Abstract
Background:
Palliative care (PC) is an added layer of support provided concurrently with cancer care and serves to improve wellbeing and sustain quality of life. Understanding what is meaningful and a priority to patients, their families, and caregivers with lived experience of cancer and PC is critical in supporting their needs and improving their care provision. However, the impacts of engaging cancer patients within the context of PC research remain unknown.
Objective:
To examine the impacts of engaging individuals with lived experience of cancer and PC as partners in PC research.
Methods:
An a priori systematic review protocol was registered with PROSPERO (CRD42021286744). Four databases (APA PsycINFO, CINAHL, EMBASE, and MEDLINE) were searched and only published, peer-reviewed primary English studies aligned with the following criteria were included: (1) patients, their families, and/or caregivers with lived experience of cancer and PC; (2) engaged as partners in PC research; and (3) reported the impacts of engaging cancer PC patient partners in PC research. We appraised the quality of eligible studies using the Critical Appraisal Skills Program (CASP) and GRIPP2 reporting checklists.
Results:
Three studies that included patient partners with lived experience of cancer and PC engaged at all or several of the research stages were identified. Our thematic meta-synthesis revealed impacts (benefits and opportunities) on patient partners (emotional, psychological, cognitive, and social), the research system (practical and ethical) and health care system (service improvements, bureaucratic attitudes, and inaction). Our findings highlight the paucity of evidence investigating the impacts of engaging patients, their families and caregivers with lived experience of cancer and PC, as partners in PC research.
Conclusions:
The results of this review and meta-synthesis can inform the more effective design of cancer patient partnerships in PC research and the development of feasible and effective strategies given the cancer and PC context patient partners are coming from.
Keywords
Introduction
Both challenges and opportunities within health care services can be strategically leveraged to advance knowledge and strengthen the quality and provision of patient care. Including and prioritizing patients’ voices, needs, and urgencies, and partnering with them in health research that informs practice and care is instrumental to achieving holistic person-centered care. The evidence is now substantive and wide-ranging that the inclusion of patient and community members within the health research continuum can inform health care policy and practice, and benefit patients, researchers, research ecologies, and the health care system.1,2 Moreover, patient and community-informed research yields more inclusive research results that can be accessed and implemented sooner, and in more universal, useable, and equitable ways.1,2
Language about the inclusion of ‘patients’ and ‘community members’ collaborating as partners on health-related research teams is variably defined and described globally and across participatory action research (PAR) methodologies.2–5 Current international initiatives are presented in Table 1 to highlight these nuances. We will adopt the Canadian Institutes of Health Research’s (CIHR) Strategy for Patient-Oriented Research (SPOR) framework of patient engagement (PE) – the active and meaningful collaboration of patients as partners in any and/or all phases of the research continuum 1 – to promote clarity and consistency surrounding the concept of engaging patients as partners.
