Abstract
Background:
Medical assistance in dying (“MAiD”) became legal across Canada in 2016, and in Québec in 2015. Provincial/territorial regulatory bodies play a critical role in MAiD as they can issue binding requirements on health practitioners. Law and regulatory standards are the “twin pillars” of MAiD regulation, yet the content of MAiD practice standards for physicians is unstudied.
Design:
This article analysed MAiD guidance for physicians from Canadian medical regulators (often called the “College of Physicians and Surgeons”), using a qualitative descriptive approach, informed by regulatory space theory.
Methods:
We identified MAiD-specific regulatory documents (practice standards and related documents) using web-based searches and follow-up inquiries. We analysed the documents using qualitative descriptive analysis and the Framework Method, facilitated by NVivo. The analysis focused on identifying areas where regulators issued guidance beyond the law.
Results:
We identified 15 regulatory documents from 11 of the 13 provinces and territories. We determined that these documents primarily outline the law without detailed guidance on how to apply it. We identified eight areas for which regulators provided guidance that went beyond the MAiD-specific legislation, most relating to core aspects of medical practice, such as competency, documentation, and patient-centred care. The rights and obligations of conscientious objectors were a predominant focus in all documents. The documents largely lacked guidance about the meaning of terms in the legislation. There was also variation in standards between provinces and territories; the documents focused on similar topics but varied in their policy choices. Physicians in each province/territory are therefore subject to differing expectations (to some extent).
Conclusion:
This study highlights a gap in guidance on the meaning of legal terms in the
Plain language summary
In Canada, medical assistance in dying (MAiD) became legal in 2016, with Québec legalizing it in 2015. In each province and territory, regulatory bodies like the “College of Physicians and Surgeons,” play a key role in guiding physicians on how to carry out MAiD. However, there has been little research on regulators’ practice standards for physicians. This study examined MAiD-related guidance for physicians from Canadian medical regulators. We found 15 documents from 11 provinces and territories that were specific to MAiD. The goal was to see how these documents went beyond the law and provided additional requirements and advice on how to handle MAiD in practice. The results showed that most documents mainly restate the law, with limited extra guidance. However, there were eight areas where regulators offered advice that went beyond the legal requirements. These areas included medical competencies, proper documentation, and patient-centered care. One key focus in all documents was the rights of physicians who object to MAiD for personal or moral reasons. The study also found that the guidance varied between provinces and territories. While most documents covered similar topics, the specific expectations differed somewhat. This may create some confusion for physicians, as they may face different standards depending on where they practice. Overall, the study points out the lack of clear guidance on the meaning of certain legal terms and highlights the variation in MAiD regulation for physicians across Canada.
Keywords
Introduction
A growing number of jurisdictions, including the Netherlands, Belgium, Switzerland, Canada, Colombia, Spain, Germany, Austria, Portugal, New Zealand, 7 of 8 jurisdictions in Australia, and 11 jurisdictions in the United States, have legalized some form of assisted dying (“AD”). 1 AD involves a physician (or in some jurisdictions, a nurse practitioner) administering or prescribing a lethal substance to a patient who is suffering, at their request, to end their life.
Debates regarding AD are polarized, and jurisdictions around the world have engaged in significant deliberation about whether to permit it and in what circumstances. Beyond the question of whether to legalize AD, jurisdictions must also consider how to regulate it. What constitutes “regulation” is contested, and it is increasingly seen as broader than just law.
2
Regulatory scholar Julia Black defines regulation as: . . .the sustained and focused attempt to alter the behaviour of others according to defined standards or purposes with the intention of producing a broadly identified outcome or outcomes, which may involve mechanisms of standard-setting, information-gathering and behaviour-modification.
