Abstract
Background:
Each year millions are diagnosed with cancer, impacting both patients and caregivers. Few interventions target both patients and family caregivers together, despite their shared experiences. While dyadic psychoeducational programs are gaining attention, evidence on developing and implementing these in international trials is limited. The DIAdIC trial faced unique challenges requiring innovative solutions to maintain study integrity.
Objectives:
To present our experiences with the development and implementation of two dyadic psychoeducational home-based programs, FOCUS+ and iFOCUS in the context of a randomized controlled trial (RCT) in six European countries.
Design:
A case report detailing our experiences in the development and implementation of two dyadic psychoeducational home-based programs (one face-to-face and one web-based) across multiple countries, highlighting the challenges and mitigating strategies in an international context.
Methods:
A chronological narrative describing experiences with the development and implementation of iFOCUS and FOCUS+.
Results:
The FOCUS+ and iFOCUS programs were successfully developed for the European context through rigorous translation and adaptation processes. Despite recruitment challenges including COVID-19 restrictions and administrative hurdles, 431 patient-caregiver dyads were enrolled across 6 European countries. Quantitative and qualitative data assessed the outcomes of FOCUS+ and iFOCUS interventions, including the primary endpoints of emotional functioning and self-efficacy. Fidelity was evaluated using audio recordings, checklists, and user data. Challenges in trial management were addressed with flexible timelines and technical support.
Conclusion:
The international DIAdIC trial developed and implemented two psychoeducational dyadic programs for patients with advanced cancer and their family caregivers. Based on our experiences we share several insights for future similar studies. These relate to the attention needed for context-specific adaptations when using existing interventions or programs, the translation of human-facilitated programs to standalone eHealth versions, the challenges of adopting a dyadic focus in the study, the pragmatic challenges of conducting an RCT and evaluating implementation and effects, and the technology used for study management.
Keywords
Introduction
In the EU, a total of 3.7 million people are newly diagnosed with cancer each year and about 33.9 million people are estimated to be living with cancer. 1 Advancements in cancer treatments such as early detection and new therapies change the illness trajectory and prognosis of patients with cancer, enabling patients diagnosed with advanced cancer to experience extended periods of survival.2,3 Nonetheless, a large body of evidence has demonstrated the negative effects of advanced cancer on the quality of life of patients. Besides the symptom burden, patients with advanced cancer also report emotional and spiritual suffering, challenges in their daily functioning, and a decline in their overall quality of life, resulting in serious psychosocial care needs.4 –6 Although patients with cancer often receive state-of-the-art medical care for their physical symptoms, they seldom have access to care that addresses the detrimental effects of the illness on their quality of life. The impact of advanced cancer extends beyond the patient, as family caregivers report similar levels of role adjustment problems and emotional distress as patients do, with anxiety and depression being present in up to 40% of family caregivers. 7 This reflects the family caregivers’ pivotal role in care provision, often providing complex care at home; sometimes with little support from others.8,9 Moreover, the sustainability of family caregiving is challenged by demographic shifts toward an aging population, increased chronic health conditions among the elderly, and the impending shortfall of family caregivers relative to the number of potential care recipients.10,11
Research indicates that the responses to the illness of patients with advanced cancer and their family caregivers are interrelated. 12 In addition, there are strong indications that dyadic interventions which target patients and family caregivers as one unit of care could empower the dyad, and improve their quality of life, resulting in better outcomes for both parties and could be more cost-effective than single-target interventions.13 –15 At the same time, there is a substantial body of evidence that demonstrates the benefits of early integrated palliative care to patients and family caregivers, even when provided alongside disease-modifying treatments. 16 Nevertheless, there are few psychosocial or educational intervention programs that help dyads to manage the impact of advanced cancer and to maintain their quality of life. 17 One such intervention that provides dyadic psychoeducational care and support to patients with advanced cancer and their family caregivers is the FOCUS intervention developed by Laurel Northouse. 18 The FOCUS intervention is a face-to-face dyadic psychoeducational intervention for patient-caregiver dyads in a cancer trajectory and consists of five modules: Family involvement (F), Optimistic outlook (O), Coping effectiveness (C), Uncertainty reduction (U), and Symptom Management (S). 18 Additionally one component of the FOCUS intervention (F) was translated to a web module. Research shows that both face-to-face and web-based FOCUS interventions could have a positive effect on the patient and caregiver’s quality of life and self-effectiveness in managing the impact of the disease.19,20
For the H2020 DIAdIC project, we developed and evaluated the face-to-face FOCUS+ program and the web-based iFOCUS program, which are both based on the US FOCUS interventions. As part of the DIAdIC trial, large-scale international three-arm superiority trial we researched the effectiveness, cost-effectiveness, and mechanisms of change of iFOCUS and FOCUS+. Despite the growing interest in psychoeducational support programs, there is limited evidence regarding the development and implementation of such interventions within the framework of an international multicenter three-arm trial. Considering that the DIAdIC trial took place during the COVID-19 pandemic introduced additional, unprecedented challenges that required creative solutions to ensure the continuity and integrity of the study. Therefore, within this reflexive paper we present our experiences, challenges, and strategic approaches related to (1) the development process of FOCUS+ and iFOCUS for European application and (2) the implementation of FOCUS+ and iFOCUS in the context of a randomized controlled trial (RCT) in six European countries (Belgium, Denmark, Italy, the Netherlands, Ireland, and the United Kingdom).
