Abstract
Background:
Palliative sedation is a key component of palliative care for end-stage cancer patients with refractory symptoms. In mainland China, midazolam-based titrated palliative sedation is rarely used clinically, with limited data and studies reported.
Aim:
To illustrate the development of clinical practice patterns for midazolam-based palliative sedation in advanced cancer patients in mainland China, identify clinical regularities, and summarize clinical experience to promote the administration of palliative sedation for terminally ill cancer patients in mainland China.
Design:
A retrospective observational study was conducted using clinical records of terminal cancer patients who underwent midazolam-based palliative sedation from 2016 to 2025.
Setting/participants:
Terminal cancer patients who underwent palliative sedation in the oncology ward of Hangzhou Hospital of Traditional Chinese Medicine, a tertiary medical center in Eastern mainland China.
Results:
The overall implementation rate of palliative sedation in terminal cancer patients was 7.98% (55/689). Refractory dyspnea was the primary indication for sedation (47.3%). The proportion of patients receiving adequate sedation was significantly higher in the titrated group than in the non-titrated group (p < 0.001). The mean survival time of the 50 patients who received adequate sedation was 67.45 h. Among them, the mean survival time in the dyspnea subgroup (38.88 h) was significantly shorter than in the pain (125.42 h) and agitation (120.33 h) subgroups (p < 0.001). The mediation analysis further demonstrated that the indication for palliative sedation (presence or absence of dyspnea) fully mediated the association between the patient’s disease diagnosis (presence or absence of lung cancer) and survival time. In this study, 66.7% of family members had never previously heard of palliative sedation, and their attitudes toward this therapy were quite ambivalent.
Conclusion:
Effective communication of palliative sedation helps improve family members’ acceptance of this therapy. Patients with refractory dyspnea, especially lung cancer patients with this symptom (who may have more severe underlying disease), may have shorter survival times than those with agitation or pain. Rigorous pre-procedural risk communication with families is recommended before sedation, and close monitoring and assessment during sedation are essential for safety.
Introduction
With the ongoing development of mainland China, terminal cancer patients and their families are gradually changing their views on life. While pursuing disease control and longer survival, they now pay more attention to maintaining quality of life during treatment.
Palliative sedation, a key part of palliative care, helps end-stage cancer patients whose severe symptoms cannot be relieved by standard treatments.1,2 Studies show palliative sedation effectively reduces both physical and mental suffering without shortening patients’ survival.3,4 However, this treatment is rarely used for terminal cancer patients in mainland China due to the country’s specific medical environment, with three primary barriers.
Traditional Chinese culture and palliative sedation: How family protection prolongs treatment decision
Traditional Chinese culture is characterized by a strong collectivist orientation and places significant emphasis on family unity. Families are seen as a key source of support, with the duty to protect every member. In European countries, the decision-making authority for palliative sedation typically rests with the patients themselves. 5 However, in China, families frequently request physicians to partially or fully withhold information from patients and make medical decisions on behalf of patients, which reflects a cultural preference for shared family responsibility over individual decision-making in crises. While such practices are intended to reduce patients’ suffering, the decentralized decision-making framework prioritize families’ perspectives over patients’ actual needs. Many families lack basic knowledge about palliative sedation, and their well-intentioned actions often overlook patients’ personal experiences and genuine requirements, resulting in adequate understanding and support for patients requests for palliative sedation. Some families even equate palliative sedation with “giving up” or “euthanasia” 6 and exert pressure on other families by invoking traditional Chinese ethical values; such conflicts prolong the decision-making process and thus act as a key barrier to the timely implementation of palliative sedation.