2
Modern scholars like Black see regulation as polycentric, involving state and non-state-based actors that interact in complex ways, using a range of mechanisms including law, policy, guidelines, ethical codes, training, and funding.2,3
AD regulation varies internationally both in its content and in the instruments used to steer behaviour. For example, Belgium and the Netherlands have concise legislation supplemented by other instruments, including professional standards and institutional ethics policies.4,5 Additionally, in Belgium, many healthcare institutions mandate patients requesting AD to consult with a specialized palliative care team, even though this requirement is not part of the AD legislation (known as the “palliative filter” norm). 5 The legislation in Australia, which varies by jurisdiction, is much more comprehensive. Each state and the Australian Capital Territory has a lengthy and prescriptive AD statute that governs each step of the AD process. 6 Nevertheless, some key issues, such as institutional objection in the state of Victoria, are left to policy.7,8 In Canada, criminal law is federal, while healthcare implementation (including the practice of AD) is the responsibility of the provinces and territories. 9 Canada’s AD regulatory system involves both law and professional practice standards, 10 which are supplemented by other mechanisms including regional health authority policies, 11 and professional association guidelines. 12
Given their binding nature, we can conceive of law and regulatory standards as the “twin pillars” of Canadian assisted dying regulation. There are two primary legal frameworks for medical assistance in dying (“MAiD”) in Canada (summarized in Table 1). Québec passed the
Overview of medical assistance in dying provisions in Canada’s
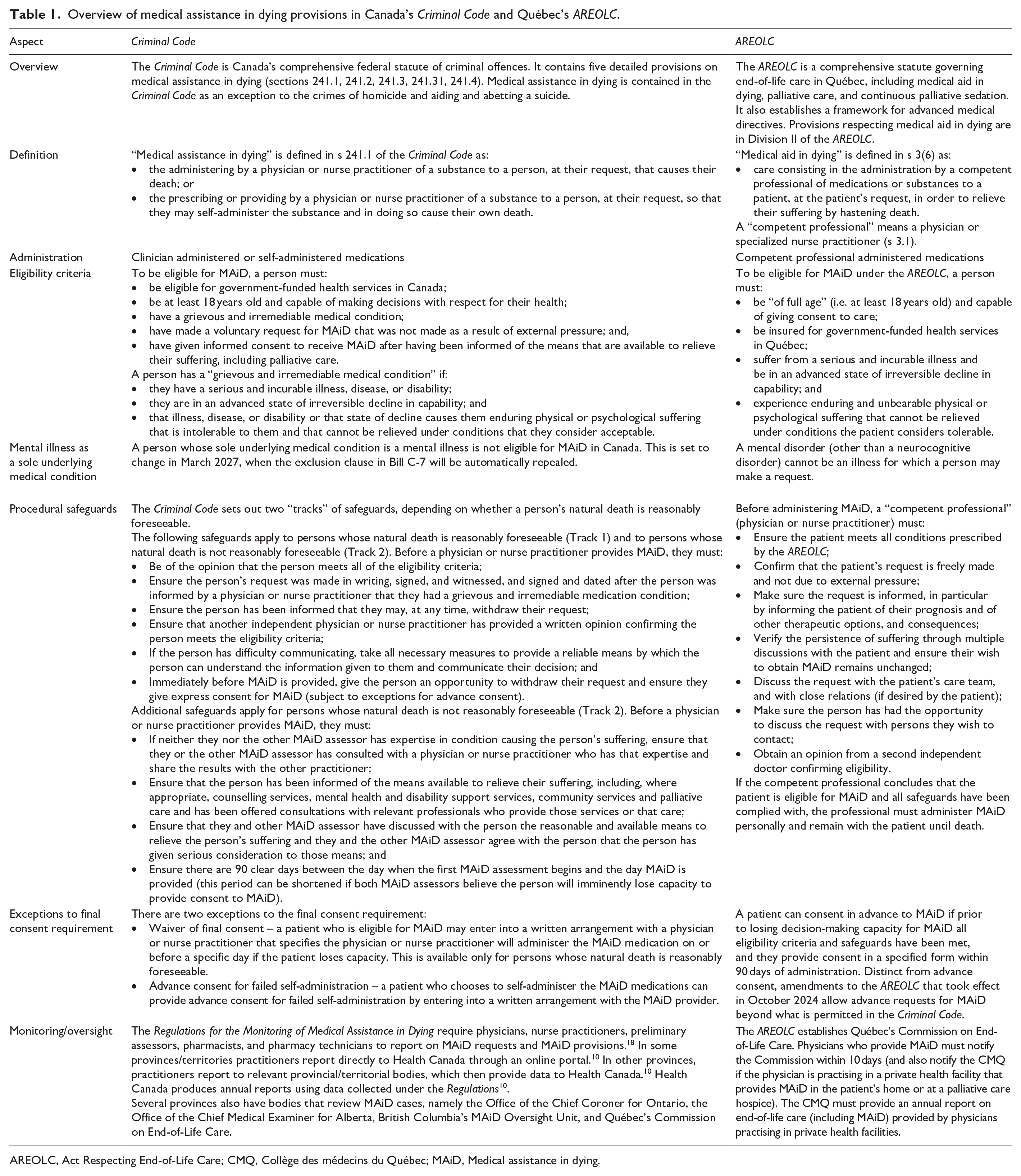
AREOLC, Act Respecting End-of-Life Care; CMQ, Collège des médecins du Québec; MAiD, Medical assistance in dying.
