeling is reversed. This has resulted in improved survival and decrease in major adverse cardiovascular events (MACE) during follow-up. Conversely, revascularization in patients not having viable myocardium has not shown any favorable structural alteration and clinical benefits.
Mechanism of Chronic LV Dysfunction
In normal animals, subendocardium governs transmural contraction and there is sensitive coupling between subendocardial myocardial blood flow (SE-MBF) and 5 cardiac function. Normally SE-MBF is much more per unit of cardiac muscle, compared to subepicardial MBF. In conditions of reduced MBF (as in CAD), the SE-MBF is more reduced than Epi- MBF. Further, reflex hyperemia that occurs following relief of obstruction, there is misdistribution—with more flow to subepicedium and continued low MBF to SE (SE-MBF). Thus, both during ischemia and reactive hyperemia that follow, there is a marked decrease in SE-MBF, and consequently in muscle dysfunction. These alterations in SE-MBF at rest in patients with hibernating myocardium (HM) have been well documented using cardiovascular magnetic resonance imaging (CMR), as compared to areas with significant coronary stenosis in the same patient which improved (SE-MBF and LV function) following revascularization. Further, coronary flow reserve (CFR) is almost always reduced in viable but dysfunctional myocardium, more severely in segment with low flow at rest.
It is worth remembering that resting myocardial blood flow and CFR are unaffected in coronary artery stenosis up to 40% of diameter reduction, between 40% and 80% resting myocardial blood flow is unaffected but the CFR is decreased, and coronary artery stenosis 80% and beyond not only causes marked decrease basal resting blood flow but also CFR. 6
The severity of reduced CFR impacts on the ability of viable myocardium to improve contraction (contractile reserve) on ionotropic stimulation as these variations in resting MBF impact the degree of CFR—higher the resting MBF, greater is the CFR, with no CFR in segments with least resting MBF. This forms the basis of myocardial response to positive ionotropic drugs—like dobutamine as in dobutamine stress echocardiogram. Other factors that influence myocardial response to drugs include structural alteration in cardiac myocyte (due to chronic decrease in MBF) and downregulation of beta receptors. Thus, the ability of myocardium to recover its function is not only influenced by resting MBF (consequently CFR) but also by 7 structural alterations in cardiac myocytes, extracellular matrix, and microcirculation. The structural alterations in cardiomyocytes include depletion of contractile elements and cytoskeletal proteins, dysfunction and depletion of subcellular elements like sarcoplasmic reticulum, and T-tubules. These structural alterations are time dependent with severe changes in longstanding hibernation, hence there is urgency in treating these patients.
The extracellular matrix shows increased amounts of type-I collagen, type-III collagen, and fibronectin. In addition, there may be a rarefaction of capillaries. All these changes influence unfavorably the recovery of contractile function even after effective revascularization. Recovery of function, following revascularization, is influenced by volume of scar (inversely proportional) or its converse, the mass of viable cardiac myocytes or their indirect parameters—thickness of myocardial wall and LV volumes. However, these indirect measures (wall thickness and LV volumes) cannot distinguish between viable and remodeling myocardium (from nonviable), especially in remote areas (from ischemic segments) and thus lack specificity. One vessel disease can also result in changes, in remote areas without associated CAD. These remote areas (from obstructive CAD) show structural and functional alteration (reduced CFR and altered metabolism) similar to that seen in obstructive CAD in experimental study. Revascularization of occlusive CAD disease has resulted in recovery of function in remote areas.