Safety concerns of palliative sedation from healthcare providers and their diminished professional fulfillment
Numerous literature analyses have suggested that palliative sedation does not shorten the survival time of terminal cancer patients.7–10 Midazolam as one of the four cornerstone agents for the promotion of quality care in dying patients, is widely utilized in EU countries for palliative sedation in terminally ill cancer patients. 11 Its rapid onset and short duration of action align with the European Association for Palliative Care guidelines for symptom-targeted pharmacotherapy in end-stage disease management.7,12 However, the Chinese Palliative Care Practice Guidelines did not mention midazolam in its recommendations for symptom control in end-of-life patients (including dyspnea, pain, and agitation).13,14 The absence of domestic data has led Chinese medical teams to remain concerned about the safety of palliative sedation, 15 thereby hindering its further promotion. Particularly for patients with advanced cancer suffering from refractory dyspnea, the risk of midazolam-induced excessive sedation or respiratory depression, if unintentionally overdosed, makes many clinicians hesitant to administer palliative sedation. Additionally, primary doctors tend to have more negative attitudes and greater psychological distress when implementing palliative sedation. 16
Systemic deficiencies in legal frameworks and institutional support mechanisms
The lack of well-established protocols and legislation further complicates the decision-making process for healthcare providers, as they fear potential legal disputes that may arise from the use of palliative sedation, thereby increasing their reluctance to consider this treatment option for their patients. Concurrently, we urge policymakers to accelerate the development of nationally recognized palliative sedation regulations and strengthen related legislative frameworks to ensure equitable protection of stakeholder rights.
The authors have pioneered midazolam-based palliative sedation application for refractory symptom control since 2016. This study conducted a retrospective observational analysis of palliative sedation recipients during the past 9 years. Key findings and clinical experiences are systematically reported, as follows.
Methods
Data collection
A retrospective observational study was conducted on the terminal cancer patients who received palliative sedation in the Department of Oncology at Hangzhou Hospital of Traditional Chinese Medicine between June 2016 and November 2025. Clinical records of enrolled patients were thoroughly reviewed. Key data subsets included: (1) demographic, (2) primary tumor sites, (3) Eastern Cooperative Oncology Group Performance Status (ECOG PS), (4) combination of analgesic, (5) indications for palliative sedation, (6) Richmond Agitation Sedation Scale (RASS) changes before and through palliative sedation, (7) daily dose of midazolam, (8) duration of palliative sedation, (9) efficacy in refractory symptom management, (10) survival time from the initiation of palliative sedation to death, (11) Serious Adverse Events (SAEs) during sedation, and (12) family evaluation after palliative sedation.
Clinical indications
At least two physicians independently assessed the patient’s condition. The clinical indications for palliative sedation included: (1) the terminal stage of cancer, (2) expected survival time is <30 days, as assessed by the dying patient condition assessment sheet, 17 (3) persistent refractory pain, agitation, or dyspnea that cannot be alleviated by conventional treatments, and (4) the RASS score was ⩾0. 18
Decision-making process
In contrast to the patient-centered decision-making model adopted in European Union countries, family members in China play a dominant role in decisions regarding palliative sedation, while also representing the greatest barrier to the timely implementation of such therapy. So, doctors must fully inform them about palliative sedation to gain their support. Finally, an informed consent form must be signed when patients and all family members have reached an agreement to initiate sedation.
Criteria for assessing the efficacy in refractory symptom management
When the patient’s RASS score was <0, the assessment of sedation effectiveness should be determined according to different indications:
For analgesia: the Faces Pain Rating Scale (FPS-R) score ⩽3.
For agitation: RASS score <0.
For dyspnea: peripheral oxygen saturation improved without increasing the oxygen flow rate via nasal cannula.
The identification of appropriate outcome measures for palliative sedation still lacks a well-recognized evaluation standard; therefore, our efficacy assessments remain exploratory, limited by insufficient statistical data and requiring further validation.
Sedation titration protocol
During the very beginning of 5 years (2016–2021), due to limited experience, the initial dosage of midazolam varied widely, and the titration protocol was also absent during palliative sedation, leading to persistent insufficient or excessive sedation in some patients.
Therefore, we launched the second phase of palliative sedation in December 2021. The protocol was as follows: An initial slow intravenous bolus of 2 mg midazolam was administered over 2 min, followed by a continuous maintenance infusion at 0.04 mg/kg/h. Hourly assessments were then performed to evaluate facial expressions, limb movements, verbal communication ability, peripheral oxygen saturation, and other vital signs. These assessments allowed us to determine the Wong–Baker FPS-R and RASS scores, thereby judging whether adequate sedation depth was achieved (Table 1) and whether refractory distress was effectively relieved. If the RASS score was >0 and no SAEs occurred, the infusion rate was increased by 50%–100%. This titration process was repeated until refractory symptoms improved or the RASS score reached −4.
Sedation depth assessment and management form.

RASS: Richmond Agitation Sedation Scale; SAE: Serious Adverse Event.
