Abstract
Background:
Pain is a common symptom in people with dementia living in nursing homes, but cognitive impairment, including language and communication difficulties, challenges pain assessment and the ability to self-report pain.
Objectives:
This study aimed to identify and summarize patterns, advances, and gaps in research literature describing pain assessment in people with dementia living in nursing homes.
Design:
We conducted a scoping review following Arksey and O’Malley’s methodological framework.
Methods:
Systematic searches were conducted in CINAHL, Embase, MEDLINE, and PsycINFO. We included studies describing pain expressions in people with dementia and/or healthcare personnel assessment of pain in people with dementia, in a nursing home context. Charted data included demographics, methodological descriptions, ethical and quality assessment and relevant findings. Relevant findings were summarized using thematic analysis, and an overview of patterns, advances, and gaps in the research literature is presented.
Results:
Thirty-nine studies were included. The results describe three patterns: (1) pain awareness; (2) suspected pain and (3) pain mapping. Collectively, these patterns constitute a process of pain assessment, integrating pain expressions of people with dementia. Important perspectives on self-reporting are touched upon in several of the included studies, though direct descriptions of attempts to capture the residents’ own experience of pain are sparse.
Conclusion:
This scoping review provides a comprehensive description of pain assessment in people with dementia living in nursing homes as a process in three steps. We identified several knowledge gaps in the understanding of this process and provide concrete recommendations for further research. The results underpin the importance of pain assessment approaches that incorporate the flexibility to meet residents’ varying and potentially fluctuating ways of communicating pain.
Trial registration:
This scoping review is registered in the Open Science Framework (https://osf.io/8kaf5/).
Background
The nursing home population is characterized by a high degree of multimorbidity
1
and polypharmacy
2
and a large proportion of nursing home residents have a moderate-to-severe degree of dementia.3,4 Studies have documented a pain prevalence in people with dementia living in nursing homes of 35%–43%,2
–4 but a possible prevalence of 60%–80%.
5
Thus, pain assessment is an important part of care for this population.
6
The International Association for the Study of Pain defines pain as ‘
The use of assessment tools can supplement challenging pain assessment and support residents’ limitations in communicating verbally. Numerous observational assessment tools targeting pain in people with dementia have been developed and evaluated10,14 and systematic use of standardized observational tools has been recommended.6,15,16 However, assessment tools only capture fragments of the overall picture. 9 The ability of people with dementia to self-report is an individual resource that healthcare personnel (HCP) should engage, promote and support. 17 At some point in the dementia trajectory, extensive cognitive impairment will make self-reporting so difficult that HCP must depend on for instance observational assessment tools. 18 Nevertheless, HCP can work purposefully to use valid self-reporting for as long as possible.19,20
A scoping review by Pringle et al. exploring the complexity of pain recognition, assessment and treatment for people living in nursing homes, found a need for training and detailed guidelines for appropriate assessment of pain in the nursing home population in general. 21 However, they did not investigate people with dementia in particular, nor focused on knowledge and tools that emphasize accounting for individual variation and the ability to self-report. A systematic review by Tsai et al. investigated the effectiveness of interventions to improve pain assessment and management in people with dementia. 22 They found that comprehensive pain models improve nurses’ pain assessment and management. However, none of the included interventions emphasized a structured approach to safeguard individuals’ residual capacity to self-report, and the review was concerned about people with dementia in general and not particularly the nursing home population. 22 Hence, to the best of our knowledge, no study has reviewed the research literature with a comprehensive perspective on pain assessment in people with dementia living in nursing homes, and how the residents’ expressions of pain are integrated into the clinical practice of HCP. Thus, the aim of this scoping review was to identify and summarize patterns, advances and gaps in research literature describing pain assessment in people with dementia living in nursing homes.
Methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) extension for Scoping Reviews checklist was used to prepare this manuscript. 23 The procedure presented in this section is derived and extended from a peer-reviewed protocol. 24 Two or more of the authors were involved in every step of the process, and methodological decisions were discussed extensively. We utilized the five first stages of Arksey and O’Malley’s methodological framework for scoping reviews, with Levac et al.’s recommendations for each stage: (1) Identifying the research questions; (2) Identifying relevant studies; (3) Study selection; (4) Charting the data and (5) Collating, summarizing and reporting the results.25,26 The method was additionally advanced by using the PAGER framework (Pattern, Advances, Gaps, Evidence for Practice and Research Recommendations). 27
Stage 1: Identifying the research questions
We searched an overview of pain assessment in people with dementia based on the clinical practice of HCP, and how it integrates pain expressions of people with dementia. To clarify the focus of the scoping review, we developed two research questions to target the broad aim of the review:
RQ1: How is the clinical practice regarding pain assessment in people with dementia living in nursing homes described in the research literature?
RQ2: How are pain expressions of people with dementia living in nursing homes described and included in the clinical practice regarding pain assessment?
Stage 2: Identifying relevant studies
A systematic search in the CINAHL, Embase, MEDLINE and PsycINFO databases was conducted. No time limit for publication was specified. We formed three main blocks in the search strategy: people with dementia (Population), pain expressions in people with dementia and/or HCP’s assessment of pain (Concept of interest) and nursing homes (Context). 28 The search strategy combines MeSH terms and synonyms within the respective blocks. When developing the search strategy, we observed that the utilization of the search terms in population and context sufficiently reduced the search results, enabling us to apply broad terms for the concept of interest, preventing the exclusion of relevant studies. The search strategy went through several rounds of revision and quality assurance in collaboration with experienced librarians and the full search strategy is available as Supplemental Material (Additional File 1). The main search was carried out in December 2022 and updated in May 2024. The reference lists of the included studies were manually searched.
Stage 3: Study selection
Inclusion and exclusion criteria are presented in Table 1.
Eligibility criteria guiding study selection.

SE and CKO independently reviewed the first 300 abstracts prior to discussing and reaching consensus on the discrepancies. CKO solely reviewed the remaining abstracts. Rayyan 30 was used as a tool for team-based screening, and sources that subsequently matched the inclusion criteria were obtained for full-text assessment. If the relevance of a study was unclear from the title and abstract, the full article was reviewed. All full texts were independently assessed for eligibility by two researchers. Several calibration meetings were held during the selection process, and disagreements were discussed until consensus was reached.
Stage 4: Charting the data
Data from 13 studies, randomly selected among the included, were extracted and reviewed by two researchers (CKO and SE) to determine consistency in the understanding of the studies’ compatibility with the research questions and aim. Data from the remaining studies were charted by CKO alone. The final data-charting form was reviewed and approved by all authors, including demographics, aim and research questions, methodological descriptions and relevant findings. Levac et al. argue the importance of quality assessment in scoping reviews to achieve information on the quality of existing knowledge. 26 Therefore, all authors made an informal assessment of quality during the full-text review and noted any quality deficiencies. Study quality was then assessed using the Mixed Methods Appraisal Tool (MMAT). 31 SE and CKO independently assessed 10 studies, and CKO solely assessed the remaining. Reflecting the rationale for quality appraisal in scoping reviews, no studies were excluded based on the appraisals. 26 The importance of ethical awareness in reviews has been emphasized. 32 In response, we conducted an ethical mapping inspired by Westerdahl et al. 33 The ethical mapping considered the description of ethical approval, informed consent, data protection, financial support and conflict of interest.
