Abstract
Background:
Rates and patterns of inpatient palliative care (IPC) use in metastatic upper urinary tract carcinoma (mUTC) are largely unknown.
Objectives:
To assess temporal trends and predictors of IPC in patients with mUTC.
Design:
Relying on the National Inpatient Sample (2008–2019), we identified 3563 mUTC United States inpatients.
Methods:
Estimated annual percentage changes (EAPC) analyses as well as logistic regression models addressing IPC use were fitted. Subgroup analyses addressed IPC use according to the number and location of metastatic sites.
Results:
Of 3563 mUTC inpatients, 540 (15%) received IPC. Overall, the rate of IPC increased from 2 to 22% between 2008 and 2019 (EAPC +10%). In subgroup analyses, the highest increase in IPC use was observed in patients with liver metastases (from 3 to 33%; EAPC + 11%), in patients with bone metastases (from 3 to 29%; EAPC: +10%), and in patients with three or more metastatic sites (from 0.5 to 31%; EAPC: 10%; all p ⩽ 0.003). In multivariable logistic regression models, liver metastases (odds ratio (OR) 1.91, 95% confidence interval (CI) 1.53–2.39), bone metastases (OR 1.83, 95% CI 1.46–2.28), and contemporary year of admission (OR 1.93, 1.57–2.38; all p < 0.001) independently predicted higher IPC rates. Limitations include the retrospective nature of the inpatient database.
Conclusion:
The IPC rate in mUTC was very low (15%). However, this rate increased to approximately 22% in 2019. The highest IPC rates were recorded in contemporary liver (33%), bone (29%), and three or more metastatic sites (31%) United States inpatients. Therefore, clinicians should be sensitized to early IPC use in mUTC patients in general as well as in other metastatic distribution patterns.
Plain language summary
Early implementation of palliative care in cancer treatment represents a well-established guideline recommendation for patients with advanced cancers. However, patterns of inpatient palliative care use in patients with metastatic upper urinary tract carcinoma are largely unknown.
The research team examined temporal trends and predictors of palliative care use among inpatients with metastatic upper tract urinary tract carcinoma in the United States between 2008 and 2019.
Of 3563 United States inpatients with metastatic upper urinary tract carcinoma, only a minority of 15% received inpatient palliative care. Between 2008 and 2019, the rate of inpatient palliative care significantly increased from 2 to 22%. In particular, the highest average annual increase in inpatient palliative care use was observed in inpatients with multiple metastases as well as those with bone or liver metastases. Even after accounting for other clinical and tumor characteristics, liver metastases, bone metastases, and contemporary years of admission were associated with use of inpatient palliative care.
In the United States, inpatient palliative care use in metastatic upper urinary tract carcinoma was very low. Moreover, this study identified important differences in inpatient palliative care use according to the number as well as the location of metastatic sites. In consequence, the current observations validate the need to sensitize clinicians towards guideline-recommended implementation of inpatient palliative care use, regardless of the number and location of metastatic sites.
Introduction
Palliative care focuses on the optimal management of distressing symptoms and may thereby improve patients’ quality of life.1 –3 Moreover, access to inpatient palliative care (IPC) is considered a favorable indicator of high quality of health care.4 –7 Therefore, the implementation of IPC represents a well-established guideline recommendation for advanced malignancies,3,8 including genitourinary carcinoma, such as metastatic bladder cancer and metastatic upper urinary tract carcinoma (mUTC).9 –11 Despite the overall low rate of IPC use in mainly urological as well as non-urological malignancies, advances evidenced by a higher rate of IPC use have been documented in metastatic bladder cancer.12 –14 However, the field of IPC use is largely unknown in mUTC patients.
We addressed this knowledge gap and hypothesized that rates of guideline-recommended IPC use may have increased over time in mUTC patients. In addition, we proposed that the number and location of metastatic sites may also represent predictors of IPC use in mUTC patients. This hypothesis was based on a previous publication that demonstrated that liver and bone metastases may be associated with worse survival. 15 To test these hypotheses, we relied on a contemporary population-based cohort of mUTC patients within the United States.