Markedly dilated LV (volume greater than twice the normal) usually caused by large number of scarred segments are less likely to demonstrate improved LV function (as measured by increase in EF by >5%) after revascularization. The involvement of >4 segments of LV by scar also identifies the LV, that is unlikely to improve following revascularization just as the restrictive pattern of mitral flow (E/A ratio >2 and short EDT). Tissue Doppler (rather than strain) with its high temporal resolution may allow measurement of pre- ejection velocity which, if preserved, is sign of viability and is a predictor of functional recovery. Thickness (at end diastolic) of LV wall >0.5to 0.6 cm is more likely to recover its function if contractile reserve is present. The combination of wall thickness, with presence of CR has a sensitivity of 88% and specificity of 77% for prediction of recovery. Perfusion and metabolic imaging by SPECT and PET that measure MBF require presence of viable myocytes. Technetium-based perfusion tracers—sestamibi and tetrafosmin—are lipophilic cations and are retained in mitochondria and serve as markers of cellular viability. Administration of nitrates to improve resting MBF improves sensitivity and specificity, with positive predictive value of 90%. Decreased regional myocardial trace uptake at rest may represent either lack of cell membrane integrity (like scar) or markedly decreased MBF (as in hibernation). Metabolic imaging can distinguish between the two as metabolism is preserved in viable cells, unlike in scar. Thus, in HM (markedly decreased MBF), viable myocytes show perfusion–metabolism mismatch 8 unlike in scar where these two (perfusion and metabolism) are matched. The extent of PET mismatch corelate with improvement in LV function and symptoms, following effective revascularization. The shift of myocytes metabolism from fatly acid to glucose, under ischemic condition can also be shown by decreased 9 uptake of BMI123 PP (Beta123 methyl iodophenyl penta decanoic acid)—a fatty acid analog. A decreased BMI PP long after resolution of ischemic symptoms, ischemic memory is scintigraphic imprint of previous ischemia and is clue to the etiology of LV dysfunction.
Other methods of evaluation of viability include evaluating microcirculation by contrast echo and contractile reserve (either by echo or cardiac MRI using dobutamine as stressor. All these tests should not only evaluate the presence of viable myocardium but also its extent (of LV myocardium) as it is an important determinant of recovery of LV function, symptom relief, and adverse CV events in the follow-up.
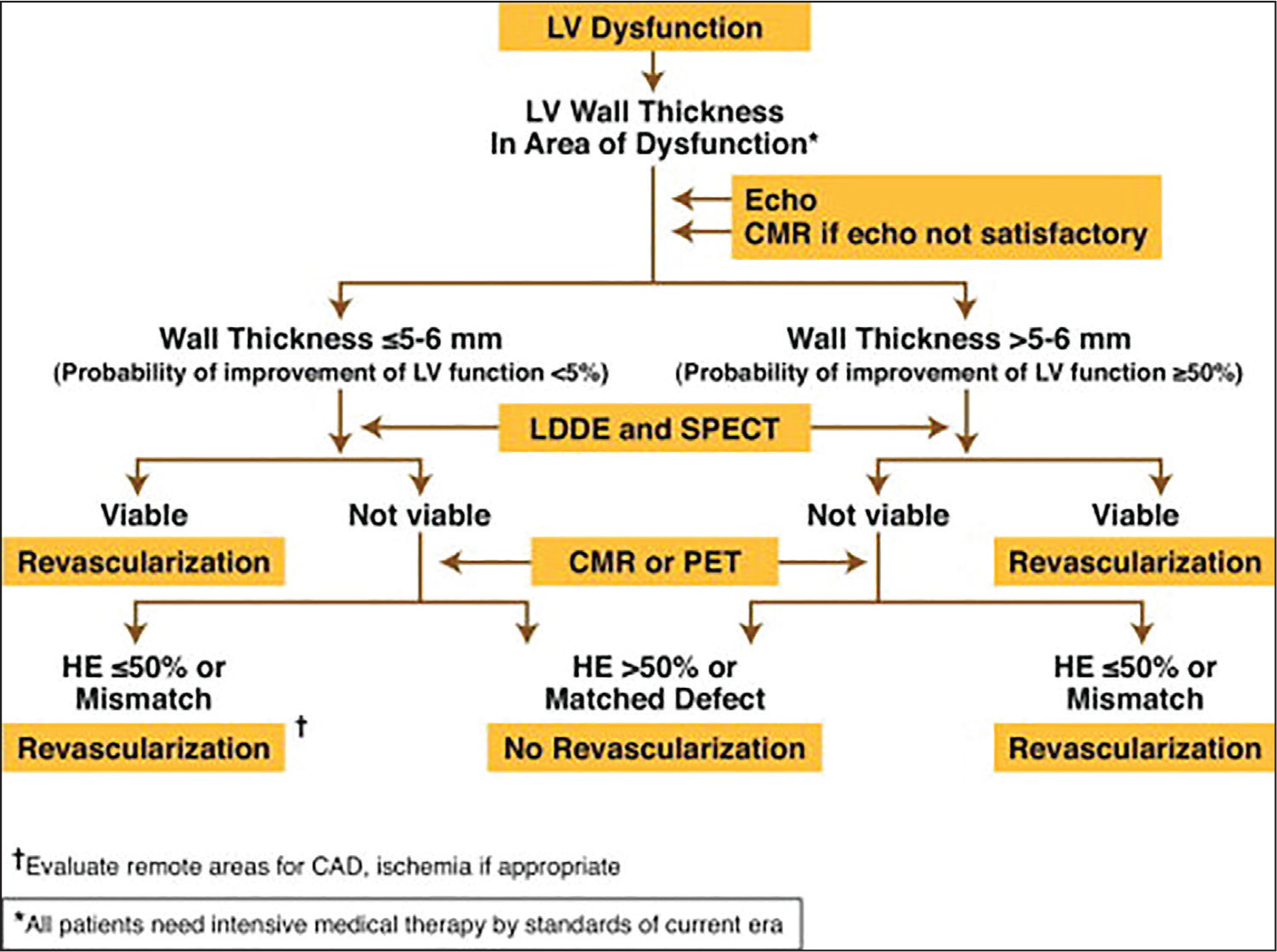
Evaluation of Viability