Research has tended to focus on MAiD law and its implementation,19 –25 with comparatively less attention paid to standards. Existing regulatory studies have examined guidance for nurses, 16 pharmacists, 17 health authority policies, 11 practices surrounding death certificates, 26 and rural considerations in regulatory documents. 27 These studies have found that, like other healthcare practices, there is considerable variation in how each province and territory regulates MAiD, a function of Canada’s federated structure.11,15,16,28 Some argue that variation in regulation by province and territory is problematic; it wastes resources, and may confuse health professionals and the public.29 –31 On the other hand, regulatory variation may reflect and be responsive to the specificity of jurisdictional needs and priorities.
To our knowledge, there is no research to date on MAiD regulatory documents for physicians in Canada. This is an important gap to fill. Physicians and nurse practitioners are the only two professions authorized to assess and provide MAiD, with physicians providing the majority of MAiD procedures (90.6% in 2022). 10 Non-compliance with practice standards is serious; it can subject physicians to investigation and discipline, up to and including the loss of their licence to practice. 13 Additionally, as illustrated by a 2018 legal case challenging the Ontario College of Physicians and Surgeon’s “effective referral” requirement for objecting physicians, the framing of practice standards can directly impact patient access to MAiD. 32 Canada’s regulatory approach of law scaffolded by practice standards may also provide insights for other countries considering or implementing AD, particularly those that have some separation of regulatory authority, such as Australia.
This article examines practice standards and related documents (i.e. “regulatory documents”) issued by each Canadian province and territory’s medical regulator. As a threshold point, we note that while there is a diversity of opinions on whether MAiD should be permitted, and in what circumstances, this article steps beyond these discussions and recognizes that MAiD is now legal in Canada. The aim of this article is to investigate how medical regulators have responded to the law and what guidance they have provided to physicians. The study investigated three key research questions:
(1) What MAiD-specific regulatory documents have been released by provincial/territorial medical regulators?
(2) For which topics do these documents aim to regulate behaviour beyond legal obligations?
(3) Does this content vary across provinces and territories, and if so, how?
Regulation of the medical profession in Canada
In Canada, the medical profession is self-regulating. With some exceptions, each province and territory has delegated the authority to regulate physicians to “Colleges,” 33 non-profit corporations empowered by legislation to act as the governing professional organization (in the Yukon, the regulator is called the Yukon Medical Council rather than a “College”; Table 2). The Northwest Territories and Nunavut do not have Colleges; the government is the regulatory authority. This article refers to these bodies as “regulators.”
Canadian medical regulators by jurisdiction.
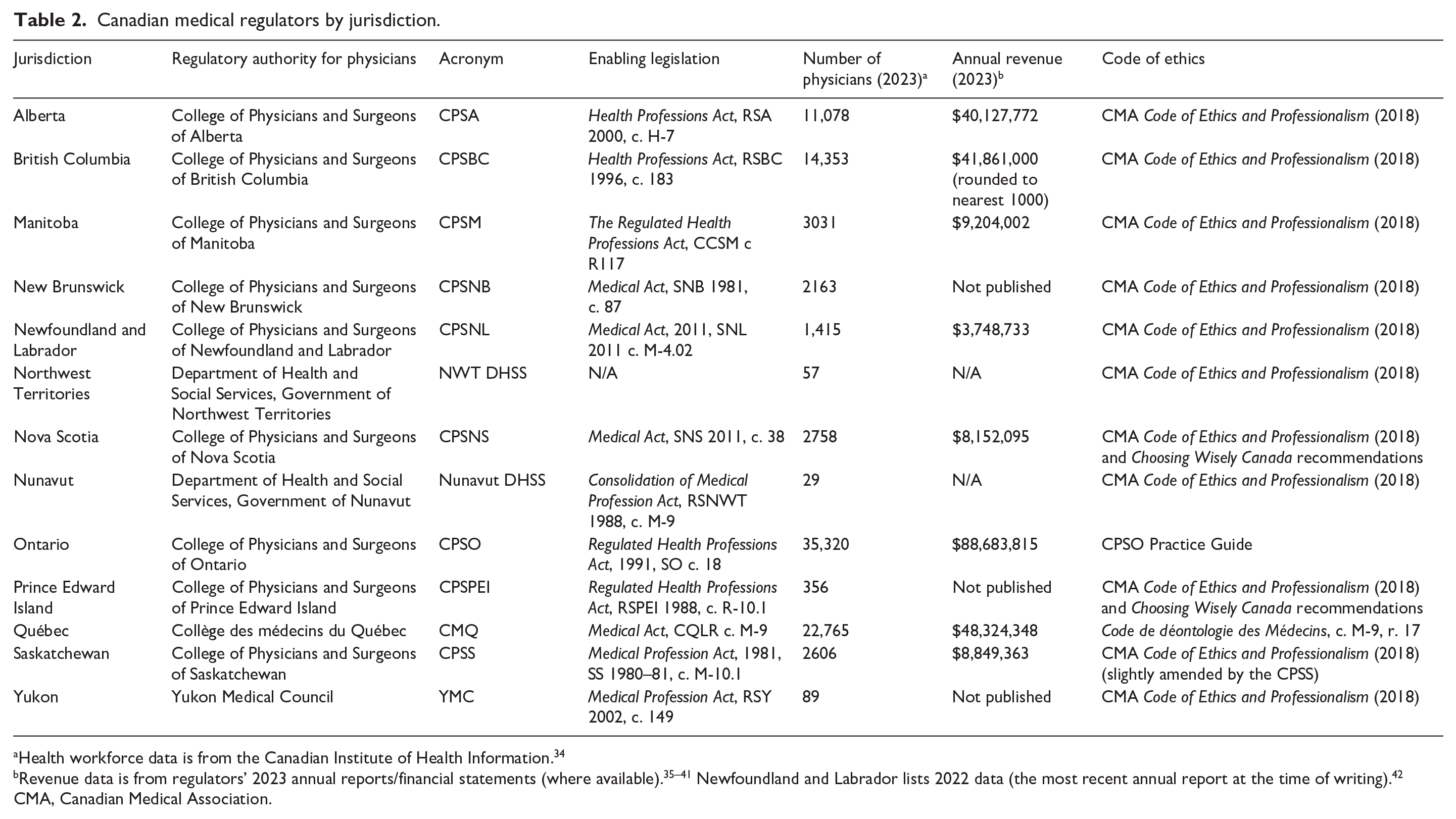
Health workforce data is from the Canadian Institute of Health Information. 34
Revenue data is from regulators’ 2023 annual reports/financial statements (where available).35 –41 Newfoundland and Labrador lists 2022 data (the most recent annual report at the time of writing). 42
CMA, Canadian Medical Association.
