Abstract
Objectives
We sought to examine the prevalence of prospective registration and publication rates in digital health trials.
Materials and Methods
We included 417 trials that enrolled participants in 2012 and were registered in any of the 17 WHO data provider registries. The evaluation of the prospective trial registration was based on the actual difference between the registration and enrollment dates. We identified existing publications through an automated PubMed search by every trial registration number as well as a pragmatic search in PubMed and Google based on extracted metadata from the trial registries.
Results
The prospective registration and publication rates were at (38.4%) and (65.5%), respectively. We identified a statistically significant (
Conclusion
The adherence of investigators to the best practices of trial registration and result dissemination is still evolving in digital health trials. Further research is required to identify contributing factors and mitigation strategies to low compliance rate with trial publication and prospective registration in digital health trials.
Keywords
Introduction
In the realm of scientific research, study results should be made available and accessible to the broader research community to assess the evidence around the efficacy and potential harm of healthcare interventions.
1
Emanuel et al.'s framework defines seven ethical guidelines for clinical research involving human subjects.
2
The first of which describes the value derived from disseminating research results as “
A number of studies have examined the ramification of publication bias and debated a number of contributing factors to non-publication of clinical trial results such as recruitment challenges, funding sources and study design.3–10 Other challenges pertaining to investigators were also identified as contributing factors to non-publication of clinical trial results, such as lack of time or disagreement with coauthors.11,12
The notion of clinical trial registration was conceived in 1986, when Simes suggested that the act of registration would help control the risk of publication bias by providing a new data source for trial information and results.13–15 The trial registries would also help mitigate selective reporting of positive outcome by comparing outcomes reported in trial publication versus outcome measures indicated in the trial registration.16–21 In 2004, the International Committee of Medical Journal Editors (ICMJE) introduced a new mandate to promote prompt registration of all clinical trials. 22 In 2005, the World Health Organization (WHO) started an initiative to standardize trial registrations and trial registry datasets across multiple national and international registries. 23 In October 2008, the 7th revision of the Declaration of Helsinki (DoH) was adopted with new requirements focusing on the importance of prospective registration of clinical trials and reporting of their results. 24 The prospective registration of clinical trial means that investigators should register their trials prior to the enrollment date of the first trial participant, otherwise, the registration would be considered retrospective. In 2015, the WHO announced a new statement on public disclosure of clinical trial results with more guidelines on trial registration, publication of results, and the inclusion of the trial registration number (TRN) in respective publications to enable linking of trial reports with clinical trial registry information. 25
Two studies reported publication rates between 66% and 68% for clinical trials registered in the US-based clinical trials registry, ClinicalTrials.gov.26,27 Another study reported publication rates at 73% for clinical trials registered in at least one of several clinical trial registries (ClinicalTrials.gov, Current Controlled Trials, WHO International Clinical Trials Registry Platform (ICTRP), EU Clinical Study Register, and Indian, Australian-New Zealand, and Chinese Clinical Trial Registries). 28 Three other studies investigated prospective trial registration and the quality of registration in the International Standard Randomized Controlled Trial Number (ISRCTN) registry and the ICTRP registries.29–31 The results of these studies reported that prospective trial registration was between 37.8% and 53.4%. A recent global study published in the British Medical Journal in 2020 reported that only 41.7% of the included randomized controlled trials were registered prospectively. 32
However, investigators could be overwhelmed with challenges that may be particularly problematic in digital health trials, such as the usability of the intervention under test, participant recruitment, and retention challenges that may contribute to non-publication rate and prospective trial registration.33–39 To our knowledge, this is the first global review to analyze the non-publication rate and prospective registration of digital health clinical trials across the seventeen WHO data provider registries. 40
Research objectives
The primary research objective was to examine the prospective trial registration and publication rates of digital health randomized clinical trials registered in any registry that is part of the WHO data provider registries. The secondary research objectives were (1) to investigate the compliance with recruitment and inclusion of the TRN in the published trials, (2) to explore the relationship between publication rates, trial size and the duration from the enrollment of the first participant to the publication of trail results (enrollment-to-publication duration), and (3) to analyze the relationship between retrospective trial registration and the duration from trial registration to the publication of trial results (registration-to-publication duration).