Methods
This case report outlines our experiences, challenges, and mitigating strategies during the development and implementation of two dyadic psychoeducational home-based programs across multiple countries as part of the DIAdIC trial. Using a narrative, chronological approach, we first describe the planned development process, followed by a reflection on the implementation of the programs within an international RCT. The report highlights key obstacles encountered and the strategies employed to address them. The CARE guideline from the EQUATOR network was used to report this study and the completed checklist is included in Supplemental Appendix 1.
Results
Experiences during development of the FOCUS+ and the iFOCUS programs
Developing FOCUS+: Participatory and Iterative Process Framework for Language Adaptation
The US FOCUS program, developed in 2002 has been tested for dyads with different types of cancer across the United States.19,21 Nevertheless, the effectiveness of a program may vary greatly depending on the national and cultural context in which it was developed and implemented.22 –24 It is likely that there are numerous differences in health-related norms, societal values, and the organization of healthcare between the United States 20 years ago and Europe nowadays. Therefore, to create a contemporary suitable version of the FOCUS program, fitting to a European context it was essential to conduct a comprehensive process of translation and content adaptation. 25
The US FOCUS program functioned as a base for the development of the FOCUS+ program. To achieve fit to Belgian, Danish, Irish, Italian, Dutch, and English contexts, the Participatory and Iterative Process Framework for Language Adaptation (PIPFLA) method was used. 26 The PIPFLA method consists of multiple steps: (1) preparation, (2) forward translation, (3) backward translation, (4) review of backward translation, (5) harmonizing, (6) review by reference group, (7) harmonizing, (8) review by reference group, (9) harmonizing, (10) proofreading, and (11) final language adaptation.25,26
In contrast to the original PIPFLA method, our approach involved a preliminary evaluation of the program’s appropriateness in each country. We also expanded the involvement of the reference groups, deviating from PIPFLA’s guidance where these groups (which provide an emic and etic perspective) are less involved. Additionally, program stakeholders such as nurses, clinicians, patients, and family caregivers from all six countries reviewed the program materials on multiple occasions. Using a template, we collected feedback from these stakeholders. To not deviate too much from the theory underlying the original FOCUS intervention, Professor Laurel Northouse (the developer of the original FOCUS intervention), was involved in the analysis of stakeholder feedback. A “TIDieR checklist (Template for Intervention Description and Replication),” was used to give a detailed description of the resulting FOCUS+ program (Supplemental Appendix 2). The translation and adaptation process was undertaken from May 2019 to December 2020. More information about the FOCUS+ development process can be found elsewhere. 26
Following the slightly adjusted PIPFLA process with greater involvement of reference groups resulted in an adapted version of the original US FOCUS program: the FOCUS+ program. This also included a FOCUS+ conversation manual for use by the nurses implementing the FOCUS+ program. This manual was developed to allow for uniformity across all countries while incorporating country-specific language nuances. Modifications to the wording of the original US FOCUS program were permitted for sensitive subjects such as the end of life and sexuality if they were more culturally appropriate and likely to be better accepted by participants. Adaptations to the original FOCUS manual target six topics and are summarized in Box 1.
Summary of adaptations in FOCUS+ conversation manual for nurses.

The original US FOCUS program also provided diverse information resources to dyads such as leaflets, booklets, and websites on cancer therapies, consequences of cancer, and referrals. Recognizing the impracticality of translating all US materials and the need for adaptation to diverse European healthcare services, each country selected national equivalents of the materials. These materials were compared and evaluated, leading to the decision from the coordination team to integrate them into a booklet. This booklet aligned with the structure of the FOCUS+ session and served as a reference for dyads throughout the intervention, featuring content, including exercises on dyadic communication.