Most regulators have a similar governance structure determined by legislation, with a board (or council) as the primary governing and standard-setting body, and committees that have powers and duties exercised on behalf of the board. The size and budget of each regulator varies widely (Table 2). The regulator is responsible for: licensing postgraduate medical trainees and physicians; setting policies, guidelines, and standards for safe and ethical practice; and responding to complaints from the profession and the public, including conducting investigations and hearings, and disciplining members who fail to meet the regulator’s standards and expectations. 13
The central mandate of the regulators, like other professional regulatory bodies, is to promote and protect the public interest. For example, Alberta’s
Most Colleges limit their role to the regulatory functions of licensing, quality assurance, and discipline, and leave advocacy, position-taking, and collective bargaining with the government to provincial/territorial medical associations.
13
Notably, the Collège des médecins du Québec (CMQ) has a more active role than its counterparts in other provinces and territories.
13
It takes positions in the media and has its own
The regulators guide medical practice using a range of instruments, including codes of ethics45,46 and standards (called “standards of practice” or “policies”), which set out the minimum requirements and recommendations across a range of practices. For example, the College of Physicians and Surgeons of Alberta has over 40 standards of practice on issues ranging from prescribing to maintaining patient records to relationships with industry and patients.
47
The requirement to develop standards is set out in legislation (e.g., Ontario’s
Method
Analytical framework
This study is a qualitative content analysis of MAiD regulatory documents for physicians. It is part of a broader project that aims to develop an optimal holistic model for AD regulation, 50 premised on Black’s broad definition of “regulation” 2 and “regulatory space” theory, which suggests actors and their regulatory tools interact to shape behaviour.3,51
We defined “regulatory documents” as documents that set standards and offer physicians MAiD-specific guidance. We included supplementary documents like “advice to the profession” statements, as they inform the interpretation of practice standards and can be considered a component of standard-setting. We excluded general medical practice standards from our analysis (e.g. those pertaining to informed consent), as the focus was on MAiD-specific standards. It is important to emphasize, however, that MAiD-specific standards are situated in a network of relevant general medical practice standards, to which all MAiD assessors/providers are expected to adhere. This is reinforced by the
Search strategy
The authorial team was comprised of three legal academics and a psychiatrist, all with MAiD-related regulatory expertise. Regulators were identified through legislative review (Table 2). We employed a two-stage search strategy: (1) searches of each regulator’s website and grey literature; and (2) email inquiries (where required). The lead author (EC) searched each regulator’s website for current MAiD regulatory documents using drop-down menus and a variety of search terms individually and in combination, including: medical assistance in dying, MAiD, standards of practice, guidance, policy, advice, advice to the profession, and FAQs. Repeated searches were conducted from April 2022 and were finalized on 1 September 2023. EC also identified and examined relevant literature to triangulate searches on medical regulator websites.