Material and methods
Data source
We relied on discharge data from the National Inpatient Sample (NIS) to assess trends and predictors of IPC use in mUTC patients admitted to the hospital between 2008 and 2019. The NIS is a set of longitudinal hospital inpatient databases included in the Healthcare Cost and Utilization Project (HCUP) and formed by the Agency for Healthcare Research and Quality (AHRQ) through a federal-state partnership. 16 All diagnoses and procedures were coded based on the International Classification of Diseases (ICD) 9th revision Clinical Modification (ICD-9-CM), ICD 10th revision Clinical Modification (ICD-10-CM), as well as ICD 10th revision Procedure Coding System (ICD-10-PCS).
Study population
We identified inpatients aged ⩾18 years with a diagnosis of malignant neoplasm of renal pelvis or ureter (ICD-9-CM code 189.1, 189.2 and ICD-10-CM codes C65, C65.1, C65.2, C65.9, C66, C66.1, C66.2, C66.9) and a secondary diagnosis of metastatic disease (ICD-9-CM codes 196.0–196.1, 196.3–196.5, 196.7–196.9, 197.x, 198.x, and ICD-10-CM codes C77.0–C77.1, C77.3–C77.4, C77.6–C77.9, C78.x, C79.x; Supplemental Table 1). Since contiguous invasion and synchronous or metachronous urinary lesions are common in mUTC and do not meet the criteria for distant metastases, patients with exclusive kidney metastases (ICD-9-CM codes 198.0, and ICD-10-CM codes C79.0x) and exclusive metastases in the urinary tract (ICD-9-CM codes 198.1, and ICD-10-CM codes C79.1x) in absence of any other metastatic site were excluded according to previously reported methodology (Supplemental Table 1). 17 In addition, one patient with missing information related to sex was excluded.
Definition of variables for analyses
The primary end point of the study, IPC use, was identified by ICD-9-CM code V66.7 and ICD-10-CM Z51.5, according to prior validation studies.18 –20 The Deyo modification of the Charlson Comorbidity Index (CCI) was used to account for comorbidities based on coding algorithms for defining comorbidities in ICD-9-CM and ICD-10-CM codes by Quan et al. 21 Covariates that were used for adjustment in multivariable logistic regression models consisted of age at admission (years, continuously coded), sex (male vs female), race/ethnicity (Other vs African American vs Caucasian), CCI (≥9 vs 7–8 vs 6), hospital region (South vs Northeast vs Midwest vs West), teaching hospital status (teaching vs nonteaching), and hospital size (small (<200 beds) vs medium (200–399 beds) vs large (≥400 beds)), as well as number (three or more sites vs two sites vs single site) and location of metastatic sites (liver (yes vs no), bone (yes vs no), lymph nodes (yes vs no), lung (yes vs no), and brain (yes vs no)). Critical care therapy (CCT) was defined according to previously used methodology as invasive mechanical ventilation, percutaneous endoscopic gastrostomy tube, dialysis for acute kidney failure, total parenteral nutrition, and tracheostomy (Supplemental Table 1).6,12,22 For univariable and multivariable analyses, year of admission was dichotomized in contemporary (2015–2019) versus historical year of admission (2008–2014). Total hospital charges (in $USD) were inflation-adjusted to the year 2019.
Statistical analyses
First, descriptive characteristics were tabulated. For continuously coded variables, medians and interquartile ranges (IQRs) were reported. Wilcoxon rank sum test examined the statistical significance of differences in medians. For categorical variables, frequencies and proportions were reported. Pearson’s Chi-square test assessed the statistical significance of differences in proportions. Second, estimated annual percentage changes (EAPC) were tested with the linear regression based on log-rates in the overall cohort and in subgroup analyses addressing the number and location of metastatic sites, as well as teaching hospital status and hospital bed size. Third, univariable and multivariable logistic regression models with generalized estimating equations accounting for clustering at the hospital level were fitted for the prediction of IPC use. Adjustment variables included patient and hospital characteristics to account for and to reduce confounding and selection bias.
Analyses and reporting of this study followed the STROBE guidelines for reporting observational studies (Supplemental File 1). 23 For statistical computing and graphics, the R software environment was used (R version 4.3.2; R Foundation for Statistical Computing, Vienna, Austria). 24 Specifically, for logistic regression models with generalized estimating equations analyses, we relied on the “geepack” package (Version 1.3.10). All tests were two sided, with a significance level set at p < 0.05.