Stage 5: Collating, summarizing and reporting the results
In this stage, we prepared an overview and summary of the extracted information, which is presented in the results section. The review includes both quantitative and qualitative data. The quantitative results have been transposed into descriptive phrases, and the descriptive summary is formulated in text. Our results are described and discussed in line with the PAGER framework. 27 Hence, a descriptive thematic analysis of the key findings, was conducted to identify patterns in the research literature; reading, rereading and coding the data, then generating initial themes, which were reviewed and refined several times. 34 As a scoping review intends to summarize, not synthesize, the results are presented descriptively on a semantic level, using the same terms as those used in the referenced studies where feasible. 25
Results
A total of 3954 unique records were assessed by title/abstract after duplicates were removed. The selection process is documented in a PRISMA flowchart (Figure 1). 35
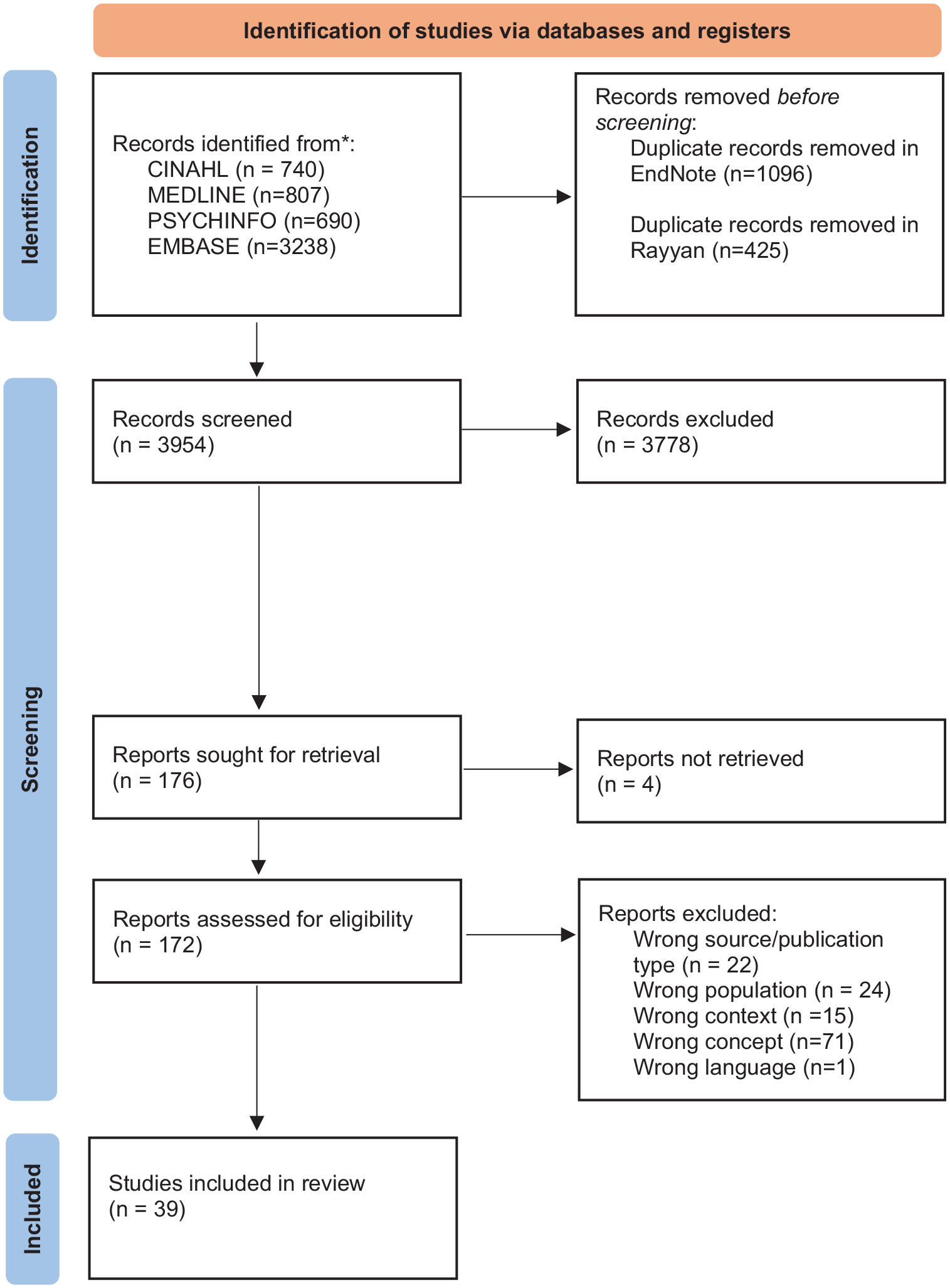
Overview of study selection process.
Thirty-nine studies met all the inclusion criteria. Twenty-three had a quantitative approach, seven a qualitative approach and nine had a mixed or multiple-method approach. The studies were published between 2002 and 2024, in Asia (
Presentation of studies included.