EC located publicly available regulatory documents on medical regulator websites in each province and territory, except Québec, New Brunswick, and Nunavut. In these provinces, EC followed up with email inquiries. In Québec, the CMQ provided its MAiD policy. On 12 October 2022, EC again emailed the relevant regulators in New Brunswick and Nunavut to request any MAiD regulatory documents but received no response (these jurisdictions do not have a MAiD standard).
Analysis
For context, the research team reviewed the mandate of each regulator as set out in the relevant legislation (Table 2) and as stated on their websites, noting the contrast between the CMQ and counterparts in other provinces and territories. The research team also drew on their expertise with
All documents were uploaded into NVivo qualitative data analysis software (release 1.6, QSR International). We employed Sandelowski’s qualitative descriptive analysis, 52 which has been used in previous studies of nursing and health authority regulatory documents.11,16 This approach produces “thematic surveys,” with the analysis and interpretation of the findings remaining descriptive and “data-near.” 52 To manage the data, we used the Framework Method of qualitative content analysis, a method increasingly used in policy studies to code and systematically chart data into a matrix to facilitate comparison and analysis.53,54
EC conducted the initial analysis. EC read all documents in full and then coded each one, focusing on elements that were not merely a restatement of the law, but rather new obligations or guidance from the regulator. Based on discussions with all authors and reference to EC’s reflexive journal created while coding, EC grouped codes into themes. EC then charted the data into a matrix to aid in comparison and analysis. The authors iteratively discussed the matrix and refined the final themes. The manuscript was written in accordance with the SRQR reporting guidelines (Supplemental Material). 55
Results
Sample of regulatory documents and their characteristics
We identified 15 regulatory documents on MAiD from 11 regulators (Table 3). The regulatory documents were labelled:
standards of practice (seven documents);
policy (four documents; which effectively function as standards);
guidelines (one document); and
advice to the profession (three documents).
The documents ranged from 4 to 102 pages (median = 12 pages) and had reasonably consistent style. Most documents were between 4 and 17 pages and focused on legal requirements and professional expectations for physicians. In contrast, documents from Québec and the Northwest Territories were considerably longer, at 102 pages and 35 pages, respectively.56,57 The Northwest Territories document was addressed to multidisciplinary teams, not just physicians. 57 The Québec document contained much more clinical guidance than the other jurisdictions and referenced a medication protocol developed by the Institut national d’excellence en sante et en service sociaux (“INESSS”), at the CMQ’s request (this document is in a “restricted access” section of the INESSS website, which one of the authors (M.G.) had access to by virtue of being a physician licensed in Québec). 58 The documents cross-referenced other sources including College standards and ethical codes.
Overview of provincial/territorial medical regulatory documents on medical assistance in dying.

The College of Physicians and Surgeons of Ontario (CPSO) issues “policies,” “statements,” and “positions.” It indicates policies: “set out expectations for the professional conduct of physicians practising in Ontario.”
58
Advice to the profession documents “are intended to provide physicians with additional information and general advice in order to support their understanding and implementation of the expectations set out in policies.”
59
In
The CPSPEI issues “policies” and “guidelines.” It indicates policies: “reflect the minimum professional and ethical behaviour, conduct or practice expected by the College of Physicians and Surgeons of PEI. Physicians licensed with the College are required to be familiar with and comply with the College policies.” 60
The CMQ’s regulatory document was on a secure website and was only available to the public on request. At the time of the analysis, there were two versions of the CMQ’s document, one in French (updated November 2019) and one in English (updated April 2018).55,61 The foreword to the document in French indicates that the November 2019 updates concerned revisions to discrete areas: obtaining medicines and dealing with the pharmacist; the medical prescription; clarifications regarding administration of the MAiD substance; and updates to forms. 61 In May 2022, the pharmacological guidelines of the November 2019 update were replaced by a new national medical protocol developed by INESSS at the CMQ’s request. 57 On 24 January 2023, the CMQ confirmed via email correspondence that the English version of the CMQ regulatory document contains the same content the document in French, but for the updates listed in the foreword. Given the lead author of the study is English-speaking, and the updates were only to these discrete areas, the analysis was conducted using the English version of the CMQ document. The lead author also reviewed a DeepL English translation of the French version to check for any unique content relevant to this analysis.
The CPSS uses policy to set out expectations of the College for physicians practising in Saskatchewan.62,63
CMQ, Collège des médecins du Québec; CPSA, College of Physicians and Surgeons of Alberta; CPSBC, College of Physicians and Surgeons of British Columbia; CPSM, College of Physicians and Surgeons of Manitoba; CPSNL, College of Physicians and Surgeons of Newfoundland and Labrador; CPSNS, College of Physicians and Surgeons of Nova Scotia; CPSO, College of Physicians and Surgeons of Ontario; CPSPEI, College of Physicians and Surgeons of Prince Edward Island; CPSS, College of Physicians and Surgeons of Saskatchewan; MAiD, medical assistance in dying; NWT DHSS, Department of Health and Social Services, Government of Northwest Territories; YMC, Yukon Medical Council.