Participatory action research (PAR) initiatives and approaches.
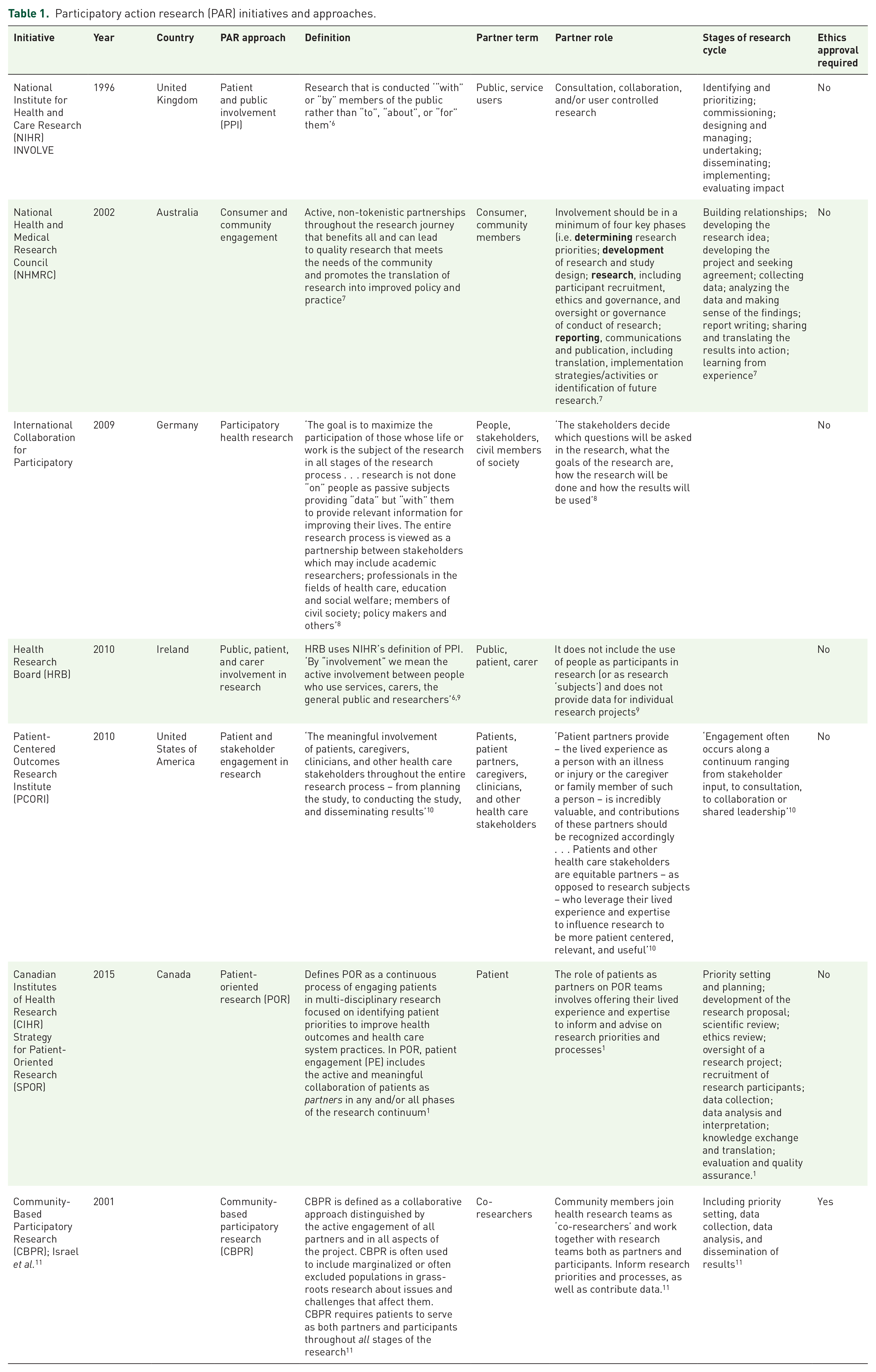
Despite the lack of consensus in language across the international landscape, engaging patients as partners moves patients beyond the traditional view of mere data contributors (i.e. participants) to a more involved collaboration; the research focus shifts from being ‘on or about’ those impacted to research ‘with’ those the research impacts. Existing primary studies and synthesis reviews have reported on the impacts of partnering with patients, with various disease-specific lived experiences (e.g. acute, chronic, cancerous, non-cancerous)12,13,14,15 and across various health care contexts (e.g. non-palliative,16,17 palliative). 18 However, to our knowledge, there are no studies that have studied the impacts across multiple contexts (i.e. cancer and palliative).
Palliative care (PC) is an added layer of support provided concurrently with cancer care and serves to improve wellbeing and sustain quality of life. 19 A new public health approach to PC, involving PAR, has been recommended to address the needs of people with complex and advanced conditions. 20 While previous studies report on how cancer patients, or PC patients, separately, have served as partners, the impacts of engaging patients with lived experience of both cancer and PC within the context of PC research remains unknown. Current knowledge gaps exist regarding whether patients with lived experience of cancer and PC have unique, disease-specific needs and experiences affecting their involvement in serving as research partners compared with patients with other health conditions (e.g. asthma, diabetes, cancer). Understanding what is meaningful and a priority to patients, their families, and caregivers with lived experience of cancer and PC is critical in supporting their needs and improving their care provision.
The objective of this systematic review is to examine the impacts of engaging patients with lived experience of cancer and PC as partners in PC research. We aim to learn how individuals with lived experience of cancer and PC have been engaged as partners in PC health research projects and to understand the impacts of this engagement on: (1) patient partners, (2) research projects, and (3) health care systems. We will use the findings from this knowledge synthesis to generate recommendations for the future design and conduct of meaningful research within cancer and PC research.
Methods
This systematic review’s protocol has been registered with PROSPERO (CRD42021286744) and the review has been reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement. 21
Adopted definitions and framework
CIHR’s SPOR 1 definitions of a patient, PE, and patient-oriented research (POR) were adopted for this review. A ‘patient’ refers to an individual (e.g. patient, family member, friend, caregiver) with lived experience of cancer and PC. 1 Thus, we refer to patient partners with lived experience of cancer and PC as Cancer Palliative Care Patient Partners (CPCPP). Our definition of ‘patient’ is linked and comparable with other studies using PE in Alberta.13,22 We used and modified the Art and Humanities Research Council’s (AHRC) 23 definition of ‘impacts’ to include anything that influenced the research and affected patient partners and/or researchers at an individual, community, and/or policy development level.