We identified three patterns in the thematic analysis in which HCP are assessing pain in people with dementia living in nursing homes: (1) pain awareness; (2) suspected pain and; (3) pain mapping. Collectively, these patterns constitute a
Pattern 1: Pain awareness
HCP must actively search for pain in people with dementia. 39 ‘Pain awareness’ concerns how HCP are aware that pain might occur, as well as their alertness, knowledge and understanding of the situation. To discover pain, it must be prioritized, and it requires a combination of familiarity with the resident and professional expertise with pain and dementia.40,41 Pain awareness can also have a preventive and protective aspect, for example by checking positions to avoid painful pressure ulcers.41,42
Pattern 2: Suspected pain
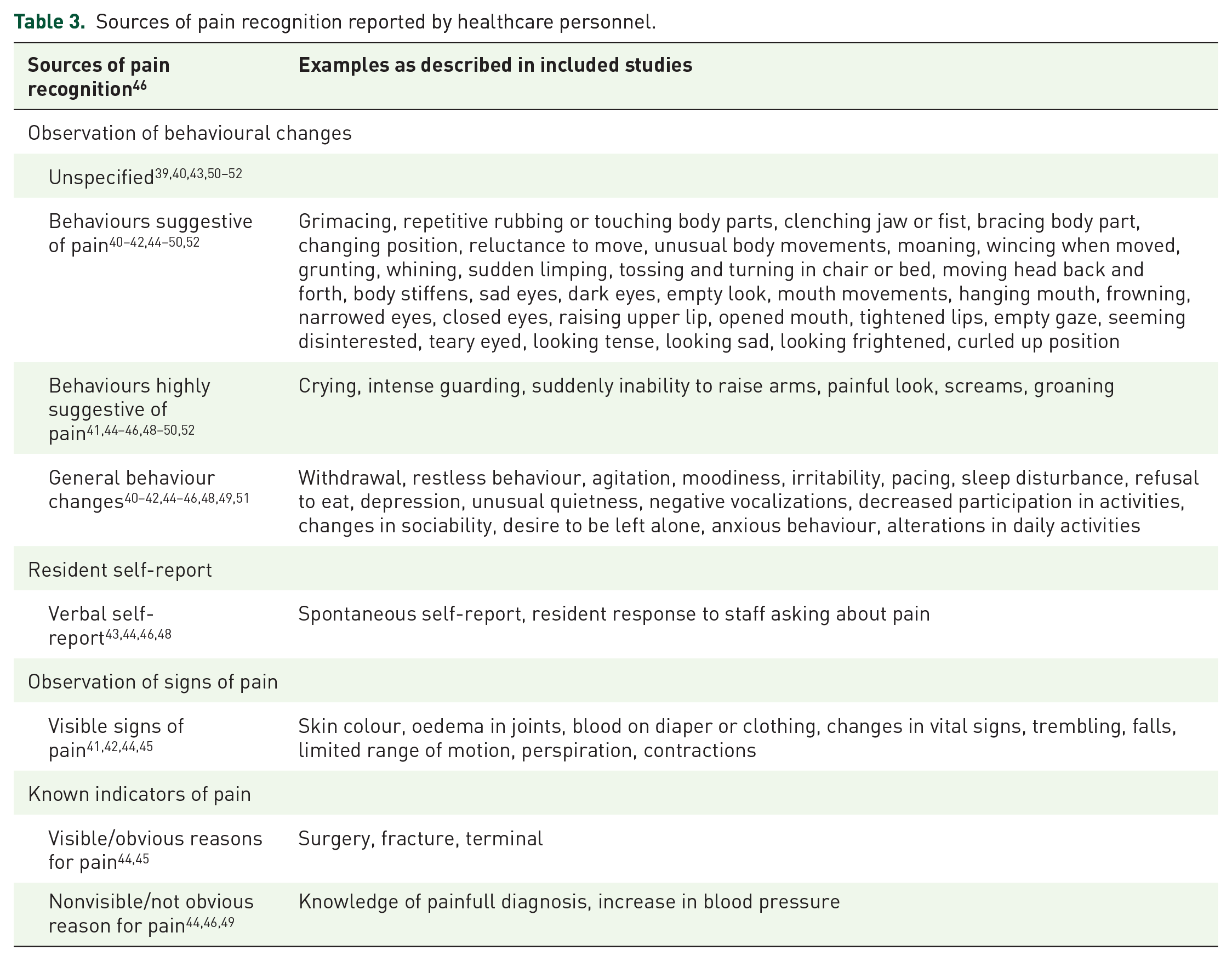
‘Suspected pain’ refers to the moment when HCP recognize that a person with dementia might be in pain. The included studies describe several sources of suspected/recognized pain: (a) observation of behavioural changes39 –54; (b) verbal self-reports43,44,48,53,54; (c) observation of signs of pain 41,42,44,45,54 and (d) known indicators of pain.44,46,49 This categorization is based on the conceptual model of how HCP engage in identifying and deciding whether to treat the residents’ pain, developed by Gilmore-Bykovskyi and Bowers. 46 The model describes how the presence or absence of an obvious reason for pain, influences HCP’s levels of certainty regarding pain. Behavioural change in people with dementia might result in suspected pain but with a high degree of uncertainty – especially in the absence of an obvious reason. 46 Gilmore-Bykovskyi and Bowers present three groups of behavioural indicators: behaviours suggestive of pain (e.g. repetitive rubbing of a body part), behaviours highly suggestive of pain (e.g. intense guarding with care) and general behaviour changes (e.g. withdrawal or agitation). 46 Ford et al. compared behavioural pain expressions across different ethnic groups and found no significant differences, only the words used to describe pain. 55