The development process of FOCUS+ resulted in a home-based face-to-face intervention consisting of two home visits of 90 min and one online video session of 30 min, conducted by a trained nurse, with 4 weeks between each session. The nurses had access to a conversation manual to provide guidance and support during the sessions.
Developing iFOCUS: using the Scrum methodology
The FOCUS+ program served as the foundation for the development of the web-based iFOCUS program. An extensive development process was essential for iFOCUS to become a self-managed, user-friendly psychoeducational web-based program for patients with advanced cancer and their family caregivers while retaining the essence of FOCUS+.
The development of iFOCUS adhered to the four phases of the agile Scrum methodology namely: design (1), development (2), go-live (3), and maintenance and support (4). In line with the Scrum methodology, we applied timeframes of rapid program development and evaluation, also called “sprints.” 27 For the development of iFOCUS we developed a seven-step development process (Figure 1), which could be fully integrated into the four-phased Scrum methodology. The seven steps of development were (1) concept design, (2) development of mock-ups, (3) obtaining feedback from the international consortium, (4) technical development of the program, (5) creating country-specific versions for each of the six participating countries, (6) preliminary testing through user and functional testing, and (7) implementing the final version for the RCT. Five teams were involved in the development of iFOCUS: (1) a core development group consisting of academics, clinicians, and healthcare professionals who were actively involved in the daily development of program content and flow, (2) a web-development team, (3) the DIAdIC international consortium, (4) audio-visual experts, and (5) potential end-users from each participating country (i.e., patients with advanced cancer and family caregivers). 27

iFOCUS development flow. 27
Each of the seven development steps followed sequentially on the previous one, but flexibility was allowed to enable continuous refinement and correction. The collaborative efforts of different teams and the integration of diverse perspectives enabled the identification and resolution of errors and bugs throughout the development process of iFOCUS.
The primary challenges we faced during the development of iFOCUS included converting the FOCUS/FOCUS+ “identity” (theoretical program background) into functional features, the spreading of content within and between sessions, program tailoring, ensuring both program safety and a user-friendly login procedure, incorporating materials in the iFOCUS program and establishing an onboarding procedure. Based on our experiences and review of available literature, the challenges we faced were either specific to the conceptualization of iFOCUS (program-specific challenges) or general to the development of eHealth programs (general challenges). The user testing (step 6) included 42 participants (20 patient-family caregiver dyads and 2 bereaved family caregivers) who reviewed the iFOCUS program. While potential end-users found the iFOCUS program to be acceptable and usable, minor content, tailoring, and program flow adaptations were required.
The development process of iFOCUS resulted in a self-management psychoeducational eHealth program, consisting of four sessions of approximately 60 min each, over a 12-week period—with a 3-week gap between each session, that could be used autonomously by the patient-caregiver dyad together. More details about the development process are described in Chapter 2 of this dissertation. 27
Experiences during the implementation of the FOCUS+ and iFOCUS programs in an international RCT
Information on the DIAdIC trial study design, setting, and participants can be found in Chapter 3 of this dissertation, which encompasses the DIAdIC trial protocol. 28
Recruitment and sample size
Patients were recruited and enrolled via participating hospitals. Hospital staff screened patients for the inclusion and exclusion criteria. Eligible patients received a first oral introduction to the trial, preferably in attendance of the family caregiver. If the dyad agreed to participate, a data collector made an appointment with the dyad to obtain their informed consent. Patients and family caregivers were invited to complete the baseline (T0) measurement independently from each other. Only those who completed the baseline measurement were randomized and considered as enrolled in the study. The recruitment period started in February 2021 and was foreseen to last 12 months.
Inclusion of patient-caregiver dyads within this trial also made recruitment more complex. There were cases in which the patient was interested to participate but was unable to identify a family caregiver. Another challenge pertained to the motivation of the patient or family caregiver to join and how they could influence one another. In most cases, if the patient wanted to participate, then the family caregiver followed and vice versa, but there were also cases where they deterred one another from participation. As a result, the recruitment of dyads may make the participating partner a de facto gatekeeper.
While we intended to recruit 156 dyads in each participating country, recruitment went considerably slower than anticipated. The following reasons for this delay were identified:
The trial took place during the protective measures taken by governments and healthcare organizations in view of the COVID-19 pandemic. One of the measures implied limited access to hospitals for external staff such as data collectors but also for family caregivers, often making it impossible for family caregivers to accompany patients to consultations. This led to challenges in presenting the study orally to the dyad. COVID-19 also led to increased pressure on healthcare organizations, limiting the available time, and means to support recruitment. Research directly aimed at COVID-19 was prioritized, effectively halting other research.