We used the SPIDER framework 24 to operationalize our research question. The sample included adult patients, and/or their families and caregivers with lived experience of cancer and PC. Our phenomenon of interest was the involvement of this sample as partners in PC research. We anticipated that through our review we would obtain studies that were qualitative or mixed-methods in design and evaluate the impacts of involving CPCPPs in the PC research process.
Applied methods of PE
Based on the scope and aim of our systematic review, we determined that it would not be appropriate to partner with patients in any stage of our study.
Search strategy
A systematic search strategy was developed with the support of a health research librarian in MEDLINE and translated to three other electronic databases (i.e. APA PsycINFO, CINAHL, and EMBASE) on November 14, 2021 (Table 2). Four comprehensive concepts related to our question of interest were combined to obtain relevant search results. Each query was comprised terms related to one of the following concepts: (1) PC, (2) patient/family/caregiver, (3) patient-oriented and PE research, (4) impacts, and (5) cancer. Our search strategy included two search combinations, one with and without the cancer concept. We imported results based on the search strategy without cancer and manually screened for the concept of cancer to ensure a more comprehensive search. We identified additional candidate studies from the reference lists of eligible studies and excluded gray literature and conference abstracts. Only English language full-text primary studies, regardless of publication date, were considered for inclusion.
OVID MEDLINE search strategy.

Study eligibility and selection
Following the execution of the search strategy, search results from each electronic database were imported to Covidence. 25 Two reviewers (AP and IN) independently screened titles and abstracts in duplicate, met regularly to discuss any eligibility criteria-related inquiries that arose, and consulted a third senior reviewer (MS) to resolve any disagreements. Full-text screening followed a similar procedure and full agreement was obtained.
Study eligibility criteria are highlighted in Table 3. We only excluded titles and abstracts, and full-text studies if: (1) only patients with lived experience of non-cancer illnesses and/or PC were engaged; (2) patients with lived experience of cancer and PC were solely engaged as participants instead of partners in PC research; (3) patients with lived experience of cancer and PC were engaged in research evaluating PC services; or (4) if they evaluated a PC intervention. Therefore, we only included published, peer-reviewed primary studies in the English language that aligned with our inclusion criteria: (1) adult patients, their families, and/or caregivers with lived experience of cancer and PC; (2) engaged as partners in PC research; and (3) reported on the impacts of engaging CPCPPs in PC research. We also included articles if there was a mix of patient partners with lived experience of cancer and non-cancer illnesses.
Systematic review eligibility criteria.

Patient partners were differentiated from participants if they assisted in carrying out the study at any point (e.g. research planning, development, recruitment, data collection, analysis, dissemination, knowledge translation) during the research process and did not contribute any data. Studies that did not fit these criteria were not included. In addition, there were no limits for the following: (1) the study publication date; (2) study design (i.e. qualitative, quantitative, mixed-methods); (3) geographical context; or (4) patient partner characteristics (e.g. sex, gender, age, type of cancer diagnosis, stage of patient’s cancer).
Data extraction and quality assessment
Data were extracted from studies that met our inclusion criteria using a standardized form developed based on the protocol. One reviewer (AP) pilot tested this data extraction form with a single included study and then extracted data on the remaining included studies. Subsequently, a second reviewer (IN) verified the extracted data for accuracy and comprehensiveness. Data extracted from each study were related to three domains: (1) study demographics (i.e. year of publication, country study was conducted); (2) study information (i.e. research questions, aims/objectives; theoretical underpinnings; research type; study design; patient partners’ characteristics including sex, age, sample size, type of patient partner, type of cancer diagnosis, stage of patient’s cancer; study methodology and methods; data analyses; key study findings; reported study strengths, limitations, future research directions and recommendations stated by the authors); and (3) the impacts (e.g. on patient partners, researchers, the research, and/or health care system) of cancer PE in PC research.
To maintain consistency in our approach, the quality of all included studies was independently assessed by two reviewers (AP and IN) using two evidence-based assessment tools. Any discordance was resolved through discussion or by a third senior reviewer (MS). The qualitative methodological quality of included studies was assessed using the Critical Appraisal Skills Program (CASP). 26 To assess the quality, transparency, and consistency of the international PE evidence base reported by included studies, the Guidance for Reporting Involvement of Patients and Public (GRIPP2) short-form checklist was used. 27 Despite our anticipation of eligible mixed-methods studies, we did not encounter any studies using this design that fit our inclusion criteria and, therefore, did not need to use the Mixed-Methods Appraisal Tool (MMAT) Version 2018. 28
Data synthesis
Given all the studies that met our eligibility criteria were qualitative, the heterogeneity of, and lack of reported empirical data in our included studies, precluded the statistical pooling of findings for a meta-analysis and any subgroup analyses. Instead, study characteristics were tabulated and narratively synthesized to integrate and explore relationships within the data. We used a qualitative meta-synthesis approach; 29 specifically, a thematic synthesis (i.e. line-by-line coding of primary study findings; the development of descriptive and analytical themes) 30 to thematically translate, integrate, and describe all relevant qualitative findings from our eligible studies. 30 One reviewer (AP) independently and inductively coded each line of verbatim text (e.g. original authors’ findings) that reported impacts of PE in cancer PC research. Themes were developed by systematically ‘going beyond the findings’ of eligible studies and generating additional concepts or understandings. 30 Team discussions were held to check for interpretation consistency, further refine emergent descriptive and analytical themes, and verify themes in relation to our review aim to ensure robustness.