Observable signs of pain are emphasized, and the most described are (a) behavioural changes that differ from baseline behaviour42,44 –46,49,52,56; and (b) facial expressions of pain.41,42,45,47,52 –54,57 ‘Knowing the person’ is highlighted as a crucial prerequisite for recognizing changes from baseline, to identify unique individual pain behaviours and detecting and interpreting pain-related changes in people with dementia.40 –42,44 –46,48,52 Family members are described as important resources,39,48,56 as they may be familiar with the residents’ earlier behaviours, and capable of interpreting their present behaviours. 48 However, though HCP can distinguish behavioural changes from baseline, the behavioural changes might have other causes.46,50,51 As Alzheimer’s dementia progresses, observable pain behaviours might diminish and the observation of pain behaviour will be even more difficult. 58
The different sources of pain identification reported by HCP in the included studies are presented in Table 3.
Sources of pain recognition reported by healthcare personnel.
Pattern 3: Pain mapping
Pain mapping is complex and refers to the specific and more comprehensive part of pain assessment. Pain mapping can be both regulatory-driven (i.e. ‘on admission’) or patient-driven (i.e. ‘the person appears to be in pain’),
52
where HCP builds upon their suspicion of pain, and/or attempts to determine the underlying cause of the residents stated pain or behaviour that suggests pain. One study found that pain assessment driven by regulation was prevalent.
52
The state of knowledge is unclear, but there is insufficient evidence to conclude that regular pain mapping using a pain assessment tool is
There are high validity, reliability and agreement between physicians in the pain assessment of people with dementia with mild/moderate levels of cognitive impairment, but these dropped in the assessment of residents with severe cognitive impairment. 60 Assistive personnel assigned less pain intensity and affective distress to the person in pain when the person was described as severely ill with Alzheimer’s dementia, compared to when the stage of dementia was not stated. 61
The perspective of pain mapping in dementia will further be described according to: (a) pain assessment tools; (b) a combination of pain mapping strategies; and (c) self-reporting.