A major factor that has impacted recruitment was the impact of the COVID-19 crisis on the healthcare sector. In some hospitals, family caregivers were no longer allowed to accompany the patient to consultations, which made it impossible to orally present the study to the patient-caregiver dyad and invite them both to participate. Additionally, hospital staff also reported having less time to be involved in the study than anticipated. Based on our experiences and supported by other research, hospital staff, and potential trial participants were also hesitant to engage in clinical trials due to fears of contracting COVID-19. 29
Stricter regulations, an excessive administrative burden to obtain approvals for the study, and stricter rules for the screening and invitation of patients to studies caused delays and fewer study referrals in certain countries.
Variable recruitment strategies in hospitals played a significant role, with some achieving targets through active data collector involvement in the screening and identification of eligible patients, while other hospitals imposed barriers, leading to lower-than-expected recruitment.
Acquiring informed consent from both patients and their family caregivers for dyadic participation posed an extra challenge, particularly in cases where one party was unwilling to participate.
The requirement for computer access and internet familiarity posed a challenge, reducing willingness to participate among those unfamiliar with computer-administered interventions.
Various other issues such as the consequences of Brexit, regional healthcare reorganization, and staff shortages increased pressure on time and resources, resulting in a lower priority for psychoeducational research.
Mitigating strategies
To address recruitment delays, the consortium implemented several strategies:
The recruitment period was originally foreseen to last a total of 12 months and the data collection period was 18 months (to complete T2 for all dyads). However, the slower-than-anticipated progress in recruitment led the DIAdIC consortium to extend the recruitment period to 24 months and the data collection period to 30 months, providing more time for countries with lower rates (see Figure 2). The overall project period was also extended by 6 months without affecting deliverables.
Additional recruitment sites (hospitals) were included in some countries (see Table 1).
If patients were unaccompanied at the hospital, recruitment staff tried to obtain family caregiver contact details and personally introduce the study via phone.
In the event of tightened COVID-19 measures, the face-to-face FOCUS+ sessions were permitted to be conducted online via a GDPR-compliant platform (e.g., MS Teams and Zoom). Figure 3 illustrates the decision-making process. As a consequence, the mode of delivery of the FOCUS+ intervention can differ between dyads, that is, either face-to-face or via an online video platform. Therefore, each partner institution is responsible for recording the mode of delivery of the FOCUS+ intervention for each dyad in each country, which allows the monitoring of within-group variations in effectiveness.
Monthly bilateral meetings were organized between the coordinator and all the RCT partners to enhance local screening strategies and reduce gatekeeping in some hospitals. Successful strategies, such as oncology nurses conducting screenings instead of physicians, were shared within the consortium.
Monthly online gatherings for data collectors and recruitment staff were established to exchange knowledge and best practices, promoting successful recruitment strategies.

Initial and extended recruitment period.

Decision-making process in method of delivery of FOCUS+ intervention in light of possible COVID-19 developments.
Recruitment sites per country and recruitment starting dates.

Additional recruitment sites.
Even when applying different strategies to mitigate the lower-than-anticipated recruitment numbers, we did not reach the anticipated number of recruited dyads in all countries. At the end of the recruitment period (end of February 2023), a total number of 431 of the intended 936 dyads were enrolled in the DIAdIC trial (see Table 2).
Recruitment numbers per country.

Outcomes, measurements, and data collection procedures
During the DIAdIC trial, both qualitative and quantitative data were collected. Quantitative data was collected three times from patient-caregiver dyads; T0, the baseline measure before randomization, T1, or first follow-up (T0+12 weeks), and T2, or second follow-up (T0 + 24 weeks). Emotional functioning and self-efficacy in managing the impact of cancer on patients and family caregivers at T1 were the primary endpoints. Emotional functioning was measured using a 10-item short form based on the European Organization for Research and Treatment of Cancer emotional functioning item bank. Self-efficacy was measured with 17 items from the Lewis Cancer Self-Efficacy Scale.
The secondary endpoints were the primary outcomes at T2, and other outcomes such as quality of life, benefits of illness, coping, dyadic communication, and ways of giving support at T1 and T2. At T1 both the patient and the family caregiver filled in questions to evaluate the effectiveness and satisfaction with the programs. Figure 4 shows the participant timeline and the data collection. An overview of the instruments, underlying measured concepts, and their timing is added as a Supplemental Appendix 3.