Results
Descriptive characteristics
Within the NIS database (2008–2019), we identified 3563 mUTC inpatients (Table 1). Of those, 540 (15%) received IPC. Patients using IPC were more frequently treated in contemporary years (63 vs 46%; p < 0.001). Moreover, IPC patients more frequently exhibited liver metastases (43 vs 28%; p < 0.001), bone metastases (43 vs 30%; p < 0.001), multiple metastatic sites (53 vs 42%; p < 0.001), and higher comorbidity burden (CCI ⩾ 9, 28 vs 22%; p = 0.011). While total hospital charges were comparable between the two groups (p = 0.5), IPC patients had a longer median length of stay (median 6 vs 5 days; p < 0.001) and a higher in-hospital mortality rate (26 vs 4%; p < 0.001).
Baseline characteristics of 3563 metastatic upper urinary tract carcinoma (mUTC) patients, stratified according to inpatient palliative care (IPC) use.
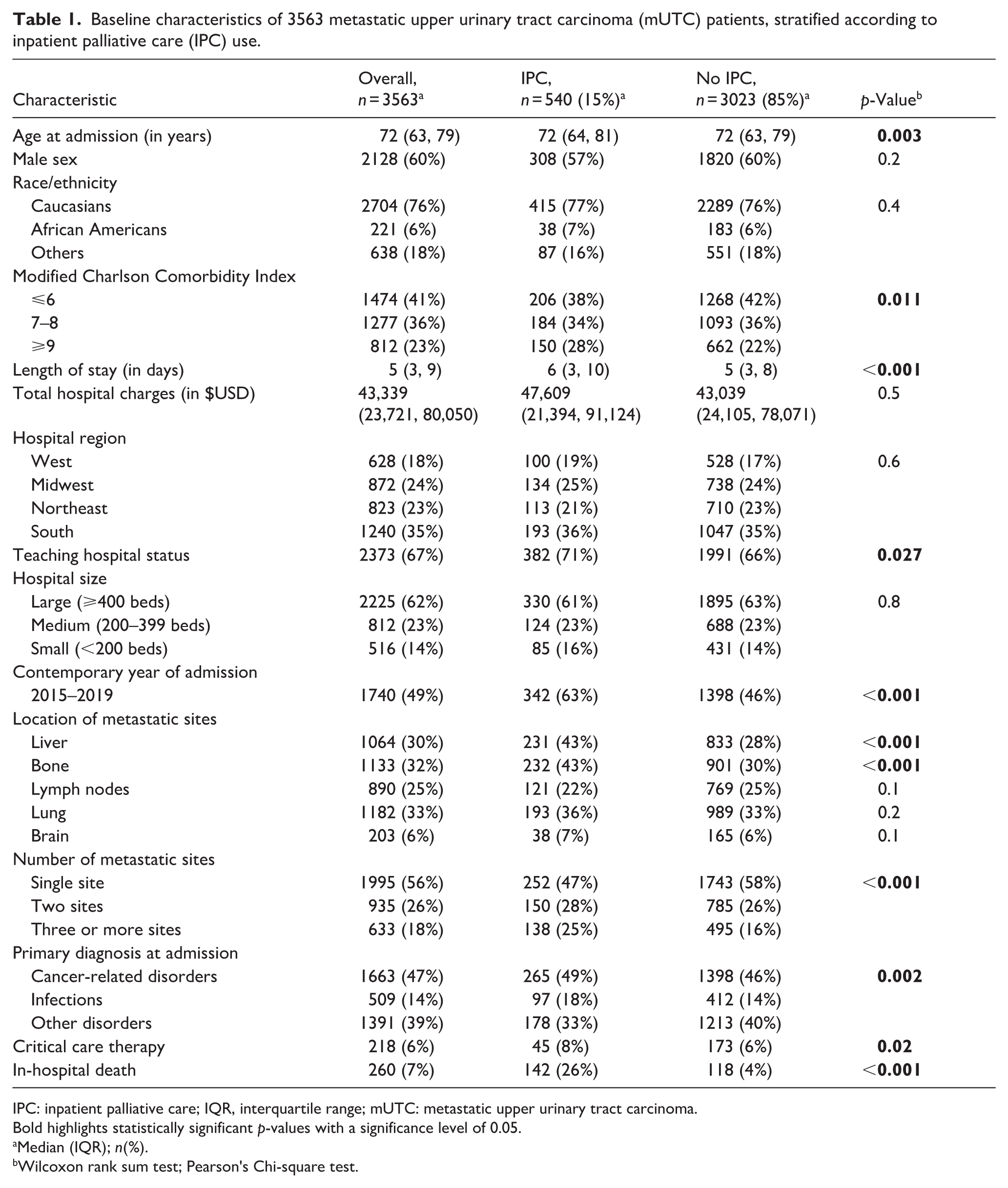
IPC: inpatient palliative care; IQR, interquartile range; mUTC: metastatic upper urinary tract carcinoma.