Results
Description of included studies
Study flow
Overall, a total of 1,957 possibly relevant studies were identified through the four electronic databases: APA PsycINFO (n = 212), CINAHL (n = 486), EMBASE (n = 791), and MEDLINE (n = 472). Covidence removed 676 duplicates leaving a new total of 1,281 potentially eligible studies for titles and abstract screening. Of these, only 35 full texts underwent retrieval, uploading, and secondary screening. The primary reasons for excluding most of the full-text studies included: partnering with a non-cancer and/or pediatric population; using PE language incorrectly (e.g. if patients were described as ‘partners’ or ‘co-researchers’ but did not actually collaborate on research processes and only contributed data that were subsequently analyzed by researchers without lived experience expertise); employing a non-PAR study design; or study aims that were not focused on PE impacts. A total of three studies were included in the final data extraction and synthesis phase. Figure 1 displays the PRISMA flow diagram of the selection of articles through the different phases of the systematic review.

PRISMA (2020) flow diagram of the systematic review process.
Study characteristics
We identified three studies31–33 that engaged CPCPPs at any or all the research stages defined in patient and public involvement (PPI) 6 and POR. 34 As highlighted in Table 4, the included studies were published between 2006 and 2016, and all three studies: (1) gathered data in the United Kingdom; (2) implemented a PPI approach; (3) employed a qualitative study design; and (4) were situated within non-Indigenous cultural contexts.
Study characteristics.

Patient partner and engagement characteristics
Demographics
Of the three included studies, the number of patient partners recruited to engage in the PC research were 2, 32 4, 33 and 15. 31 While most studies reported on the sex of their participants, Forbat et al. 33 study was the only one to report on both the sex and age of their patient partners (i.e., females, over 18 years of age). Thus, most studies did not report on patient partner characteristics, including race and ethnicity.
Lived experiences
As aligned with our inclusion criteria, all individuals involved as partners in each study had lived experience of both cancer and PC. Aside from Forbat et al. 33 mentioning that their patient partners were 2 years post-bereaved, the authors of all three studies did not describe patient partners’ years of cancer and PC lived experience. Regarding the type of patient partners involved, Cotterell et al. 32 did not specify, while Forbat et al. 33 partnered with former carers, and Wright et al. 31 partnered with both patients and carers but did not specify whether their carers were current, former, or both. Similarly, Wright et al. 31 and Cotterell et al. 32 did not report who their partners cared for, while Forbat et al. stated that their patient partners cared for ‘their parents (n = 3), spouse (n = 1), and child (n = 1)’ 33 (p. 762). Except for Wright et al. 31 mentioning that two of their patient partners were in receipt of PC services, Cotterell et al. 32 and Forbat et al. 33 did not describe whether their partners were currently experiencing cancer and PC. In addition, none of the three studies provided information regarding the type of cancer(s) patient partners were affiliated with. Only Wright et al.’s 31 study stated patient partners were affiliated with advanced-stage cancer.
Roles and stages of engagement
The authors of each included study referred to their patient partners as ‘co-researchers’31,33 and ‘service user researchers’. 32 Table 5 displays the focus of each included study, the process for patient partner involvement, patient partner roles or contributions, and the levels of engagement and impact (i.e. on the patient partners, research projects, and health care system). Included studies reported a spectrum of PE in research activities (i.e. priority setting and planning, development of the research proposal, scientific review, ethics review, oversight of a research project, recruitment of research participants, data collection, data analysis and interpretation, knowledge exchange and translation, and evaluation and quality assurance), which we aligned with CIHR’s research lifecycle 34 in Table 6. Wright et al.’s 31 reference-group members were engaged with the study design and development of patient information sheets; later, they joined as patient and carer co-researchers. Cotterell et al. 32 involved service user researchers and a research advisory, comprised experienced researchers in service user involvement, and patients with lived experience of cancer and PC who advised on the study and data interpretation.