Our analysis focused on obligations beyond the law, but we note that all documents included information about the law. The legal content was descriptive; the documents generally avoided interpreting or defining legal terms, instead referring physicians to seek legal advice if unsure (although a few documents did define key legal terms, discussed further in Theme 4). Uniquely, the CMQ’s document was designed to help clinicians reconcile the federal law in the
Consistent with the regulators’ mandate, the documents stated their purposes were to:
provide information on legal and procedural requirements for MAiD (for physicians and the public); and
set out the law and guidance about physicians’ professional responsibilities.
We identified eight themes involving guidance and additional standards beyond the law (Table 4). Key aspects where the documents aligned and varied are set out in Table 4.
Topics that Canadian medical regulators have issued guidance on regarding medical assistance in dying.

MAiD, medical assistance in dying.
Theme 1: Balancing patient access with physicians’ objections to MAiD
All documents addressed conscientious objection, a refusal to participate because of personal, moral, or religious convictions. Nova Scotia referenced “non-participation,” encompassing conscientious objection and “various practical reasons such as lack of availability or lack of expertise.” 65
The
a.
Expectations varied somewhat in the third category, despite similar language. For example, the Manitoba document instructed physicians to provide: “timely access to a resource [such as other registrants, healthcare providers, and publicly available resources] that will provide accurate information about MAiD.” This aligns with provincial legislation which confirms that professional regulatory bodies in Manitoba cannot require participation: “a professional regulatory body must not make a regulation, by-law, rule or standard that requires a member of the regulated profession to provide or aid in the provision of medical assistance in dying”71, 74 British Columbia and Nova Scotia required an “effective transfer of care”: . . .advising patients that other practitioners may be available to see them, suggesting the patient visit an alternate physician or service, and if authorized by the patient, transferring the medical records as required.
72
In Nova Scotia, “effective transfer of care” also requires the practitioner to transfer the patient to a person who is willing to accept the transfer and . . .positive action to ensure the patient is connected in a timely manner to a non-objecting, available, and accessible physicians, other health-care professional, or agency that provides the service or connects the patient directly with a health-care professional who does.59,60
Six medical regulators explicitly required the physician to transfer the patient’s records (Table 5).
b.
c.
d.
Overview of obligations to patients requesting MAiD for physicians who have a CO.

CMQ, Collège des médecins du Québec; CO, conscientious objection; CPSA, College of Physicians and Surgeons of Alberta; CPSBC, College of Physicians and Surgeons of British Columbia; CPSM, College of Physicians and Surgeons of Manitoba; CPSNL, College of Physicians and Surgeons of Newfoundland and Labrador; CPSNS, College of Physicians and Surgeons of Nova Scotia; CPSO, College of Physicians and Surgeons of Ontario; CPSPEI, College of Physicians and Surgeons of Prince Edward Island; CPSS, College of Physicians and Surgeons of Saskatchewan; MAiD, medical assistance in dying; NWT DHSS, Department of Health and Social Services, Government of Northwest Territories; YMC, Yukon Medical Council.
Theme 2: Ensuring requisite care, knowledge, and skill
The
a.
b.
c.
Theme 3: Facilitating patient-centred decision-making and care
Guidance on facilitating patient-centred decision-making and care related to five distinct aspects. These centred on who to involve and the practical aspects of MAiD provision.
a.
b.
c.
d.
e.
f.
Theme 4: Assisting physicians to satisfy legislative criteria and safeguards
The
a.
b.
c.
d.
e.
f.
f.
Theme 5: Safe and effective MAiD provision
The
a.
b.
Theme 6: Documentation and accountability
Regulators emphasized thorough documentation and compliance with accountability mechanisms to protect the patient, the physician, and the public.
a.
b.
c.
Theme 7: Professional ethics
The
a.
b.
c.
Theme 8: Support for physicians involved in MAiD
Regulators “encouraged,”65,70 or “strongly encouraged”68,75 professional support including support for the emotional challenges of being involved in MAiD.
a.
b.