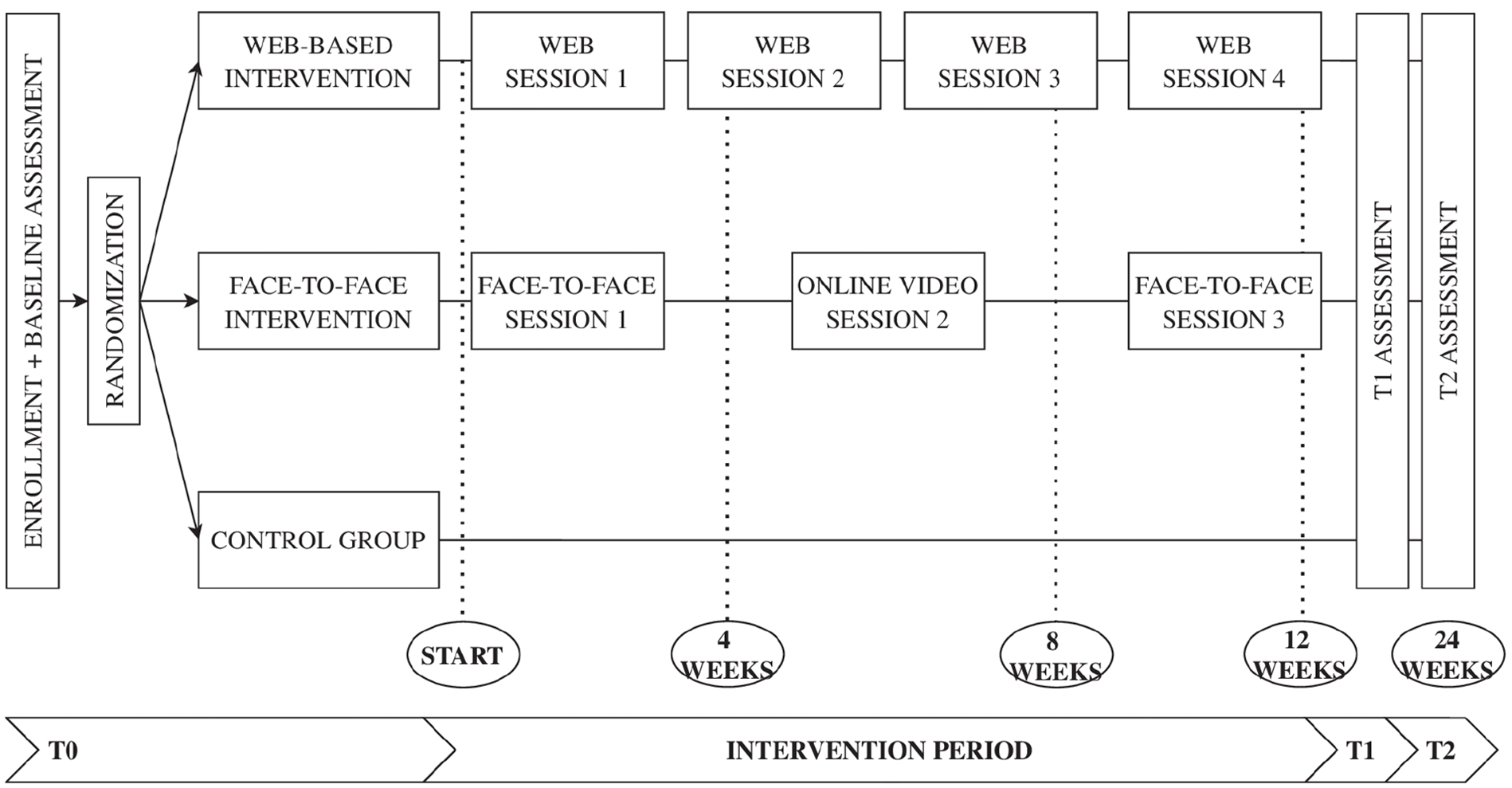
Participant timeline. 28
Additionally, a process evaluation was conducted to assess program fidelity and to identify and understand the influence of possible mechanisms of change within the interventions.
Process evaluation data that was collected included:
Audio recording of the FOCUS+ (face-to-face) sessions to measure FOCUS+ fidelity: an objective assessment (1) where a research team member used a checklist on 20% of randomly selected dyads every 3 months, and a reflective self-assessment (2), where intervention nurses used a checklist on a small sample of dyads they worked with in the initial 3 months of the trial.
Logging of iFOCUS (web-based) sessions to measure iFOCUS fidelity: The following information was recorded: number of logins per session (dyads may complete a session over multiple occasions), time between login and logout, number of clicks in each session (giving indication of how interactive/participating dyads are in the sessions), time spent per slide, number of clicks on the personal workbook, which videos were watched and, how many times the audio files (voice-overs) were listened to. This information could be drawn from the iFOCUS platform in an Excel format and reflected important aspects of program fidelity.