Patient partner characteristics in included studies. a

Table column headings replicate those used by Bird et al. 36
Level of Engagement framework adopted from Manafo et al. 2
Summary of stages of patient engagement in palliative care research lifecycle.

Research partnership characteristics
Recruitment strategies
The three studies employed different strategies for identifying and recruiting patient partners. Wright et al. 31 stated that they developed a reference group through patient forums of UK cancer networks where members later joined as co-researchers. In addition, co-researchers were identified from a participating hospice day care service using a targeted approach and through collaboration with a hospice clinical team. 31 Forbat et al. 33 only reported opportunity sampling by recruiting their co-researchers from carer organizations, while Cotterell et al. 32 did not specify how their patient partners were identified and recruited.
Training and compensation
As one of CIHR’s 1 guiding pillars of PE, support of patient partners in research collaborations can be reflected in the training opportunities offered and provided to patient partners. Wright et al. 31 reported individually and group training (i.e. 90 minutes long) their co-researchers. In addition, Wright et al. 31 held mock focus groups with their co-researchers and provided additional sessions upon request to accommodate co-researchers while receiving PC services. Forbat et al. 33 reported training their co-researchers in conducting qualitative interviews and trained interview respondents who chose to join as a co-researcher. Forbat et al. 33 also mentioned that co-researchers were not instructed regarding the extent to which they should share details about themselves or ‘what not to say’. By contrast, Cotterell et al. 32 did not report on any training of their service user researchers. Forbat et al. 33 and Cotterell et al. 32 did not mention providing their patient partners with any financial compensation, and while Wright et al. 31 reported that financial resources were given to patient partners, they did not provide any details regarding what compensation was given.
Quality reporting assessment
Qualitative methodology assessment
Quality appraisal scores for the qualitative methodology of all included studies are shown in Table 7. As all three included studies utilized a qualitative study design, Cotterell et al. 32 and Wright et al. 31 used focus groups, while Forbat et al. 33 employed interviews. Two reviewers (AP and IN) independently assessed whether each of the studies appropriately reported each of the following sections: (1) a clear statement of aims, (2) suitable qualitative methodology, (3) research design, (4) recruitment strategy, (5) data collection, (6) researcher-participant relationship considerations, (7) ethical considerations, (8) rigorous data analysis, (9) clear statement of findings, and (10) extent of the study’s value. Studies by Wright et al. 31 and Forbat et al. 33 demonstrated high quality across all ten domains; Cotterell et al. 32 demonstrated high quality across nine of ten domains but did not clearly report on any ethical considerations. Given the congruence between the qualitative components of the included studies, a strong overall quality reporting score resulted for each study.
Quality appraisal of qualitative design in included studies using CASP (2018) Checklist. 26

Note: Y, Yes; CT, Can’t Tell; N, No.
PE reporting assessment
A quality assessment of the reporting of PE in our included studies can be found in Table 8. Again, for each of the included studies, two reviewers (AP and IN) independently assessed five components of quality PE reporting: (1) aims, (2) methods, (3) study results, (4) discussions and conclusions, and (5) reflections and critical reflections of PE. Since the studies by Forbat et al. 33 and Wright et al. 31 were the only two studies to addressed each of these components, we allocated a strong quality score for them both. Although Cotterell et al. 32 reported on four of the five components, they did not provide discussion, conclusions, or critical reflections on their PE. As a result, we gave this study a moderate quality score.
Quality appraisal of patient and public involvement (PPI) reporting in included studies using GRIPP2 Short-Form Checklist (2017). 27

Note: Page numbers are displayed to indicate the page each component is reported on; NR, not reported.
Thematic meta-synthesis on patient partnership impacts
Reported impacts related to cancer PC patient partnerships in PC research from each of the included studies were thematically synthesized into benefits (i.e. anything that was identified as a positive influence, produced a helpful outcome, or promoted the wellbeing of the CPCPPs, researchers, research, and/or the health care system) and opportunities (i.e. anything that was identified as a need for improvement or to advance the wellbeing of the CPCPPs, researchers, research, and/or the health care system). As illustrated in Figure 2, eight sub-themes emerged from our thematic meta-synthesis.

Emergent model from meta-synthesis.
Impact on patient partners
Partnering with cancer PC patients in PC research has been associated with various emotional, psychological, cognitive, and social experiences. Impacts to each of these four domains will be discussed in relation to the benefits, opportunities, or both.