Pain assessment tools
Several studies report the use of pain assessment tools as part of pain assessment in clinical practice.40,53,37,45,46,47,49 However, the included studies provide limited descriptions of the relationship between the clinical use of assessment tools, degree of dementia and residual capacity to self-report. There are significant differences in HCP use of standardized assessment tools, both interpersonal62,63 and between different types of assessment tools. 64 Registered nurses and assistive personnel using standardized assessment tools largely agreed on the presence of pain at the moment but agreed to a lesser extent on how often pain occurred in the past week. 63 One study reported poor agreement between tools based on observation compared to self-reports. 64 Registered nurses reported the use of assessment tools to a greater extent than assistive personnel. 49
A study by Closs et al found that two-thirds of the participants with moderate or severe dementia were able to use simple self-report assessment scales. 65 Many of those who when asked, claimed to have no pain indicated that they had pain when they used pain scales. 65 In contrast, another study found that participants with moderate-to-severe dementia unable to use verbal tools often could use nonverbal tools. 54
Combination of pain mapping strategies
Several of the included studies describe a combination of strategies, where HCP assess and integrate information from various sources including review of medical records40,50, physical examination44,45,66,67, medical history 44 and intuition. 48 The scope of registered nurses’ pain assessment increased with severe dementia and a high degree of uncertainty. 68 A study investigating the combination and weighting of different sources in pain assessment, found that mapping multiple indicators of pain was not necessarily more appropriate than one single proxy report. 69 Team meetings with interdisciplinary evaluations of pain for people with dementia report less pain than assessment with standardized observational tools. 66
Several of the included studies described trialling different combinations of pharmacological and nonpharmacological interventions targeting various potential underlying causes of changed behaviour, including pain.39,44,46,50,68 This is described as ‘trial and error’, and the goal is that the person with dementia will return to baseline functioning with the reduction or elimination of their behavioural symptoms.44,46
Self-reporting of pain
The use of self-reports was highlighted as the most meaningful, when possible. 40 At the same time, several of the included studies describe the difficulties HCP experience when communicating with people with dementia, and this is one of the major barriers to recognizing and assessing pain in the group.39,41,42,46,48,51 There are different points of view when it comes to self-reporting of people with dementia. Two studies stated that a large proportion of the included people with dementia were unable to verbally self-report,64,70 and 78% of HCP believed that people with dementia could not accurately provide a self-report of pain, 56 another study (44%) stated that people with dementia could verbalize at least ‘some pain’ if their pain management were ineffective. 71 Three of the included studies compared HCP reports of pain with the residents’ reports of pain, and the findings are contradictory.63,72,73 People with dementia reported higher prevalence, 63 intensity and frequency 72 compared to HCP. On the other hand, assistive personnel is found to score pain as significantly higher than the people with Alzheimer’s dementia themselves. 73 One study found no significant differences between the prevalence of self-reported pain symptoms when comparing people with and without dementia. People with dementia reported higher pain intensity, were less likely to tell HCP about their pain, and fewer reported that HCP asked about their pain, compared to people without dementia. 74
Two studies found that a large proportion of the included people with dementia were unable to verbally self-report.64,70 Cohen-Mansfield found significantly higher scores on the Mini-Mental State Examination 29 in the responders to self-report questions, than in non-responders. 64 Chen and Lin’s findings indicate that people with dementia with up to a moderate level of cognitive impairment may be able to self-report, despite limitations in communication and self-awareness. They highlight that HCP should accept the pain reports of people with dementia to promote adequate pain management, and in addition, use a multifaceted approach for those in the later stages of dementia. 72
Integrating the patterns into a coherent process of pain assessment
Collectively, the three identified patterns constitute a pain assessment process.
This process is largely characterized by uncertainty due to cognitive impairment affecting the person’s ability to verbally express pain, and difficulty establishing certainty regarding the underlying causes of pain.39,46,48,50,51,68,75 Significantly fewer pain assessments are carried out on people with dementia in nursing homes, compared to people without dementia. 36
The process of pain assessment involves different HCP disciplines and roles.39,40,42,52 To connect the various aspects, the process relies on continuity in relation to communication and information.39,40,52,60 Pain assessment is described as a complex network of communication channels in the nursing home, and communication between different disciplines is problematized in several studies.40 –42,52,67 Poor or inaccurate documentation and communication could be a barrier to effective pain assessment. 51 Andrews et al. found that 83% of the pain episodes investigated contained documentation only about the problem and the intervention. 43 The use of a pain management protocol may address these challenges, as it may provide a common language for staff to talk about pain across disciplines and help to strengthen the communication of pain observations.67,76 The use of an electronic systematic pain assessment protocol to help HCP identify visual patterns in pain scores over time has been promoted. This could also be a faster and easier way to store and access data. 67
Summary of results
We identified three patterns describing the current state and advances of research concerning the pain assessment process in people with dementia living in nursing homes: (1) pain awareness; (2) suspected pain and (3) pain mapping. Patterns, advances and gaps in the research literature concerning pain assessment in people with dementia living in nursing homes are summarized in Table 4.
Patterns, advances and gaps in the included studies.