Material and methods
Data source
The WHO ICTRP is a free online portal that provides unified access to trial registration information across different clinical trial data provider registries.[40,41] To identify the registered digital health trials, we utilized the advanced search feature in the ICTRP search portal. We applied our search terms and phrases to the [Title] or [Intervention] field and downloaded the matching trials in XML format for further analysis. To identify journal publications of included trials we searched trial papers in PubMed and Google as described in the “Identification of Publication” subsection.
Statistical analysis
We used Pearson Chi-square statistic for bivariate analyses. These statistical analyses were performed in SPSS Statistics version 24 (IBM Corporation, Armonk, NY). We used Fisher's exact test statistic for bivariate analyses where the expected cell value was less than 5. The Fisher's exact test was performed in STAT Statistics version 14.2 (StataCorp LLC, TX).
Inclusion criteria
We included all eHealth, mHealth, telehealth, and digital health-related randomized clinical trials that are registered in any of the 17 trial registries within the ICTRP database and include any information and communication technology component, such as:
Online web and mobile application Internet, websites and personal computer application Digital games and social media application Telehealth and telemedicine components
We included trials irrespective of their recruitment status or trial phases. The enrollment date in the trial registries was used to indicate the start date of the included trials. The inclusion criteria were limited to trials that started the participants enrollment between January 1st, 2012 and December 31st, 2012 as described in Online Appendix I.
Exclusion criteria
We excluded registered clinical trials that were not randomized or did not include any digital components in their intervention. Trial publications were only identified in peer-reviewed journals, excluding pre-print servers and conference abstracts. Trials that merely utilized short messages service (SMS), phone calls, emails or video communication without any other interactive components were also excluded. We excluded trials that only reported on computerized or online surveys and questionnaires.
Search terms for digital health clinical trials
We developed a comprehensive list of 86 search terms and phrases through an iterative process as explained in Online Appendix II.
Data extraction
We downloaded the XML files for the 417 matching clinical trials in the ICTRP search portal. These files were transformed into a tabular format and imported into a local relational SQL Server Database for further data extraction and analysis. The details regarding extracting the trial condition, primary sponsor and major technology are provided in Online Appendix III.
Prospective trial registration
The data export from the ICTRP dataset did include a field to indicate whether a trial was registered retrospectively.41,56 However, our analysis showed that this field did not include correct information for a substantial number of trials within our sample of 417 included trials. We therefore chose to evaluate prospective trial registration based on the actual difference between the registration and enrollment dates as described in Online Appendix IV.
Identification of randomized trials
We were able to identify randomized trials through a text match search in the following three fields in the data export from the ICTRP dataset: “Study_design,” “Public_title,” and “Scientific_title”. We searched for matching words that included the term ‘Random’ and did not include the term ‘Non-Random’.
Identification of publication
We identified existing publications through a two-step iterative process. The first step included an automated identification process that included a PubMed search by every TRN. The second step included a pragmatic search in PubMed and Google based on extracted metadata from the trial registries; trial title, investigators, location/city and institution. We only considered trial publications that reported at least one of the primary outcome measures of the underlying trials. Complete details of the publication identification processes are described in Online Appendix V.
Results
We utilized the advanced search functionality in the ICTRP search portal to apply our 86 search terms and phrases including all trial phases and recruitment status. There were 22,859 unique trials that matched our search terms, within which, 15,096 trials were randomized, and 1018 trials were enrolled in 2012. After screening against our inclusion and exclusion criteria, 417 trials were included, and 601 trials were excluded as per the following breakdown (Figure 1).

Included trials from the search results.