Bold highlights statistically significant p-values with a significance level of 0.05.
Median (IQR); n(%).
Wilcoxon rank sum test; Pearson's Chi-square test.
Temporal trends of inpatient palliative care use and inpatient mortality
Overall, the rate of IPC use in mUTC patients increased from 2.2% in 2008 to 21.5% in 2019 (EAPC +9.8%; 95% CI: +6.4 to +13.5%; p < 0.001; Figure 1). Conversely, no statistically significant difference in in-hospital mortality was observed over time (EAPC −1.0%, 95% confidence interval (CI) −4.8 to +2.8%; p = 0.6). In subgroup analyses addressing the number of metastatic sites, the rate of IPC use increased from 1.3 to 18.3% in patients with single metastatic site (EAPC +9.7%, 95% CI +5.9 to +14.1%; p < 0.001), from 4.5 to 20.4% in patients with two metastatic sites (EAPC +10.1%, 95% CI +4.1 to +17.4%; p = 0.01), and from 0.5 to 31.1% in patients with three or more metastatic sites (EAPC +9.5%, 95% CI +5.1 to +14.6%; p = 0.003; Figure 2). Focusing on the location of metastases, IPC use increased from 2.5 to 33.1% in patients with liver metastases (EAPC +10.8%, 95% CI +5.9 to +16.7%; p = 0.002), from 3.0 to 28.9% in patients with bone metastases (EAPC +10.3%, 95% CI +6.4 to +14.8%; p < 0.001), from 1.9 to 21.4% in patients with lymph node metastases (EAPC +9.3%, 95% CI +5.6 to +13.4%; p < 0.001), and from 3.6 to 21.4% in patients with lung metastases (EAPC +7.7%, 95% CI +3.7 to +12.2%; p = 0.005) between 2008 and 2019 (Figure 3). After stratification for teaching hospital status, IPC use increased from 0.7 to 22.5% in patients treated in teaching hospitals (EAPC +10.2%, 95% CI +6.1 to +14.8%; p < 0.001) as well as from 3.7 to 17.7% in patients treated in nonteaching hospitals (EAPC +8.6%, 95% CI +4.1 to +13.8%; p = 0.006) between 2008 and 2019 (Figure 4(a) and (b)). Similarly, IPC use increased from 2.7 to 22.9% in patients treated in hospitals with medium size (200–399 beds: EAPC +10.2%, 95% CI +5.3 to +16.1%; p = 0.003) and from 2.6 to 20.9% in patients treated in hospitals with large size (⩾400 beds: EAPC +9.7%, 95% CI +5.3 to +14.7%; p = 0.002; Figure 4(c) and (d)).

Rates of IPC use and mortality over time in mUTC patients within the NIS from 2008 to 2019.

Rates of IPC use over time in mUTC patients within the NIS from 2008 to 2019, stratified according to the number of metastatic sites (single vs two vs three or more sites).