Through continuous evaluation, we observed that refractory pain and dyspnea could not be effectively relieved when RASS scores were above 0, as patients remained relatively alert. In contrast, the proportion of SAEs was excessively high at RASS −5. For most family members, maintaining a certain level of communication during palliative sedation is preferred, so excessive sedation is not desirable. From the physicians’ perspective, adequately reducing the patient’s level of consciousness is essential to effectively relieve the perception of refractory symptoms. Therefore, classifying RASS scores from −1 to −4 as adequate sedation reflects a shared decision-making process among clinicians, patients, and families that balances therapeutic efficacy, safety, and psychological acceptability. In clinical practice, the criteria for judging whether adequate sedation has been achieved should follow an individualized principle. Once the patient’s refractory symptoms are relieved (see the chapter Criteria for Assessing the Effectiveness of Palliative Sedation), adequate sedation can be considered achieved; a lower RASS score is not necessarily better. This is another reason why the RASS score range defining adequate sedation is set relatively broadly to allow greater flexibility.
Combination therapy
Patients receiving strong opioids for analgesia before sedation may undergo dose conversion to fentanyl patches or co-administration of equivalent morphine with midazolam using a syringe pump for combined analgesia and sedation.
For patients with prominent agitation and delirium, a combination of haloperidol and midazolam may be used for enhanced sedation.
Special considerations
For patients with pulmonary, hepatic, or renal impairment, the initial titration dose of midazolam should be reduced to 1 mg, and dose adjustment should be even more conservative to avoid excessive sedation. Caution was advised when using midazolam in patients taking drugs or fruits that inhibit CYP3A4 enzymes, such as macrolide antibiotics, itraconazole, ketoconazole, nicardipine, and grapefruit, since these may decrease midazolam clearance.
Management of adverse effects
Midazolam-induced respiratory depression may exacerbate respiratory failure. If respiratory rate decreases to 10 breaths/min, or the oxygen saturation (SpO2) <94%, immediately pause the midazolam infusion. In cases of airway obstruction and rapid desaturation, flumazenil 0.5 mg intravenous injection can rapidly reverse the sedative effects of midazolam as an emergency resuscitation. Other adverse effects such as delirium, hallucinations, and hypotension usually resolve after slowing or discontinuing the infusion.
Family member satisfaction survey
During the bereavement period following the passing of sedated patients, we contacted their families via telephone follow-up to listen to and document their evaluations and feedback on palliative sedation.
Analyses
Statistical analyses were performed using Microsoft Office Excel and IBM SPSS Statistics 25.0 software. Categorical variables including gender and primary tumor sites were compared between groups using the chi-square test, while differences were analyzed with nonparametric tests (Mann–Whitney U test). Changes in RASS scores before and after sedation were evaluated using the Wilcoxon signed-rank test. The Kaplan–Meier method combined with the log-rank test was employed to compare differences in time from admission to death between subgroups. Using the multivariate Cox proportional hazards regression model, we assessed the effect of palliative sedation and multiple factors that may influence mortality risk. A two-tailed p-value <0.05 was considered statistically significant. Mediation analysis was performed to examine the potential mediating role of sedation indicators in the relationship between diagnosis (lung cancer or not) and survival time.
Results
The differences between non-titrated and titrated sedation in terms of demographic, clinical characteristics, efficacy, and safety
A total of 689 terminal cancer patients received hospice care in our department, of whom 55 (7.98%) received palliative sedation. 74.5% of sedation consent forms were signed by the patients themselves, while 25.5% were signed by an authorized family member, after all families had provided unanimous assent prior to sedation. Among 55 sedation patients, 62% were men. Lung cancer patients accounted for the highest proportion (43.6%). 70.9% of patients received concurrent strong opioids for analgesia. The most common indication for sedation was dyspnea (47.3%), which differs from the previous literature reports. 19
Among the 16 patients who received non-titration protocol, the adequate sedation rate was 68.75% (11/16), the efficacy in refractory symptoms management was 81.25% (13/16), and the incidence of SAEs was 12.5% (2/16). Analysis of data from three patients with insufficient sedation and poorly controlled refractory symptoms revealed that the primary cause was overly conservative adjustment of midazolam dosage, resulting in inadequate sedation (RASS = 1) and failure to effectively relieve patients’ dyspnea and pain.