Prospective trial registration and publication rates
In summary, 160/417 (38.4%) trials were registered prospectively and 273/417 (65.5%) trials were associated with identified outcome publications. Of the 273 identified trial publications, 8 were identified in references in the ClinicalTrials.gov registry, 199 were identified in PubMed and 66 were identified through Google search as described in Figure 2.

Publication identification results.
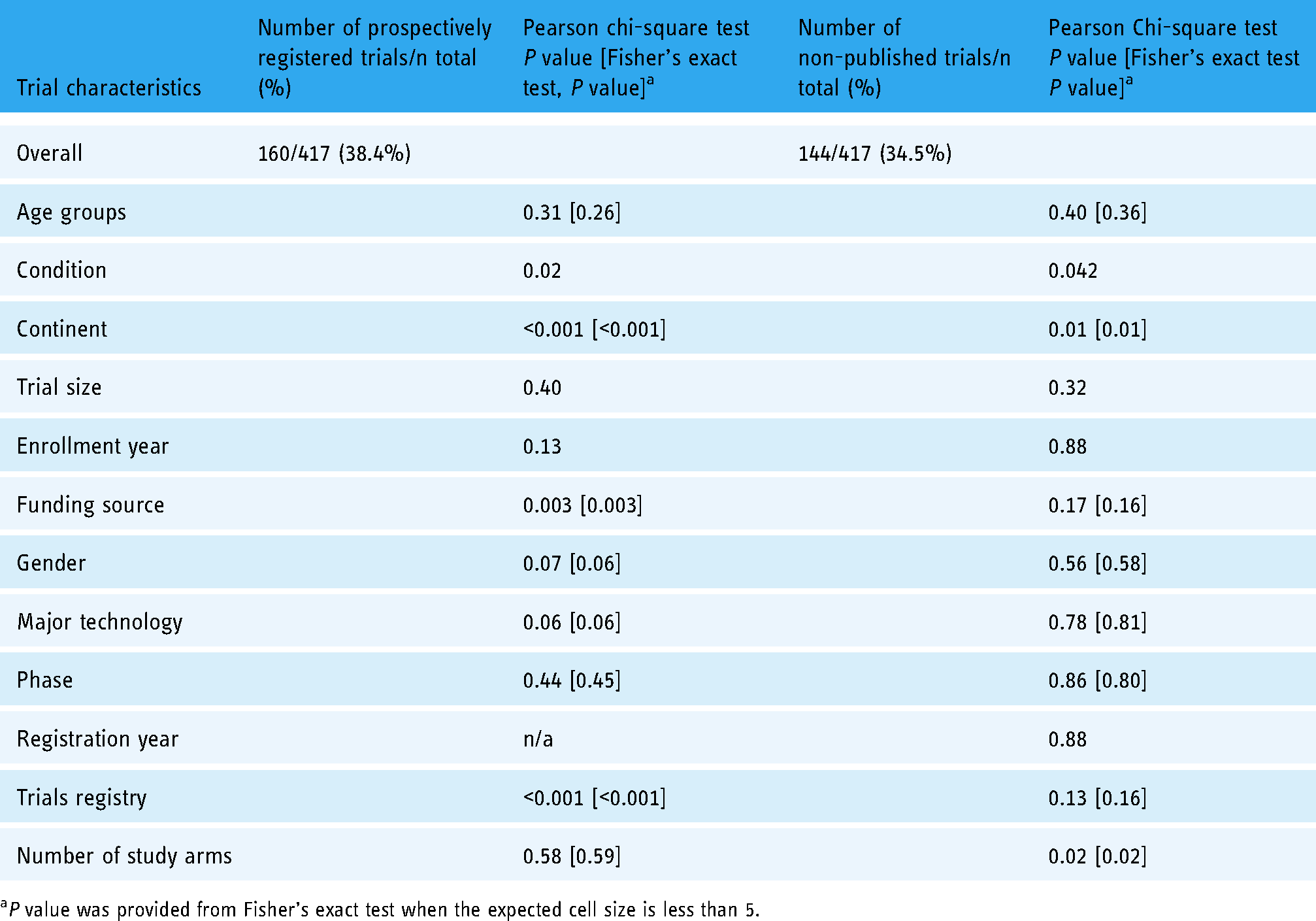
We examined the relationship between trial characteristics and the non-publication rate and prospective registration of all included trials as summarized in Table 1 and detailed in Online Appendix VI.