To explore participants’ perceptions of the interventions, their user experiences, core elements central to their experiences, and the mechanisms of change, we conducted interviews: Per country, we conducted 6 interviews with intervention nurses, 6 interviews with research staff, and 10 dyads who received the intervention and 1 dyad non-completer.
Both quantitative and qualitative findings will be integrated as part of a mixed-method evaluation of the FOCUS+ and iFOCUS programs. The starting point of the mixed-method approach is the quantitative findings (effect of iFOCUS/FOCUS+), followed by consulting other data sources to explain the results. This integration of quantitative and qualitative findings will offer deeper insights into the effectiveness of these programs and help in formulating recommendations for future program development and implementation. If necessary, in light of the COVID-19 pandemic, these interviews could take place online.
While the involvement of a third party with expertise in technical development and maintenance of digital platforms was necessary, this created reliance on the web-development team during development and during the trial implementation of iFOCUS and FOCUS+. Considering the significant role of the iFOCUS platform in the DIAdIC RCT, serving as both one of the intervention programs and as a platform for management and data collection, we became dependent on the web-development team when encountering issues and bugs related to the registration of data such as enrollment, allocation, questionnaires, and process evaluation data. To cope with this challenge a ticketing system was established using an online platform for collaborative project development. Errors or bugs were reported in this system with a clear description of the bug and its location. Potential solutions were discussed in weekly meetings.
Adhering to the trial timeline presented challenges, requiring flexibility in the delivery of FOCUS+, iFOCUS, and when conducting data collection. These challenges could be attributed to various factors such as the disease progression of participating dyads, their treatment trajectory, and the availability of both patients with advanced cancer and their family caregivers for sessions or questionnaires. Although adherence to the timeline was strongly encouraged, some flexibility was permitted, allowing dyads to deviate from the intended 12 weeks after T0 if needed, and prioritizing the completion of all program sessions.
Discussion
As part of the international DIAdIC trial, we developed and implemented two psychoeducational programs for patients with advanced cancer and their family caregivers: a face-to-face program (FOCUS+) and a web-based program (iFOCUS). We encountered various challenges in relation to the development of the digital program, recruitment of dyads, and adherence to the project as described in the protocol, not least due to the wide-ranging impacts of the COVID-19 pandemic. Various mitigating strategies were implemented during the development and implementation of FOCUS+ and iFOCUS as part of the DIAdIC trial.
Our experiences with the development and implementation of the two programs in an international RCT, as described in this article, provide some insights that are likely generalizable to other contexts and therefore can provide useful considerations for future similar studies.
First, starting from existing (in our case American) interventions or programs with proven effectiveness provides advantages, but also has its challenges. We started the development process not from zero but rather opted to take a proven effective psychoeducational program as the foundation for the development of FOCUS+ and iFOCUS and make adaptations. Given the accumulating evidence on effective interventions, starting a development process from proven effective programs can increase the likeliness of achieving a positive effect in a new setting or population and might be a more cost-effective and efficient approach to program development.30 –33 Nevertheless, when starting a development process, other factors than potential effectiveness such as the cultural fit and program fit to the population need to be taken into consideration.34,35 Considering that interventions hold a degree of context specificity, transferability from interventions to a new location or to a new setting and expecting the same positive outcomes is often uncertain.22 –24 More so, research indicates that unadapted interventions and programs are likely to be less effective than contextually adapted or locally developed interventions. 36 Hence for our development, which translated the program to a new context, we used the PIPFLA framework which took into account the contextual differences in cancer care in the United States around 2002 and Europe in 2021. 26
Second, in regard to context specificity, we need to take into account that the development and implementation of the iFOCUS and FOCUS+ programs largely took place during the COVID-19 pandemic. Considering the unique and unprecedented challenges that presented themselves during the pandemic, temporal bias can not be ruled out. One example of a potential influence is that stress, anxiety, and depression rates were elevated among the general population during the COVID-19 pandemic, which could have impacted participant recruitment and influenced the study outcomes. 37
Third, translating programs that build on interpersonal interaction and dyadic coping and discussion to an eHealth format is challenging. Our experience has been that the development of a digital psychoeducational program comes with specific challenges, both in terms of program requirements and eHealth development in general. 27 While described as possibly more cost-effective than face-to-face interventions 38 we found the development of a digital psychoeducational program to be a time and resource-intensive process, even when based on existing programs. Due to limited resources and time we had to make pragmatic choices such as involving potential end-users in a consultancy role as part of user testing rather than as part of a co-development trajectory. Nevertheless, available evidence encourages active user involvement in all stages of eHealth development as this is likely to create a web program that fits better to their context.39,40 However, user input may also be inherently biased, as it is based on personal experiences, internalized routines, and assumptions. 41 To mitigate this, we included a sizable and diverse sample of participants from six different countries in the user-testing phase for the development of iFOCUS. However, recruitment bias on a national level can not be entirely ruled out. Additionally, there is a conundrum between the ideal eHealth development trajectory, which actively involves end-users across all phases of development, and project limitations concerning available time and resources. Therefore, during the development of an eHealth program, we recommend to make pragmatic which takes into consideration program requirements and project limitations, actively involves users during eHealth development, and thorough documentation of underrepresented and overrepresented populations in the program.