The documents suggested resources to support well-being, including those from the CMA, provincial/territorial medical associations, and employee assistance programs. Québec provided additional suggestions, including turning to colleagues, support groups in an institution, team meetings, and an individual consultation with a psychologist or social worker. 56
Discussion
This article provides important insight into the obligations of physicians who encounter patients seeking MAiD. It also showed how different regulators have approached the same federal law (the MAiD framework in the
MAiD regulation, like other aspects of healthcare, involves a broad range of bodies and actors that steer the system. 2 Provincial and territorial medical regulators are a powerful player in Canada’s MAiD “regulatory space.”51,79 They can investigate and sanction physicians and are an important mediator between the public and the profession. As a result, they can be seen as the “next level down” from the law. They have the potential to provide needed guidance on MAiD to physicians and to bridge some of the federal-provincial/territorial divide. However, this study suggests that the regulators do not occupy this middle space in some key respects. We found that regulatory documents focus primarily on describing (but not defining or offering interpretations of) the MAiD law, largely reflect existing medical practice, and defer to guidance from other regulatory actors such as provincial health departments, Health Canada, and CAMAP.
Professional standards are centred on the MAiD law
A key part of developing MAiD competency is learning to apply the legal framework to clinical practice.16,21 Canada’s MAiD provisions in the
Professional standards reflect existing medical practice
The regulators provided some guidance where the law (particularly the
Professional standards connect with and defer to other “regulatory actors”
This study supports the view that MAiD regulation in Canada, while centred on twin pillars of the law and professional standards, also reflects “networked governance,” through an interconnected set of actors across organizations. 3 The documents reinforced guidance from Health Canada, provincial/territorial governments, and other professional organizations, including CAMAP. Alignment in guidance from various regulatory actors helps physicians to know what to do, because they receive consistent normative guidance and so do not have to reconcile competing positions. However, when it comes to facing challenging and complex cases, silence from medical regulators leaves physicians to look to other regulatory actors for guidance, such as their provincial/territorial government, local authority, healthcare institution, and professional societies.
Causes of variation in standards
The documents focused on broadly similar topics, yet regulators made distinct policy choices on a range of issues (Table 4). Several factors may account for this variability. As a threshold point, variation in provincial/territorial standards is not unique to MAiD, given Canada’s approach to health professional regulation (which some see as outdated).33,82 Each medical regulator operates independently,
33
with its own local context, policies, and interactions with other key actors in the regulatory space, such as health authorities, and regulators of other health professions. Budget differences may also influence resources that various regulators have available for the development of MAiD-specific standards and guidelines. For example, Ontario, a large province, has nearly 20 times the revenue of the regulator in Newfoundland and Labrador (Table 2). Another factor is regulators’ obligation to consult when changing practice standards, a model that has been criticized as less responsive and flexible than in other countries.
82
Variability may also be a product of the regulators playing distinct roles in MAiD reform and implementation. Québec’s CMQ was deeply involved, actively initiating reflective inquiry into MAiD and taking a lead role in civil society engagement.
13
The
Implications of findings for policy and practice
This study highlighted a gap in guidance about the meaning of legal terms in the
A key implication of this study is that polycentric regulation can lead to fragmentation. Other studies have demonstrated that this creates uncertainty and is challenging for physicians to navigate.22,31,80,83 Gaps in guidance and variability (whether from the constitutional division of powers or polycentric regulation) leave physicians to try their best to determine who they should be following among a range of regulatory actors, including the federal and provincial/territorial governments, local health authorities, professional organizations, and healthcare institutions. Coupled with the prospect of
Whose role then is it to provide practitioners with guidance on the meaning of the MAiD law, and what form should it take? The federal government could have done so through defining the key terms in the legislation. It did not do so. The courts could provide an authoritative interpretation, but other than in the
In 2023, a Model Practice Standard was released by Health Canada, designed to help regulators align their practice and “ensure the protection of the public in . . . complex cases.” 10 Yet, at the time of writing this article the regulators had not adopted this standard wholesale or agreed to harmonize their MAiD standards. Is variation in regulatory documents problematic? On one hand, some argue that inconsistency can be confusing to physicians and the public,11,30 and represents duplicative resource investment. 29 Aspects of the documents may also impact patient access. There are regional differences in MAiD prevalence in Canada, 78 and internationally,85,86 and reasons for these differences are still emerging (Québec is actively exploring the question of why its rates of MAiD are higher than some other provinces/territories. 87 ) Existing research suggests that cultural factors, and factors that facilitate or hinder a patient’s ability to connect to the MAiD system, including regulatory and institutional policies, may play a part in these differences.86,88 For instance, Canadian jurisdictions requiring effective referral connect patients to MAiD services, whereas those with an “information-only” approach risk less access by placing the burden on patients to take positive steps (even when they do not have the information or power to do so). On the other hand, variability in regulatory documents may not necessarily be problematic as it may reflect a jurisdiction’s unique cultural context. Variation is inevitable in healthcare more generally, 91 and other areas of medicine also lack uniform national standards. Variation between jurisdictions can also lead to systems improvements provided regulators are having “regulatory conversations” 89 and are open to observing and learning from each other. 3 This reflects the idea of provinces/territories as “laboratories of democracy,” testing and refining policies (an approach that is also seen in other international jurisdictions, such as Australia) 90 .