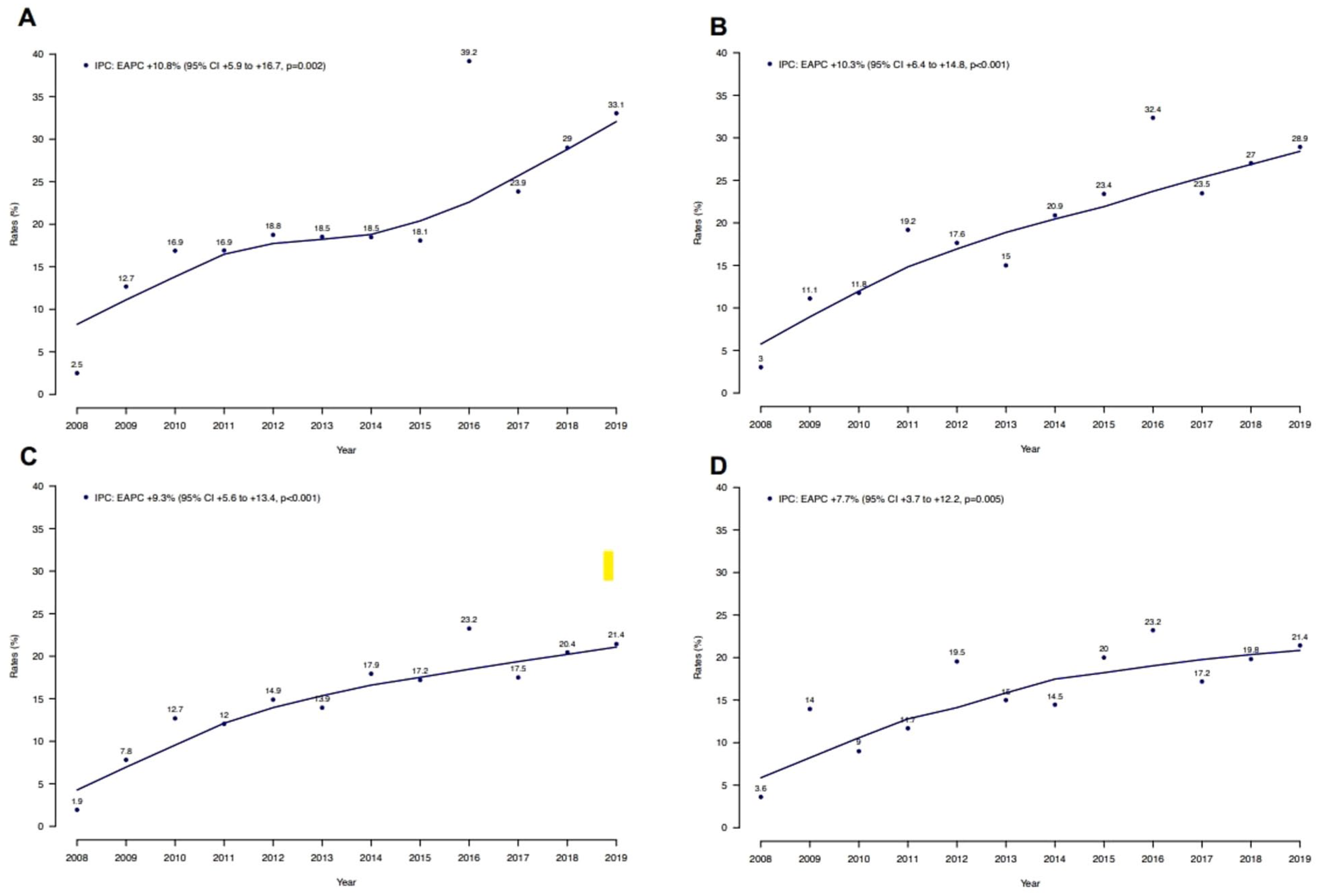
Rates of IPC use over time in mUTC patients within the NIS from 2008 to 2019, stratified according to the location of metastatic sites: (a) liver, (b) bone, (c) lymph node, and (d) lung metastases.