Fourth, deliberately adopting a dyadic focus was important but also came with challenges. Other research has suggested that many aspects of stress and coping in relation to cancer and its treatment have an interdependent impact on patient and family caregivers and that dyadic programs are more aligned to that reality and are therefore more likely to have positive outcomes for both patients and family caregivers.42,43 However, this comes with particular challenges as the recruitment of dyads involves specific additional challenges compared to the recruitment of individual patients or individual family caregivers separately. Moreover, the exclusion of dyads who can not identify a family caregiver is a concern in dyadic trials.
Fifth, considering the challenges during recruitment, our experiences reiterate the question of whether the RCT paradigm, assuming rigidity and strict control, matches well with the real-world contexts in which people deal and cope with the illness, treatments, and complex consequences on all aspects of life. Pragmatic trial designs have been put forward as more fit approaches to these contexts.44,45 However, even when adopting a pragmatic trial design, questions about permitting flexibility in aspects not initially identified will inevitably arise. Therefore, to gain insights in these processes and mechanisms of change we conducted a process evaluation using both qualitative and quantitative data as part of the DIAdIC trial.
Sixth, the potential challenges of the IT technology that is relied on for the study need to be considered. Because of the complexity and different components of our study, we decided, together with an eHealth-specialized company, to develop a web platform that integrated the different aspects of the study, including study management aspects such as enrollment, questionnaire administration, randomization, reminders but also including the iFOCUS program. However, this created a dependence on the web-development team to resolve bugs, errors, and issues before starting the data collection (and to a lesser extent resolvable minor ones during the data collection). Separating the eHealth intervention (in our case iFOCUS) from study management technology may be a better option. For instance, programs like REDCap (Research Electronic Data Capture), which are specifically designed for data collection in research studies, are widely used and have proven to be reliable and secure and can offer a less concerning basis for study management. 46
Conclusion
The international DIAdIC trial developed and implemented two psychoeducational dyadic programs for patients with advanced cancer and their family caregivers: a face-to-face version (FOCUS+) and a web-based version (iFOCUS). While minor cultural differences between countries arose during development, these were easily resolved, allowing the core content of the programs to remain unchanged. The greater challenges related to cultural differences emerged during trial recruitment, where local legislation, agreements, and bureaucratic hurdles significantly hindered the process, which may impact the interpretation of the results across countries. Other important insights for future studies relate to the attention needed for context-specific adaptations when using existing interventions or programs, the translation of human-facilitated programs to standalone eHealth versions, the challenges of adopting a dyadic focus in the study, the pragmatic challenges of conducting an RCT and evaluating implementation and effects, and the technology used for the study management.