Relationship between trial characteristics and prospective registration and non-publication rates of included trials (details provided in Online Appendix VI).
a
There were significant relationships (
Only 160/417 (38.4%) of the included trials were registered prospectively. There were significant relationships (
Enrollment-to-publication duration and trial size
We postulated that smaller trials would be easier to conduct and publish as they have less recruitment and enrollment challenges. To support our postulate, we found statistically significant differences (

Relationship between trial enrollment-to-publication duration and trial size, number of published trials and non-publication rate.
Registration-to-publication duration and retrospective registration
We examined the prevalence of retrospective trial registration and its relationship to trial publication and the registration-to-publication duration. Overall, 168/273 (61.5%) of published trials were registered retrospectively. We found a statistically significant relationship (
Relationship between trial registration-to-publication duration and retrospective trial registration.

Non-cumulative values. The number of retrospectively registered trials, the number of published trials, and the reported percentage is not cumulative within every year level.
Compliance with participants recruitment and inclusion of the TRN in published trials
When evaluating the compliance with participants recruitment, we found that 111/273 (40.7%) of the published trials reported fewer subjects who were actually recruited than the target size indicated in the trial registry, 96/273 (35.2%) published trials reported actual recruitment matching the trial target size, and 66/273 (24.2%) published trial recruited more participants (over-recruitment) than the target size in the trial registry. Ensuring adequate participant recruitment is critical to support the statistical power and internal validity of the trial results. Enrollment of fewer participants could introduce type II errors in reported study results. 61 We suggest that over-recruitment could be appropriate and would empower further assessment of secondary hypotheses. Therefore, we considered trial recruitment as compliant if the actual recruitment was equal to or more than the target size as defined in the trial registration.
We verified whether a reference to the TRN was indicated in the papers of the 273 published trials. Nearly half of the trials 142/273 (52%) provided a reference to their respective TRN in their publications. We examined the relationship between trial characteristics and the compliance with participants recruitment and inclusion of the TRN in published trials as summarized in Table 3 and detailed in Online Appendix XI.
Compliance with participants recruitment and inclusion of trial identification in published trials (details provided in Online Appendix XI).

a
Discussion
We found that 34.5% of all included trials remain unpublished six years after the enrollment of trial participants. Nearly half of our included trials 56.1% were registered in the ClinicalTrials.gov registry, of which 36.3% remain unpublished. Our non-publication rate was higher compared to a 2018 study that reported a 27% non-publication rate for digital health trials registered in ClinicalTrials.gov. 62 Although both studies focused on examining non-publication rates in digital health trials, their study design, data source and approach were quite different, hence a direct comparison would not be valid. The current study has a broader scope including 417 trials that started participants recruitment in 2012 and were registered in any of the seventeen WHO data provider registries. The former 2018 study included 556 trials that were registered in the ClinicalTrials.gov registry only and were completed within three years, between 2010 and 2013. 62
Our finding is also higher than that reported in another study with a non-publication rate at 29% for large randomized clinical trials, i.e. trials with at least 500 enrolled participants. 3 Other studies reported that nearly half of the included trials were not published, which is considerably higher than our own findings.1,4,28 To explain these differences in non-publication rates, we postulate that the rapidly evolving technology elements of digital health trials could introduce extrinsic motivation to investigators to publish their results and stay ahead of the technology innovation curve.