In addition, two cases of SAEs (severe respiratory depression) due to excessive sedation (RASS = −5) were identified: one occurred in an underweight, frail female patient (BMI = 17.3, ECOG 4), and the other in a male patient with airway obstruction. As such characteristics are common in sedated patients, we believe that more intensive monitoring and assessment can help reduce the occurrence of SAEs.
Based on the above results and feedback from patients and their families, we adopted 2 mg as the initial dose of midazolam and implemented a structured titration protocol with hourly assessment and dose adjustment, aiming to improve sedation efficacy and reduce serious adverse events (SAEs). Among the subsequent 39 patients who received titrated sedation, 100% achieved adequate sedation. The efficacy rate for refractory symptom management was 92.3% (36/39). The incidence of respiratory depression was 5.13% (2/39), and all events were rapidly reversed by intravenous flumazenil bolus, further confirming the safety of this protocol.
Data statistics showed no significant differences between non-titrated and titrated patients in age, gender, primary cancer sites, ECOG PS, combination analgesic use, sedation indications, sedation duration, daily midazolam dose, efficacy in refractory symptom management, incidence of SAEs, or survival time. In contrast, the proportion of adequate sedation was significantly higher in titrated patients than in non-titrated patients, and the difference was statistically significant (p < 0.001). The mean change of RASS scores in the non-titrated group 3 (2, 4) was significantly weaker than those in the titrated group 5 (4, 5) and difference was also statistically significant (p = 0.008; Table 2).
The differences between non-titrated and titrated sedation patients.

RASS: Richmond Agitation Sedation Scale.
Survival time means the time from the initiation of palliative sedation to death.
Factors influencing survival time of adequate sedated patients
The mean survival time of the 50 adequate sedated patients was 67.45 h, which is comparable to data reported in relevant literature (Figure 1).20–24 If categorized by the indications for sedation, the mean survival time in the dyspnea subgroup was 38.88 h, significantly shorter than that in the analgesia subgroup (125.42 h) and the agitation subgroup (120.33 h; p < 0.001; Figure 2). When analyzing the Cox regression model with sedation indications, gender, disease diagnosis (lung cancer vs non-lung cancer), and opioid analgesic use as independent variables, the results showed that sedation indications served as an independent protective factor. Dyspnea had a significant positive impact on survival risk (HR = 3.743, 95% CI: 1.651–8.483), and it was significantly different from analgesia (p = 0.026, HR = 0.34, 95% CI: 0.131–0.881) and agitation (p = 0.002, HR = 0.277, 95% CI: 0.118–0.606).
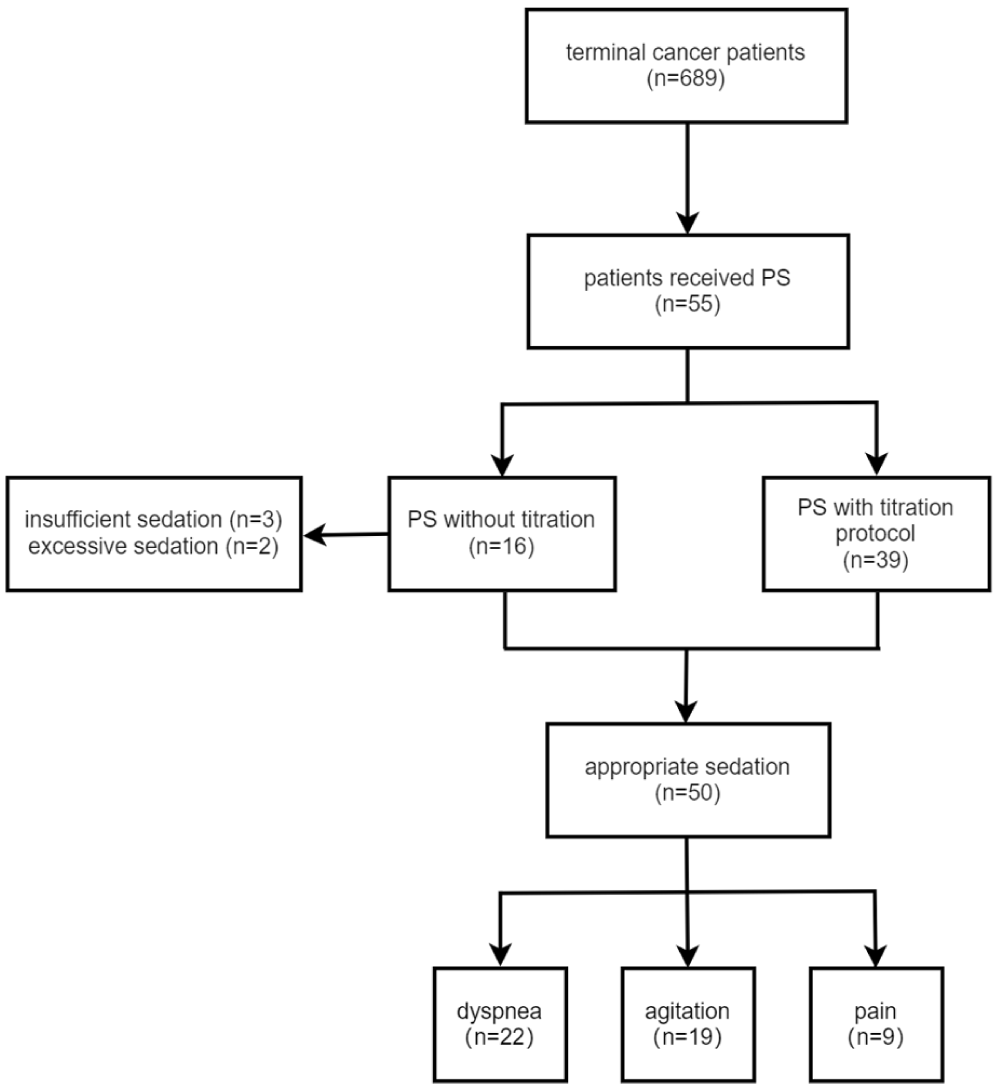
Study flow diagram.

Analysis of survival curves across sedation indication subgroups.