Tests for viability and coronary angiography are essential prior to a consideration for revascularization. There is no perfect test with 100% sensitivity and specificity, hence will need a combination of tests for decision-making. Which test to be used in a given medical center is influenced by the availability of test and the skill in performing and interpreting the result. One of the suggested formats for diagnosis and therapy of revascularization in case of chronic severe LV dysfunction is given in Figure 1.
Cost effective therapy of ischemic LV dysfunction is to avoid major investigation in patients who are least likely to have viability and thus long-term benefits.
This is a proposed predictive model but not validated in any clinical trial. 10
Multi Vessel Disease with Global LV Dysfunction
(LV function—unlikely to recover if any one Categories is present)
A- 4 major criteria
B- 3 major + 1 minor
C- 2 major + 2 minor
LV wall thickness < 5 to 6 mm LV EF: <20%
No response to LDDE LV EDDI: >5.5 cm/m2
CMR – LGE >50% LV EDVI: 170mL/m2 -Echo
SPECT – Negative for viability ESVI: > 90mL/m2 -Echo
PET – Negative for HM by LV angio
EDVI = >200 mL/m2
ESVI = >120 mL/m2
Revascularization—Ischemic Heart Failure
The scientific basis of revascularization in ischemic cardiomyopathy is based on the following facts:
Revascularization of ischemic myocardium improves left ventricular regional and global mechanical and metabolic function. Successful revascularization has potential to induce reverse LV remodeling—with its associated decrease in LV EDV and LV ESV, and increase in EF. Improvement of LV function after revascularization will impact long-term prognosis. The Benefit of effective and complete revascularisation is proportional to the magnitude of HM (expressed as % of LV) with larger benefit in patients with higher percentage of after effective and complete revascularization.
Viability studies have revealed that the annual mortality rate is markedly decreased (3.2% after revascularization), as compared to 11 16% with optimal medical therapy (OMT) nearly 80% decrease in mortality after effective and complete revascularization. Most of the studies of revascularization have been nonrandomized with observational data and compared CABG with OMT and very few with PCI.
Clinical evidence of LV functional recovery
CABG—Medical Therapy in CAD
Three RCT's, done between 1970 and 1980, by Veterans Administration12, 13 CO-OP Study, Coronary Artery Surgery Study, and European Coronary Surgery Study
14
showed superiority of CABG over medical management in patients
15
with multivessel disease, more so in patients with LV dysfunction with perioperative mortality less than 6%. The improved long-term prognosis is due to the recovery of function by stunned and HM.