Numerous studies have described challenges with implementing MAiD, and how resources for practice have developed over time.19,21,22 In Australia, for example, where both criminal law and health care are regulated at the state level, each state’s voluntary-assisted dying (“VAD”) statute requires VAD assessors and providers to complete mandatory training on the law. 92 Law reform bodies deemed this training necessary because VAD was new and without an established body of practice. 92 Recent education initiatives have been aimed at addressing the gaps in Canada that we identified in this study, but their non-binding nature means that gaps in the regulatory space remain. In 2024, CAMAP launched a comprehensive accredited training program for MAiD assessors and providers, funded by Health Canada. 93 This can help promote shared approaches to practice and help practitioners to feel more confident in their knowledge of the law and its application. However, in constrast to Australia, the training is not mandatory.
Implications for other jurisdictions
Canada’s model of MAiD regulation with its “twin pillars” (law and professional standards) has strengths and weaknesses. The federal law provides national consistency in MAiD eligibility criteria and procedural safeguards. In the provinces and territories, the regulators are potentially a powerful source of additional MAiD regulation. They have a more direct understanding of clinical care than legislators and can shape issues relevant to their local contexts. However, this model gives significant discretion to the regulators in choosing whether to issue a standard and what policy choices to make on discrete issues. Practice-near guidance is left to other actors. This has the potential to result in regulatory fragmentation and practice variability, both of which pose challenges for MAiD practitioners.
The study’s findings can support regulators and policy makers in other countries that are considering legalizing AD or are in the process of implementing it to identify issues to regulate. They may wish to consider what regulatory tools to use to give effect to the topics discussed in this paper, and the implications of these choices. We found that in Canada, there is potential for certain issues relevant to the practice of MAiD – such as the definition of key terms in the law – to fall between cracks in the regulatory space, with no one body being responsible for giving clinicians the guidance they seek. Countries may wish to ensure greater clarity in the terms used in their laws or may wish to identify those regulatory actors responsible for providing guidance on the meaning and interpretation of law. This study surfaces the need to consult with clinicians to ensure that legislative terms and associated guidance is meaningful and understandable to clinicians.
Limitations
This study examined regulatory documents as of 1 September 2023. Some documents may have been revised since. Additionally, this study did not examine practice standards from medical regulators that were not specific to MAiD but might nonetheless influence practice. The study is a document analysis and does not capture the impact, or perceived impact, of these documents in practice, or the context of the policy development. Three of the authors (EC, JD, BW) are currently undertaking a qualitative study exploring key stakeholders’ perceptions of MAiD regulation in practice, which will investigate these issues further.
Conclusion
This study analysed regulatory documents on MAiD for physicians from Canadian regulators. The documents were focused on similar topics, but there was variation in medical regulators’ policy choices. Physicians in each province and territory are therefore subject to differing obligations (in some respects) depending on where they practice, despite the unifying framework of the federal
Supplemental Material
sj-docx-1-pcr-10.1177_26323524251338859 – Supplemental material for Medical assistance in dying in Canada: A review of regulatory practice standards and guidance documents for physicians
Supplemental material, sj-docx-1-pcr-10.1177_26323524251338859 for Medical assistance in dying in Canada: A review of regulatory practice standards and guidance documents for physicians by Eliana Close, Mona Gupta, Jocelyn Downie and Ben P. White in Palliative Care and Social Practice
Footnotes
Acknowledgements
We gratefully acknowledge research assistance from Katie Cain.
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Australian Research Council Future Fellowship (project number FT190100410:
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.G. and J.D. were on Working Groups that developed modules for the federal MAiD National Curriculum (funded by Health Canada and developed by the Canadian Association of MAiD Assessors and Providers). M.G. chaired the Canadian Task Group that developed the Model Practice Standard, and J.D. was a member (mandated by Health Canada to develop a model practice standard for the regulation of MAiD by provincial/territorial health professional regulatory bodies). J.D. is on the Advisory Board for the Completed Life Initiative, a national MAiD advocacy and education initiative in the United States. B.P.W. was contracted by the Victorian, Western Australian, and Queensland governments to provide the legislatively mandated training for health practitioners involved in voluntary assisted dying in those states. E.C. was employed on these projects. All views expressed in this article are those of the authors and not the organizations they are affiliated with.
Data availability statement
The regulatory documents used in this study have been cited in the manuscript. Those which are no longer publicly available on regulators’ websites can be obtained from the lead author, on request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