Emotional
Several benefits surrounding CPCPPs’ emotional health and wellbeing were synthesized across the included studies. Emotional benefits, as an emergent sub-theme, can include living with purpose, having a sense of personal achievement, being able to express emotions in a safe environment, and displaying emotional agility. Some CPCPPs viewed the collaborative approach in PC research as offering them an opportunity to make active and meaningful contributions – for their own benefit as they approached the end of their lives, and for those coming after them in utilizing PC services and the research community. 32 PE in cancer PC research encouraged CPCPPs to live with purpose, ‘be part of shaping new and more appropriate treatment for others going through a similar experience’, and pay back service to the health professionals and system that treated and supported them. 32 Taken together, CPCPPs described their engagement as helping them ‘live well with cancer’ and ‘refocus their lives in a positive, purposeful, and productive way’ by supporting their cancer survivorship. 32
CPCPPs also shared a few engagement opportunities regarding their emotional wellbeing. Some felt they were unable to achieve all they desired in their engagement roles given a lack of clarity, vision, aim, and understanding about their involvement and role expectations. 32 This invoked frustration and emotional distress among CPCPPs collaborating in PC research. Other CPCPPs reported emotional burdens related to ‘hearing accounts and discussing their own personal experiences’, 31 compounded by cancer discussions with clinicians and researchers who came across as negative, insensitive, and dismissive of CPCPPs’ needs. Some health professionals seemed void of emotion when discussing project and health care-related issues and information that were profoundly emotional to CPCPPs. 32 Thus, some research teams ensured emotional supports were available to CPCPPs. 31
Psychological
Numerous psychological benefits were reported by the included studies. Psychological health and wellbeing can include sense of self, self-confidence, mental health, and inspiration and motivation. CPCPPs reported a sense of enhanced self-confidence based on their research involvement, sensing all that they could achieve, and sharing their lived experiences with others. 32 Partnering in PC research also enabled CPCPPs to negate the effects of their cancer illness while their overall mental health improved. For instance, CPCPPs reported their involvement was ‘a positive way to keep active, combat depression and loneliness, and deal with their cancer diagnosis and treatment’, dispel feelings of hopelessness, and help them differentiate between the disease and the person. 32
CPCPPs’ research involvement also positively impacted study participants. During interviews, participants viewed CPCPPs as role models for their perseverance and engagement in research. 32 This inspired both participants and CPCPPs to ‘fight’ their cancer. CPCPPs were highly motivated to improve PC services and make meaningful contributions to PC research. 32
A critical opportunity related to patient partners’ engagement and psychological wellbeing involved one’s concept of self in relation to one’s diagnosis. ‘It cannot be assumed that co-researchers are at ease in conducting research with other patients on account of their diagnosis alone’ 31 (p. 824). It may be demotivating and a trigger for CPCPPs to relive their ‘trauma’, especially with those who do not share in their lived experiences. Patient partners with lived experiences of both cancer and PC are recommended. 31
Cognitive
Cognitive health and wellbeing are promoted through continuous learning and involve displaying attentiveness, information processing, and mental flexibility to carry out daily activities. Collaborating with CPCPPs in PC research created learning opportunities and a positive space for participants and CPCPPs to engage in enriching discussions. 32 When CPCPPs co-interviewed participants, a ‘co-construction’ of their caregiving identities emerged. 33 Moreover, providing appropriate, iterative, effective, and collective training was cited as a ‘necessary’ benefit when collaborating with CPCPPs. 31 Training was reported to support CPCPPs’ involvement in research activities, build their research knowledge in addition to their experiential knowledge, and support their ability to be ‘valuable contributors to the research process’.31,33
CPCPPs also shared engagement opportunities regarding their cognitive wellbeing. While CPCPPs expressed the importance of opportunities to gain experience in data collection, they acknowledged it was not always practical or feasible for advanced cancer patients receiving PC to travel to training events. 31 Similarly, CPCPPs reported considerations regarding limited attention span and restricted physical movement due to advanced stages of cancer, making it important to find tasks that could accommodate their current deteriorated state. 31
Social
Social health is another sub-theme that emerged from our meta-synthesis and can be described as relationship building and understanding, connectedness with others, and community, communication, and support. CPCPPs and experienced researchers reported an appreciation for building and sustaining their ongoing team relationships. 31 CPCPPs acknowledged how some experienced researchers displayed understanding regarding their unspoken needs and challenging circumstances. For instance, when CPCPPs displayed exhaustion while co-interviewing participants, researchers alleviated CPCPPs from their role; there was ‘an understanding that they could leave the study at any time without giving a reason’ 31 (p. 823).