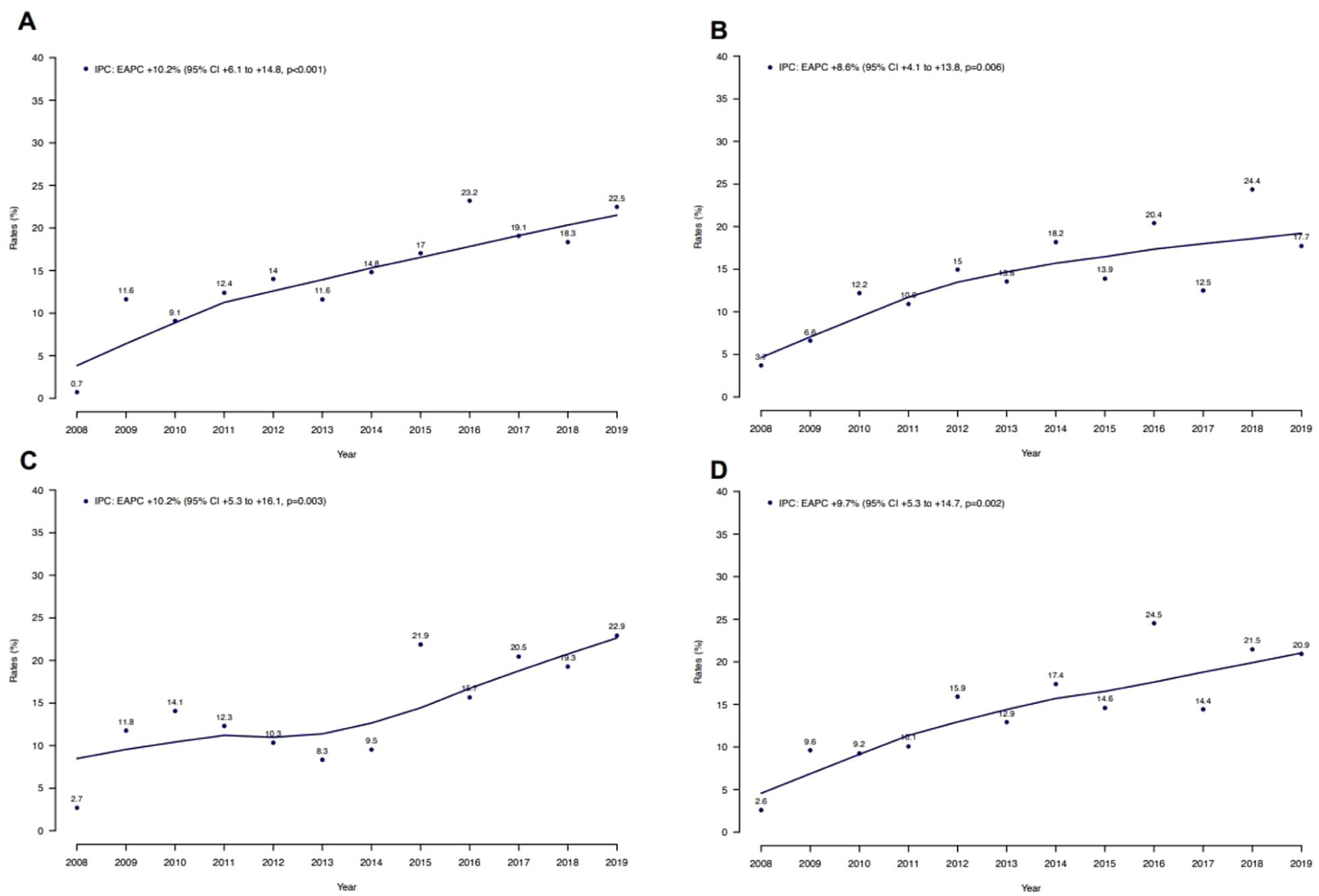
Rates of IPC use over time in mUTC patients within the NIS from 2008 to 2019, stratified according to (a) teaching hospital status, (b) nonteaching hospital status, (c) hospitals with medium bed size (200–399 beds), and (d) hospitals with large bed size (⩾400 beds).
The association between metastatic sites and inpatient palliative care use
In univariable logistic regression models, higher comorbidity burden (CCI ⩾ 9: univariable odds ratio (OR) 1.33, 95% CI 1.06–1.66; p = 0.013), two metastatic sites (OR 1.32, 95% CI 1.07–1.63; p = 0.009) and three or more metastatic sites (OR 1.90, 95% CI 1.52–2.39; p < 0.001), the presence of liver metastases (OR 1.95, 95% CI 1.62–2.34; p < 0.001), the presence of bone metastases (OR 1.75, 95% CI 1.45–2.10; p < 0.001), and admission in contemporary study years (OR 1.97, 95% CI 1.62–2.40; p < 0.001) were associated with IPC use (Table 2). After adjustment for patient and hospital characteristics, only the presence of liver metastases (multivariable OR 1.91, 95% CI 1.53–2.39; p < 0.001), the presence of bone metastases (OR 1.83, 95% CI 1.46–2.28; p < 0.001), and admission in contemporary study years (OR 1.93, 95% CI 1.57–2.38; p < 0.001) remained independent predictors of IPC use.
Univariable and multivariable logistic regression models predicting IPC use in mUTC patients after adjustment for clustering at the hospital level using generalized estimating equation methodology.

CI: confidence interval; IPC: inpatient palliative care; mUTC: metastatic upper urinary tract carcinoma; OR: odds ratio; Ref: reference.
