Abstract
Aortic stenosis (AS) is characterized by gradual thickening, fibrosis, and calcification leading to reduced opening of aortic valve leaflets. Aortic valve narrowing increases the afterload on left ventricle (LV), which consequently leads to hypertrophy and myocardial fibrosis which over a period of time leads to diastolic dysfunction. LV diastolic dysfunction is an independent predictor of heart failure rehospitalization and mortality in post-aortic valve replacement patients. Chronic pressure overload leads to development of systolic dysfunction due to afterload mismatch. Patients with severe symptomatic AS and depressed LV function without significant myocardial scarring will derive maximum benefit after percutaneous valve replacement and thus show immediate improvement after reduction of LV afterload. We report two such cases which had good LV function recovery immediately after transcatheter aortic valve replacement (TAVR).
Introduction
Aortic valve degeneration occurs as a part of highly regulated inflammatory, fibrotic, and osteogenic process and not just due to wear and tear. Initiation of degenerative process begins with endothelial injury, lipid infiltration, and chronic inflammatory response. This later on leads to calcification in a regulated manner. However, statins and other anti-inflammatory drugs failed to halt the process of degeneration. 1 Chronic pressure overload due to valve narrowing leads to myocardial hypertrophy to maintain the wall stress and cardiac output. 2 The hypertrophied myocardium eventually develops cell death and myocardial fibrosis leading to left ventricular dysfunction and heart failure. Approximately one-third of patients with severe symptomatic AS have left ventricle (LV) systolic dysfunction. 3 Reduction of ejection fraction can be due to various reasons like coexisting coronary artery disease, chronic afterload mismatch or intrinsic contractile dysfunction. If the LV dysfunction was due chronic pressure overload, it can be reversed after valve replacement before irreversible myocardial fibrosis develops. Development of LV dysfunction leads to high perioperative risk for surgical aortic valve replacement (AVR), and with advancement of technology, minimally invasive transcatheter AVR (aortic valve replacement) (TAVR) provides an alternative form of therapy with equal long-term results in high and intermediate risk patients. Previous studies have shown favorable results on LV function recovery after TAVR.4-6 Here, we present two case reports of reversible LV dysfunction post TAVR.
Case History—Case 1
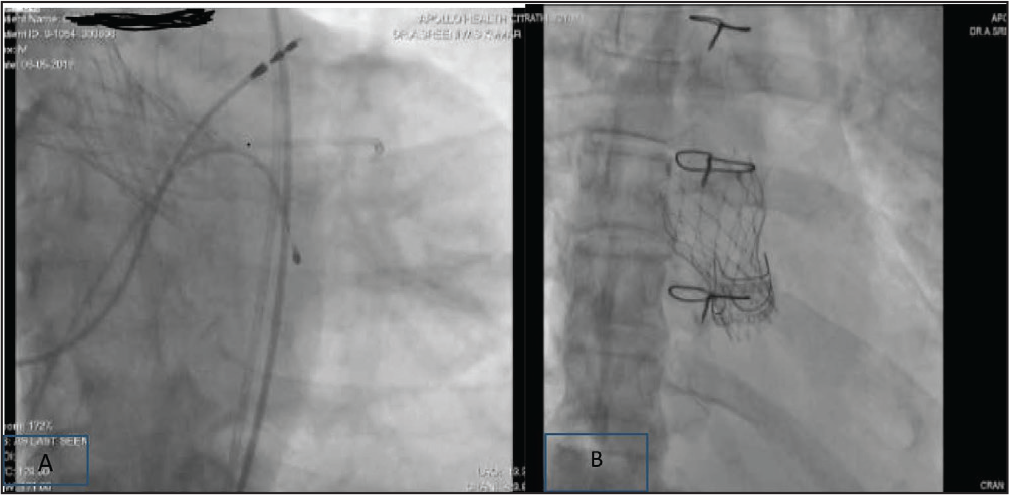
An 81-year-old Hypertensive male with Hypothyroidism underwent dual chamber pacemaker for sick sinus syndrome 1 year back. Now, he presented with complaints of exertional breathlessness of NYHA class III with paroxysmal nocturnal dyspnoea (PND). He was hospitalized for worsening breathlessness 3 months back and treated with intravenous diuretics and other supportive medication and improved symptomatically. ECG was suggestive of atrial fibrillation (AF) with paced rhythm. Two-dimensional echocardiogram was done with EPIQ (Philips, Netherlands) ultrasound machine. It showed that severe calcific aortic stenosis (AS) with aortic valve mean gradient was 50 mm Hg along with mild aortic regurgitation (AR). The patient had global hypokinesia of LV with moderate LV dysfunction with an ejection fraction of 38%. Left ventricular ejection fraction (LVEF) was calculated with the Simpson’s method, transvalvular pressure gradient determined by the Bernoulli formula, and aortic valve area (AVA) calculated with the continuity equation. Two-dimensional echocardiogram was repeated after TAVR before discharge on third post-procedure day. The pre-procedure serum creatinine was 1.8 mg dl–1 and estimated glomerular filtration rate (eGFR) was 38.7 ml min–1. Coronary angiogram was done which showed normal epicardial coronary arteries. He was planned for AVR (aortic valve replacement) in view of recent LV failure. His STS score for surgical AVR mortality was 9.2% and euroSCORE was 4.56%. After heart team discussion in view of high risk for surgical AVR planned for TAVR. Sizing of TAVR valve was performed with multidetector computer tomography and analysis was done using 3mensio software. After giving loading doses with dual anti platelets and adequate hydration, he was taken for procedure through femoral access under local anesthesia and systemic sedation. TAVR was done through right femoral artery with 29 mm self-expanding Medtronic CoreValve (Evolut R) (Figure 1A). The prosthesis was deployed retrograde over a stiff guide wire placed in LV, under fluoroscopic guidance. Hemostasis was secured with 2 pro-glides after the procedure. After procedure, his serum creatinine (1.43) had reduced and vitals were stable. Repeat echocardiogram was done before discharge from hospital and his LV function had improved with EF of 55% and mild paravalvular leak. Mean aortic valve gradients had decreased to 8 mm Hg after TAVR. He was discharged with single antiplatelet and oral dabigatran in view of AF. He was in regular follow-up since 4 months with no further episodes of LV failure and in functional class II.