Mediation analysis demonstrated that the regression coefficient a (quantifying the association between diagnosis (lung cancer vs non-lung cancer) and sedation indications (presence vs absence of dyspnea)) was 0.534 (p = 0.039). The regression coefficient b (representing the association between sedation indications and survival time) was 98.323 (p = 0.003). Additionally, the direct effect coefficient c′ (for the association between diagnosis and survival time after adjusting for sedation indications) yielded a value of −67.089 (p = 0.237). The mediating effect accounted for 52.54% of the total effect, with the non-significant direct effect indicating that sedation indications fully mediate the relationship between disease diagnosis and survival time in this study (Figure 3).

Full mediating effect of dyspnea between lung cancer diagnosis and survival time.
Discussion
Due to the vast population and the increasing degree of aging, the incidence and mortality rates of cancer in China are higher than those in Western countries. According to authoritative statistical data, the number of deaths due to malignant tumors in China in 2019 has reached approximately 2.8 million, 25 creating a significant demand for palliative care, including palliative sedation. However, this field has long remained marginalized and has not received adequate attention. Until 2016, in response to the growing demand for palliative care, the Chinese government began to promote the construction of a palliative care system. 26 Despite these efforts, significant bottlenecks persist in advancing palliative care in mainland China.
Strategies to improve family acceptance of palliative sedation
At the initial stage of diagnosis, we gradually introduce the concept of palliative care to patients with advanced cancer and their families, hoping that they can gradually accept the reality psychologically. Nevertheless, when it comes to making decisions regarding palliative sedation, depression, and resistance among families remain extremely common. On the one hand, they wish to relieve the patient’s suffering; on the other hand, they want the patient to remain as conscious as possible. As sedative medications gradually erase the patient’s consciousness, families often feel that their loved one is being slowly taken away. This gap between expectation and reality makes it profoundly difficult for them to accept. Due to the lack of professional psychologists and social workers in our team, there was no effective intervention available to address families’ psychological needs, leading to frequent delays or even abandonment of sedation therapy as decisions were difficult to reach promptly. In the initial years, the rate of palliative sedation implementation in our center was lower than the average rates documented in the EU and neighboring East Asian countries/regions.27–31 During 9 years of clinical practice in palliative sedation, we have continuously refined our protocols and have addressed this challenging issue through joint efforts from two parties.