Supplemental Material
sj-docx-2-pcr-10.1177_26323524241310458 – Supplemental material for Developing and implementing a nurse-delivered and a web-based dyadic psychoeducational program for people with advanced cancer and their family caregivers: sharing experiences from a three-arm international randomized controlled trial (DIAdIC)
Supplemental material, sj-docx-2-pcr-10.1177_26323524241310458 for Developing and implementing a nurse-delivered and a web-based dyadic psychoeducational program for people with advanced cancer and their family caregivers: sharing experiences from a three-arm international randomized controlled trial (DIAdIC) by Aline De Vleminck, Vincent Van Goethem, Sigrid Dierickx, Orphé Matthys, Kim Beernaert, Mogens Gronvold, Philip Larkin, Monica Guberti, Erica Witkamp, Joanne Reid, Katherine Bristowe, Luc Deliens, Lore Lapeire, Peter Hudson and Joachim Cohen in Palliative Care and Social Practice
Supplemental Material
sj-docx-3-pcr-10.1177_26323524241310458 – Supplemental material for Developing and implementing a nurse-delivered and a web-based dyadic psychoeducational program for people with advanced cancer and their family caregivers: sharing experiences from a three-arm international randomized controlled trial (DIAdIC)
Supplemental material, sj-docx-3-pcr-10.1177_26323524241310458 for Developing and implementing a nurse-delivered and a web-based dyadic psychoeducational program for people with advanced cancer and their family caregivers: sharing experiences from a three-arm international randomized controlled trial (DIAdIC) by Aline De Vleminck, Vincent Van Goethem, Sigrid Dierickx, Orphé Matthys, Kim Beernaert, Mogens Gronvold, Philip Larkin, Monica Guberti, Erica Witkamp, Joanne Reid, Katherine Bristowe, Luc Deliens, Lore Lapeire, Peter Hudson and Joachim Cohen in Palliative Care and Social Practice
Supplemental Material
sj-pdf-1-pcr-10.1177_26323524241310458 – Supplemental material for Developing and implementing a nurse-delivered and a web-based dyadic psychoeducational program for people with advanced cancer and their family caregivers: sharing experiences from a three-arm international randomized controlled trial (DIAdIC)
Supplemental material, sj-pdf-1-pcr-10.1177_26323524241310458 for Developing and implementing a nurse-delivered and a web-based dyadic psychoeducational program for people with advanced cancer and their family caregivers: sharing experiences from a three-arm international randomized controlled trial (DIAdIC) by Aline De Vleminck, Vincent Van Goethem, Sigrid Dierickx, Orphé Matthys, Kim Beernaert, Mogens Gronvold, Philip Larkin, Monica Guberti, Erica Witkamp, Joanne Reid, Katherine Bristowe, Luc Deliens, Lore Lapeire, Peter Hudson and Joachim Cohen in Palliative Care and Social Practice
Footnotes
Acknowledgements
The authors wish to thank DIAdIC members and contributors not already indicated in the authorship and group authorship.
Declarations
Author’s note
Evaluation of Dyadic Psychosocial Interventions for People with Advanced Cancer and their Informal Caregivers (DIAdIC): An international randomized controlled trial—“DIAdIC.” Group authorship: Kim Eecloo (Department of Family Medicine and Chronic Care, Vrije Universiteit Brussel, Brussels, Belgium), Ulrik Sørensen Schmidt (Department of Public Health, University of Copenhagen, Copenhagen, Denmark; Palliative Care Research Unit, Bispebjerg and Frederiksberg Hospital, Copenhagen, Denmark; University College Dublin, National University of Ireland, Dublin, Ireland), Suzanne Guerin (University College Dublin, National University of Ireland, Dublin, Ireland), Catherine Jordan (University College Dublin, National University of Ireland, Dublin, Ireland), Paul D’Alton (University College Dublin, National University of Ireland, Dublin, Ireland), Michael Connolly (University College Dublin, National University of Ireland, Dublin, Ireland), Monika Pilch (University College Dublin, National University of Ireland, Dublin, Ireland), Massimo Costantini (Research and EPB Unit, Health Professions Department, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy), Agnes van der Heide (Erasmus MC, University Medical Center Rotterdam, Rotterdam, the Netherlands), Judith Rietjens (Erasmus MC, University Medical Center Rotterdam, Rotterdam, the Netherlands), Maaike van der Wel (Erasmus MC, University Medical Center Rotterdam, Rotterdam, the Netherlands), Kevin Brazil (The School of Nursing and Midwifery, Queen’s University of Belfast, Belfast, UK), David Scott (The School of Nursing and Midwifery, Queen’s University of Belfast, Belfast, UK), Gillian Prue (The School of Nursing and Midwifery, Queen’s University of Belfast, Belfast, UK), Lydia Tutt (Cicely Saunders Institute of Palliative Care, Policy and Rehabilitation, King’s College London, London, UK), Richard Harding (Cicely Saunders Institute of Palliative Care, Policy and Rehabilitation, King’s College London, London, UK), Peter May (Centre for Health Policy and Management, Trinity College Dublin, Dublin, Ireland), Samantha Smith (Centre for Health Policy and Management, Trinity College Dublin, Dublin, Ireland), Charles Normand (Cicely Saunders Institute of Palliative Care, Policy and Rehabilitation, King’s College London, London, UK; Centre for Health Policy and Management, Trinity College Dublin, Dublin, Ireland), Chiara Puglisi (Research and Statistics Infrastructure, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy), Federica Sforacchi (Research and Statistics Infrastructure, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy), Valeria Soffientini (Research and Statistics Infrastructure, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy), Stefania Tortella (Research and Statistics Infrastructure, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy), Elena Turola (Research and Statistics Infrastructure, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy), Silvia Zini (Research and Statistics Infrastructure, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy), and Silvia Di Leo (Psycho-Oncology Unit, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