Case 2
A 70–year-old nondiabetic and nonhypertensive male underwent surgical bioprosthetic AVR in 2009 (Perimount Valve). He presented to hospital with dyspnoea on effort since 2 months and progressive from class II-III with paroxysmal nocturnal dyspnoea (PND) since 1 month. He was not able to sleep in supine position since 2 weeks despite using diuretics. There was no history of chest pain, palpitation, and syncope. His ECG was showing normal sinus rhythm with incomplete LBBB (Left bundle branch block) with infrequent VPCs. (Ventricular premature complexes) Two-dimensional echocardiogram was done which showed degeneration of previously implanted bioprosthetic valve with severe AS and moderate aortic valve regurgitation. ECHO was suggestive of dilated LV with global hyperkinesia of LV with moderate LV dysfunction EF 40% and mild MR, mild TR and moderate PAH with dilated IVC. His baseline renal parameters were elevated with serum creatinine 2.04 mg dl–1 and e GFR was 34.5 ml min–1. His STS score for redo surgical AVR mortality was 10.2% and euroSCORE was 9. After heart team discussion, patient was planned for TAVR. In view of previous surgical AV valve ring, under sizing of TAVR valve was done after CT scan based measurements. We had chosen 23 mm Evolut R ( Figure 1 B) Medtronic valve and procedure was completed with only 30 ml of contrast and under sedation. Post procedure, the aortic valve mean gradients had decreased from 54 mm Hg to 7 mm Hg and no paravalvular leak. Post procedure, he had dramatic improvement in symptoms and no further PND episodes. Predischarge echocardiogram was suggestive of improved LV function, and EF was 52% before discharge. He is in regular follow-up since 9 months and in NYHA class I.
Discussion
Aortic Stenosis (AS) is not only disease of valve but also of the LV. Development of LV dysfunction with coexisting severe aortic valve stenosis was associated with higher perioperative risk. 7 Previous observational studies have showed an increased mortality (9% to 19%8, 9) in perioperative period in patients who have developed myocardial fibrosis and left ventricular systolic impairment. Successful TAVR procedure was associated with sustained clinical and functional long-term benefits up to 4 years of follow-up. 10 TAVR allows an early recovery of LV unlike surgical AVR as LV is not exposed to extracorporeal circulation in TAVR. 11 In previous studies, time taken for recovery of LV function is variable after AVR ranging from 3 months to 1 year.12-14 Even in PARTNER A trail, ≈50% of patients demonstrated early LVEF recovery (≥10% absolute increase) in EF at 30-day follow-up after SAPIEN TAVR. 15 Those without LV function recovery in immediate period after TAVR still show some improvement over a period of time. Dauerman et al studied 156 patients from the CoreValve Extreme and high-risk trials with LVEF ≤ 40% at baseline who had 30-day LVEF data. Approximately two-thirds of patients showed a ≥10% increase in EF in the first 48 h after TAVR. Patients were more likely to have early LVEF recovery if they had a mean gradient of >40 mm Hg and less baseline myocardial damage. 16 Our two patients fall in high-risk TAVR group with low baseline LV EF and severe AS with mean gradients more than 40 mm Hg. There was an improvement in functional status and LV ejection fraction immediately after afterload reduction with TAVR. TAVR offers an attractive and promising form of therapy in these high-risk subsets. However, LV EF measurement alone may not be an adequate test for assessment of LV recovery after TAVR. Detailed analysis of LV myocardial mechanics, myocardial remodeling, and flow dynamics is required to understand the pathology and likely benefit after TAVR. Strain tissue echocardiography can provide diagnostic and prognostic information and it can help identify those patients likely to benefit from TAVR.
(A) 80 Year Male Post TAVR and Post DDDR Pacemaker. (B) 70 Year Male with Valve-in-Valve TAVR
Conclusions
Patients with severe symptomatic AS and depressed LV function without significant myocardial scarring will derive maximum benefit after percutaneous valve replacement and will show immediate improvement after reduction of LV afterload by TAVR.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