Enhance the efficiency of communication with patients’ families
We launched an electronic questionnaire on palliative sedation in 2024. Through a series of targeted questions, the families can not only release psychological stress during the answering process but also gain a comprehensive understanding of the goals, benefits, and risks associated with palliative sedation. The questionnaire results showed that 66.7% of the families had no prior knowledge of palliative sedation before. However, after completing the questionnaire, 86.7% of them expressed support for the sedation. Moreover, we found that gaining support from core family members can effectively expedite the sedation decision-making process.
Optimizing the palliative sedation workflow
Compared with early non-titrated sedation, the rate of inappropriate sedation was significantly reduced in patients managed with a titration protocol. Improved sedation quality enhanced physicians’ confidence, making them more persuasive when recommending sedation to patients and their family members.
Through the above interventions, the proportion of patients receiving palliative sedation has gradually increased from 1.96% to 21.7% over the past 9 years (Figure 4). Families’ satisfaction rate with sedation therapy was 95.2%.

Annual trend in sedation rate.
Clinical outcomes and safety of titrated sedation in patients with refractory dyspnea
Significant differences in survival time were observed among patients with different indications for sedation. Patients with refractory dyspnea had a markedly shorter survival time than those with pain or agitation. Mediation analysis demonstrated that patients with end-stage lung cancer and concomitant refractory dyspnea who received sedation had a significantly shorter survival time than those with other complications. This finding was consistent with our clinical observation that terminally ill cancer patients with refractory dyspnea often presented with unstable vital signs, indicating a more critical condition.
Notably, of the 18 patients with respiratory dyspnea who underwent titrated sedation, 16 (88.6%) showed improvement in peripheral oxygen saturation without any elevation in nasal cannula oxygen concentration. We also observed that as the level of consciousness decreased, restlessness and physical agitation were alleviated, and tachypnea gradually returned to a normal respiratory rate. These changes may contribute to reduced oxygen consumption, thereby contributing to the improvement in oxygen saturation, similar to the mechanism by which morphine relieves respiratory distress. 32 Nevertheless, sedation was associated with an attenuated cough reflex, and some patients required endotracheal suctioning to prevent airway obstruction. Therefore, before initiating palliative sedation in such high-risk patients, clinicians must fully inform family members of the potential risks, especially respiratory depression. During sedation, intensive monitoring is mandatory, and drug dosage should be titrated with extreme caution to maintain an optimal balance between symptom relief and safety.
However, it is important to acknowledge that these findings were obtained from a retrospective observational analysis of historical data, which may be subject to residual confounding and indication bias. The findings from our analysis should be interpreted with caution and require further validation. In the future, we will continue to refine the operational details of palliative sedation for such high-risk patients, balancing sedation quality, and safety. Meanwhile, we will further explore communication strategies tailored to the cognitive characteristics of Chinese patients and their families, so as to facilitate the promotion of palliative sedation therapy for terminally ill cancer patients in the Chinese mainland.
Conclusion
The implementation rate of palliative sedation for terminal cancer patients in mainland China is lower than in the EU and neighboring East Asian countries/regions. Education about palliative sedation should be based on a full understanding of traditional Chinese family culture and psychological needs, so that it can be better accepted by more patients and their families. Midazolam-based titrated sedation significantly increases adequate sedation rates and sedation quality. Patients with refractory dyspnea, especially lung cancer patients with this symptom (who may have more severe underlying disease), may have shorter survival times than those with agitation or pain. Rigorous pre-procedural risk communication with families is recommended before sedation, and close monitoring and assessment during sedation are essential for safety.
Footnotes
Acknowledgements
We gratefully acknowledge the expert guidance provided by clinicians from St. Christopher’s Hospice, St. John’s Palliative Care Centre, and the Department of Palliative Medicine at Charing Cross Hospital, St. Mary’s Hospital, and Hammersmith Hospital during the formative phase of our palliative care practice development. None of the authors have any conflicts of interests to report.
Ethical considerations
All methods were conducted under the ethical standards of the Declaration of Helsinki. The study was approved by the Research Ethics Committee of Hangzhou Hospital of Traditional Chinese Medicine under the number 202001291746000301573.
Consent to participate
This was a retrospective observational study, and informed consent was waived by the ethics committee. The electronic questionnaire for family members and the bereavement telephone follow-up were conducted with prior verbal consent from the participants, but were not approved by the Institutional Review Board (IRB).
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable.