Discussion
In this review, we aimed to identify and summarize patterns, advances and gaps in research literature describing pain assessment in people with dementia living in nursing homes. We included and examined 39 studies, finding that pain assessment is described as a process, facilitated by uninterrupted information transfer. We identified perspectives of importance on self-reporting, but direct descriptions of self-reporting and attempts to capture the patient’s own experience of pain were sparse.
Evidence for practice and research recommendations
Our findings highlight and illuminate aspects of pain assessment that are important to reflect on in clinical work with this patient group. Bradbury-Jones states that the evidence for practice using the PAGER framework also targets a broader understanding of the practice field, involving stakeholders beyond clinicians (e.g. researchers). 27 Evidence to inform practice and research recommendations seen in such a context can contribute by providing concrete recommendations for further research responding to identified knowledge gaps. 27 The gaps that need to be addressed are presented in Table 4, and the most prominent are elaborated and discussed in this section.
People with dementia’s limited ability to verbally communicate, constitute major challenges and this is highlighted in the literature as a problem that must be addressed. 19 Hence, the literature is focused on objective assessment alternatives when self-reporting cannot be carried out: these alternatives include the development, testing and implementation of assessment tools. 10 However, there are nuances between ‘fully capable of self-reporting’ and ‘not at all capable of self-reporting’. Our findings show limited descriptions of how to support people with dementia to communicate their subjective experiences of pain; how HCP can assess the ability/residual ability for self-reporting and how to integrate different pain assessment strategies at different degrees of the residual capacity of the target group to self-report. Self-reporting is mainly described as whether or not the person is able to confirm or deny the presence of pain and to describe the severity of the pain. Descriptions of self-reported pain in the included studies are largely quantified. Qualitative descriptions of the subjective experience of pain are not emphasized, either in those with mild or moderate dementia. Quantitative pain measures are vital in pain management but often overlook important attributes of the subjective experience, such as personal context and meaning, which can have a major impact on the experience of pain. 9 There is a knowledge gap regarding the promotion of systematic individualized pain assessment and how to place reported pain, the results of assessment tools or clinical examinations into a larger context. Wideman highlights the need for assessment models that specifically emphasize how to address subjectivity related to pain in general. 9 Our results show that this might be even more challenging in people with dementia. Nevertheless, we claim that models of pain management in this group and context can have the flexibility to meet individual residents’ varying and potentially fluctuating ways of communicating pain, as well as their individual need for assessment, intervention and evaluation.
The results describe ‘trial and error’ strategies: the use of interventions as part of an assessment to find the underlying cause of behavioural changes. Due to risk of delayed treatment, ‘trial and error’ should follow a thorough pain mapping. However, we found that pain mapping will not eliminate all uncertainty, and ‘trial and error’ can be appropriate for instances where uncertainty cannot be eliminated. There is a lack of knowledge concerning strategies for systematic implementation and evaluation of ‘trial and error’, where this is unavoidable. Sandvik et al. discuss how people with dementia receive painkillers as much as or more than people without dementia, in contrast to an earlier trend of undertreating pain due to assessment challenges. 77 People with dementia in nursing homes constitute a population with a high degree of multimorbidity that is vulnerable to pharmacological side effects.1,78 The evaluation of implemented measures is therefore particularly important. These factors highlight the importance of further developing and implementing models that facilitate the systematic evaluation and informational continuity of any pain intervention: both as a result of a specific pain assessment or ‘trial and error’.
We found that pain awareness in particular was described as having a preventive function. Systematic work to prevent pain in this population is described in the included studies to a limited extent. Pain prevention is outside the scope of this review, but in a patient group with such a high prevalence of pain, prevention should be a priority in both clinical practice and future research. 10 Liao et al. state that there is a lack of knowledge about dementia and pain among HCP, which can be solved with easy access to ongoing training. 79 Although competence-enhancing measures were outside the scope of this review, we acknowledge this as an important topic that should be highlighted in further studies.
Strengths and limitations
An important strength of this study was the guidance by a peer-reviewed protocol. 24 We used an established methodology25,26 and analysis method, 34 as well as standardized reporting guidelines. 23 To ensure transparency, the review process is described in detail.
This study has some limitations. First, searches, screening and selection of studies are open to error or bias. We acknowledge that this review may not have captured all relevant material, as we did not include grey literature, nor studies published in other languages than English and the Nordic languages. The search strategy resulted in a large volume and wide range of evidence. Another team of researchers might have included and chosen to emphasize other areas of the research field.