Other benefits reported by CPCPPs were enhanced self-confidence, feeling a ‘sense of belonging’, and an ‘ability to contribute’ to the research. 32 CPCPPs expressed that their research engagement enabled them to: (1) achieve personal and collective goals; (2) be part of a supportive community where their experiences were accepted and understood; and (3) be inspired by other CPCPPs as role models. 32 Similarly, former carers (i.e. CPCPPs) were able to co-construct caregiving identities through interactional sequences of shared experiences while co-interviewing current carers with lived cancer and PC experience. 33
Other forms of social support reported to be important for patient partner engagement included offering alternatives that could fit with their needs and circumstances; for instance: regular check-ins with patient partners to reassess their needs; clearly detailing their partner roles; offering flexible training sessions (individual based and/or as a collective); recording focus groups, so that, those who are unable to attend can listen to them on their own time and still feel connected to the team and involved in the research; and obtain travel insurance to cover the transportation of CPCPPs in researchers’ cars. 31
There were also opportunities around how to navigate relationships between CPCPPs and experienced researchers, and CPCPPs and participants. CPCPPs perceived there to be power imbalances, which made them feel: (1) undervalued, undermined, used, and marginalized; (2) that their contributions were not perceived as credible; and (3) that they were peripheral to decision-making, core research activities, and priorities. 32 Furthermore, CPCPPs sensed that the staff had ‘tokenistic attitudes about their involvement’ in the research, and that there could be better job networking opportunities for them. 32
Due to a lack of understanding and clarity around CPCPPs’ roles, some additional tensions and interactional difficulties in sharing experiences were reported. For example, when CPCPPs spoke about their experiences during interviews, participants would change the topic of conversation and adopt ‘antithetical stances constructing contrary rather than collaborative accounts’ 33 (p. 766). On other occasions, it was reported that the barrier between experienced researchers and CPCPPs became blurred when CPCPPs shared similar experiences and were known to participants. 31
Impact on research
Impacts on the PC research projects due to cancer PC patient partnerships involved practical (i.e. recruitment practices; characteristics and retention of engaged patient partners) and ethical (i.e. maintaining anonymity) benefits, opportunities, or both.
Practical
The sole practical research benefit identified from the collaborative cancer PC patient partnership involved the co-generation of data by both the interviewer (i.e. CPCPPs) and interviewee. 33 This produced a richer description and understanding of the lived experiences of cancer and PC.
Practical research opportunities that emerged from the meta-synthesis, involved the tensions between balancing the desire to involve CPCPPs at all stages of the research cycle (i.e. from initial design and recruitment, from data collection and analysis, to writing up and dissemination) with the realities advanced CPCPPs face when receiving PC services. 31 The recruitment and data collection stages with advanced CPCPPs will take longer than is usual or expected; these processes will be interrupted as the CPCPP’s health changes over time, thus impacting the generation, production, and dissemination of meaningful results from the study. 31 In addition, longer completion times will require additional funding supports and should include the sharing of emerging findings with patient partners over shorter time spans. 31
Ethical
One ethical opportunity that was reported as a research impact involved maintaining participant and CPCPP anonymity. In research, identity anonymity ensures dynamics are not upset and responses inhibited during discussions, such as through focus groups. 33 However, in the cancer and PC community, it is not uncommon for CPCPPs and research participants to ‘know each other’. 31
Impact on health care system
Due to the original aims of the included studies, we only identified two sub-themes (i.e.
Discussion
Summary of the results
Our systematic review revealed a dearth of research on PE in cancer and PC in general, but especially over the last 7 years, as the most recent studies identified were conducted in 2010 and 2016. While research has been conducted internationally (i.e. the United Kingdom), our synthesis shows that PE has not been applied within other national cancer and PC research contexts. The quality of identified studies, as assessed by CASP 26 and GRIPP2, 27 were congruent with all areas of conducting rigorous qualitative research and addressed all the CIHR’s 1 four pillars of PE; however, there is room for improvement in the reporting of PE to align with the GRIPP2 form checklist. 27
Our systematic review and meta-synthesis explored and described an array of impacts (i.e. benefits, opportunities) on patient partners (i.e. emotional, psychological, cognitive, social), the research system (i.e. practical, ethical) and health care system (i.e. service improvements, bureaucratic attitudes and inaction), based on established cancer PC research partnerships (Figure 2). All three of the studies included in our systematic review and meta-synthesis engaged CPCPPs as active and equal research team members which coincide with the definitions and foundations of both the UK’s PPI 6 and CIHR’s SPOR. 34 Impacts were disproportionately represented in our emergent themes depending on the studies’ original focus and more detailed reporting and transparency of their application of PPI. Still, all themes were derived from our three included studies and are interconnected. For instance, CPCPPs’ engagement in PC research elicits meaningful contributions which in turn can motivate CPCPPs to fight their cancer and feel a sense of personal achievement. Not only were CPCPPs impacted by their engagement, but participants were also impacted by other CPCPPs’ engagement, as were the research and health systems. This is particularly shown through the inspiration and motivation, and relationship and power dynamics concepts from the emotional and social health and wellbeing themes.