Our results also showed that the vast majority 96.6% of digital health trials are funded by non-industry sponsors, such as universities, hospitals, medical institutes and research centers. We also found that trials funded by university sponsors were 1.5 times more compliant with trial recruitment compared to trials funded by any other sponsors. This finding indicates that investigators of university sponsored trials are excelling at adopting best practices and strategies in trial design to improve participant recruitment, which is to be expected within the academic context of the university sponsors. We speculate that industry sponsors are more interested in the broader opportunity in the digital health marketplace beyond the realm of academia and best practices of randomized trials design.
The lowest compliance with prospective trial registration was at 14.3% for industry-funded trials. In contrast to our present study, other studies assessing the prospective registration of clinical trials found that industry-funded trials were more likely to be compliant with prospective trial registration compared to non-industry-funded trials.63–70 However, we also found that only 14 (3.4%) trials of the included 417 trials were industry-funded which may limit the generalizability of our finding in comparison to other studies.
The compliance with participants recruitment was at 59.3%, which is higher than that of the McDonald et al. study that reported 31.1% and 46.7% compliance in participants recruitment compared to the original and revised target size respectively.71,72 However, the design of that study was limited to 114 muti-center trials which has its unique recruitment challenges.
73
The compliance with trial recruitment was significantly associated with the trial size (
Selective registration bias of digital health clinical trials
Despite the emphasis on prospective trial registration introduced by the 2004 ICMJE mandate and the 2008 DoH, we found that only 38.4% of all included trials were registered prospectively.22,24 Similar findings were reported by two independent studies indicating that compliance with prospective trial registration was at 31%.74,75
We hypothesize that investigators may be more inclined, or biased, to registering their trial only when submitting their results to peer-reviewed journals for publication. To support our hypothesis, we found a statistically significant relationship (
The vast majority 95.7% of trials registered within one year before publication were registered retrospectively. Our results showed that investigators, who did not register their trials promptly prior to enrollment, were required or motivated to do so only when they submit their results to scholarly journals. There may be a number of contributing factors to this selective registration bias. Firstly, the investigator may have deferred the trial registration task until the completion of the trial and only when the results are finalized and ready to be published. Secondly, the investigator may also be not aware of the ethical expectation to register their trial promptly. Lastly, journal editors and peer-reviewers are likely to suggest registration of the submitted trial publication prior to accepting the publication submission. 76 It's important to establish and broaden the adoption of prompt trial registration requirement in ethical approval and guidelines of clinical trial design at the institutional and academic level.
Challenges in oncology trials
The trial condition groups were statistically associated with the non-publication rate (
Compliance with prospective trial registration in Australia and New Zealand
The ANZCTR leads with 89.7% compliance with prospective trial registration. Also, trials from Australia and New Zealand were leading with 89.5% compliance with prospective trial registration (the 0.2 percent decline is due to one trial that was registered in the ANZCTR registry and was not located in Australia or New Zealand). The high compliance with prospective trial registration in the ANZCTR registry was acknowledged by another study indicating an incremental trend in prospective trial registration from 48% to 63% for trials registered in the ANZCTR registry between 2006 and 2012. 63
Compliance with participants recruitment in the Middle East
Digital health trials in the Middle East had the lowest non-publication rate and the highest compliance with participants recruitment at 26.7% and 81.1%, respectively. These results are influenced by the Iranian trials registered in the IRCT registry with the lowest non-publication rate and the highest compliance with participants recruitment at 25% and 88.9% respectively. All trials registered in the IRCT were funded by university sponsors. This finding aligns with another study that reported that 97% of IRCT trials were funded by university and other governmental institutions. 52 The academic and public sponsorship may be a contributing factor to low non-publication rate and high compliance in recruitment of trials registered in the IRCT as they would likely encourage a culture of adherence with best practices in trial design.
Several studies discussed the legacy of the IRCT and its role in upholding best practices in the field of clinical research in Iran.67–69 However, we found that prospective trial registration was the lowest at 8.3% for trials registered in the IRCT. Our results support findings from another study that reported on the rationale behind the low prospective trial registration rate at 8.3% for trials registered in the IRCT between 2008 and 2011. 70 The low rate in prospective trial registration in the IRCT may have contributed to highest compliance in participants recruitment. When registering the trial retrospectively, in particular towards the end of the trial, the authors would have the opportunity to adjust the target trial size in the submitted registry information to match the actual number of participants.