We conducted an assessment of quality and ethical standards. Levac et al. argue how quality appraisal is an important aspect of mapping and identifying gaps in the existing literature, giving comprehensive information on the nature and extent of those gaps. 26 The MMAT guidelines are standardized. 31 However, the appraisal is vulnerable to bias, as the result depends on the interpretation of the researcher. We sought rigour by involving all members of the research team in the quality appraisal. Studies with low methodological quality are not excluded in this scoping review, following methodological recommendations, 26 which contributes to a complementary description of the research field. Hence to this, a second limitation is that studies with less robust evidence and a high risk of bias are not excluded, and results must be used cautiously.
Conclusion
This scoping review provides a comprehensive picture of the existing research on pain assessment in people with dementia living in nursing homes as a process with three steps; it also contributes to the understanding of highly complex nursing processes in this group and context. It has identified several knowledge gaps in the understanding of this process and provides concrete recommendations for further research. The phenomenon of self-reporting in people with dementia is insufficiently explored, and there is limited knowledge on how HCP relates to varying degrees of residual capacity to self-report. The results underpin the importance of pain assessment approaches that have sufficient flexibility to meet individual residents’ varying and potentially fluctuating ways of communicating pain.
Supplemental Material
sj-docx-1-pcr-10.1177_26323524241308589 – Supplemental material for The process of pain assessment in people with dementia living in nursing homes: a scoping review
Supplemental material, sj-docx-1-pcr-10.1177_26323524241308589 for The process of pain assessment in people with dementia living in nursing homes: a scoping review by Caroline Kreppen Overen, Maria Larsson, Adelheid Hummelvoll Hillestad, Ingela Karlsson and Siren Eriksen in Palliative Care and Social Practice
Supplemental Material
sj-docx-2-pcr-10.1177_26323524241308589 – Supplemental material for The process of pain assessment in people with dementia living in nursing homes: a scoping review
Supplemental material, sj-docx-2-pcr-10.1177_26323524241308589 for The process of pain assessment in people with dementia living in nursing homes: a scoping review by Caroline Kreppen Overen, Maria Larsson, Adelheid Hummelvoll Hillestad, Ingela Karlsson and Siren Eriksen in Palliative Care and Social Practice
Supplemental Material
sj-docx-3-pcr-10.1177_26323524241308589 – Supplemental material for The process of pain assessment in people with dementia living in nursing homes: a scoping review
Supplemental material, sj-docx-3-pcr-10.1177_26323524241308589 for The process of pain assessment in people with dementia living in nursing homes: a scoping review by Caroline Kreppen Overen, Maria Larsson, Adelheid Hummelvoll Hillestad, Ingela Karlsson and Siren Eriksen in Palliative Care and Social Practice
Supplemental Material
sj-docx-4-pcr-10.1177_26323524241308589 – Supplemental material for The process of pain assessment in people with dementia living in nursing homes: a scoping review
Supplemental material, sj-docx-4-pcr-10.1177_26323524241308589 for The process of pain assessment in people with dementia living in nursing homes: a scoping review by Caroline Kreppen Overen, Maria Larsson, Adelheid Hummelvoll Hillestad, Ingela Karlsson and Siren Eriksen in Palliative Care and Social Practice
Supplemental Material
sj-pdf-5-pcr-10.1177_26323524241308589 – Supplemental material for The process of pain assessment in people with dementia living in nursing homes: a scoping review
Supplemental material, sj-pdf-5-pcr-10.1177_26323524241308589 for The process of pain assessment in people with dementia living in nursing homes: a scoping review by Caroline Kreppen Overen, Maria Larsson, Adelheid Hummelvoll Hillestad, Ingela Karlsson and Siren Eriksen in Palliative Care and Social Practice
Footnotes
Appendix
Acknowledgements
We would like to thank Annelie Ekberg-Andersson and Linda Borg (Information Specialists at Karlstad University) and Kari L. Mariussen (Head Librarian at Lovisenberg Diaconal University College) for their contribution to the search strategy presented in this scoping review. We would also like to thank the staff at the library at Lovisenberg Diaconal University College for helping us obtain articles to which we did not have access.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