Bold highlights statistically significant odds ratios and p-values with a significance level of 0.05.
Adjusted for age at admission, sex, race/ethnicity, modified Charlson Comorbidity Index, hospital region, teaching hospital status, hospital size, metastatic sites, critical care therapy, and year interval.
Discussion
Temporal trends and predictors of IPC use in mUTC patients are largely unknown. Based on previous observations in patients with advanced bladder and urethral cancer,12 –14,25 we hypothesized that rates of IPC use in mUTC patients may have increased over time. Additionally, we proposed that the number and location of metastatic sites may represent predictors of IPC use in mUTC patients, based on previous observations where certain metastatic sites are predisposed to higher mortality rates. 15 We relied on a contemporary population-based cohort of mUTC and made several important observations.
First, in mUTC patients, the overall rate of IPC use was only 15%. Over time, the IPC rate increased from 2% in 2008 to 22% in 2019 (EAPC +10%; p < 0.001). The currently observed rate of IPC is considerably higher than the IPC rate of 4% previously reported by Joyce et al. 7 in a historical cohort of mUTC treated between 2004 and 2012. Therefore, the present observations are encouraging and imply stronger adherence to guideline recommendations.3,8 However, the IPC rate recorded in the current study is significantly lower than the palliative care use among patients with mUTC reported by Connors et al. (23%) within the National Cancer Database (NCDB 2004–2020). 26 Likewise, the currently recorded IPC rate is lower than the ones described by Han et al. (28%) and Mazzone et al. (25%) in metastatic bladder cancer patients.12,14 In addition, the current IPC rate in mUTC patients is significantly lower than the IPC rate in patients with advanced non-urological malignancies such as metastatic lung cancer (50%) and metastatic breast cancer (22%).27,28 Based on the above observations, IPC rates recorded in metastatic bladder cancer or metastatic lung cancer should ideally be regarded as a benchmark that should represent the objective of IPC use in other metastatic cancers, including mUTC.
Second, we identified important differences in IPC use according to the number of metastatic sites. Patients receiving IPC more frequently exhibited multiple metastatic sites (53 vs 42%; p < 0.001). Although rates of IPC use increased over time in all subgroups (single site: EAPC +9.7, two sites: EAPC +10.1%, and three or more sites: EAPC 9.5%), IPC rates in patients with multiple metastatic sites were generally higher than those in patients with single-site metastases. Since the presence of multiple metastatic sites is associated with less favorable survival in mUTC patients, 15 higher IPC rates in those patients may indicate that the likelihood of mortality or worse survival outcomes may prompt IPC use. These considerations are also supported by Connnors et al., 26 who reported an association between more frequent palliative care use in patients with mUTC and an overall survival of less than 6 months. Moreover, these findings may suggest that palliative care involvement in mUTC patients, contrary to guideline recommendations, occurs not in the early but rather in an advanced stage of disease.3,8
Third, we identified important differences in the use of IPC according to the location of metastases in mUTC patients. IPC patients more frequently exhibited liver metastases (43 vs 28%; p < 0.001) and bone metastases (43 vs 30%; p < 0.001). In separate temporal trend analyses addressing IPC use over time, the highest increase in IPC use was also observed in patients with liver metastases (EAPC +11%) and bone metastases (EAPC +10%; all p ⩽ 0.002). Moreover, even after adjustment for patient and hospital characteristics, including the number of metastatic sites, both the presence of liver metastases and bone metastases remained independent predictors of IPC use in mUTC patients. Specifically, the rate of IPC was two-fold in patients with liver metastases and bone metastases than in others. Taking the differences in IPC use according to the number of metastatic sites into account, these findings suggest that not only the metastatic burden but rather the location of metastases plays a crucial role in access to IPC services. To the best of our knowledge, we are the first to record differences in access to IPC according to the location of metastases in mUTC patients. Therefore, a direct comparison with previous studies cannot be done. However, similar observations by Mazzone et al. and Han et al. in metastatic bladder cancer patients12,14 suggest that metastatic patterns may not only influence IPC use rates in mUTC, but also in other more prevalent metastatic cancers. The present observations may be explained in several ways. First, liver and bone metastases are frequently associated with acute, high-burden symptoms that prompt unplanned hospitalizations. Liver metastases can lead to clinical deterioration through hepatic dysfunction, jaundice, intractable nausea, abdominal pain, or encephalopathy. Similarly, bone metastases may cause pathologic fractures, spinal cord compression, severe pain, and impaired mobility, requiring urgent inpatient treatment. In these situations, palliative care may be consulted to assist with symptom control or goals-of-care discussions. Second, liver and bone metastases are often clinically perceived as more aggressive compared to other metastatic patterns. The clinical perception of less favorable prognosis may further increase IPC use. Taking these considerations into account, higher IPC use rates in mUTC patients with liver and bone metastases imply that initiation of IPC frequently occurs as a response to advanced symptomatic decline rather than as a proactive integration at an early disease stage, as recommended by guidelines. In consequence, the current observations further validate the need to sensitize clinicians toward guideline-recommended implementation of IPC use, regardless of the number and location of metastatic sites.