Some of the impacts to CPCPPs were tied to the research and health system impacts and are consistent with previous health research findings. Like our meta-synthesis, others have reported on how patient partners perceive a sense of personal achievement in paying forward the meaningful contributions they make (i.e. emotional benefits) which, in turn, helps keep them ‘actively distracted’ and able to separate their ‘sense of self’ from the ‘disease’ (i.e. psychological benefits).35–39 PE has been found to offer a sense of generativity (versus stagnation) in creating an opportunity to build and expand upon research knowledge and skills (i.e. cognitive benefits), and develop a sense of community and belonging (i.e. social benefits).15,40,41,17 However, there is evidence that a lack of role clarity can negatively impact the engagement of health research partners and create frustration and emotional distress (i.e. emotional and cognitive opportunities).13,17
Noteworthy, there are also unique findings that emerged from our meta-synthesis. For instance, CPCPPs experienced several emotional and physical burdens that inhibited their involvement capacity. CPCPPs reported that sometimes listening to others or recounting their own personal health stories, and experiencing various dismissive and insensitive responses from clinicians and researchers, resulted in distress (i.e. emotional opportunity). CPCPPs expressed that simply possessing a disease diagnosis should not be presumed to equate to their comfort in collecting data from patients with other disease diagnoses (i.e. psychological opportunity). Even though most remained engaged, due to the complex nature of their lived experiences (cancer and PC) and deteriorating health, CPCPPs’ training, involvement capacity, and duration in the research were limited (e.g. cognitive opportunity). Still, co-constructing caregiving identities through interactions and various research innovations (e.g. recording focus groups, paying for travel insurance for transportation), enabled them to remain connected to the team and research process (i.e. social opportunity).
Systematic review and meta-synthesis study limitations and strengths
Despite our use of rigorous and previously established systematic review methods, there are some study limitations worth considering. First, the differences in language used to define and engage patients as partners in the research studies we reviewed, may have resulted in us missing relevant articles, thereby impacting the comprehensiveness of our synthesis. Relatedly, we may have omitted eligible studies from our synthesis by excluding non-English language studies. Finally, although representative of the current state of the literature, there is a disproportionate representation of relevant studies conducted worldwide; all three of our included studies were conducted in the United Kingdom, which may limit the generalizability of our findings to other geographical contexts.
Aside from these limitations, there are also notable strengths of our work. One strength of this review is that we developed an a priori protocol (ID# CRD42021286744) and submitted it for registration to maintain clarity, transparency, and reproducibility. Second, despite the paucity of published research on the cancer PC patient population, our review findings illustrate the various impacts of engaging them as partners in PC research. Finally, the combination of our convergent and unique findings on CPCPPs suggests feasible methods and recommendations that may enhance future research in this area.
Recommendations and future directions
Our systematic review and meta-synthesis findings highlight the importance of PE in cancer PC and strengthening research training programs (e.g. training on co-conducting focus groups and interviews) for CPCPPs and researchers, to create more accommodating and flexible partnerships throughout the entire research process and to enhance the quality of the data collected. We recommend that future research look at additional, feasible, and effective strategies for engaging CPCPPs in PC research even amid their illness reality. Researchers should ask CPCPPs what they need to be adequately and appropriately supported, prior to their study engagement and subsequently revisit this throughout each stage of their involvement. In addition, we suggest more diverse perspectives (e.g. abilities, ages, ethnicities, gender identities, geographic locations, language groups, racial communities, sexual orientations) 42 be invited to partner in cancer PC research to ensure equitable and meaningful engagement. Based on observations from our study, Table 9 presents a compiled list of recommendations on the collection and reporting of information for future PE cancer PC research, which should be followed, whenever possible.
Reporting recommendations.

Given the international diversity in the language of PE frameworks, we encourage patient partners and health care researchers to strive for consensus building in nomenclature. We also recommend offering CPCPPs more appropriate, flexible, and adaptable PE frameworks for a more responsive approach to the unique opportunities CPCPPs bring to a PC research project. Manafo et al. 2 state that patient partners are usually engaged in the beginning of the research process. Although Wright et al. 31 and Cotterell et al. 32 engaged CPCPPs at the priority setting stage, all three included studies31–33 primarily engaged CPCPPs in the later stages (e.g. data collection) of the PC research projects. This observation calls for an investigation of the specific reasons why CPCPPs were not engaged throughout all stages of the research process.
Conclusion
Taken together, our study highlights the impacts on CPCPPs and participants involved in PC research and the health care system. Findings from our review highlight the need to consistently apply a PE framework, which might increase the uptake and inclusivity of CPCPPs in future PE cancer PC research. The results of this review can inform the more effective design of cancer PC patient partnerships in PC research and the development of feasible and effective strategies given the cancer and PC context patient partners are coming from.