The inclusion of TRN in published trials was the lowest for trials registered in the IRCT at 22.2%. Combined with the lowest prospective trial registration of those trials, we postulate that investigators of IRCT trials may be unaware of the ICMJE recommendation for prospective trial registration and the WHO re-affirmation to include the TRN in the trial publication.
Adherence to best practices in clinical trials from Europe
In our study, Europe was the region with (1) the largest number (43.6%) of digital health trials, (2) the highest compliance with the inclusion of TRN (68.7%), and the second-lowest non-publication rate (28%) tied with the Middle East The overall lead in European trials is influenced by the Netherlands National Trial Register which had the second-lowest non-publication rate at 25%, and the highest compliance with inclusion of TRN at 87.5%.
Enrollment to publication duration
The majority (88.3%) of the published trials in our study were published within five years after the enrollment of the first participant. We postulated that small trials would be easier to conduct as they have fewer recruitment and enrollment challenges, hence they would likely be published in a shorter time compared to large trials. To validate our postulate, we found statistically significant differences (
Limitations
Our study is the first to our knowledge that analyzed global digital health clinical trials registered in all the WHO data provider registries. 40 Our study was limited to 417 trials registered in any of the seventeen WHO data provider registries and did not examine other registries, such as the federal office of public health's portal for human research in Switzerland, and the Philippine health research registry.77,78 As such, when identifying published trials in peer-reviewed journals, PubMed and Google search were utilized. We acknowledge that not considering trials registries other than the WHO data provider registries, not including databases other than PubMed and Google, and the limited number of included trials may have impacted the external validity of our study results. There are known sensitivity limitations with the ICTRP search portal. 79 We followed the recommendation and utilized the advance search function as described in Appendix II.
Despite the ICMJE and WHO emphasis on trial registration, not all randomized trials are registered.75,80 We did not include unregistered trials in our analysis, which may impact the internal validity of our results.
We considered the enrollment and/or trial start date as provided in the registration information of the included trials. The registration information is provided manually and voluntarily by the registering investigators who are often overwhelmed with competing priorities and limited resources. Therefore, the enrollment date provided in the trial registries may not always be up-to-date or maintained promptly.
Lastly, our study only included registered trials enrolled in 2012. Our findings predate the recent 2015 WHO calls for improving public disclosure of trial results and the linkages between these results and the trial registry entries. 25
Conclusion
In the field of digital health randomized clinical trials, the adherence of investigators to the best practices of trial registration and result dissemination is still evolving. Further research is required to identify contributing factors and mitigation strategies to low compliance rate with trial publication and prospective registration in digital health clinical trials.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-1-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-2-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-2-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-3-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-3-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-4-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-4-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-5-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-5-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-6-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-6-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-7-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-7-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-8-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-8-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-9-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-9-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-10-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-10-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Supplemental Material
sj-docx-11-dhj-10.1177_20552076221090034 - Supplemental material for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries
Supplemental material, sj-docx-11-dhj-10.1177_20552076221090034 for Prospective trial registration and publication rates of randomized clinical trials in digital health: A cross-sectional analysis of global trial registries by Mustafa Al-Durra, Robert P Nolan, Emily Seto and Joseph A Cafazzo in Digital Health
Footnotes
Conflict of interest
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Contributorship
The lead author (MAD) and senior author (JC) conceived the study idea and developed the study design with contributions from all authors. MAD executed the search queries, extracted data, analyzed the results, and drafted the manuscript. All authors interpreted results were involved in the reporting of the study, reviewing and editing the manuscript, and approved the final version of the manuscript. All authors agree to take responsibility for the work.
Ethical approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Guarantor
MAD.
Supplemental material
Supplemental material for this article is available online.