Despite its novelty, this study has limitations. First, we relied on a large retrospective inpatient database. Despite systematic adjustment for biases and confounders, a potential for selection and reporting biases remained, as is the case with all studies relying on retrospective databases such as the Surveillance, Epidemiology, and End Results and the NCDB.7,13,15,26,29 For example, higher in-hospital mortality observed among patients receiving IPC likely reflects indication bias, as IPC is more likely initiated for individuals with a more advanced stage of disease (e.g., multiple metastatic sites), higher symptom burden, or clinical decline. Second, we relied on previously established and validated methodology based on both ICD-9 and ICD-10 codes for the identification of IPC patients.18 –20,25,30,31 However, different IPC definitions or methodological approaches are possible. Therefore, the use of other approaches, as well as different study cohort selection, may result in IPC rates that may not be directly comparable. Third, palliative care can be delivered in inpatient, outpatient, or home-based settings.1,4 Since the NIS exclusively provides inpatient data, the current study may underestimate overall rates of palliative care, including outpatient and inpatient care. Moreover, compared to a patient-level or cancer-specific registry, the NIS captures hospitalizations rather than unique individuals. Therefore, it is not possible to distinguish between repeat admissions for the same patient or to identify index admissions. As a result, IPC rates may be overestimated if patients with complex diseases or treatment-related complications contribute multiple discharges. Last but not least, the NIS provides only a limited amount of detail. Specifically, the NIS lacks detailed information regarding tumor characteristics, such as tumor stage, molecular characteristics, recurrence rates, or survival outcomes. This limits the ability to adjust for cancer severity, to evaluate disease-specific prognostic factors, or to track care of individual patients over time. In addition, other patient-related characteristics unavailable in the NIS database, such as social support and cultural background, may have influenced medical decision-making, including IPC use. However, multivariable adjustment relied on important and clinically meaningful characteristics available in the NIS database.
Conclusion
The overall rate of IPC in mUTC was very low (15%). However, this rate increased to 22% in 2019. Moreover, the highest rates of IPC were recorded in contemporary United States inpatients with liver (33%), bone (29%), and three or more metastatic sites (31%). Therefore, clinicians should be sensitized to early IPC use in mUTC patients in general as well as in other metastatic distribution patterns.
Supplemental Material
sj-docx-1-pcr-10.1177_26323524261416927 – Supplemental material for Temporal trends and predictors of inpatient palliative care in metastatic upper urinary tract carcinoma: An observational study
Supplemental material, sj-docx-1-pcr-10.1177_26323524261416927 for Temporal trends and predictors of inpatient palliative care in metastatic upper urinary tract carcinoma: An observational study by Carolin Siech, Lukas Scheipner, Andrea Baudo, Mario de Angelis, Letizia Maria Ippolita Jannello, Francesco Di Bello, Fred Saad, Shahrokh F. Shariat, Nicola Longo, Luca Carmignani, Ottavio de Cobelli, Sascha Ahyai, Alberto Briganti, Cristina Cano Garcia, Luis A. Kluth, Felix K. H. Chun and Pierre I. Karakiewicz in Palliative Care and Social Practice
Footnotes
Acknowledgements
Carolin Siech was awarded a scholarship by the STIFTUNG GIERSCH.
Ethical considerations
Analyses and reporting followed the National Inpatient Sample reporting rules. Due to the anonymously coded design of the National Inpatient Sample, study-specific ethics approval was not required by the institutional review board.
Consent to participate
The Healthcare Cost and Utilization Project – National Inpatient Sample is deidentified, thus consent is not required.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