○ Rate of death from any cause was lower amongst patients who underwent CABG than PCI over a period of 4 years. ○ Complete revascularization was higher with CABG than PCI.
The data is derived from large prospective multicentric database of over 25 000 patients for stable multivessel disease and heart failure (stage C/D ACC/AHA HF category) 3-year mortality, morbidity was similar for both PCI, and CABG when SYNTAX score were low (<23) and intermediate (23-32), with survival advantage when score was high >32. Repeat revascularization was high with PCI (HR = 9.42).
STICH Trial (Surgical Treatment for Ischemic Heart Failure) 21
This trial addressed revascularization therapy (basically CABG vs intensive medical therapy) in patient with variable degree of ischemic heart failure, with primary endpoint of death from any cause. The secondary end point was death from CV cause or composite of death or hospitalization for cardiovascular cause. Furthermore, there was selection criteria for operators (surgeons) who have to show ability to perform CABG with heart failure with ejection fraction of <40% on at least 25 patients with mortality of 5% or less.
The results at the end of 5 years showed no significant difference between intensive medical therapy alone and medical therapy with CABG. CABG showed better secondary outcomes as compared to intensive medical therapy. The extended study STICH-ES showed at the end of 10 years, rates of death from any cause or death from CV causes or hospitalization for CV causes were significantly lower in CABG group (76.6% vs 87%, HR = 0.72, P = <.001) (Limitation of STICH trial, was they studied variable severity of heart failure with no standardized protocol for assessment of viability. Further, they excluded patients with LMCA disease who benefit maximally with revascularization. 500 patients (out of 1200) had only 1 or 2 vessel disease with no known benefit from CABG. Further, 11% surgical group had concomitant mitral value replacement, which increased mortality and morbidity. Further, there were crossovers from medical therapy to CABG which diluted the benefit of CABG over medical therapy by the intention to treat analysis. When this was accounted by per protocol analysis, there was 24% (P = .005) relative risk reduction in mortality with CABG.
Recent meta-analysis of 22 21 studies involving, 16 191 patients comparing OMT with PCI and CABG in patients with heart failure and EF <40% revealed paucity of RCT in ischemic cardiomyopathy. This meta-analysis showed significant reduction of mortality with CABG (HR = 0.66, 95% CI—0.61 to 0.72, P = <.001 and PCI–HR 0.73, CI 0.62 to 0.90, P = <.001) over medical therapy. Revascularization is better than medical therapy with significant reduction in mortality independent of viability testing.
Another study by Ross et al in 2006 showed that revascularization was better than medical therapy. The risk-adjusted survival was best with CABG (HR = 0.44 vs CI 0.38-0.52) vs no revascularization as compared to survival benefit with PCI (HR = 0.58 vs CI 0.49-0.69) v/s no revascularization.
In another analysis of registry data of New York by Bangalore et al comparing CABG with PCI (DES stent) with LV EF 35% and median follow-up of 2 to 9 years revealed equal long-term survival, with repeated revascularization higher in PCI group, due to increased occurrence of MI, but with lower risk of stroke. 23 It must be remembered that revascularization procedures—CABG and PCI—are associated with higher immediate mortality, for long-term survival benefit.
European guideline-driven recommendations in the context of heart failure.
2018 ESC7EACT5 Guidelines on myocardial revascu larization
Recommendations on revascularizations in patients with chronic heart failure and systolic left ventricular dysfunction (ejection fraction

American guideline-driven recommendations in the context of heart failure.
ACC/AATS/AH A/ASE/ASNC/SCAl/SCCT/STS 2017 Appropriate Use Criteria for Coronary Revascularization in Patients with Stable Ischemic Heart Disease
Revascularization to improve survival compared with medical therapy in the anatomic setting of left ventricular dysfunction

Until more randomized clinical data are available, it is desirable to follow ESC, AHA/ACC guidelines with respect to revascularization in ischemic heart failure (Tables 1 and 2).
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
